4 results
Prior cultures predict subsequent susceptibility in patients with recurrent urinary tract infections
- Marissa Valentine-King, Barbara Trautner, Roger Zoorob, George Germanos, Jason Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s67
-
- Article
-
- You have access Access
- Open access
- Export citation
-
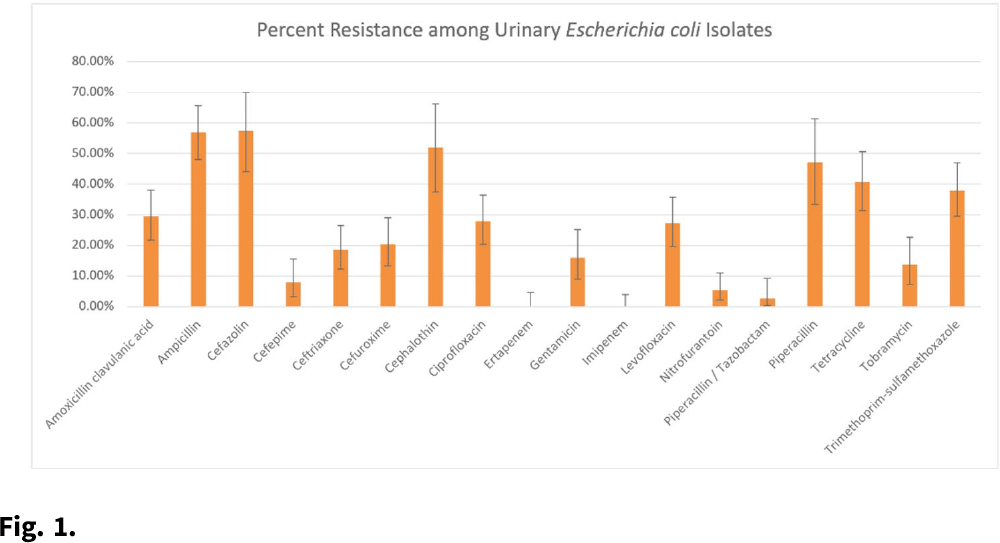
Background: Patients with recurrent urinary tract infections (rUTI) experience frequent exposure to antimicrobial regimens, leaving them at higher risk for developing antibiotic resistance. Little information on the prevalence of antibiotic resistance among patients with rUTI has been published. Although the IDSA recommends using a prior culture to guide empiric treatment, studies have not examined the predictive ability of a prior culture among patients meeting rUTI criteria. We constructed an antibiogram and evaluated test metrics, including sensitivity, specificity, and positive predictive value (PPV) and negative predictive values (NPV) of a prior culture (any organism), on predicting resistance (PPV) or susceptibility (NPV) of a future culture among patients with uncomplicated rUTI in an outpatient setting. Methods: We retrospectively extracted electronic health record data from outpatients aged ≥18 years who had an ICD-10 code for cystitis listed twice in 6 months or thrice in 12 months between November 1, 2016, and December 31, 2018. Patients sought care at either urology or primary care practices within an academic medical center in Houston, Texas. Patients with functional or structural abnormalities of the genitourinary tract, signs or symptoms of pyelonephritis, or pregnancy were excluded. Antibiogram data were reported for uropathogens with ≥30 isolates, and intermediate results were considered resistant. Test metrics and Bayes’ PPV and NPV were calculated using standard formulas. Results: We included 597 visits from 232 unique patients. Most were White (63%) and female (92%), and the cohort had a median age of 58 (IQR, 41–68). Among 310 rUTI episodes with a urine culture, 189 (61%) had at least 1 uropathogen isolated, and Escherichia coli (n = 130, 66%) was most common among all 196 uropathogens. E. coli isolates had >20% resistance to 10 of 18 antibiotics (Fig. 1). E. coli resistance to ciprofloxacin was 27.9%, resistance to nitrofurantoin was 5.5%, and resistance to trimethoprim-sulfamethoxazole was 38.0%. The PPVs for predicting resistance were highest for ceftriaxone (0.86; 95% CI, 0.60–0.96), ciprofloxacin (0.84; 95% CI, 0.63–0.94), and levofloxacin (0.84; 95% CI, 0.63–0.94). NPVs of resistance were highest for gentamicin (0.97; 95% CI, 0.83–1.00), ceftriaxone (0.94; 95% CI, 0.86–0.98), and cefepime (0.94; 95% CI, 0.84–0.98), whereas NPVs for cefuroxime, ciprofloxacin, levofloxacin, and nitrofurantoin were all >0.83. Conclusions: We detected considerable antibiotic resistance among patients with rUTI to commonly prescribed antibiotics. Prior urine culture susceptibility demonstrated moderate-to-high PPVs for predicting future resistance to ceftriaxone and fluoroquinolones as well as high NPVs for several cephalosporins and fluoroquinolones, which could inform empiric prescribing choices.
Funding: This investigator-initiated research study was funded by Rebiotix, a Ferring Company.
Disclosures: None
Analysis of recurrent urinary tract infection management in women seen in outpatient settings reveals opportunities for antibiotic stewardship interventions
- Marissa A. Valentine-King, Barbara W. Trautner, Roger J. Zoorob, George Germanos, Michael Hansen, Jason L. Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 17 January 2022, e8
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objectives:
We characterized antibiotic prescribing patterns and management practices among recurrent urinary tract infection (rUTI) patients, and we identified factors associated with lack of guideline adherence to antibiotic choice, duration of treatment, and urine cultures obtained. We hypothesized that prior resistance to nitrofurantoin or trimethoprim–sulfamethoxazole (TMP-SMX), shorter intervals between rUTIs, and more frequent rUTIs would be associated with fluoroquinolone or β-lactam prescribing, or longer duration of therapy.
Methods:This study was a retrospective database study of adult women with International Classification of Diseases, Tenth Revision (ICD-10) cystitis codes meeting American Urological Association rUTI criteria at outpatient clinics within our academic medical center between 2016 and 2018. We excluded patients with ICD-10 codes indicative of complicated UTI or pyelonephritis. Generalized estimating equations were used for risk-factor analysis.
Results:Among 214 patients with 566 visits, 61.5% of prescriptions comprised first-line agents of nitrofurantoin (39.7%) and TMP-SMX (21.5%), followed by second-line choices of fluoroquinolones (27.2%) and β-lactams (11%). Most fluoroquinolone prescriptions (86.7%), TMP-SMX prescriptions (72.2%), and nitrofurantoin prescriptions (60.2%) exceeded the guideline-recommended duration. Approximately half of visits lacked a urine culture. Receiving care through urology via telephone was associated with receiving a β-lactam (adjusted odds ratio [aOR], 6.34; 95% confidence interval [CI], 2.58–15.56) or fluoroquinolone (OR, 2.28; 95% CI, 1.07–4.86). Having >2 rUTIs during the study period and seeking care from a urology practice (RR, 1.28, 95% CI, 1.15–1.44) were associated with longer antibiotic duration.
Conclusions:We found low guideline concordance for antibiotic choice, duration of therapy and cultures obtained among rUTI patients. These factors represent new targets for outpatient antibiotic stewardship interventions.
Analysis of Recurrent Urinary Tract Infection Management in Outpatient Settings Reveals Opportunities for Antibiotic Stewards
- Marissa Valentine-King, Barbara Trautner, Roger Zoorob, George Germanos, Jason Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s34
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Studies of antibiotic prescribing choice and duration have typically excluded women with recurrent UTI (rUTI), yet the Infectious Disease Society of America (IDSA) UTI treatment guidelines are applicable to recurrent and sporadic cystitis. We sought to better understand prescribing practices among uncomplicated rUTI patients in terms of choice of drug, duration of therapy, and the risk factors for receiving guideline-discordant therapy. Methods: We performed a retrospective database study by extracting electronic health record data from adults seen at academic primary care, internal medicine, or urology practices between November 2016 and December 2018. Inclusion criteria included having ≥2 or ≥3 International Classification of Diseases Tenth Edition (ICD-10) cystitis codes recorded within a 6- or 12-month period, respectively. We excluded patients with ICD-10 codes indicating any structural or functional genitourinary comorbidities, interstitial cystitis, vaginosis, compromised immune systems, or pregnancy in the prior year. Patients were also excluded if they had signs or symptoms of pyelonephritis at presentation. Results: Overall, 232 patients presented for 597 outpatient visits. Most were married (52.2%), non-Hispanic white (62.9%), and female (92.2%), with a median age of 58 years (IQR, 41–68). Only 21% of visits with an antibiotic prescribed for treatment consisted of a first-line therapy agent prescribed for the recommended duration. In terms of antibiotic choice, these agents were prescribed in 58.4% of scenarios, which primarily included nitrofurantoin (37.8%) and trimethoprim-sulfamethoxazole (TMP-SMX) (20.3%). Guideline-discordant choices of fluoroquinolones (28.8%), and β-lactams (11.2%) were the second and third most commonly prescribed drug categories, respectively. Multinomial logistic regression identified age (OR, 1.02; 95% CI, 1.002–1.04) or having a telephone visit (OR, 3.17; 95% CI, 1.54–6.52) as independent risk factors for receiving a β-lactam. The duration exceeded the 3-day guideline recommendation in 87.6% of fluoroquinolones and 73% of TMP-SMX (73%) prescriptions, and 61% of nitrofurantoin prescriptions exceeded the recommended 5-day duration. Multiple logistic regression analysis revealed that seeking care at a urology clinic (OR, 2.81; 95% CI, 1.59–5.17) served as an independent factor for therapy duration exceeding guideline recommendations. Conclusions: This retrospective study revealed shortcomings in prescribing practices in the type and duration of therapy for rUTI. rUTI as well as sporadic UTI are important targets for outpatient antibiotic stewardship interventions.
Funding: This investigator-initiated research study was funded by Rebiotix Inc, a Ferring Company.
Disclosures: None
Effectiveness of Stewardship Intervention for Urinary Tract Infections in Primary Care: A Difference in Differences Study
- Larissa Grigoryan, George Germanos, Roger Zoorob, Mohamad Sidani, Haijun Wang, Mohammad Zare, Melanie Goebel, Barbara Trautner
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s515-s516
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Adherence to 2011 Infectious Diseases Society of America (IDSA) guidelines for urinary tract infections (UTIs) remains low in primary care. Fluoroquinolones are commonly prescribed to treat uncomplicated cystitis, and most antibiotic prescriptions have durations that exceed current recommendations. We performed a difference-in-differences study to assess the effectiveness of a stewardship intervention in a family medicine clinic at an academic outpatient center from August 2016 to March 2019. During our intervention period, the FDA released 2 additional warnings about the side effects of fluoroquinolones. Methods: The study had 2 sites (intervention and comparison) and 3 periods: baseline, before the intervention, and the intervention. During the first 2 years, we obtained baseline data and performed interviews (preintervention period) exploring provider prescribing decisions for cystitis at both sites. During the intervention period at the intervention site only, we presented an educational lecture including an overview of the IDSA guidelines, definitions for various UTI syndromes and actual clinical examples, and instruction on use of a decision aid. During the audit and feedback phase, providers were contacted once per month in person or by phone to provide follow-up on whether their treatment decision adhered to the IDSA guidelines. We performed a log-binomial regression analysis of the primary outcome, adherence to the IDSA guidelines for management of uncomplicated cystitis, both to antibiotic choice and duration of therapy. Results: We performed 156 audit-and-feedback sessions with 13 providers during the intervention period (March 2018–2019). Patients in both sites were similar in terms of age and Charlson comorbidity index. Adherence to the guidelines for antibiotic choice and duration increased in the intervention period at both sites (Fig. 1). The treatment of cystitis in the intervention period of the intervention site was 11.5 times (95% CI, 6.1–21.6) as likely to be guideline-adherent as the treatment in the baseline period of the comparison site (Fig. 2). Conclusions: Adherence to IDSA guidelines for the choice of antibiotic and duration increased in both intervention and comparison sites. Even though the intervention site started with higher compliance, improvement was also greater in the intervention site. FDA warnings about the side effects of fluoroquinolones released during the intervention period may have contributed to the avoidance of fluoroquinolones at both sites. Our intervention was effective at improving antibiotic choice and duration, so our future plans include incorporating our decision-support algorithm into the electronic medical record.
Funding: This study was supported by Zambon Pharmaceuticals.
Disclosures: None




