Volume 57 - Issue 2 - April 2009
Article
The transformation of synthetic hectorite in the presence of Cu(II)
- Håkon Fischer, Peter G. Weidler, Bernard Grobéty, Jörg Luster, Andreas U. Gehring
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 139-149
-
- Article
- Export citation
-
The influence of Cu(II) on the hydrothermal and thermal transformations of a synthetic hectorite was investigated by a combined approach using mainly X-ray diffraction, thermal analyses, and electron paramagnetic resonance spectroscopy. The presence of Cu(II) during hydrothermal treatment increased the crystallite size. Copper (II) was both structure-bound and associated with the inner surfaces of the particles. Upon heating, structural destabilization of the hectorite began at ∼400°C as indicated by the formation of free radicals. Between 600 and 700°C, the hectorite converted to enstatite, and in the presence of Cu(II), to enstatite and richterite. The formation of richterite as an additional conversion product is explained by the creation of structural weakness due to structure-bound Cu(II) in F-containing hectorite. Our results suggest that traces of Cu(II), typical of natural environments, may influence the conversion products in high-temperature geochemical systems.
Swelling behavior of Na- and Ca-montmorillonite up to 150°C by in situ X-Ray diffraction experiments
- Shoji Morodome, Katsuyuki Kawamura
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 150-160
-
- Article
- Export citation
-
The effects of temperature on the swelling properties of smectites are important for a variety of different geological conditions, but studies on this topic have been rather limited. The purpose of this study was to investigate the swelling behavior of Na- and Ca-montmorillonite at various temperatures greater than room temperature, up to 150°C, using in situ X-ray diffraction (XRD) analysis. A sample chamber was designed, the temperature and humidity of which were controlled precisely, for environmental in situ measurements. The XRD measurements were performed at small relative humidity (RH) intervals for precise observation of the swelling behavior.
The swelling behavior of Na-montmorillonite showed distinct zero-, one-, and two-layer hydration states. The basal spacings of Na-montmorillonite changed continuously with RH for various temperatures in the transition region between the zero- and one-layer hydration states, and the swelling curves of the transition region moved to greater degrees of RH with increasing temperature. The basal spacings jumped from the one- to two-layer hydration states for all temperatures at almost the same RH.
The basal spacings of Ca-montmorillonite changed continuously from the zero- to the two-layer hydration states at all temperatures. This behavior is remarkably different from that of Na-montmorillonite. At low-RH conditions, the d001 value of Ca-montmorillonite decreased with increasing temperature. The swelling curves of Ca-montmorillonite did not show a plateau at any temperature for the one-layer hydration state. The swelling curves of Ca-montmorillonite moved to greater RH with temperature, similar to the transformation region between the zero- and one-layer hydration states in Na-montmorillonite. These differences between Na- and Ca-montmorillonite are related to the hydration powers of exchangeable cations.
Enhancement of dissolution rates of amorphous silica by interaction with amino acids in solution at pH 4
- Motoharu Kawano, Tamao Hatta, Jinyeon Hwang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 161-167
-
- Article
- Export citation
-
Amino acids are present in various geochemical environments and they interact with mineral surfaces. To evaluate the effects of amino acids on mineral dissolution at pH conditions less than their isoelectric points (pI), dissolution experiments of X-ray amorphous silica in solutions containing 10.0 mmol/L of various amino acids (cysteine, asparagine, serine, tryptophan, alanine, threonine, histidine, lysine, and arginine) at pH 4 were performed. The results confirmed that basic amino acids (histidine, lysine, and arginine) produce an 8- to 8.5-fold enhancement of the rate of dissolution of amorphous silica compared with an amino acid-free control. Neutral amino acids (cysteine, asparagine, serine, tryptophan, alanine, and threonine) enhanced rates of dissolution by a factor of ∼3 to 3.5. The rate-enhancement effects of amino acids are controlled by concentrations of the amino acid’s cationic species which interact with the negatively charged >SiO− sites at the surface of the amorphous silica.
Chemostratigraphic correlation of sediments containing expandable clay minerals based on ion exchange with Cu(II) triethylenetetramine
- Tomáš Grygar, Jaroslav Kadlec, Anna Žigová, Martin Mihaljevič, Tereza Nekutová, Richard Lojka, Ivo Světlík
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 168-182
-
- Article
- Export citation
-
Copper(II) triethylenetetramine [Cu(trien)]2+ is an agent suitable for the 1-step determination of the cation exchange capacity (CEC) of many geomaterials using a procedure much less laborious than other, commonly used methods. It is also suitable for the determination of the composition of original exchangeable cations. In contrast to other common ions used for CEC analysis, the Cu(II) complex with triethylenetetramine, [Cu(trien)]2+, is specific for expandable clay minerals. The robustness of [Cu(trien)]2+ analysis was verified using reference clays, ion-exchanged reference clays, sediments, and soils. The [Cu(trien)]2+-based CEC of expandable clay minerals is not influenced significantly by ferrihydrite, goethite, manganite, birnessite, calcite, and gypsum. Birnessite, calcite, and gypsum admixtures affect the composition of the evolved cations. [Cu(trien)]2+ does not recover the entire CEC of soils (but rather that of the clay minerals only) which contain components other than clays which contribute to the CEC, e.g. soil organic matter. In a series of loess with buried paleosols and recent soils the [Cu(trien)]2+-based CEC ranged from 30 to 110% of total CEC obtained by traditional BaCl2 methods. The relative ratio of Ca to Mg, the prevailing exchangeable cations in soils and sediments in exogenic environments, are similar after [Cu(trien)]2+ and conventional BaCl2 treatments. The Ca/Mg ratio in the exchangeable fraction was used successfully for chemostratigraphic correlation of paleolacustrine sediments from a large lake in the Upper Carboniferous basins of eastern equatorial Pangaea and a series of recent flood plain sediments of the meandering Morava River in the Czech Republic. The Ca/Mg ratio obtained by [Cu(trien)]2+ analysis is proposed as a novel tool for the chemostratigraphic correlation of sediment series containing expandable clay minerals.
Crystallographic features and cleavage nanomorphology of chlinochlore: Specific applications
- Giovanni Valdrè, Daniele Malferrari, Maria Franca Brigatti
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 183-193
-
- Article
- Export citation
-
Natural and synthetic micas have been used widely as substrates to study biological systems; but, as in the case of negatively charged DNA, anionic charge repulsion may render micas a less than ideal templating surface for many biological systems. The purpose of this study was to investigate the potential for the chlorite clinoclore, which contains a positively charged interlayer octahedral sheet, to serve as a substrate for DNA adsorption. The relationships between clinochlore cleavage characteristics, in terms of nano-morphology, and surface potential are investigated, as are its average crystal chemistry and topology. That the structural features of clinochlore can be used successfully to condense, order, and self assemble complex biomolecules, such as DNA is also proven.
A natural IIb-4 clinochlore [\$\end{document}
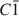 symmetry, unit-cell parameters a = 0.53301(4); b = 0.92511(6); c = 1.4348(1) (nm); α = 90.420(3); β = 97.509(3); γ = 89.996(4) (°)] with chemical composition \$\end{document}
symmetry, unit-cell parameters a = 0.53301(4); b = 0.92511(6); c = 1.4348(1) (nm); α = 90.420(3); β = 97.509(3); γ = 89.996(4) (°)] with chemical composition \$\end{document}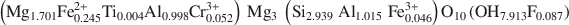 was selected. The octahedral sites of the silicate layer (<M(1)−O> = 0.2080 nm and <M(2)−O> = 0.2081 nm) are equal and occupied by Mg, whereas the octahedral sites in the interlayer M(3) and M(4) (<M(3)−O> = 0.2088 nm and <M(4) − O> = 0.1939 nm) show different sizes and are mostly completely occupied by divalent (Mg2+ and Fe2+) and trivalent (Al3+) cations, respectively.
was selected. The octahedral sites of the silicate layer (<M(1)−O> = 0.2080 nm and <M(2)−O> = 0.2081 nm) are equal and occupied by Mg, whereas the octahedral sites in the interlayer M(3) and M(4) (<M(3)−O> = 0.2088 nm and <M(4) − O> = 0.1939 nm) show different sizes and are mostly completely occupied by divalent (Mg2+ and Fe2+) and trivalent (Al3+) cations, respectively.The clinochlore cleaved surface is present in two forms: (1) the stripe type (0.40 nm in height, up to several micrometers long and ranging from some nanometers to a few microns in lateral size); and (2) the triangular type (0.40 nm in height). Both features may result either from interlayer sheets whose cleavage weak directions are related to the different M(3) and M(4) site occupancy, or from weak interlayer bonding along specific directions to the 2:1 layer underneath. The cleaved surface, particularly at the cleaved edges, presents high DNA affinity, which is directly related to an average positive surface and ledge potential.
Interlayer conformations of intercalated dodecyltrimethylammonium in rectorite as determined by FTIR, XRD, and TG analyses
- Zhaohui Li, Wei-Teh Jiang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 194-204
-
- Article
- Export citation
-
Intercalation of a wide variety of alkylammonium cations into the interlayer spaces of swelling clay minerals leads to many different applications, ranging from surface-charge measurements to rendering the clay compatible for the preparation of clay nanocomposites, but knowledge of the exact conformation of the intercalated organic species is still incomplete, thus preventing a full understanding of this process. The purpose of this study was to investigate the interlayer conformation of dodecyltrimethylammonium (DDTMA) bromide in rectorite as affected by the amounts of DDTMA intercalated, using a battery of physical and spectroscopic methods. The capacity of rectorite to intercalate DDTMA was equivalent to 1.67 times the cation exchange capacity (1.67 CEC) of the mineral even though the initial input was as much as 5.00 CEC. When the DDTMA intercalated was <0.50 CEC of the mineral, minimal counterion sorption was associated with DDTMA intercalation. Derivative thermogravimetric (DTG) analyses revealed a single-peak decomposition temperature (Tpeak) at 430°C. X-ray diffraction (XRD) analyses indicated a flat-lying monolayer of the intercalated molecules, while shifts in Fourier Transform infrared (FTIR) bands confirmed gauche conformation. These results suggested that cation exchange was the dominant mechanism. At the maximum intercalation, the DDTMA adopted a horizontal trilayer arrangement with mainly gauche conformation as determined by FTIR and XRD. Meanwhile a second Tpeak appeared at 255°C, similar to the Tpeak of solid DDTMA. Counterion bromide sorption accompanying DDTMA intercalation reached a capacity of 310 mmol/kg. The results indicated that van der Waals interaction was responsible for the DDTMA uptake at the amount beyond 0.50 CEC. When the amount of DDTMA intercalated was between 0.5 and 1.67 CEC, the XRD patterns showed non-integrality, i.e. the 002 reflection was split into two non-integral peaks with 2 × d002 < d001 and 3 × d003 > d001. They became integral at 1.67 CEC. The results suggest that the mineral might be composed of mixed layers of a monolayer intercalated rectorite and a trilayer intercalated rectorite, without a bilayer as intermediate, when the amount of DDTMA intercalated was between 0.5 and 1.67 CEC.
Bacteria-clay interaction: Structural changes in smectite induced during biofilm formation
- Alexandra Alimova, A. Katz, Nicholas Steiner, Elizabeth Rudolph, Hui Wei, Jeffrey C. Steiner, Paul Gottlieb
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 205-212
-
- Article
- Export citation
-
Bacteria play an important role in determining the properties and behavior of clay minerals in natural environments and such interactions have great potential for creating stable biofilms and carbon storage sites in soils, but our knowledge of these interactions are far from complete. The purpose of this study was to understand better the effects of bacteria-generated biofilms on clay interlayer expansion. Mixtures of a colloidal, 2-water hectorite clay and Pseudomonas syringae in a minimal media suspension evolve into a polysaccharide-rich biofilm aggregate in time-series experiments lasting up to 1 week. X-ray diffraction analysis reveals that upon aggregation, the clay undergoes an initial interlayer contraction. Short-duration experiments, up to 72 h, result in a decrease in the d001 value from 1.50 to 1.26 nm. The initial interlayer contraction is followed in long-duration (up to 1 week) experiments by an expansion of the d001 value of 1.84 nm. The expansion is probably a result of large, biofilm-produced, polymeric molecules being emplaced in the interlayer site. The resultant organo-clay could provide a possible storage medium for carbon in a microbial colony setting.
The role of clay minerals in the preservation of organic matter in sediments of Qinghai Lake, NW China
- Bingsong Yu, Hailiang Dong, Hongchen Jiang, Guo LV, Dennis Eberl, Shanying Li, Jinwook Kim
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 213-226
-
- Article
- Export citation
-
The role of saline lake sediments in preserving organic matter has long been recognized. In order to further understand the preservation mechanisms, the role of clay minerals was studied. Three sediment cores, 25, 57, and 500 cm long, were collected from Qinghai Lake, NW China, and dissected into multiple subsamples. Multiple techniques were employed, including density fractionation, X-ray diffraction, scanning and transmission electron microscopy (SEM and TEM), total organic carbon (TOC) and carbon compound analyses, and surface area determination. The sediments were oxic near the water-sediment interface, but became anoxic at depth. The clay mineral content was as much as 36.8%, consisting mostly of illite, chlorite, and halloysite. The TEM observations revealed that organic matter occurred primarily as organic matter-clay mineral aggregates. The TOC and clay mineral abundances are greatest in the mid-density fraction, with a positive correlation between the TOC and mineral surface area. The TOC of the bulk sediments ranges from 1 to 3% with the non-hydrocarbon fraction being predominant, followed by bitumen, saturated hydrocarbon, aromatic hydrocarbons, and chloroform-soluble bitumen. The bimodal distribution of carbon compounds of the saturated hydrocarbon fraction suggests that organic matter in the sediments was derived from two sources: terrestrial plants and microorganisms/algae. Depth-related systematic changes in the distribution patterns of the carbon compounds suggest that the oxidizing conditions and microbial abundance near the water-sediment interface promote degradation of labile organic matter, probably in adsorbed form. The reducing conditions and small microbial biomass deeper in the sediments favor preservation of organic matter, because of the less labile nature of organic matter, probably occurring within clay mineral-organic matter aggregates that are inaccessible to microorganisms. These results have important implications for our understanding of mechanisms of organic matter preservation in saline lake sediments.
Polarized ATR-FTIR investigation of Fe reduction in the Uley nontronites
- Bryan R. Bzdek, Molly M. McGuire
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 227-233
-
- Article
- Export citation
-
Reduction of structural Fe in smectites affects the surface chemical behavior of the clay, but the underlying mechanism and changes in clay structure are still in need of investigation, particularly with respect to changes in the tetrahedral sheet. The purpose of this study was to probe changes in the tetrahedral sheet that occur when structural Fe is reduced in the Uley nontronites, NAu-1 and NAu-2, using polarized attenuated total internal reflection Fourier-transform infrared spectroscopy. Despite the differences in their structures — NAu-2 has tetrahedral Fe3+ while NAu-1 does not — the changes observed in the Si-O stretching region were quite similar. Reduction results in a shift of the in-plane Si-O stretching modes to lower frequencies, while the out-of-plane Si-O stretch shifts to higher frequencies. The magnitude of these shifts is greater in NAu-2 than in NAu-1, but the crystallinity of the tetrahedral silicate sheet of NAu-2 is preserved upon reduction. In both nontronites, the orientation of the out-of-plane Si-O bond changes and becomes completely perpendicular to the basal (001) surface of the clay, indicating the formation of trioctahedral domains wherein the individual tetrahedra reorient relative to the plane of the clay layer.
Simultaneous incorporation of Cr, Zn, Cd, and Pb in the goethite structure
- Navdeep Kaur, Markus Gräfe, Balwant Singh, Brendan Kennedy
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 234-250
-
- Article
- Export citation
-
In order to improve our understanding of how the goethite crystal structure is affected by the incorporation of metals (and by variations in the amount of the incorporation), and to review any possible synergistic and antagonistic effects of co-metals, the present investigation focused on the incorporation of multiple (di-, tri-, and tetra-) metals, i.e. Cr, Zn, Cd, and Pb, in the goethite crystallographic structure. A series of single- and multi-metal M-Cr/Zn/Cd/Pb-substituted goethites with M/(M+Fe) molar ratios = 0.10 were prepared. The general sequence of metal entry in single-metal substituted goethites was Zn = Cr > Cd > Pb and in multi-metal-substituted goethites was Zn > Cr ⩾ Cd > Pb. Simultaneous incorporation of Cr, Zn, Cd, and Pb up to 10.5 mole % was achieved in goethite. Synchrotron X-ray diffraction and extended X-ray absorption fine structure (EXAFS) techniques were employed to assess the structural characteristics of the synthesis products. Rietveld refinement of XRD data showed small changes in unit-cell parameters and Fe/M-Fe/M distances due to M substitution(s). A typical goethite-like crystalline structure remained intact, however. The unit-cell parameters were mutually, linearly correlated, though Fe/M-Fe/M distances were not, indicating that complex changes occurred at the local scale. In single-metal substituted goethites, incorporation of Cr reduced the unit-cell volume by 0.13% while that of Zn, Cd, and Pb increased it by 1.09, 3.58, and 0.56%, respectively. The changes in multi-metal-substituted goethites appeared to be the complex combination of that of the individually incorporated metals. The X-ray absorption near edge structure study of Pb-substituted goethites showed that the majority of associated Pb was Pb2+, while Pb4+ was preferred over Pb2+ in the bulk structure. Measurements by EXAFS at the Fe K-edge indicated that the Fe polyhedra contracted in the presence of Cd2+ and Pb2+, providing room for the substitution of larger cations. Measurements by EXAFS at the Cr and Cd K-edges indicated symmetric Cr/Cd polyhedra with single Cr/Cd-O distances and, at Fe and Zn K-edges and the Pb LIII-edge, indicated asymmetric polyhedra with two sets of Fe/Zn/Pb-O distances. The Zn octahedra were possibly Zn(OH)4O2, which enlarged the metal-metal corner-sharing distance to 3.86 Å. This configuration of ligands around the Zn2+ cation might occur to balance local charges. Symmetric polyhedra appeared to reduce steric strains in the structure, compared to the asymmetric polyhedra. The result was that Cr enhanced the incorporation of Zn, Cd, and Pb, while the converse was true for Zn.
Anisotropy of magnetic susceptibility study of kaolinite matrix subjected to biaxial tests
- Aniruddha Sengupta
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 251-263
-
- Article
- Export citation
-
The potential for structural failure of consolidated clay materials, which is of great importance in many applications, typically are assessed by measuring the localized strain bands that develop under anisotropic load stress. Most methods are precluded from providing a full understanding of the strain anisotropy because they only give two-dimensional information about the stressed clay blocks. The purpose of the present study was to investigate three-dimensional strain localization in a kaolinite matrix, caused by strain anisotropy due to a biaxial plane-strain test, using a relatively new method known as Anisotropy of Magnetic Susceptibility (AMS). This method involves induction of magnetism in an oriented sample in different directions and measurement of the induced magnetization in each direction. The AMS analyses were performed on core samples from different parts of the deformed kaolinite matrix. The degree of magnetic anisotropy (P′), which is a measure of the intensity of magnetic fabric and a gauge of strain intensity, was shown to be greater in cores containing shear bands than in those containing none. A threshold value for P′ for the deformed kaolinite matrix was identified, above which shear bands may develop. The comparison of the shape parameter (T), obtained from undeformed and deformed samples, illustrated a superimposition of prolate strain over the original oblate fabric of the kaolinite matrix. The orientation of the principal strain axis revealed that reorientation or rotation of the principal axis occurred along the shear bands.
Mineralogical and physicochemical investigation of Mg-smectite from Jbel Ghassoul, Morocco
- Abdelaziz Benhammou, Boumediene Tanouti, Lahbib Nibou, Abdelrani Yaacoubi, Jean-Pierre Bonnet
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 264-270
-
- Article
- Export citation
-
‘Ghassoul’ clay is a Mg-rich clay from Morocco which is of great industrial use and interest, but its characterization is still incomplete. The purpose of this study was to provide further details regarding the structure and characteristics of this important commercial clay mineral. Mineralogical and physicochemical characterizations of the raw form of ‘Ghassoul’ clay from Jbel Ghassoul in Morocco, and of its <2 µm size fraction, purified and Na+-saturated, were carried out using X-ray diffraction (XRD), X-ray fluorescence (XRF), infrared spectroscopy (IR), and differential and thermogravimetric analysis (DTA/TG). The XRD data revealed that the raw Ghassoul clay consists mainly of a Mg-rich trioctahedral smectite, stevensite, together with quartz and dolomite. The IR spectra consisted of vibrations typical of trioctahedral smectites. The DTA/TG curves were also similar to those of trioctahedral smectites. Chemical analysis confirms that this Mg-rich trioctahedral smectite is a stevensite characterized by a limited isomorphic substitution of Si4+ by Al3+. The thermal transformations examined by in situ XRD as a function of the firing temperature from 100 to 1200°C indicated that stevensite was transformed to enstatite (MgSiO3) at temperatures >800°C and that quartz was transformed to cristobalite when the temperature exceeded 1100°C. These transformations were irreversible. The specific surface area and cation exchange capacity (CEC) of the ‘Ghassoul’ clay are 133 m2/g and 75 meq/100 g, respectively. The main exchangeable cation is Mg2+ (53 meq/100 g).
Melt synthesis and characterization of synthetic Mn-rich tainiolite
- Alexander Baumgartner, Christian Butterhof, Sebastian Koch, Ruslan Mariychuk, Josef Breu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 271-277
-
- Article
- Export citation
-
Large transition-metal contents add desirable physical properties, such as redox reactivity, magnetism, and electric or ionic conductivity to micas and make them interesting for a variety of materials-science applications. A Mn- and F-rich tainiolite mica,
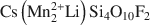 , was synthesized by a high-temperature melt-synthesis technique. Subsequent annealing for 10 days led to a single-phase and coarsegrained material. Single-crystal X-ray diffraction studies were performed and characteristic geometric parameters were compared to the analogous ferrous compound, synthetic Fe-rich tainiolite,
, was synthesized by a high-temperature melt-synthesis technique. Subsequent annealing for 10 days led to a single-phase and coarsegrained material. Single-crystal X-ray diffraction studies were performed and characteristic geometric parameters were compared to the analogous ferrous compound, synthetic Fe-rich tainiolite, 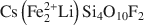 . Both tainiolite structures are outside the compositional stability limits for the 2:1 layer structure, and incorporating the relatively large cation Mn2+ requires significant structural adjustments in both the octahedral and tetrahedral sheets. As expected, increasing the ionic radius of the octahedral cation from 0.78 Å (VIFe2+) to 0.83 Å (VIMn2+) reduces the octahedral flattening angle from <Ψ> = 57.05° to <Ψ> = 56.4°, the smallest value ever observed for a tetrasilicic mica. However, even this small <Ψ> value is insufficient to match the lateral sizes of the tetrahedral and octahedral sheets and, in addition, unusual structural adjustments in the tetrahedral sheet are required. The average tetrahedral bond length <T-O> is much greater (1.643 Å) than the average value observed for tetrasilicic micas (1.607 Å,) and a significant difference between the <T-O>apical (1.605 Å) and the <T-O>basal bond lengths (1.656 Å) and an enlarged basal flattening angle (τbas = 106.29°) are noted. These parameters indicate: (1) that the 2:1 layer might be more flexible than previously thought, to allow matching of the lateral dimensions of the tetrahedral and octahedral sheets; and (2) that many other compositions that appear interesting from a materials-science point of view might be accessible.
. Both tainiolite structures are outside the compositional stability limits for the 2:1 layer structure, and incorporating the relatively large cation Mn2+ requires significant structural adjustments in both the octahedral and tetrahedral sheets. As expected, increasing the ionic radius of the octahedral cation from 0.78 Å (VIFe2+) to 0.83 Å (VIMn2+) reduces the octahedral flattening angle from <Ψ> = 57.05° to <Ψ> = 56.4°, the smallest value ever observed for a tetrasilicic mica. However, even this small <Ψ> value is insufficient to match the lateral sizes of the tetrahedral and octahedral sheets and, in addition, unusual structural adjustments in the tetrahedral sheet are required. The average tetrahedral bond length <T-O> is much greater (1.643 Å) than the average value observed for tetrasilicic micas (1.607 Å,) and a significant difference between the <T-O>apical (1.605 Å) and the <T-O>basal bond lengths (1.656 Å) and an enlarged basal flattening angle (τbas = 106.29°) are noted. These parameters indicate: (1) that the 2:1 layer might be more flexible than previously thought, to allow matching of the lateral dimensions of the tetrahedral and octahedral sheets; and (2) that many other compositions that appear interesting from a materials-science point of view might be accessible.



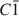
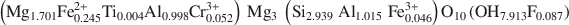
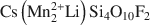 , was synthesized by a high-temperature melt-synthesis technique. Subsequent annealing for 10 days led to a single-phase and coarsegrained material. Single-crystal X-ray diffraction studies were performed and characteristic geometric parameters were compared to the analogous ferrous compound, synthetic Fe-rich tainiolite,
, was synthesized by a high-temperature melt-synthesis technique. Subsequent annealing for 10 days led to a single-phase and coarsegrained material. Single-crystal X-ray diffraction studies were performed and characteristic geometric parameters were compared to the analogous ferrous compound, synthetic Fe-rich tainiolite, 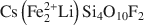 . Both tainiolite structures are outside the compositional stability limits for the 2:1 layer structure, and incorporating the relatively large cation Mn
. Both tainiolite structures are outside the compositional stability limits for the 2:1 layer structure, and incorporating the relatively large cation Mn