Volume 57 - Issue 1 - February 2009
Article
Significance of phyllosilicate mineralogy and mineral chemistry in an epithermal environment. Insights from the palai-islica Au-Cu deposit (Almería, SE Spain)
- Javier Carrillo-Rosúa, Salvador Morales-Ruano, Iñaki Esteban-Arispe, Purificación Fenoll Hach-Alí
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-24
-
- Article
- Export citation
-
Phyllosilicate mineralogy is key to understanding hydrothermal processes within accepted epithermal deposit models but little information has been published about the mineral chemistry of epithermal deposits. X-ray diffraction, optical and electronic microscopy (scanning and transmitted), electron microprobe, and Fourier transform infrared spectroscopy were used in this work to study phyllosilicates in the Palai-Islica Au-Cu epithermal, volcanic-hosted deposit, in order to link phyllosilicate mineralogy and mineral chemistry to ore genesis. Different phyllosilicate assemblages are characteristic of two types of mineralization, and related hydrothermal alteration. Chlorite and mica appear in polymetallic quartz veins with sulfides, and in the related chloritic and sericitic hydrothermal alteration. These minerals have notable textural and chemical differences (i.e. Fe/(Fe+Mg), Si and Al in chlorite and illitic and phengitic components in mica) amongst veins and altered rocks, revealing different genetic conditions. These chemical features also distinguish propylitic and regional, non ore-related, low-temperature alteration. Hot hydrothermal fluids of near-neutral pH are responsible for vein mineralization and alteration. Illite, interstratified illite-smectite, kaolinite, and pyrophyllite are characteristic, with a distribution pattern by zones, of the intermediate argillic and advanced argillic alteration around areas of silicification. In the latter, native gold appears associated with interstratified illite-smectite, suggesting a relatively low-temperature formation. Hot, low-pH fluids are responsible for this mineralization and alteration assemblage. The present study contributes to epithermal models showing the co-existence of two different alteration styles in the same hydrothermal system.
X-Ray diffraction line-broadening study on two vibrating, dry-milling procedures in kaolinites
- Pablo Pardo, Joaquín Bastida, Francisco J. Serrano, Rafael Ibáñez, Marek A. Kojdecki
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 25-34
-
- Article
- Export citation
-
Due to the great technological importance of the microstructure of kaolinite, characterizing its evolution during dry milling of kaolin and analyzing the microstructural information obtained from different methods were the main aims of this work. The microstructural alteration of kaolinite is evaluated by X-ray diffraction and electron microscopy methods, comparing the results obtained and analyzing the correlations between them. The Warren-Averbach and Voigt-function methods of X-ray diffraction microstructural analysis have been applied successfully to the study of the effects of two different, vibrating-cup dry-milling configurations in the microstructure of kaolinite from the reflections corresponding to (001) diffracting planes. The crystallite-size estimates obtained from the two methods correlate well. Field emission scanning electron microscopy measurements of kaolinite particle thickness are in good agreement with the crystallite size estimated by the two methods. The Warren-Averbach method also provided estimates of the contribution to line broadening. Vibrating-cup milling has been proved to be a more efficient method of strain comminution of kaolinites than other milling techniques, reaching extensive microstructural degradation within seconds.
Rapid and direct synthesis of spherical organotalc
- Maguy Jaber, Fabrice O. M. Gaslain, Jocelyne Miehé-Brendlé
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 35-39
-
- Article
- Export citation
-
Organotalcs, in which organic moieties are covalently bonded to Si atoms belonging to the tetrahedral sheets, are usually prepared by a sol-gel process starting from ethanolic solution of Mg nitrate, organo-alcoxysilanes, and aqueous sodium hydroxide solution. In this case, gypsum-like particles are obtained. In this work, evaporation-induced self-assembly within aerosols was used for the first time in order to prepare organotalc spheres. These hybrid lamellar materials can be used as environmental barriers, as polymer fillers, and as catalytic supports. Using octyltriethoxysilane as a source of Si, spherical particles with sizes ranging from 20 nm to 1 µm are obtained. X-ray diffraction and transmission electron microscopy images show that the d001 value equals 2.8 nm meaning that, in this case, organic moities are either alternatively distributed or identically tilted in the interlayer space. Compared to the classical synthesis at room temperature or in autoclaves, and besides being a continuous process, the reaction time is reduced to several minutes instead of hours. Homogeneous forms are obtained by aerosol whereas irregular shapes are obtained in the classical synthesis.
Structural characterization of (Cu2+,Na+)- and (Cu2+, NH4+\$\end{document}
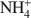 )-exchanged bentonites upon thermal treatment
)-exchanged bentonites upon thermal treatment
- Torsten Seiffarth, Christian Kaps
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 40-45
-
- Article
- Export citation
-
Bentonites are excellent materials for sequestering various metal cations because of the cation fixation ability of the constituent montmorillonite layers, but sometimes, such as in the case of Cu2+, the exact location of cation fixation with respect to the clay layers is difficult to determine. Na-montmorillonite was prepared from the <2 µm fraction of the bentonite Calcigel (from Bavaria, Germany) and exchanged by Cu2+ and Na+ or by Cu2+ and \$\end{document}
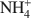 cations. The resulting materials (bi-ionic Cu-Na and Cu-NH4 samples, respectively, as well as homo-ionic forms with Cu2+, Na+ and \$\end{document}
cations. The resulting materials (bi-ionic Cu-Na and Cu-NH4 samples, respectively, as well as homo-ionic forms with Cu2+, Na+ and \$\end{document}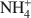 ) were heated for 24 h at temperatures of 300 and 450°C and the structural evolution characterized using X-ray diffraction (XRD) analysis, Fourier Transform Infrared (FTIR) spectroscopy, and differential scanning calorimetry (DSC) analysis.
) were heated for 24 h at temperatures of 300 and 450°C and the structural evolution characterized using X-ray diffraction (XRD) analysis, Fourier Transform Infrared (FTIR) spectroscopy, and differential scanning calorimetry (DSC) analysis.The XRD patterns showed that the Cu sample and the Cu-Na sample have basal spacings of 12.5 Å. Upon heating at 300 and 450°C, the layers collapsed to 9.5 Å. In contrast, the d001 value in the NH4 sample and Cu-NH4 sample decreased to 10.0 Å and 10.2 Å, respectively, during the heat treatment. The Cu2+ ions migrated irreversibly into the montmorillonite structure.
For the NH4 and the Cu-NH4 samples, DSC analyses show that NH3 evolved at between 300 and 400°C though the octahedral sheet was not altered substantially by the H+ generated. Infrared spectra show that the bands of the Si-O and OH vibrations of all samples were changed upon heating due to the movement of the dehydrated cations into the hexagonal holes of the tetrahedral sheet. Apparently no Cu2+ was trapped in the octahedral sheet. In the case of the Cu-NH4 form, both Cu2+ fixation and de-ammonization occurred during the heat treatment. Other than maintaining the basal spacing, no effect of the presence of \$\end{document}
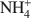 on the Cu2+ fixation could be found for the montmorillonite studied.
on the Cu2+ fixation could be found for the montmorillonite studied.
Magnetic enhancement during the crystallization of ferrihydrite at 25 and 50°C
- E. Cabello, M. P. Morales, C. J. Serna, V. Barrón, J. Torrent
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 46-53
-
- Article
- Export citation
-
Soil formation usually results in an increase in magnetic susceptibility. The magnetic properties of the products of transformation of ferrihydrite, a typical precursor of other soil Fe oxides, were examined in the present work. Synthetic 2-line ferrihydrite was aged at two temperatures (25 and 50°C) and two different relative humidities (80 and 100%) in the presence of silicate, phosphate, citrate, and tartrate as adsorbed ligands (molar anion/Fe ratio = 1–3%). The ligands delayed or prevented the transformation of ferrihydrite to hematite. The magnetic susceptibility of the ferrihydrite transformation products increased with aging, the rate of increase depending on the type of ligand added and its concentration. The largest increase in magnetic susceptibility, sixfold, was obtained with ferrihydrite in a citrate/Fe ratio of 1%, after 1500 days. The resulting magnetic products exihibited superparamagnetic behavior at room temperature and high coercivity at 5 K. The formation of an intermediate ferrimagnetic phase in the ferrihydrite-to-hematite transformation might explain the magnetic enhancement observed in many aerobic soils lacking other sources of magnetic minerals.
Hydrogen bonding and vibrational spectra in kaolinite-dimethylsulfoxide and -dimethylselenoxide intercalates — A solid-state computational study
- Eva Scholtzová, L’ubomír Smrčok
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 54-71
-
- Article
- Export citation
-
The aims of this study were to obtain accurate structural information on the dimethyl sulfoxide (DMSO) and dimethylselenoxide (DMSeO) kaolinite intercalates, paying close attention to the hydrogen-bond geometries, and to provide a detailed interpretation of the individual vibrational modes of intercalates under study and relate their energies to the formation of the hydrogen bonds. Accurate positions of all the atoms in the structures of kaolinite:dimethylsulfoxide (K:DMSO) and kaolinite:dimethylselenoxide (K:DMSeO) intercalates have been obtained by the total energy minimization in solid state at density functional theory (DFT) level of the theory. The bond distances and angles in the kaolinite 1:1 layer are in good agreement with those reported in the most recent single-crystal refinement of kaolinite. Computed geometries of DMSO and DMSeO agree well with the high-quality diffraction data and independent theoretical ab initio calculations. The organic molecules are fixed in the interlayer space mainly by three moderately strong O-H⋯O hydrogen bonds, of different strengths, with the O⋯O contact distances being within 2.739–2.932 Å (K:DMSO) and 2.681–2.849 Å (K:DMSeO). Substantially weaker C-H⋯O and O-H⋯S(Se) contacts play only a supporting role. The optimized atomic coordinates were used to calculate the individual vibrational modes between 0 and 4000 cm−1. The maximum red shifts of the OH-stretching modes caused by the formation of the O-H⋯O hydrogen bonds were 407 cm−1 (K-DMSO) and 537 cm−1 (K-DMeSO), respectively. The Al-O-H bending modes are spread over the large interval of 100–1200 cm−1, but the dominant contributions are concentrated between 800 and 1200 cm−1. Theoretically calculated energies of the OH- and CH-stretching modes show good agreement with the previously published figures obtained from the infrared and Raman spectra of these intercalates.
A new massive deposit of allophane raw material in Ecuador
- Stephan Kaufhold, Annette Kaufhold, Reinhold Jahn, Salomón Brito, Reiner Dohrmann, Rainer Hoffmann, Hartmut Gliemann, Peter Weidler, Manfred Frechen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 72-81
-
- Article
- Export citation
-
In Ecuador, DINAGE (known today as the Servicio Geológico Nacional) and the German Federal Institute for Geosciences and Natural Resources have discovered a huge allophane deposit covering an area of >4000 km2. This study presents the results from an investigation of a 16-m thick vertical sequence from this deposit, supposedly the weathering product of two different volcanic ash deposits. In particular, the distribution of alkali metals within the uppermost layer indicates that the weathering process is still ongoing.
According to the mineralogical composition, an allophane-rich layer (allophane facies) could be distinguished from the underlying halloysite-rich layer (halloysite facies). A 2-m thick transition zone is characterized by the presence of gibbsite and intermediate specific surface area values. Only a few imogolite fibers could be identified (by scanning electron microscopy), indicating the dominance of allophane over imogolite in the allophane facies. Single allophane particles were investigated by atomic force microscopy, though this method was less accurate than transmission electron microscopy with respect to the determination of the primary particle diameter. Carbon isotope analysis (14C) suggested an age of ∼20,000 y for the allophane layer.
Within the allophane facies, a 4-m thick layer occurs containing 70–80 wt.% allophane with an N2-BET specific surface area of >300 m2/g. Based on infrared and energy-dispersive X-ray diffraction measurements, an Al/Si ratio of 1.3–1.4 was established for this allophane, which is between Al-rich and Si-rich allophane. The allophane layer may be of economic value due to the large allophane content, the small amount of organic matter, and the significant thickness of the deposit.
Bromide-ion distribution in the interlayer of the layered double hydroxides of Zn and Al: Observation of positional disorder
- S. V. Prasanna, A. V. Radha, P. V. Kamath, S. Kannan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 82-92
-
- Article
- Export citation
-
Because of the anisotropy in bonding, layered hydroxides crystallize with extensive structural disorder due to the incorporation of stacking faults. In contrast, the loss of crystallinity in Br−-ion intercalated layered double hydroxides (LDHs) arises due to the positional disorder of Br− in the interlayer. The structure of the interlayer in other LDHs is poorly understood due to the low X-ray scattering power of the commonly found anions such as Cl− and \$\end{document}
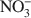 relative to that of the metal hydroxide layers. On heating to 175°C, the Br− ion migrates from positions of lesser site degeneracy to those of greater site degeneracy as dehydration of the interlayer opens up access to positions hitherto occupied by intercalated water molecules. The new (18h) site is situated closer to the proton of the metal hydroxide layer (1.809 Å) compared to the 6c site (2.402 Å). This shows a pre-association of the bromide ion with the proton of the hydroxide layer leading to the release of HBr upon decomposition of the bromide-containing LDHs. The fact that Cl−-containing LDHs also decompose with the evolution of HCl shows that such a redistribution of the atoms in the interlayer is more common than is generally recognized.
relative to that of the metal hydroxide layers. On heating to 175°C, the Br− ion migrates from positions of lesser site degeneracy to those of greater site degeneracy as dehydration of the interlayer opens up access to positions hitherto occupied by intercalated water molecules. The new (18h) site is situated closer to the proton of the metal hydroxide layer (1.809 Å) compared to the 6c site (2.402 Å). This shows a pre-association of the bromide ion with the proton of the hydroxide layer leading to the release of HBr upon decomposition of the bromide-containing LDHs. The fact that Cl−-containing LDHs also decompose with the evolution of HCl shows that such a redistribution of the atoms in the interlayer is more common than is generally recognized.
Extraction of diagenetic and detrital ages and of the 40Kdetrital/40Kdiagenetic ratio from K-Ar dates of clay fractions
- Marek Szczerba, Jan Środoń
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 93-103
-
- Article
- Export citation
-
Illite age analysis (IAA) is a classical method for extracting diagenetic and detrital ages from mixed ages measured by K-Ar. This approach is based on measuring the masses of diagenetic and detrital illitic components in a few different grain-size fractions of one rock sample and measuring the mixed ages of these fractions. The 1Md illitic polytype is usually considered to be diagenetic, while 2M1 is considered detrital. A plot of the function: exp(λt)−1 (where t is time and λ is the decay constant) vs. weight percent of the detrital fraction is constructed. On the basis of linear extrapolation to end-member fractions, the diagenetic and the detrital age is obtained. This approach does not take into account various K contents in different polytypes (%Kdetrital and %Kdiagenetic). In order to do that, the detrital mass fraction (wt.%detrital) should be recalculated into the percentage of detrital K (%Id(K)):
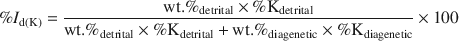
Analytical constraint of the K content of different polytypes is very difficult, so a new approach to this problem has been developed. In the present study, the plot of 40Ar*/40K vs. %Id(K) for a precisely determined ratio of 40Kdetrital/40Kdiagenetic was observed to be linear. On the basis of this observation, a computer program, MODELAGE, was written in the Java programming language using as input a few measured detrital illite mass fractions along with the mixed K-Ar ages of the relevant grain fractions. It then calculates the end-member ages and the 40Kdetrital/40Kdiagenetic ratio using genetic algorithms.
The errors in diagenetic and detrital illite mass-fraction determination mean that the 40Kdetrital/40Kdiagenetic ratio and the end-member ages can be evaluated only with some uncertainty. The best results are obtained if the measured mass fractions represent a relatively broad range. Constraining one of the unknowns (particularly the 40Kdetrital/40Kdiagenetic ratio) improves the results significantly.
Evaluation of data obtained from the literature using the proposed approach leads to the conclusion that the 40Kdetrital/40Kdiagenetic ratio is often >1.00, and some of 1Md illite polytype materials may be of detrital origin. If this is not the case, if a broad range of mass fractions is covered, and if the differences between end-member ages are relatively small, IAA analysis still gives appropriate results, even if the true 40Kdetrital/40Kdiagenetic ratio is different from 1.00.
Clay profiling: The classification of montmorillonites
- Katja Emmerich, Felicitas Wolters, Guenter Kahr, Gerhard Lagaly
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 104-114
-
- Article
- Export citation
-
Montmorillonites, with the general composition
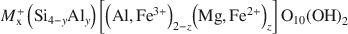 where x = ξ = 0.2–0.6, x = y+z, and y≪z, vary widely in composition and structure. The commonly used classification into five montmorillonite and two beidellite groups for the solid-solution sequence does not allow an unambiguous classification with respect to structural features and the resulting properties.
where x = ξ = 0.2–0.6, x = y+z, and y≪z, vary widely in composition and structure. The commonly used classification into five montmorillonite and two beidellite groups for the solid-solution sequence does not allow an unambiguous classification with respect to structural features and the resulting properties.The smectite structure reveals five features that allow an unambiguous description of a sample: (1) identification as either a dioctahedral or a trioctahedral smectite; (2) layer charge; (3) charge distribution between tetrahedral and octahedral sheets; (4) cation distribution within the octahedral sheet; and (5) Fe content. In addition, the nature of interlayer cations should be given as they influence certain properties of montmorillonites. Analytical methods are now available to measure and determine these structural features. Therefore, a precise classification for montmorillonites requires determination of layer charge and exchangeable cations, analysis of chemical composition, and thermal analysis (to determine the octahedral structure), in addition to X-ray diffraction analysis.
A comprehensive classification of montmorillonites based on these parameters is proposed. Ninety-six structural variations (expressed by systematic names) theoretically exist within the montmorillonite-beidellite series. Descriptive names can be used to elucidate the macroscopic properties of the montmorillonite samples in question.
A comprehensive characterization of dioctahedral smectites
- Felicitas Wolters, Gerhard Lagaly, Guenter Kahr, Rolf Nueesch, Katja Emmerich
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 115-133
-
- Article
- Export citation
-
The term ‘montmorillonite’ encompasses a wide range of chemical compositions and structures. Comprehensive and reliable characterization is essential for unambiguous classification. Twenty eight purified, Na-exchanged smectites (<0.2 µm) were characterized by layer-charge measurement using the alkylammonium method, by cation exchange capacity (CEC) measurement with Cu-triethylenetetramine, by determination of the chemical composition using X-ray fluorescence analysis, by calculation of the structural formula following determination of the octahedral structure (trans-vacant vs. cis-vacant) by simultaneous thermal analysis, and by X-ray diffraction analysis. Mössbauer spectroscopy was applied to determine the oxidation state and coordination of Fe and changes thereof during purification of the source materials.
The charge derived from chemical composition was considerably greater (by up to 30%) than the measured layer charge. The independently measured layer charge was used to calculate the structural formula. The measured CEC values, comprising the permanent charges and the pH-dependent edge charges, were consistent with measured layer charge but not with layer charge derived from the chemical composition. Therefore, the structural formula of smectites should be calculated using the measured layer charge.
The dehydroxylation temperature, which conveys information about the structure of the octahedral sheet, was correlated to the amount of Mg and Fe3+ and the location of charges. No relationship was found among the dehydroxylation temperature and the mean layer charge or the Mg content. In contrast, a clear relationship was observed between the Fe content and the dehydroxylation temperature. Montmorillonites with an Fe content <0.3/f.u. are cis-vacant and those containing Fe3+ > 0.3 mol/f.u. are trans-vacant, mostly with additional cis-vacancies. Tetrahedral substitution also appeared to be a function of the number of trans-vacancies.
The parameters analyzed provide the basis for a new descriptive classification system.
Nomenclature for stacking in phyllosilicates: Report of the Association Internationale pour l’etude des argiles (AIPEA) nomenclature committee for 2008
- Stephen Guggenheim, John M. Adams, Faïza Bergaya, Maria F. Brigatti, Victor A. Drits, Milton L. L. Formoso, Emilio Galán, Toshihiro Kogure, Helge Stanjek, Joseph W. Stucki
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 134-135
-
- Article
- Export citation



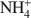
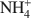
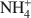
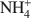
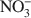
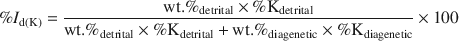
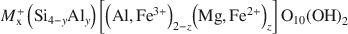 where
where