2 results
A dissipative random velocity field for fully developed fluid turbulence
- Rodrigo M. Pereira, Christophe Garban, Laurent Chevillard
-
- Journal:
- Journal of Fluid Mechanics / Volume 794 / 10 May 2016
- Published online by Cambridge University Press:
- 04 April 2016, pp. 369-408
-
- Article
- Export citation
-
We investigate the statistical properties, based on numerical simulations and analytical calculations, of a recently proposed stochastic model for the velocity field (Chevillard et al., Europhys. Lett., vol. 89, 2010, 54002) of an incompressible, homogeneous, isotropic and fully developed turbulent flow. A key step in the construction of this model is the introduction of some aspects of the vorticity stretching mechanism that governs the dynamics of fluid particles along their trajectories. An additional further phenomenological step aimed at including the long range correlated nature of turbulence makes this model dependent on a single free parameter,
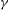 ${\it\gamma}$, that can be estimated from experimental measurements. We confirm the realism of the model regarding the geometry of the velocity gradient tensor, the power-law behaviour of the moments of velocity increments (i.e. the structure functions) including the intermittent corrections and the existence of energy transfer across scales. We quantify the dependence of these basic properties of turbulent flows on the free parameter
${\it\gamma}$, that can be estimated from experimental measurements. We confirm the realism of the model regarding the geometry of the velocity gradient tensor, the power-law behaviour of the moments of velocity increments (i.e. the structure functions) including the intermittent corrections and the existence of energy transfer across scales. We quantify the dependence of these basic properties of turbulent flows on the free parameter 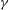 ${\it\gamma}$ and derive analytically the spectrum of exponents of the structure functions in a simplified non-dissipative case. A perturbative expansion in power of
${\it\gamma}$ and derive analytically the spectrum of exponents of the structure functions in a simplified non-dissipative case. A perturbative expansion in power of 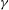 ${\it\gamma}$ shows that energy transfer, at leading order, indeed take place, justifying the dissipative nature of this random field.
${\it\gamma}$ shows that energy transfer, at leading order, indeed take place, justifying the dissipative nature of this random field.
12 - Genetics of human susceptibility to infection and hepatic disease caused by schistosomes
-
- By Alain J. Dessein, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Sandrine Marquet, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Carole Eboumbou Moukoko, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Hèlia Dessein, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Laurent Argiro, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Sandrine Henri, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Dominique Hillaire, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Christophe Chevillard, Immunologie et Génétique des Maladies Parasitaires, Institut National de la Santé et de la Recherche Médicale, Laboratoire de Parasitologie-Mycologie, Faculté de Médecine, Marseille, France, Nasureldin El Wali, Institute of Nuclear Medicine and Molecular Biology, University of Gezira, Wad Medani, Sudan, Mubarak Magzoub, Institute of Nuclear Medicine and Molecular Biology, University of Gezira, Wad Medani, Sudan, Laurent Abel, Génétique Humaine des Maladies Infectieuses, Institut National de la Santé et de la Recherche Médicale, Faculté de Médecine Necker, Paris, Virmondes Rodrigues, Jr, Faculty of Medicine do Triangulo Mineiro, Ubéraba, Brazil, Aluizio Prata, Faculty of Medicine do Triangulo Mineiro, Ubéraba, Brazil, Gachuhi Kimani, Kenya Medical Research Institute, Biomedical Sciences Research Centre, Nairobi, Kenya
- Edited by Richard Bellamy, Kintampo Health Research Centre, Ghana
-
- Book:
- Susceptibility to Infectious Diseases
- Published online:
- 14 August 2009
- Print publication:
- 22 December 2003, pp 337-360
-
- Chapter
- Export citation
-
Summary
Schistosome infections cause much suffering in millions of people living in tropical regions of Africa, Asia, and South America (Prata, 1987; Chitsulo et al., 2000). The most severe clinical symptoms affect the kidneys and urinary tract. However, schistosomes also cause various other disorders such as heart failure and neurological diseases. Three species of schistosome are responsible for most human infections (Schistosoma mansoni, Schistosoma japonicum, and Schistosoma haematobium). These species are found in different geographical locations, have different vectors, and cause different symptoms. Schistosomes are multicellular parasites that are disseminated as free swimming larvae (cercariae) in ponds, lakes, and rivers by snails. Humans become infected when they stay in contaminated water for a few minutes. The cercariae penetrate the human skin and develop into male or female adult schistosomes within 5 or 6 weeks. These small worms (Fig. 12.1A) can live in the vascular system of their vertebrate host for 2 to 5 years. Schistosomes do not multiply within their vertebrate host. The female worms, however, lay hundreds of eggs per day in the mesenteric or vesical veins of their host. Most of the symptoms associated with these infections are caused by the inflammation that is induced by the immunogenic and toxic substances produced by the eggs. The chronic cellular reaction that develops around the eggs is organised in a granuloma (Von Lichtenberg, 1962; Warren et al., 1967).