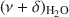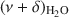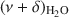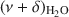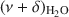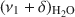Volume 64 - Issue 5 - October 2016
Article
Sequestration of Catechol and Pentachlorophenol by Mechanochemically Treated Kaolinite
- Valeria Ancona, Paola Di Leo, Maria Donata Rosa Pizzigallo
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 513-522
-
- Article
- Export citation
-
The pollution of soils by organic contaminants, such as phenols, is a serious problem because of the high toxicity and persistence in the environment. Mechanochemical treatments (MTs) of polluted soils with minerals, such as clays and oxides, which have surfaces that exhibit catalytic properties, have been suggested to be a new useful strategy to promote both organic and inorganic pollutant degradation. Nevertheless, much still remains to be studied about the capability of clays to promote pollutant removal by means of the mechanochemical activation of the mineral surfaces. This work investigates the efficiency of the mineral kaolinite in promoting the sequestration of catechol (CAT) and pentachlorophenol (PCP) by MT. A well crystallized kaolinite (KGa-1b) was milled for prolonged times with different amounts of organic molecules so as to obtain two different clay:organic compound ratios. Prolonged grinding and a higher clay mineral:organic compound ratio were found to be more effective in promoting a stronger removal than simple contact. After 1 h of mechanochemical treatment, the PCP and CAT removal percentages were 32% and 20%, respectively. Additionally, a 7-day undisturbed incubation of the milled mixtures produced a trend for increased CAT removal (up to 40%). The interaction mechanism between kaolinite and each organic compound (i.e. CAT and PCP) after a MT was inferred by integrating information from spectroscopic, diffractometric, and chromatographic analyses. X-ray diffraction and Fourier-transform infrared data suggested a strong interaction between CAT and KGa-1b. This interaction mechanism likely occurs through the formation of an inner-sphere complex by H-bonding between the organic molecules and the oxygens of the kaolinite tetrahedral sheet. On the other hand, a weak interaction (i.e. van der Waals type) can occur between the KGa-1b O-planes and the PCP molecules, which likely bind to the external surfaces of KGa-1b.
XRD and TEM Studies on Nanophase Manganese Oxides in Freshwater Ferromanganese Nodules from Green Bay, Lake Michigan
- Seungyeol Lee, Huifang Xu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 523-536
-
- Article
- Export citation
-
Freshwater ferromanganese nodules (FFN) from Green Bay, Lake Michigan have been investigated by X-ray powder diffraction (XRD), micro X-ray fluorescence (XRF), scanning electron microscopy (SEM), high-resolution transmission electron microscopy (HRTEM), and scanning transmission electron microscopy (STEM). The samples can be divided into three types: Mn-rich nodules, Fe-Mn nodules, and Fe-rich nodules. The manganese-bearing phases are todorokite, birnessite, and buserite. The iron-bearing phases are feroxyhyte, goethite, 2-line ferrihydrite, and proto-goethite (intermediate phase between feroxyhyte and goethite). The XRD patterns from nodule cross sections suggest the transformation of birnessite to todorokite. The TEM-EDS spectra show that todorokite is associated with Ba, Co, Ni, and Zn; birnessite is associated with Ca and Na; and buserite is associated with Ca. The todorokite has an average chemical formula of Ba0.28p2+(Zn0.14p+2Co0.05p3+Ni0.02p2+) (Mn4.99p4+Mn0.82p3+Fe0.12p3+Co0.05p3+Ni0.02p2+) O12·nH2O. Barium is the main cation in the structural tunnels of the todorokite. The average chemical formula of birnessite and Ca-buserite are: Na0.14p+Ca0.19p2+(Mn1.48p4+Mn0.52p3+0.52) O4·nH2O and Ca0.27p2+(Mn1.46p4+Mn0.54p3+)O4·nH2O, respectively. Most nodules have a concretionary structure of alternating Fe/Mn layers, commonly with a core of reddish feldspar containing phyllosilicates and hematite micro-crystals. Other cores consist of goethite, cristobalite, tridymite, hercynite, hematite-bearing quartz, coal, and chlorite-bearing rock fragments. The hexagonal or hexagonal-like structures of hematite micro-crystals and clay minerals in the cores may serve as heterogeneous nucleation sites for the Mn-oxides and Fe-(oxyhydr)oxides. The alternating Fe/Mn layers in FFNs might be caused by oscillatory redox condition fluctuations at the sediment-water interface due to changes in water level.
Surface Crystal Chemistry of Phyllosilicates Using X-Ray Photoelectron Spectroscopy: A Review
- Chiara Elmi, Stephen Guggenheim, Reto Gieré
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 537-551
-
- Article
- Export citation
-
The characterization of freshly cleaved mica surfaces for surface structure and chemical composition was briefly reviewed and focused on surface crystal chemistry using X-ray photoelectron spectroscopy (XPS) and other surface-sensitive techniques. This paper considers micas, which are useful as a first approximation for the behavior of many clay surfaces. Emphasis was given to phyllosilicate XPS binding energies (“chemical shift”), which were described and used to obtain oxidation state, layer charge, and chemical bonding information from the chemical shifts of different peaks. The chemical shift of the Si2p binding-energy to lower values can result from a negative charge increase because of Si4+ replacement by Al3+ and/or Fe3+. The apparent interlayer coordination number reduction from twelve to eight at muscovite and tetraferri-phlogopite (001) surfaces was indicated by the XPS measured K2p binding-energy and is consistent with bond relaxation. Although chemical shifts are valuable to distinguish chemical bonding and oxidation state, chemical shifts usually cannot distinguish between different Al coordination environments where Al is in both tetrahedral and octahedral sites.
Synthetic Zeolites Derived from Fly Ash as Effective Mineral Sorbents for Diesel Fuel Spill Remediation
- Pingqiang Gao, Yan Zhang, Lin Zhao
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 552-559
-
- Article
- Export citation
-
Development of an effective sorbent for diesel fuel spill remediation remains an important challenge in the field of synthesis due to the potential capacity of sorbents to efficiently purify contaminated sites. Fly ash, a coal combustion by-product, was used as a raw material to synthesize two inexpensive zeolites (SZ-1 and SZ-2) for oil spill remediation using an alkali fusion approach prior to hydrothermal treatment. The sorbents were characterized using scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray diffraction (XRD), and N2 adsorption/desorption. Diesel fuel sorption was used to examine the potential capacity of the synthetic zeolites to sorb oil and other petroleum products. Diesel fuel viscosity and density were determined at room temperature using a viscometer and a pycnometer, respectively. The synthetic zeolites exhibited a higher diesel fuel sorption capacity than fly ash. The SZ-1 zeolite sorbed approximately 1.43 g·g−1 and SZ-2 sorbed approximately 1.9 g·g−1. The sorption was mainly a physical process and mesopore filling seemed to play the dominant role. Sorbent textures were, therefore, vital for the sorption of petroleum products.
Adsorption of Cu(II) on Rhamnolipid-Layered Double Hydroxide Nanocomposite
- Yan Li, Hao-Yu Bi, Hui Li, Yong-Sheng Jin
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 560-570
-
- Article
- Export citation
-
A rhamnolipid-layered double hydroxide (RL-LDH) nanocomposite, derived from the rhamnolipid (RL) biosurfactant, was synthesized through a delamination/reassembling process. The adsorption characteristics of Cu(II) on RL-LDH were investigated in detail and the results indicated the potential of using RL-LDH as an environmentally friendly adsorbent to remove Cu(II). The fabricated RL-LDH nanocomposite was characterized using powder X-ray diffraction, Fourier-transform infrared spectroscopy, scanning electron microscopy, transmission electron microscopy, elemental chemical composition, and specific surface area analyses. Batch adsorption experiments were conducted to study the influence of various factors, such as contact time, initial Cu(II) concentration, temperature, initial solution pH, and electrolyte concentration on Cu(II) adsorption by the RL-LDH nanocomposite. The RL-LDH nanocomposite had a low surface area of 11.71 m2 g−1, which suggests that surface adsorption would not be important in Cu(II) adsorption. The Cu(II) adsorption data fitted the Freundlich model well at pH 5.5, whereas the adsorption kinetics were accurately described by a pseudo-second-order kinetics model. Chemical binding, that is, the formation of a RL-Cu(II) complex in the LDH interlayer, was assumed to be the rate-limiting step in the adsorption process. Thermodynamic parameters that included Gibbs free energy, enthalpy, and entropy changes were also calculated. The adsorption was found to be a spontaneous and exothermic chemisorption process. Furthermore, the adsorption properties of RL-LDH for Cu(II) were compared to Cu(II) adsorption using other adsorbents.
Near-Infrared Study of Water Adsorption on Homo-Ionic Forms of Montmorillonite
- Valéria Bizovská, Helena Pálková, Jana Madejová
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 571-585
-
- Article
- Export citation
-
The potential of near-infrared (NIR) spectroscopy to track the adsorption of water on montmorillonite saturated with different exchangeable cations is demonstrated in the present study. The Na+, K+, Ca2+, and Mg2+ forms of JP montmorillonite (Jelšový Potok, Slovakia) were first dried and then hydrated at 23, 52, 88, and 100% relative humidity (RH). The combination band of water molecules, $\end{document}
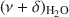 , allowed the study of the effect of exchangeable cations on the strength of H bonds between water molecules and on the amount of adsorbed water. With increasing ionic potential (IP) of the exchangeable cation, the strength of the H bonds increased and the \$\end{document}
, allowed the study of the effect of exchangeable cations on the strength of H bonds between water molecules and on the amount of adsorbed water. With increasing ionic potential (IP) of the exchangeable cation, the strength of the H bonds increased and the \$\end{document} band was shifted to lower wavenumbers. The area of the \$\end{document}
band was shifted to lower wavenumbers. The area of the \$\end{document}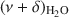 band, corresponding to the amount of adsorbed water, was compared with results from gravimetry. The good correlation (R2 > 0.97) between the two independent methods confirmed that the \$\end{document}
band, corresponding to the amount of adsorbed water, was compared with results from gravimetry. The good correlation (R2 > 0.97) between the two independent methods confirmed that the \$\end{document}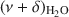 band area reflected reasonably well the amount of H2O in montmorillonite. The peak-fitting analysis of the \$\end{document}
band area reflected reasonably well the amount of H2O in montmorillonite. The peak-fitting analysis of the \$\end{document}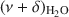 band allowed differentiation of weakly and strongly H-bonded water molecules. The position of the high-frequency component at 5260–5250 cm−1, related to H2O weakly H-bonded to basal oxygens of the tetrahedral sheets, was influenced only slightly by the exchangeable cations. Two low-frequency components were assigned to the combination modes involving asymmetric (ν3) and symmetric (ν1) stretching vibrations of strongly H-bonded H2O. Only the \$\end{document}
band allowed differentiation of weakly and strongly H-bonded water molecules. The position of the high-frequency component at 5260–5250 cm−1, related to H2O weakly H-bonded to basal oxygens of the tetrahedral sheets, was influenced only slightly by the exchangeable cations. Two low-frequency components were assigned to the combination modes involving asymmetric (ν3) and symmetric (ν1) stretching vibrations of strongly H-bonded H2O. Only the \$\end{document}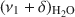 component (5055–5000 cm−1) showed significant dependence on the type of exchangeable cation and hydration level. Peak-fit analysis revealed a small effect of the type of exchangeable cation on the amount of water molecules weakly H-bonded to the siloxane surface but a pronounced effect on the content of strongly hydrogen-bonded H2O. The amount of weakly H-bonded H2O remained stable after reaching a certain level of hydration, but a gradual increase in the strongly H-bonded water molecules with increasing RH was observed.
component (5055–5000 cm−1) showed significant dependence on the type of exchangeable cation and hydration level. Peak-fit analysis revealed a small effect of the type of exchangeable cation on the amount of water molecules weakly H-bonded to the siloxane surface but a pronounced effect on the content of strongly hydrogen-bonded H2O. The amount of weakly H-bonded H2O remained stable after reaching a certain level of hydration, but a gradual increase in the strongly H-bonded water molecules with increasing RH was observed.
Clays in the Critical Zone: An Introduction
- Paul Schroeder
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 586-587
-
- Article
- Export citation
Clay Minerals in Deeply Buried Paleoregolith Profiles, Norwegian North Sea
- Lars Riber, Henning Dypvik, Ronald Sørlie, Ray E. Ferrell, Jr.
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 588-607
-
- Article
- Export citation
-
Recent discoveries of oil in deeply buried paleoregolith profiles on the Utsira High, Norwegian North Sea, was the first time basement rocks had been demonstrated to be petroleum reservoirs on the Norwegian continental shelf. The present study aimed to establish the processes responsible for the primary weathering sequence, distinguish them from other phases of alteration, and create a model for the development of reservoir properties in crystalline basement rocks.
Hand-specimen and laboratory tests revealed a link between reservoir properties in weathered granitic rocks and alteration facies. Samples were obtained from two distinct paleoregolith profiles on the Utsira High. The core samples were studied in detail by optical microscopy, X-ray powder diffraction, scanning electron microscopy, and X-ray fluorescence. In the altered coherent rock facies, porosity and permeability were mainly created by joints and fractures prior to subaerial exposure. In the altered compact rock and altered incoherent rock facies, the development of reservoir properties was increasingly affected by physicochemical interactions between the rock and percolating fluids during subaerial exposure and early diagenesis. In well 16/3-4, the altered coherent rock facies contained R0 illite-smectite (I-S), well ordered kaolinite, and a mixture of fine-grained mica and illite, produced in semi-open and closed microsystems. In the altered compact rock and altered incoherent rock facies, disordered kaolinite became more abundant at the expense of R0 I-S, well ordered kaolinite, plagioclase, and biotite, suggesting alteration in semi-open microsystems. The collapse of the rock structure and clogging of mesofractures by clays contributed to reduced permeability in the clay-rich upper part of the altered incoherent rock. In contrast, well 16/1-15 represented a more deeply truncated weathering profile compared to 16/3-4, characterized by open and interconnected mesofractures and moderate formation of clay. R0 I-S was present and kaolinite was rare throughout the profile, suggesting stagnant conditions. During burial, a porosity-reducing serpentine-chlorite Ib β = 90° polytype formed in the overlying sandstone and the regolith. Application of these results should improve the success of exploration and production efforts related to hydrocarbon reservoirs in the altered crystalline basement.
Illite-Smectite-Rich Clay Parageneses from Quaternary Tunnel Valley Sediments of the Dutch Southern North Sea — Mineral Origin and Paleoenvironment Implications
- Branimir Šegvić, Antonio Benvenuti, Andrea Moscariello
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 608-627
-
- Article
- Export citation
-
The Pleistocene sediment infill of elongated glacial incisions of the southern North Sea (SNS) is often referred to as tunnel valleys (TVs). The depositional environment is not yet fully understood and the present study addresses this challenge from the perspective of clay-mineral transformation (illite to illite-smectite) reported from the largest Elsterian TV of the SNS. Material acquired from the K14-12 borehole in the Dutch offshore was analyzed by X-ray diffraction (XRD), electron microscopy, electron microprobe, and laser particle-size analysis. Illite and illite-smectite (I-S) appeared as dominant clays along with minor amounts of kaolinite, kaolinite-smectite, and chlorite. The largest amount of I-S is recognized in the main TV portion, while in pre-glacial and uppermost deposits, I-S is less abundant. The XRD peak fitting and deconvolution suggest that I-S consists of several intermediates — ordered (well crystallized illite + R3 I-S) and disordered (R0 I-S + R0 I-SS). Given the average particle sizes (>2 µm) and Kübler index values (0.415–0.341°Δ2θ), illite as well as chlorite and kaolinite were interpreted as detrital. On the basis of the distinctive distribution, grain sizes, and compositional variations of I-S, formation by means of early diagenetic in situ smectitization of illite under a cold climate is proposed. The process operated via a series of mixed-layer intermediates derived from an illite component being converted progressively to low-charged smectite. The reaction is marked by a significant net loss of K and Al with replacement by Si in a tetrahedral coordination. Layer-charge imbalance is accommodated by Fe(III) and Mg entering an octahedral sheet, whereas Ca partly fills the interlayer sites. Smectitization rates were controlled by illite grain sizes. The results of the present study support strongly the existence of an ice-marginal freshwater depositional environment at the glacial maximum in the SNS in which early diagenesis at low temperatures resulted in incomplete conversion of illite to smectite.
Adsorption of Soil-Derived Humic Acid by Seven Clay Minerals: A Systematic Study
- Rebecca A. Chotzen, Tamara Polubesova, Benny Chefetz, Yael G. Mishael
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 628-638
-
- Article
- Export citation
-
Humic acid (HA)-clay complexes are well known for their contribution to soil structure and environmental processes. Despite extensive research, the mechanisms governing HA adsorption are yet to be resolved. A systematic study was conducted to characterize the adsorption of a soil-derived HA to seven clay minerals. Clay surfaces affected HA adsorption directly due to structural differences and indirectly by altering solution pH. The following order of HA removal was obtained for the clay minerals at their natural pH: illite ≫ palygorskite > kaolinite > sepiolite > montmorillonite = hectorite ≫ talc. Removal of HA (precipitation and adsorption) by kaolinite and illite was attributed to the low pH they induce, resulting in protonation of the clay and HA surfaces. In spite of the low pH, the zeta potential for HA remained negative, which promoted HA adsorption to the protonated clay surfaces by ligand exchange. Ionic strength did not affect HA adsorption to clay minerals with low zeta potentials, indicating that charge screening is not a major mechanism of HA adsorption for these minerals, and supporting the suggestion that ligand exchange is the main adsorption mechanism to pH-dependent sites. The increase in ionic strength did, however, promote HA adsorption to clay minerals with high zeta potentials. At pH 8–9 the order of HA affinity for clay minerals was: palygorskite >>sepiolite > montmorillonite = hectorite > kaolinite > illite > talc, emphasizing strong HA interactions with the fibrous clays. This strong affinity was attributed to their large surface areas and to strong interactions with OH groups on these clay surfaces. Results indicated that HA did not enter the intracrystalline channels of the fibrous clays but suggested that their macro-fiber structure facilitates HA adsorption. The sorption of HA to kaolinite further increased in the presence of Cu2+, and the sorption of Cu2+ increased in the presence of HA, due to a number of synergistic effects. This study emphasizes the diverse effects of clay structure and solution chemistry on HA adsorption.
Kaolinite and Halloysite Derived from Sequential Transformation of Pedogenic Smectite and Kaolinite-Smectite in a 120 ka Tropical Soil Chronosequence
- P. C. Ryan, F. J. Huertas, F. W. C. Hobbs, L. N. Pincus
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 639-667
-
- Article
- Export citation
-
Tropical soils range from nutrient-depleted lateritic soils rich in halloysite or kaolinite to Inceptisols rich in interstratified kaolinite-smectite (K-S), smectite, or related 2:1 clays. Given the strong influence of clay minerals on tropical soil quality, better understanding of factors influencing their occurrence is important for modeling and managing tropical environments. This study examines the alteration of smectite to kaolinite by way of intermediate K-S and halloysite in a 120 ka moist tropical chronosequence. Iron-rich smectite (11.6 ± 2.2% Fe2O3) is the dominant mineral in Holocene soils (1–8 ka) originating from sediments rich in plagioclase and clinopyroxene. The cation exchange capacity (CEC) of smectite is 54–84 cmolc/kg and pH is 6.1 to 7.4. Within 50 ka, smectite fixes Al-hydroxy complexes into interlayers, K+ is retained preferentially over Ca2+, and 2:1 layers are stripped of tetrahedral sheets; the resulting K-S inherits flaky smectite crystal habit and the 2:1 layers — which only expand partially — include Al-hydroxy smectite and some illite-like layers. After 50 ka, the dominant mineral is K-S, the CEC is 18–28 cmolc/kg, and the pH is 5.3. Flaky Fe-kaolinite with ~10% residual smectite layers and halloysite (7.4% Fe2O3) also occur in 50 ka soil. The 120 ka soils are dominated by flaky Fe-kaolinite (<10% residual smectite layers) and halloysite (4.9% Fe2O3), and Fe-poor hexagonal kaolinite also occurs (5–10% of soil). The CEC is 11–16 cmolc/kg and the pH is 4.7–5.3.
Changes in crystal chemistry of the soil clays (decreasing Fe, Mg, Ca, and K; increasing Al) over time reflects two reaction mechanisms: (1) cell-preserved transformation of smectite layers to kaolinite layers that accompanies conversion of smectite to K-S and eventually kaolinite; this results in the formation of flaky Fe-rich kaolinites after 50 ka; and (2) dissolution of K-S followed by crystallization of halloysite. Neoformation of hexagonal kaolinite and/or halloysite with low Fe (<3% Fe2O3) follows dissolution of Fe-kaolinite or halloysite after 100 ka. This sequence is probably common in moist tropical soils and these findings may inform modeling of soil composition in tropical landscapes where tectonic, volcanic, or geomorphic activity periodically exposes unweathered parent material, producing a range of soil ages.
Traprock Transformation into Clayey Materials in Soil Environments of the Central Siberian Plateau, Russia
- Sofia N. Lessovaia, Michael Plötze, Svyatoslav Inozemzev, Sergey Goryachkin
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 668-676
-
- Article
- Export citation
-
The study of hard rock conversion into fine earths and clayey materials in the pedosphere is important in understanding the relative proportions of recent soil features to features that were inherited from ancient epochs. Cold environments are widely thought to be areas of physical weathering, but the coexistence of physical and chemical processes have also been shown. To further examine mafic rock (dolerite) weathering in soil environments and the conversion into clayey materials, Entic Podzols formed in the cold continental climate were studied. The key study was located in the central part of the flood basalt complex, or traps (traprocks), of the Central Siberian Plateau (Russia). The qualitative mineralogy was studied using X-ray diffraction and the quantitative mineral composition was determined using X-ray diffraction and subsequent Rietveld analysis. The micromorphological characteristics of the soils were studied in thin sections. Dolerite fragments and fine earths were sampled from soil profiles underlain by dolerite. XRD analyses indicated that pyroxene and especially plagioclase contents in the dolerite fragments and fine earths decreased from the bottom to the top soil horizons mostly in the mature soil profiles that were affected by chemical weathering of dolerite. The dioctahedral and trioctahedral smectites in the soils were inherited from a dolerite previously subjected to chemical weathering. The smectite was conserved in the inherited aggregates and protected against dissolution even in acidic soil horizons. Recent pedogenesis processes fractured individual fragments, converted it into soil micromass, and slightly decreased the total smectite content of the <1 µm soil fraction. However, in soil samples collected from the bottom to the top horizons of a mature soil profile, trioctahedral smectite contents decreased as dioctahedral smectite contents increased. This suggests that dioctahedral smectites formed by pedogenic alteration of inherited trioctahedral smectites.
Characteristics of Early Earth’s Critical Zone Based on Middle—Late Devonian Paleosol Properties (Voronezh High, Russia)
- Tatiana Alekseeva, Pavel Kabanov, Andrey Alekseev, Pavel Kalinin, Veronika Alekseeva
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 677-694
-
- Article
- Export citation
-
Land colonization with vascular plants during the late Silurian—early Devonian and then arborescence during the Pragian—Givetian caused the development of new soil types. These true-rooted soils increased the rate of pedogenesis on a global scale. Since that time, soil has become a key component of the biosphere and has given rise to profound development of the Earth’s Critical Zone (CZ). Case studies of Devonian CZs have helped to record the transformation from Precambrian—Lower Paleozoic ‘proto-CZs,’ which had insufficient proto-soil cover, to modern soils with true-rooted pedosphere. Devonian (Givetian—Frasnian) paleosols from the Voronezh region of Russia are combined into pedocomplexes which occupied the top, slope, and bottom positions of a pronounced paleo-relief. Paleosols were developed from terrigenous argillites and volcanigenic-sedimentary deposits. Each pedocomplex consisted of four or more paleosols with different degrees of development and profile preservation. Paleosols exhibited several common characteristics including production and translocation of clay, ferruginization and the presence of siderite nodules, enhanced MnO/Al2O3 and (Fe2O{3}+MnO)/Al2O3 values, and in situ roots and root-system traces. The latter are siderite/goethite substituted. Stable isotope analysis of siderite shows δ13C values of between -6.1 and -13.7% indicating that CO2 had originated from C3 plants. The main mineral component of clay fractions in automorphic paleosols (top and slope of the paleorelief) is kaolinite. The important feature of these paleosols is the red-stained hematite-rich layer in their bases. These horizons developed at different depths and with different thicknesses, and marked the paleo-groundwater tables of each sub-CZ. Evidence of the imprints of vegetation is seen in the abundant in situ roots, plant fragments, and spores of rhyniophytes, lycopsids, progymnosperms, cladoxylalean ferns, and phytoleims of algae-like vascular plants. The near-equatorial location and the overall characteristics of paleosols studied suggest that the aforementioned horizons were formed in a tropically warm and humid climate. The paleo-ecological environments which accompanied pedogenesis were probably controlled by tectonic activity and volcanism.