Volume 69 - Issue 6 - December 2021
Article
Formation of Humboldtine During the Dissolution of Hematite in Oxalic Acid – Density Functional Theory (DFT) Calculations and Experimental Verification
- Paula Vehmaanperä, Bo Gong, Patrick H.-L. Sit, Riina Salmimies, Bernardo Barbiellini, Antti Häkkinen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 655-662
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Understanding the reactions taking place in the hematite-oxalic acid system is important in order to clean iron oxides from filters and to remove iron from mineral concentrates. Previous studies reported the formation of an unwanted solid phase during this process. The objective of the current work, therefore, was to visualize and rationalize the iron dissolution steps taking place in the hematite–oxalic acid reaction by combining density functional theory (DFT) calculations and experimental data. The results of DFT calculations indicated that a precipitate was formed in this reaction; XRD analysis of the solid phase after the dissolution experiment revealed the formation of humboldtine as the precipitate. The attachment of oxalate on the hematite surface and the reduction of Fe(III) to Fe(II) were key steps for humboldtine formation. Both simulations and the experimental results showed that greater oxalic acid concentrations yielded more precipitate, suggesting a simple and novel route to synthesize humboldtine, a material which is relevant to the demand for clean energy.
A Comparison of the Adsorption of Cesium on Zeolite Minerals vs Vermiculite
- D. R. Ferreira, G. D. Phillips, B. Baruah
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 663-671
-
- Article
- Export citation
-
Radiocesium was deposited on the soils of Fukushima Prefecture in Japan after the meltdown of the Fukushima Daiichi Nuclear Power Plant in 2011. The radiocesium bound to 2:1 clay minerals, such as vermiculite, common in the soil of that region and became non-exchangeable due to the strong affinity of these clay minerals for the Cs+ adsorbed. The current study generated adsorption envelopes for Cs+ on three zeolite minerals: zeolite Y, ZSM-5, and ferrierite. Two of these (ZSM-5 and ferrierite) caused monovalent cations to adsorb via a strong inner-sphere mechanism. A comparison of Cs+ adsorption on these zeolites to Na+ adsorption on the same zeolites showed that Cs+ adsorbs much more strongly than Na+, which is explained by its atomic properties. Despite the inner-sphere adsorption of Cs+ on ZSM-5 and ferrierite, the affinity of vermiculite for Cs+ is even stronger. An adsorption envelope for Cs+ on vermiculite failed to show a low-pH adsorption edge even at a pH of 1.01, with adsorption remaining at ~65% of the maximum even at this low pH. The adsorption envelopes for Cs+ on ZSM-5 and ferrierite minerals did show low-pH adsorption edges centered at pH 3.5 and 3.0, respectively, where Cs+ adsorption dropped to zero. The greater affinity of vermiculite for Cs+, even when compared with that for two zeolite minerals known to have significant affinities for monovalent ions, highlights the difficulty in removing Cs+ from contaminated Fukushima soils.
Meifuite, A New Ferrous Phyllosilicate Mineral With Modulated Tetrahedral Sheets Similar to Minnesotaite
- Shiyun Jin, Xiaochun Li, Franklin Hobbs, Stephen Guggenheim, And Huifang Xu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 672-687
-
- Article
- Export citation
-
A new ferrous phyllosilicate, meifuite, has been discovered in the Yinachang Fe-Cu-REE (rare-earth element) deposit in China. The structural formula, calculated using averaged electron probe microanalysis (EPMA) results, is K0.72Na0.20(Fe5.56Mg0.31Mn0.13)Σ6.00(Si6.95Al1.04)Σ7.99O18.84(OH)4.84 Cl1.33, with an ideal formula of KFe6(AlSi7)O19(OH)4Cl2. The structure of meifuite has a
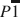 space group symmetry, with unit-cell parameters of a = 22.7773(13) Å, b = 9.5553(5) Å, c =14.3282(8) Å, α = 99.258(4)°, β = 136.750(3)°, γ = 89.899(4)°, Z = 2, and V = 2077.9(2) Å3. Meifuite has a strip-modulated 2:1 layer (T–O–T) structure similar to that of minnesotaite. About 1/8 of the tetrahedra in the T sheet are occupied by Al instead of Si, and the interlayer cavities are partially occupied by K and Na. Some of the OH sites in the octahedral sheet in the layer structure are fully or partially substituted by Cl, which is apparently the primary reason for the meifuite structure being more stable than stilpnomelane, the most common ferrous layer silicate mineral found at similar temperature and pressure conditions. An updated, more accurate structure model of minnesotaite is also provided for comparison with the meifuite structure. The mineral is named after Meifu Zhou in honor of his outstanding contributions to the field of economic geology.
space group symmetry, with unit-cell parameters of a = 22.7773(13) Å, b = 9.5553(5) Å, c =14.3282(8) Å, α = 99.258(4)°, β = 136.750(3)°, γ = 89.899(4)°, Z = 2, and V = 2077.9(2) Å3. Meifuite has a strip-modulated 2:1 layer (T–O–T) structure similar to that of minnesotaite. About 1/8 of the tetrahedra in the T sheet are occupied by Al instead of Si, and the interlayer cavities are partially occupied by K and Na. Some of the OH sites in the octahedral sheet in the layer structure are fully or partially substituted by Cl, which is apparently the primary reason for the meifuite structure being more stable than stilpnomelane, the most common ferrous layer silicate mineral found at similar temperature and pressure conditions. An updated, more accurate structure model of minnesotaite is also provided for comparison with the meifuite structure. The mineral is named after Meifu Zhou in honor of his outstanding contributions to the field of economic geology.
Interfacial X-Ray Scattering From Small Surfaces: Adapting Mineral-Fluid Structure Methods for Microcrystalline Materials
- Joanne E. Stubbs, Anna K. Wanhala, Peter J. Eng
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 688-701
-
- Article
- Export citation
-
Crystal truncation rod (CTR) X-ray diffraction is an invaluable tool for measuring mineral surface and adsorbate structures, and has been applied to several environmentally and geochemically important systems. Traditionally, the method has been restricted to single crystals with lateral dimensions >3 mm. Minerals that meet this size criterion represent a minute fraction of those that are relevant to interfacial geochemistry questions, however. Crystal screening, data collection, and CTR measurement methods have been developed for crystals of <0.3 mm in lateral size using the manganese oxide mineral chalcophanite (ZnMn3O7·3H2O) as a case study. This work demonstrates the feasibility of applying the CTR technique to previously inaccessible surfaces, opening up a large suite of candidate substrates for future study.
Polytypism of Cronstedtite from Ouedi Beht, El Hammam, Morocco
- Jiří Hybler, Zdeněk Dolníček, Jiří Sejkora, Martin Števko
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 702-734
-
- Article
- Export citation
-
The present study deals with accurate identification of polytypes, twins, and allotwins – oriented crystal associations of more than one polytype. The trioctahedral 1:1 layered silicate cronstedtite was studied using single-crystal X-ray diffraction data collected with a four-circle diffractometer equipped with a CCD detector. The sample from the skarn occurrence, Ouedi Beht, El Hammam, Morocco, was explored. It contains cronstedtite in fibrous massive aggregates in the central part, and euhedral crystals in surrounding veinlets and druses. The reciprocal space (RS) sections created by the diffractometer software and presented here were used to determine the OD (ordered-disordered) subfamilies (Bailey’s group A, B, C, D) and to identify polytypes. The chemical compositions of some crystals were determined thereafter by electron probe microanalysis (EPMA-WDS). Some crystals studied are more or less complicated allotwins. Polytypes were thus separated by cleaving crystals into smaller parts in many cases. All polytypes found belong to subfamilies A or D. The following polytypes of the subfamily A were identified: 2M1 (a = 5.49, b = 9.51, c = 14.40 Å, β = 97.30°, space group Cc), 1M (a = 5.51, b = 9.54, c = 7.33 Å, β = 104.5°, Cm), 3T (a = 5.51, c = 21.32 Å, P31), 6T2 (a = 5.50, c = 42.60 Å, P31). 2M1 and 3T were present as isolated crystals or separated by cleaving, otherwise all these polytypes are parts of allotwins. The 2M1 polytype is sometimes twinned by reticular pseudo merohedry with twin index n = 3 and 120° rotation about the chex axis as the twin operation. Allotwins of 1M + twinned 2M1 polytypes are also present. Another kind of twinning, with rotation by (2n+1)×60° about chex is rare. The subfamily D is represented mostly by 2H1 and 2H2 polytypes, a = 5.50, c = 14.25 Å, space groups P63cm (2H1), P63 (2H2). In addition, several six-layer (a = 5.49, c = 42.80 Å), mostly non-MDO polytypes were separated from allotwins by cleaving. In order to identify them, 24 possible stacking sequences were modeled, diffraction patterns calculated, graphical identification diagrams constructed, and comparisons made with actual RS sections. This simulation revealed that five pairs of sequences provided identical diffraction patterns. Polytypes actually found correspond to the following sequences: 1 (6T1), 5 (proposed Ramsdell’s symbol 6T3), 8+10 (6T5), 11+12 (6T4), 24 (6T6, trigonal polytypes, space group type P3), 22 (6R1), and 23 (6R2, rhombohedral polytypes, space group type R3c and R3, respectively). The RS section corresponding to the hexagonal polytype 6H2 (sequence 14) was also found. However, diffraction patterns geometrically indistinguishable can be produced by the twin with rotation by 180° about chex as a twin operation of the rhombohedral polytype 6R2 (sequence 23). Several aggregates with fiber texture of polytype 2H2 were separated from the central part. Use of EPMA-WDS revealed Fe and Si along with significant amounts of Mn and Mg. Crystals from veins were more Mn- and Mg-rich than these from the central part. Traces of Cl, S, and Al are present also.
Phase-Transition Process in the Hydrothermal Zeolitization of Volcanic Ash into LTA and FAU Structures
- Jorge D. Monzón, Maximiliano R. Gonzalez, Mercedes Muñoz, Andrea M. Pereyra, Elena I. Basaldella
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 735-745
-
- Article
- Export citation
-
Minerals such as quartz, present widely in various volcanic ashes, remain unaltered throughout the low-temperature hydrothermal process currently used in industry to obtain zeolites, causing an incomplete hydrothermal transformation of the starting solid. This study presents a novel and cost-effective procedure which improves the reactivity of ash toward the generation of zeolite by increasing the availability of silica and alumina components. This method leads to a final product with a large zeolite content. The transformation consisted of an ash-activation step followed by hydrothermal zeolitization. The influence of the structural, chemical, and morphological characteristics of the volcanic ash as well as the effect of the activation procedure on the ash reactivity were studied. A collected sample (VA) and an amorphous fraction obtained after VA sieving (VA40, retained on #40 mesh) were used for zeolite production. These solids were alkaline-treated separately, aged, and reacted under controlled conditions of temperature at autogenous pressure. Throughout the process, the solid phases were characterized by X-ray diffraction, energy dispersive X-ray microanalysis, scanning electron microscopy, Fourier-transform infrared spectroscopy, and N2adsorption-desorption porosimetry measurements. After activation and alkaline aging, the presence of quartz and plagioclase minerals in the natural ash seemed to improve the growth of NaAlSiO4 polymorphs, which in turn were transformed easily to zeolite structures. Even under adequate pretreatment and suitable synthesis conditions, the coarse non-crystalline fraction led to low conversion, while the highest conversions to zeolites A and X were obtained from the natural ash. The outcomes of the present study could be used to improve the conversion levels of other non-conventional aluminosiliceous minerals into zeolites.
Synthesis of a Composite Aerogel of Reduced Graphene Oxide Supported by Two-Dimensional Montmorillonite Nanolayers for Methylene Blue Removal
- Qiulin Ma, Wei Wang, Wei Ge, Ling Xia, Shaoxian Song
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 746-758
-
- Article
- Export citation
-
Two-dimensional montmorillonite nanolayers (2D Mnt) are excellent adsorbents for methylene blue due to the fully exposed active sites, but the separation of 2D Mnt from water is difficult. The objective of the present study was to assemble 2D Mnt and graphene oxide sheets into a three-dimensional aerogel (3D Mnt-rGO Gel) to achieve easy solid–liquid separation. Structural characterization demonstrated that the Mnt-rGO Gel has a porous 3D structure with Mnt nanolayers distributed uniformly within; the introduction of 2D Mnt could reduce significantly the degree of restacking of graphene sheets. Adsorption tests indicated that 2D Mnt enhances the methylene blue (MB) removal performance of Mnt-rGO Gel with a large adsorption capacity of 207 mg g–1, which may be attributed to the adsorption of MB onto 2D Mnt and the increased adsorption surface of rGO resulting from the reduced restacking of graphene sheets. The MB was removed completely by 300 mg L–1 of Mnt-rGO Gel-3 in 180 min. The adsorption process of MB onto Mnt-rGO Gel followed the pseudo-second order kinetic model and the Langmuir isotherm model. Mnt-rGO Gel also showed good reusability. Fourier-transform infrared (FTIR) and X-ray photoelectron spectroscopy (XPS) results suggested that the adsorption of MB onto Mnt-rGO Gel may be attributed to the π–π interactions between aromatic rings of MB and graphene, hydrogen bonding, and the electrostatic interactions between the nitrogen groups on the MB and oxygen-containing groups on the Mnt-rGO Gel.
Dispersion and Swellability of Ternary Surfactant Co-Modified Montmorillonites
- Wei Hua Yu, Jia Hui Liu, Min Wang, Na Li, Jun Rui Zhang, Tian Hao Huang, Chun Hui Zhou
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 759-771
-
- Article
- Export citation
-
Organo-montmorillonite (OMnt) has wide applications in paints, clay-polymer nanocomposites, biomaterials, etc. In most cases, the dispersibility and swellability of OMnt dictate the performance of OMnt in the target products. Previous studies have revealed that the properties can be improved when multiple organic species are co-introduced into the interlayer space of montmorillonite (Mnt). In the present study, single surfactant erucylamide (EA), dual-surfactants cetyltrimethyl ammonium bromide (CTAB) and octadecyltrimethyl ammonium chloride (OTAC), and ternary-surfactants EA, CTAB, and OTAC were co-introduced into Mnt by solution intercalation. The resulting OMnts were characterized by powder X-ray diffraction (XRD), Fourier-transform infrared (FTIR) spectroscopy, thermogravimetry-differential thermogravimetry (TG-DTG), water contact-angle tests, scanning electronic microscopy (SEM), laser particle-size analysis, and swelling indices. Mnt co-modified by ternary CTAB, OTAC, and EA led to a large d001 value (4.20 nm), surface hydrophobicity with a contact angle of 95.6°, swellability (50 mL/g) with small average particle sizes (2.1−2.8 μm) in xylene, and >99% of the OMnt particles were kept as <5 μm in deionized water. The formation of EA-modified-Mnt was proposed according to hydrophobic affinity, hydrogen bonding, and van der Waals forces. The nanoplatelets of the CTA+, OTA+, and EA co-modified OMnts in xylene were assembled into a house-of-cards structure by face-to-edge and edge-to-edge associations. The electrostatic attractions, electrostatic and steric repulsions, and hydrophobic interactions were responsible for the good dispersibility of OMnt in xylene. The ternary surfactant co-modified OMnt with high dispersion and swellability will make OMnt better suited for real-world applications.
Tracing Organic-Inorganic Interactions by Light Stable Isotopes (H, Li, B, O) of an Oil-Bearing Shale and its Clay Fraction During Hydrous Pyrolysis
- Norbert Clauer, Lynda B. Williams, Anthony E. Fallick
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 772-786
-
- Article
- Export citation
-
Tracing interactions during burial-induced organic maturation and associated clay-material alteration is of prime importance for understanding both the individual and combined mineral and organic processes. In the present study the light elements B, Li, O, and H of a sample from oil-prone Eocene Kreyenhagen Shale from San Joaquin Basin (California) were examined. The natural burial-induced temperature increase was simulated by pyrolysis experiments at progressively increasing temperatures (270–365°C) and for varied durations (72–216 h) applied to the whole rock and its <2 μm fraction. The illite structure as well as the K-rich interlayers of the illite-smectite mixed layers were not affected by the pyrolysis experiments and the smectite-rich interlayers did not collapse, while the soluble minerals and the organic matter were altered. The distribution pattern of the rare-earth elements (REEs) from untreated whole rock and of its pyrolyzed equivalents are within analytical uncertainty, which confirms that the changes induced by pyrolysis experiments were minimal in the bulk sample. Conversely, the REEs from the <2 μm fractions were modified significantly, suggesting that the whole rocks and the <2 μm fractions may contain different types of organic materials. Also, only the carbonates, oxides, chlorides, and organic matter were affected together with the smectite-rich interlayers of the illite-smectite structure. Bitumen coating of the smectite interlayers probably increased the amount of B of organic origin in their sites. The δ11B and δ7Li of the successively expelled hydrocarbon phases changed with increasing pyrolysis temperatures, together with the B and Li contents of the hydrocarbon-related fluids. On the basis of the δ11B and δ7Li from pyrolyzed clay fractions, the B released successively was not isotopically homogeneous, probably depending on how the type of organic matter decomposed during the successive pyrolysis steps, and on which components were released. The δ11B of organic-B increased progressively from –2‰ at low experimental temperature up to +9‰ at the highest temperature. The calculated δ7Li that was released also increased relative to the value of the outcropping sample used as a reference, but it remained almost constant from –7‰ at 310°C for 72 h to –8‰ at 365°C for 216 h. The δ18O values of the <2 μm size fractions decreased significantly during pyrolysis above 300°C, but the δD changes were rather modest. The total organic carbon (TOC) remained statistically constant after pyrolysis to 300°C, as did the δ7Li values. The pyrolysis experiments in the present study suggest the presence of bitumen-coated smectite interlayers that could have been misidentified as dehydrated smectite in the literature. Together with abnormal illite K-Ar ages, the occurrence of such bitumen-coated illite-smectite interlayers occurring in source and reservoir rocks could indicate the timing of hydrocarbon maturation relative to illitization.
Original Paper
Microstructure and Time-Dependent Behavior of STx-1b Calcium Montmorillonite Suspensions
- Yee-Kwong Leong, Pengfei Liu, Pek-Ing Au, Peta Clode, Jishan Liu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 787-796
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
CaMnt is much less important than NaMnt due to its limited commercial applications. The time-dependent property of NaMnt gel has been studied extensively as it is exploited in many applications such as drilling mud and viscosity-modifier applications. In contrast, the time-dependent property of CaMnt suspension and the factors affecting it are largely unknown. The speciation of Ca2+ ions is one such factor to be evaluated. In the current study, pH and solids concentration were examined and then used to validate a recent theory on clay gel time dependency. The results supported the theory that a strong electrostatic double layer (EDL) repulsion in the 3-D network is needed to reorganize the structure and drive it toward the state of minimum free energy. The 12 wt.% CaMnt (STx-1b) gel displayed time-dependent behavior at pH 5 but not at its natural pH of 8.4. At pH 5, the interlayer Ca2+ ions became fully hydrated and desorbed from the platelet surface. This enhanced the EDL repulsion between the platelets. At pH >6.5, the positively charged hydrolysis product Ca(OH)+ began to form and was adsorbed, partially neutralizing the permanent layer charge and weakening the repulsion. However, at 19.4 wt.% solids, the gel displayed pronounced time-dependent behavior despite the high natural pH of 8. The platelets were much closer together, allowing the EDL force to operate and effect structural reorganization. The microstructure of these CaMnt gels showed high platelet concentrations interacting to form a relatively open structure. The microstructure of a kaolin (KGa-2) suspension which showed no time-dependent behavior even after 1 day of ageing revealed the importance of particle morphology and layer charge on time dependency. Its 3-D structure was formed by relatively thick, layered platelets with a low layer charge.
Correction
Correction to: Enhancement of Amorphous Silica Dissolution byInteraction With Six-Membered Ring Heterocyclic Compounds
- Motoharu Kawano, Jinyeon Hwang
-
- Published online by Cambridge University Press:
- 01 January 2024, p. 797
-
- Article
- Export citation