Volume 58 - Issue 1 - February 2010
Article
On the Chemical Composition of Sepiolite and Palygorskite
- E. Garciá-Romero, M. Suárez
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-20
-
- Article
- Export citation
-
Many studies of the chemical composition of sepiolite and palygorskite have been carried out using analytical electron microscopy (AEM). According to the literature, a compositional gap exists between sepiolites and palygorskites, but the results presented here show that they may all be intermediate compositions between two extremes. The results of >1000 AEM analyses and structural formulae have been obtained for the samples studied (22 samples of sepiolite and 21 samples of palygorskite) and indicate that no compositional gap exists between sepiolite and palygorskite. Sepiolite occupies the most magnesic and trioctahedral extreme and palygorskite the most aluminic-magnesic and dioctahedral extreme. Sepiolite and palygorskite with intermediate compositions exist between the two pure extremes: (1) sepiolite with a small proportion of octahedral substitution; (2) palygorskite with a very wide range of substitution (the pure dioctahedral extreme is unusual); and (3) intermediate forms, Al-sepiolite and Mg-palygorskite with similar or the same chemical composition. The chemical compositions of the intermediate forms can be so similar that a certain degree of polymorphism exists between Al-sepiolite and Mg-palygorskite.
Stress-Induced Alteration of Sudoite: Structural and Chemical Modifications
- María Dolores Ruiz Cruz, María Dolores Rodríguez Ruiz, Carlos Sanz de Galdeano
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 21-36
-
- Article
- Export citation
-
The purpose of this study was to investigate the structural and chemical modifications of phyllosilicates that occur under natural conditions, using the progressive deformation of chlorite (sudoite) present in quartz-rich veins from the Internal Zone of the Rif range (Morocco) as the model system. Signs of chlorite deformation include kinks, chevron-like folds, and fractures. The samples also contain later, undeformed grains, which sealed the fractures or grew with (001) perpendicular to the compressive stress. Deformation-induced structural changes consist mainly of basal cleavages associated with ordered replacement of brucite sheets by hydrated layers, thus leading to irregular microdomains of mixed-layer chlorite-vermiculite and sudoite. Such structural modifications represent a mechanism for accommodating the compressive stress. Structural changes were accompanied by minor chemical ones, which lead from di,tri-chlorite (sudoite) to phases with a more trioctahedral character (mixed-layer chlorite-vermiculite). The hydration reaction occurred throughout a topotactic replacement of the pre-existing sudoite grains. Later, undeformed grains consist of mixed-layer chlorite-vermiculite intergrown with retrogressive kaolinite and minor Fe oxide, and are interpreted as having formed through a dissolution-precipitation process, during deformation. Retrogression of sudoite probably occurred during the latest stage of exhumation, in low-temperature conditions.
Water-Uptake Capacity of Bentonites
- S. Kaufhold, R. Dohrmann, M. Klinkenberg
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 37-43
-
- Article
- Export citation
-
The present study compares the water-vapor adsorption capacity of bentonites (natural cation population) with the Enslin-Neff method. Water-vapor adsorption at 50% r.h. (relative humidity) or 70% r.h. is known to depend heavily on the amount of permanent charge and on the type of exchangeable cation. At ~80% r.h. Na+- and Ca2+/Mg2+-dominated bentonites take up equal amounts of water. Comparing the water-uptake capacity at 80% r.h. with the cation exchange capacity (CEC) revealed a close correlation between these two variables. Appreciable scatter apparent from this plot, however, suggests that additional factors influence the water-uptake capacity.Water adsorption at external surfaces was considered to be one of these factors and was, in fact, implicated by N2-adsorption data. The ratio of external/internal water ranged from 0 to 1, which suggests that water-adsorption values cannot be applied in the calculation of the internal surface area without correction for external water.
The Enslin-Neff water-uptake capacity, on the other hand, is unaffected by microstructural features (e.g. specific surface area and porosity). The amount of exchangeable Na+ is themost important factor. However, the relationship between the Na+ content and the Enslin value is not linear but may be explained by percolation theory.
Hydrothermal Synthesis of Kaolinite and its Formation Mechanism
- Kyoung-Won Ryu, Young-Nam Jang, Soo-Chun Chae
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 44-51
-
- Article
- Export citation
-
In spite of many studies of kaolinite synthesis, questions remain as to the transformation of gel into kaolinite, the kinetics of the reaction, and the influence of solution chemistry. The purpose of the present study was to perform a hydrothermal synthesis in order to understand better the transformation from boehmite to kaolinite. Kaolinite was synthesized from amorphous SiO2 and Al(OH)3·xH2O at fixed temperature (250°C) and pressure (30 bar). The initial pH of the solution was 2. The reaction time for the synthesis was varied from 2 to 36 h. The physical properties of synthesized kaolinite were characterized by X-ray diffraction (XRD), infrared spectroscopy (IR), nuclear magnetic resonance (NMR) spectroscopy, field-emission-scanning electron microscopy (FE-SEM), transmission electron microscopy (TEM), and energy dispersive spectrometry (EDS).
The early stage of kaolinite synthesis followed activation of amorphous Al(OH)3·xH2O to initiate the reactions, i.e. ionization and subsequent crystallization of boehmite. The boehmite reacted continuously with Si4+ dissolved in solution and gradually transformed to disordered, lath-shaped boehmite. In XRD and IR patterns, the typical peaks of boehmite were weakened or disappeared following the reaction.
Structural transformation from boehmite to kaolinite occurred when the Al/Si ratio of the aluminosilicate was 1.0. The kaolinite formed was in the form of curved flakes and its crystallinity increased with reaction time. In the final stage of reaction the morphology of kaolinite changed from flaky to polygonal. The hexagonal, platy kaolinite was therefore developed to allow the gradual variation of the chemical composition, crystal structure, and morphology.
The Combined Inelastic Neutron Scattering (INS) and Solid-State DFT Study of Hydrogen-Atoms Dynamics in Kaolinite-dimethylsulfoxide Intercalate
- L’ubomír Smrčok, Daniel Tunega, Anibal Javier Ramirez-Cuesta, Alexander Ivanov, Jana Valúchová
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 52-61
-
- Article
- Export citation
-
Vibrational spectra of two kaolinite-dimethylsulfoxide intercalates, obtained using inelastic neutron scattering (INS), were analyzed with a view to understanding the dynamics of the hydrogen atoms in the structure. The main focus was on the spectral region 0–1700 cm−1, which is difficult to analyze using optical spectroscopy. The experimental vibrational spectra of kaolinite:dimethylsulfoxide and kaolinite:d6-dimethylsulfoxide collected using two different spectrometers were interpreted by means of the solid-state DFT calculations. Calculated spectra were obtained by both normal-mode analysis and molecular dynamics going beyond the harmonic approximation. The Al-O-H bending modes were found to be spread over the large interval 100−1200 cm−1, with the dominant contributions located between 800 and 1200 cm−1. The shape of the individual hydrogen spectrum depends on whether or not the respective hydrogen atom is involved in an O-H⋯O hydrogen bond and on its strength. The modes corresponding to the in-plane movements of the inner-surface hydrogen atoms are well defined and always appear at the top of the intervals of energy transfer. In contrast, the modes generated by the out-of-plane movements of the hydrogen atoms are spread over large energy intervals extending down to the region of external (lattice) modes. The C-H modes are concentrated mainly in the three regions 1200–1450 cm−1, 800–1100 cm−1, and 0–400 cm−1. While the first two regions are typical of the various deformational modes of methyl groups, the low-energy region is populated by the modes corresponding to the movements of the whole dimethylsulfoxide molecule.
Stacking Disorder in a Sedimentary Kaolinite
- Toshihiro Kogure, Jessica Elzea-Kogel, Cliff T. Johnston, David L. Bish
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 62-71
-
- Article
- Export citation
-
Although structural disorder in kaolinite has been investigated extensively, it is still not understood properly. To investigate the problem, a kaolinite specimen of sedimentary origin from Capim, Brazil, was examined, mainly by transmission electron microscopy (TEM). Selected-area electron diffraction (SAED) along the Xi ([100], [110], and [
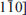 ]) directions shows various features, from completely discrete patterns to heavily streaked ones along the c* direction, suggesting that the degree of stacking disorder is variable among individual grains. High-resolution TEM images indicate that stacking faults are mainly caused by disorder of alternating t1 (~ −a/3) and t2 (−a/3 +b/3) layer displacements. Furthermore, stacking faults have been observed (1) as isolated stacking faults (e.g. insertion of an isolated t2 `fault' in an ordered sequence with t1 layer displacement) and (2) as interstratification of two kinds of multilayer blocks having regular t1 and t2 layer displacements. A mixture of grains with various degrees and modes of disorder with alternating t1 and t2 layer displacements may explain the experimental profile of the 02, 11 X-ray diffraction band.
]) directions shows various features, from completely discrete patterns to heavily streaked ones along the c* direction, suggesting that the degree of stacking disorder is variable among individual grains. High-resolution TEM images indicate that stacking faults are mainly caused by disorder of alternating t1 (~ −a/3) and t2 (−a/3 +b/3) layer displacements. Furthermore, stacking faults have been observed (1) as isolated stacking faults (e.g. insertion of an isolated t2 `fault' in an ordered sequence with t1 layer displacement) and (2) as interstratification of two kinds of multilayer blocks having regular t1 and t2 layer displacements. A mixture of grains with various degrees and modes of disorder with alternating t1 and t2 layer displacements may explain the experimental profile of the 02, 11 X-ray diffraction band.Faults related to displacement of the octahedral vacancy and/or to layer rotation were also observed in HRTEM images. The SAED patterns along the Yi ([010], [310], and [
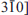 ]) directions occasionally have extra spots and/or streaks, suggesting the presence of stacking sequences with (±60°, 180°) mutual layer rotation and/or with (0, ±b/3) layer displacements. The local dickite or nacrite-like fragments formed by these faults are in qualitative agreement with recent low-temperature FTIR results from this sample, where distinct ν(OH) absorption bands reflect multiple interlayer O-H⋯O environments that are possibly ascribed to dickite and nacrite.
]) directions occasionally have extra spots and/or streaks, suggesting the presence of stacking sequences with (±60°, 180°) mutual layer rotation and/or with (0, ±b/3) layer displacements. The local dickite or nacrite-like fragments formed by these faults are in qualitative agreement with recent low-temperature FTIR results from this sample, where distinct ν(OH) absorption bands reflect multiple interlayer O-H⋯O environments that are possibly ascribed to dickite and nacrite.
Structure and Properties of Hydrogenated Nitrile Rubber/Organo-Montmorillonite Nanocomposites
- Zheng Gu, Guojun Song, Weisheng Liu, Shujing Yang, Jianming Gao
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 72-78
-
- Article
- Export citation
-
The aim of the work was to study the effect of organo-montmorillonite (OMt) on the properties of hydrogenated nitrile rubber (HNBR)/OMt nanocomposites. The nanocomposites were prepared by a melt intercalation method. The structure of the composites was studied by transmission electron microscopy (TEM) and X-ray diffraction (XRD). The behavior of stress-strain, aging resistance, solvent resistance, and the dynamic mechanical properties of HNBR/OMt nanocomposites were investigated. The TEM and XRD results showed that the OMt layers were dispersed homogeneously in the HNBR matrix. The HNBR/OMt nanocomposites showed excellent mechanical properties which were attributed to the nanometer scale dispersion and strong interaction between the HNBR and OMt. The composites possessed excellent aging resistance and oil resistance, which improved with OMt content. Dynamic mechanical analysis showed that the glass-transition temperature, Tg, of the HNBR/OMt nanocomposites was increased and the nanocomposites had a good rolling resistance in comparison to pure HNBR. The composites displayed better dynamic mechanical properties.
Determination of Exchangeable Calcium of Calcareous and Gypsiferous Bentonites
- Reiner Dohrmann, Stephan Kaufhold
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 79-88
-
- Article
- Export citation
-
The aim of the present study was to find methodological tools to obtain reasonable results for exchangable Ca2+ of gypsiferous bentonites. Cation exchange capacity (CEC) is an important property of clays. Numerous methods for calculating CEC and exchangeable cations exist; determination of exchangeable Ca2+ fails, however, when gypsiferous clays are examined because gypsum is dissolved throughout the exchange experiment, which in turn increases measureable Ca2+ concentrations. Several new methods (AgTUcalcite, CoHexcalcite, and Cu-trien5×calcite) have been developed to overcome a similar problem occurring with calcite by using exchange solutions saturated with respect to calcite prior to the experiment. In the present study these three solutions were also pre-treated with gypsum and labeled AgTUCcGp, CoHexCcGp, and Cu-trien5×CcGp. The special solutions were applied first to a gypsum- and calcite-free bentonite with known reference values for exchangeable Ca2+. The resulting exchangeable Ca2+ values obtained did not match with reference values. The solutions were then applied to natural calcareous and gypsiferous bentonites but only the proposed AgTUCcGp test method was successful. The performance of AgTUCcGp was relatively poor when applied to calcareous non-gypsiferous bentonites, the third group of test materials. Reasonable values for exchangeable Ca2+ of gypsiferous clays were obtained using a combination of two separate results: (1) calcite saturation of exchange solution (e.g. Cu-trien5×calcite) and (2) quantification of gypsum with suitable mineralogical methods. Result 1 eliminates errors caused by calcite dissolution though it is still incorrect because it contains significant amounts of Ca2+ from gypsum dissolution. After proving that gypsum was completely dissolved during the exchange experiment, result 2 was used to subtract the theoretical Ca2+ portion of gypsum from result 1. The initial concentration of gypsum of the samples studied was <1 wt.%, typical of many commercial bentonites. Using this combined procedure the sum of exchangeable cations is very close to the CEC, though it still exceeds the CEC by, on average, 3%, which is a satisfactory improvement. The resulting exchangeable Ca2+ values can be considered as operationally correct using this approach. Ca2+ saturation (Ca/CEC in %) of seven gypsiferous bentonites ranges from 1 to 69%.
Ab Initio Molecular Dynamics Study of Fe-Containing Smectites
- Xiandong Liu, Evert Jan Meijer, Xiancai Lu, Rucheng Wang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 89-96
-
- Article
- Export citation
-
In order to identify the influences imposed by Fe substitution, density functional theory-based Car-Parrinello molecular dynamics simulations were employed to study both oxidized and reduced Febearing smectites. The following basic properties were investigated: local structures in the clay layer, hydroxyl orientations, and the vibration dynamics of H and Si. Structural analyses indicated that the average Fe-O bond lengths are ~2.08 Å and 2.02 Å in the reduced and oxidized models, respectively, and the Fe substitutions did not affect the coordination structures of the Al-O and Si-O polyhedra. For hydroxyl orientations, Fe(III) substitution had no obvious influence but Fe(II) forces the coordinated hydroxyls to present a wide-angle distribution. Furthermore, the present work has shown that both substitutions can red-shift the hydroxyl in-plane bending mode. The analyses also revealed that Fe(III) substitution has no effect on the Si-O stretching, while Fe reduction causes a blue-shift of the out-of-plane stretching mode. The results provide quantitative constraints and clues for future research.
Approach to a Descriptive Model of Charge Reduction in Vermiculite by Hydrothermal Treatment
- Ana M. Campos, Sonia Moreno, Rafael Molina
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 97-109
-
- Article
- Export citation
-
Vermiculites have the potential to serve as effective catalysts if pillared with Al, but their high charge presents an obstacle to the pillaring process. The purpose of this study was to submit natural vermiculite to thermal treatments in the presence of water vapor in order to effect a reduction in the global negative charge and thereby to enhance its susceptibility to pillaring. The process of charge reduction in vermiculite under the conditions selected involved the extraction of 25% of IVAl accompanied by the extraction of structural Mg and charge-compensating cations (Ca2+, Na+, and K+). The results indicate a reduction of 35% in the global negative charge in vermiculite by the end of the treatment. Some of the VIAl content was not removed during acid washing, and probably remained in the solid in structural positions in the octahedral sheet.
Performance of Natural Zeolite and Sepiolite in the Removal of Free Cyanide and Copper-Complexed Cyanide ([Cu(CN)3]2−)
- Esra Tarlan-Yel, Vildan Önen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 110-119
-
- Article
- Export citation
-
The chemical and biological methods employed to date in the removal of free cyanide (CN−) and metal-cyanide complexes from aqueous fluids have proved expensive and problematic. A simpler and more economical approach was attempted in the present study using zeolite and sepiolite. The effectiveness of zeolite from Manisa-Gördes (Turkey) and of sepiolite Eskişehir-Sivrihisar (Turkey) at removing free and Cu-complexed cyanide, [Cu(CN)3]2− was investigated. For removal of CN−, the system performance was examined in terms of concentration, particle size, and retention time. Material with smaller particle sizes (<0.106 mm) performed better, particularly in the case of sepiolite. The maximum CN− removal capacities of zeolite and sepiolite were calculated as 571 and 695 meq/100 g for free CN adsorption, and 455 and 435 meq/100 g for Cu-complexed CN adsorption, respectively. The time to reach equilibrium was calculated as 1050 min. Acid activation, a simple cation adsorption removal method, did not improve the process, instead leading to reduced CN adsorption. Hydroxylated surfaces of metal oxides at the edges of zeolite develop charges and exchange with anions in water. Mg2+ ions located at the edges of the octahedral sheet can create complexes with CN− anions. Moreover, hydrogen bonding with anions (CN− in this case) and H+ of zeolitic water bonded to coordinated water molecules can also create complexes. These two complexes are considered to be effective mechanisms for sepiolite. The effects of both acid activation and CN adsorption were clearly observed in the Fourier-transform infrared spectra. Removal of CN was characterized by the Langmuir isotherm, indicating monolayer coverage with chemical bonding to the surface, which deteriorated during acid activation. The study indicated that zeolite and sepiolite can be used efficiently and easily for removal of free and Cu-complexed CN.
Mineralogy, Geochemistry, and Origin of Bentonite in Upper Cretaceous Pyroclastic Units of the Tirebolu Area, Giresun, Northeast Turkey
- Mehmet Arslan, Emel Abdiogğlu, Selahattin Kadir
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 120-141
-
- Article
- Export citation
-
Widespread alteration in the Upper Cretaceous pyroclastic units of the Tirebolu (Giresun) area, NE Turkey, has resulted in significant occurrences of bentonite with economic potential. No detailed geological, mineralogical, or geochemical characterization of these occurrences has been carried out to date. The aim of this study was to describe the geological background, the mineralogical, chemical, and stable-isotope characteristics of the bentonite, and major aspects of their formation, e.g. type and source of low-temperature alteration, mass balance, chemical evolution of the smectites, and geochemistry of major and trace elements. The bentonite contains abundant smectite with occasional kaolinite and mordenite, volcanogenic feldspar, quartz, biotite, hornblende, glass shards, and pumice fragments, along with the diagenetic minerals, opal-CT, and, in some locations, calcite. X-ray diffraction patterns of the clay fractions exhibit characteristics of pure montmorillonite and beidellite-type smectite. Micromorphologically, the smectite exhibits a honeycomb texture, the kaolinite occurs in both vermiform and irregular platy forms, and the mordenite occurs in fibrous form. All of these minerals are edged with devitrified volcanic glass and resorbed feldspar. Chemically, the smectites are Ca-smectite. Geochemical data indicate that alteration of the pyroclastic units took place under suboxic and anoxic environmental conditions during bentonite formation. Field observations and mineralogical, geochemical, oxygen, and hydrogen isotopic data indicate that the alteration of feldspar and volcanic glass in the pyroclastics by mixed meteoric and sea water in a shallow marine environment under alkaline and acidic conditions, respectively, controlled by environmental Al, Ca, and Na concentrations, resulted in the formation of authigenic smectite, mordenite, and kaolinite. A large Ca content in the smectite originated from surrounding units, which resulted in high alkalinity; Mg originated from seawater.



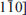 ]) directions shows various features, from completely discrete patterns to heavily streaked ones along the
]) directions shows various features, from completely discrete patterns to heavily streaked ones along the 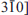 ]) directions occasionally have extra spots and/or streaks, suggesting the presence of stacking sequences with (±60°, 180°) mutual layer rotation and/or with (0, ±
]) directions occasionally have extra spots and/or streaks, suggesting the presence of stacking sequences with (±60°, 180°) mutual layer rotation and/or with (0, ±