3 results
Susceptibility reporting and antibiotic prescribing for UTIs in the inpatient setting: A nudge toward improved stewardship
- Madison Ponder, Alan Kinlaw, Lindsay Daniels, Ashlyn Norris, Kevin Alby
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s17
-
- Article
-
- You have access Access
- Open access
- Export citation
-
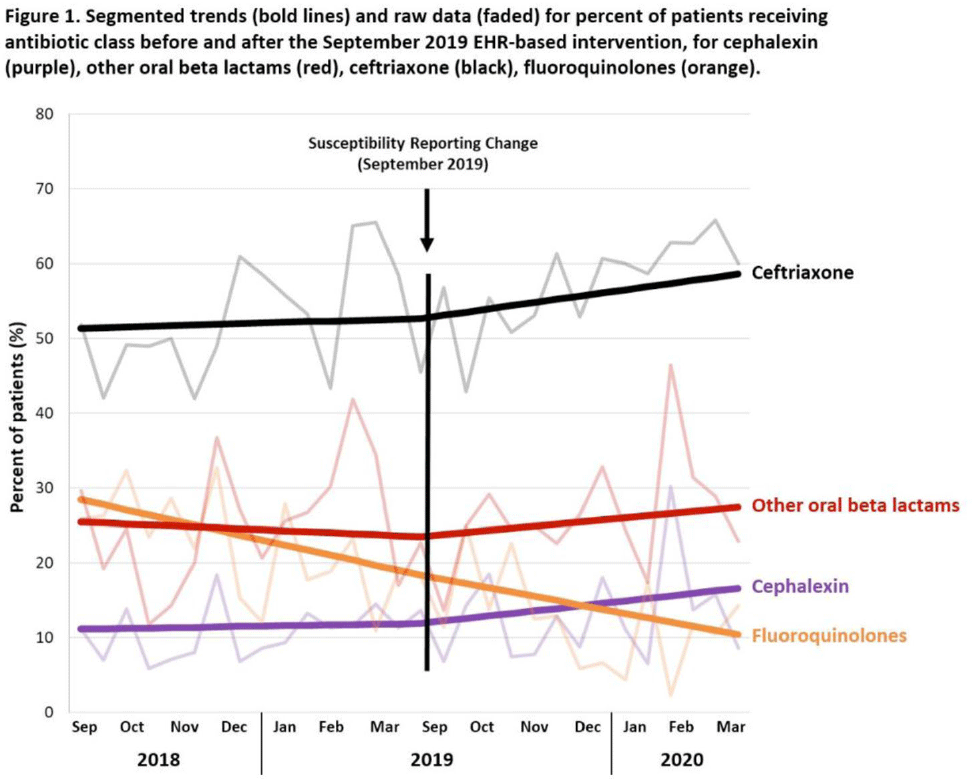
Background: Urinary tract infections (UTIs) are common in the inpatient, observation, and emergency department settings. Although many UTI-causing pathogens are susceptible to oral β-lactams, these agents are not tested directly, and susceptibility is extrapolated from other agents. To improve the use of these agents, the University of North Carolina Medical Center (UNCMC) added cephalexin to the susceptibility profile generated with urine culture results in the electronic health record (EHR). We evaluated prescribing trends of cephalexin, other oral β-lactams, fluoroquinolones, and other antibiotics for UTIs in the inpatient setting, before and after the susceptibility reporting change. Methods: An interrupted time-series analysis was conducted. Among 1,491 patients who had positive urine cultures with susceptibilities and received at least 1 antibiotic with a listed UTI indication during their inpatient stay at UNCMC, we measured the weekly prevalence (%) of patients who received each antibiotic group: cephalexin, other oral β-lactams (amoxicillin-clavulanate, cefdinir, cefuroxime), fluoroquinolones (levofloxacin, ciprofloxacin), and ceftriaxone. The study comprised a preintervention period (September 2018–March 2019) and a postintervention period (September 2019–March 2020). The prevalence of each antibiotic or group was plotted over time, and segmented linear regression was used to estimate the impact of the intervention on each antibiotic groups’ time trend. Results: At study baseline in September 2018, the weekly prevalence of antibiotic use was 11% for cephalexin, 26% for other oral β-lactams, 51% for ceftriaxone, and 29% for fluoroquinolones. Fluoroquinolone use decreased steadily throughout the study period, by 11% during the 7-month preintervention period (95% CI, −17% to −5%) and by 8% (95% CI, −13% to −3%) after the intervention (P for trend deflection, .70). In contrast, during the preintervention period, trends were flat for cephalexin, ceftriaxone, and other oral β-lactams (all P for nonzero preintervention slope were >.40). During the postintervention period, use increased for ceftriaxone (6%; 95% CI, 3%–9%). Post-intervention use also increased for cephalexin (5%; 95% CI, −3% to 12%) and other oral β-lactams (4%; 95% CI, −8%, 15%), but these trends were imprecise and not statistically significant at α = .05. Fig. 1 displays trends and raw data for each antibiotic group. Conclusions: The urine culture susceptibility reporting change was associated with small increases in cephalexin and ceftriaxone use, coincident with continued decreasing use of fluoroquinolones, for hospitalized patients with positive urine cultures and a listed UTI indication. Low-resource EHR-based interventions may confer considerable benefit for antimicrobial stewardship efforts in this clinical setting, and larger real-world studies are needed to replicate and contextualize these findings.
Funding: None
Disclosures: None
Evaluation of Penicillin Allergies and an Allergy Assessment Pilot in the Emergency Department
- Ashlyn Norris, Kalynn Northam, Lindsay Daniels, Mildred Kwan, Gary Burke, Nikolaos Mavrogiorgos, Renae Boerneke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s39
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Penicillin (PCN) allergy is one of the most frequently reported medication allergies, with ~10% of the US population reporting a PCN allergy. However, studies have shown that only 1% of the US population have a true IgE-mediated reaction to PCN. Delabeling and appropriately updating patient allergy profiles could decrease the use of alternative broad-spectrum antibiotics, rates of infectious complications [C. difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE)], antibiotic resistance, and overall healthcare cost. The emergency department (ED) is an important setting in which to assess PCN allergies and to delabel patients when appropriate because there are >130 million ED visits in the United States each year. We sought to determine the percentage of PCN allergy–labeled patients who could be delabeled through a PCN allergy assessment interview in an ED. Key secondary outcomes included the percentage of interviewed patients who could not be delabeled based on history alone but would be eligible for an amoxicillin oral challenge or a PCN skin test (PST). A prospective PCN allergy assessment pilot was performed for patients aged >18 years presenting to the UNC Medical Center ED between December 1 and December 17, 2020, with a documented PCN allergy. A pharmacist conducted penicillin allergy assessments on a convenience sample of patients presenting to the ED between 8 a.m. and 3 p.m. on weekdays. Based on patients’ reported and documented histories, charts were updated with the most accurate information and allergies were delabeled if appropriate. In total, 95 patients were assessed; 62 (65.3%) were interviewed and 15 (24.2%) were delabeled. In addition, 26 patients (41.9%) were deemed eligible for an oral amoxicillin challenge, 19 (30.6%) qualified for a PST, and 2 (3.2%) patients did not qualify for further assessment due to having a an IgE-mediated reaction in the past 5 years. Of the 15 patients who were delabeled, 6 (40.0%) received antibiotics during their admission: 4 (73.3%) of those patients received a penicillin and 2 (36.7%) received a cephalosporin, all without adverse reactions. Patient assessments took ~20 minutes to complete, including chart review, patient interview, and postinterview chart updating. The results from this pilot study demonstrate the impact of performing PCN allergy assessments in ED. Interdisciplinary opportunities should be explored to develop processes that will improve the efficiency and sustainability of PCN allergy assessments within the ED to allow this important stewardship intervention to continue.
Funding: No
Disclosures: None
Evaluation of Penicillin Allergy Prevalence and Antibiotic Prescribing Patterns for Patients within the Emergency Department
- Ashlyn Norris, Lindsay Daniels, Nikolaos Mavrogiorgos, Kalynn Northam, Mildred Kwan, Gary Burke, Renae Boerneke
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s38
-
- Article
-
- You have access Access
- Open access
- Export citation
-
As the point of entry into healthcare for many patients, the emergency department (ED) is an ideal setting in which to assess penicillin (PCN) allergies. An estimated 10% of the United States population has a reported PCN allergy; however, few studies have evaluated the prevalence and impact of PCN allergies on antibiotic selection within the ED. Patients with a documented PCN allergy are more likely to be exposed to costly alternative broad-spectrum antibiotics that have higher rates of adverse events, including C. difficile infections. We sought to determine the prevalence of PCN allergies within the UNC Medical Center ED. Key secondary outcomes included the percentage of patients with a documented PCN allergy who (1) received alternative antibiotics (carbapenems, aztreonam, fluoroquinolones, clindamycin, vancomycin), (2) received β-lactam antibiotics and experienced an allergic reaction during their ED visit, and/or (3) had received a β-lactam antibiotic during a past hospitalization or ED visit without their chart being appropriately updated. A retrospective evaluation included patients aged >18 years with a documented PCN allergy who were discharged from the ED between January 1, 2017, and December 31, 2019. Over the study period, there were 14,635 patient encounters with a documented PCN allergy that comprised 8,573 unique patients. The prevalence of PCN allergies was 14.3% for all ED encounters. PCN allergy–labeled patients received alternative antibiotics in 59.4% of ED encounters in which antibiotics were prescribed. Of the 454 β-lactam antibiotics (62 penicillins, 380 cephalosporins, 12 carbapenems) administered to PCN allergy-labeled patients within the ED, there were zero allergic reactions. Also, 18.6% of PCN allergy-labeled patients had received and tolerated a β-lactam antibiotic during prior hospitalizations or ED visits (1.7% penicillins, 14.4% cephalosporins, 2.6% carbapenems) without appropriate updated documentation to reflect β-lactam antibiotic tolerance. These findings confirm the utilization of non–β-lactam antibiotics in PCN allergy-labeled patients, highlighting the importance of accurate and updated allergy documentation in the electronic medical record. These findings also demonstrate the need for improved allergy documentation and protocols to proactively assess penicillin allergy labels while in the ED.
Funding: No
Disclosures: None




