3 results
Staphylococcus epidermidis joint isolates: Whole-genome sequencing demonstrates evidence of hospital transmission and common antimicrobial resistance
- Samantha J. Simon, Mohamad Sater, Ian Herriott, Miriam Huntley, Emma Briars, Brian L. Hollenbeck
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 45 / Issue 2 / February 2024
- Published online by Cambridge University Press:
- 15 December 2023, pp. 150-156
- Print publication:
- February 2024
-
- Article
- Export citation
-
Objective:
We investigated genetic, epidemiologic, and environmental factors contributing to positive Staphylococcus epidermidis joint cultures.
Design:Retrospective cohort study with whole-genome sequencing (WGS).
Patients:We identified S. epidermidis isolates from hip or knee cultures in patients with 1 or more prior corresponding intra-articular procedure at our hospital.
Methods:WGS and single-nucleotide polymorphism–based clonality analyses were performed, including species identification, in silico multilocus sequence typing (MLST), phylogenomic analysis, and genotypic assessment of the prevalence of specific antibiotic resistance and virulence genes. Epidemiologic review was performed to compare cluster and noncluster cases.
Results:In total, 60 phenotypically distinct S. epidermidis isolates were identified. After removal of duplicates and impure samples, 48 isolates were used for the phylogenomic analysis, and 45 (93.7%) isolates were included in the clonality analysis. Notably, 5 S. epidermidis strains (10.4%) showed phenotypic susceptibility to oxacillin yet harbored mecA, and 3 (6.2%) strains showed phenotypic resistance despite not having mecA. Smr was found in all isolates, and mupA positivity was not observed. We also identified 6 clonal clusters from the clonality analysis, which accounted for 14 (31.1%) of the 45 S. epidermidis isolates. Our epidemiologic investigation revealed ties to common aspirations or operative procedures, although no specific common source was identified.
Conclusions:Most S. epidermidis isolates from clinical joint samples are diverse in origin, but we identified an important subset of 31.1% that belonged to subclinical healthcare–associated clusters. Clusters appeared to resolve spontaneously over time, suggesting the benefit of routine hospital infection control and disinfection practices.
Surveillance of healthcare-onset clinical cultures using whole-genome sequencing reveals hidden nosocomial transmission
- Mohamad Sater, Emma Briars, Connor Parrish, Ian Herriott, Kathleen Quan, Keith Madey, Julie Shimabukuro, Linda Dickey, Shruti Gohil, Alfred Wong, Talia Hollowell, Allison Brookhart, Alison Gassett, Miriam Huntley, Susan Huang, Cassiana E. Bittencourt
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s83-s84
-
- Article
-
- You have access Access
- Open access
- Export citation
-
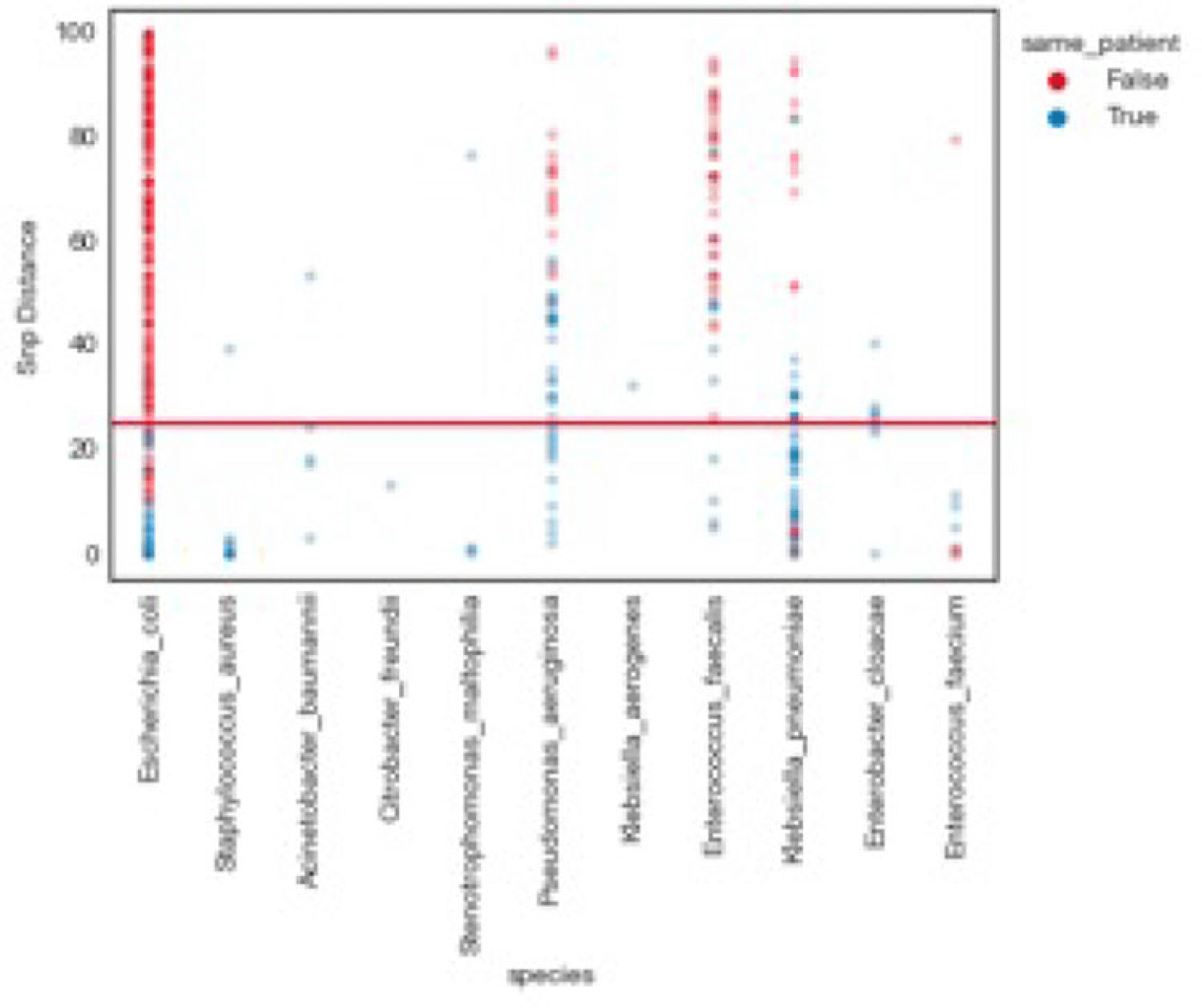
Background: Traditional hospital outbreak-detection methods are typically limited to select multidrug-resistant pathogens in a single unit, which can miss transmission of many medically important healthcare-transmissible pathogens. Whole-genome sequencing (WGS) enables comprehensive genomic resolution for accurate identification of clonal transmission. Previously, lack of scalability limited the use of WGS for hospital surveillance. Methods: We conducted prospective surveillance of select bacteria from all inpatient clinical cultures plus all bacteria from clinical cultures from ICUs and oncology units at the University of California Irvine (UCI) Clinical Microbiology Laboratory from September 2021 to February 2022. Due to pandemic stressors, this pilot test was a prelude to a real-time demonstration project. Its goal was to demonstrate the efficiency and scalability of the WGS platform when receiving samples monthly and analyzing results quarterly without the intent for real-time response. Bacterial isolates slated for discard were collected weekly and sent monthly to Day Zero Diagnostics for sequencing. In total, 1,036 samples from 926 patients were analyzed for genomic relatedness, a scalable and automated analysis pipeline already in use for rapid (days) characterization of genomic-relatedness in small and large sets of isolates. Mapping and SNP calling was performed against high-quality, best-match reference genomes. Sets of samples with pairwise distance of 2 persons with genomically related isolates and were denoted as “clusters.” Separately, we also investigated within-patient diversity by quantifying the genomic relatedness of isolates collected from individual patients. Results: Isolates represented 28 distinct species. We identified 10 Escherichia coli clusters (range, 2–4 patients; median, 2 patients), 2 Klebsiella pneumoniae clusters (range, 2–4 patients), and 1 Enterococcus faecium cluster (3 patients). All but 1 involved genomically matched isolates from multiple hospital locations. There were 4 Escherichia coli ST131 clusters spanning 4 months, including 1 with 4 patients across 3 different hospital locations. At a species level, there were distinct differences between the observed SNP distances between samples isolated from the same versus different patients (Fig. 1). All identified clusters had not been flagged by routine outbreak detection methods used by the UCI infection prevention program. Conclusions: Comprehensive WGS-based surveillance of hospital clinical isolates identified multiple potential transmission events between patients not in the same unit at the time cultures were taken. Combining WGS detection and real-time epidemiologic investigation may identify new avenues of transmission risk and could provide early warnings of clonal transmission to prevent larger outbreaks. High-volume surveillance of hospital isolates can also provide species- and context-specific clonality.
Financial support: This study was funded by Day Zero Diagnostics.
Disclosures: None
Detecting fecal microbiota transplantation–associated infection transmission using shotgun metagenomic sequencing and clonality analysis
- Emma Briars, Mohamad Sater, Nicole Billings, Ian Herriott, Emily MacLeod, Miriam Huntley
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s86
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Fecal microbiota transplantation (FMT) is a widely used modality for safe and effective treatment of recurrent Clostridium difficile infections, and FMT is being explored for the treatment of additional indications including gastrointestinal diseases and neurological disorders. Although microbiota-based therapies like FMT utilize rigorous donor screening procedures, these procedures are limited in resolution and scope, and there remains a risk of transmission of FMT-associated infectious agents from donor stool to a FMT recipient. Critically, these health concerns led the FDA to issue a 2019 safety alert for the transmission risks associated with FMT and to update its guidelines for screening and reporting. In a suspected transmission event, there is uncertainty around the source of infection; thus, methods are needed to rapidly determine whether a patient’s infection is linked to the donor stool product. Methods: Here, we developed a laboratory service sequencing and bioinformatics pipeline within our CLIA-certified laboratory for investigating suspected FMT infection transmission by measuring genomic relatedness. Our pipeline performs deep sequencing of a metagenomic sample, whole-genome sequencing (WGS) of an isolate derived from the implicated patient infection and determines the genomic relatedness between the 2 using a SNP-based analysis. The workflow was validated in silico with synthetic metagenomic samples spiked-in with WGS of clinically relevant isolate strains at varying abundance. Results: The sample and sequencing library preparation workflow was optimized across a panel of metagenomic and mock fecal microbiome samples demonstrating reproducible and reduced-bias sequencing of metagenomic samples. Our pipeline demonstrates high sensitivity and specificity for clonality calls when a spiked in isolate genome achieves 5× depth for >50% of the genome. We also demonstrated an interplay between abundance rate and sequencing depth for determining a clonality limit of detection. Conclusions: Taken together, our pipeline represents a new method that can support the clinical efforts of FMT and other microbiota-based therapies. References: US Food and Drug Administration. Important safety alert regarding use of fecal microbiota for transplantation and risk of serious adverse reactions due to transmission of multidrug-resistant organisms. Rockville, MD: Food and Drug Administration, 2019. DeFilipp Z, Bloom PP, Torres Soto M, et al. Drug-resistant E. coli bacteremia transmitted by fecal microbiota transplant. N Engl J Med 2019;381:2043–2050.
Financial support: This study was funded by Day Zero Diagnostics.
Disclosures: None




