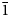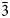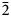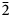3 results
Yarzhemskiite, K[B5O7(OH)2]⋅H2O, a new mineral from the Chelkar salt dome, Western Kazakhstan
- Igor V. Pekov, Natalia V. Zubkova, Oksana V. Korotchenkova, Ilya I. Chaikovskiy, Vasiliy O. Yapaskurt, Nikita V. Chukanov, Dmitry I. Belakovskiy, Inna S. Lykova, Sergey N. Britvin, Dmitry Yu. Pushcharovsky
-
- Journal:
- Mineralogical Magazine / Volume 84 / Issue 2 / April 2020
- Published online by Cambridge University Press:
- 13 December 2019, pp. 335-342
-
- Article
- Export citation
-
The new mineral yarzhemskiite, K[B5O7(OH)2]⋅H2O, was found in a halite–sylvite evaporite rock at the Chelkar salt dome, Western Kazakhstan Region, Kazakhstan. It is also associated with carnallite, polyhalite, gypsum, strontioginorite, satimolite and quartz. Yarzhemskiite occurs as separate thick tabular, short prismatic or equant crystals up to 0.5 mm × 0.7 mm × 1 mm and grains having irregular outlines up to 1 mm × 1.5 mm × 2 mm. The mineral is transparent, colourless, with vitreous lustre. It is brittle, the Mohs’ hardness is ca 2½. Cleavage is perfect on {100}. Dmeas is 2.13(1) and Dcalc is 2.112 g cm–3. Yarzhemskiite is optically biaxial (+), α = 1.484(2), β = 1.508(2), γ = 1.546(2), 2Vmeas = 75(10)° and 2Vcalc = 80°. Chemical composition (wt.%, electron microprobe, H2O was calculated by stoichiometry) is: Na2O 0.01, K2O 17.84, CaO 0.07, B2O3 67.21, H2Ocalc 13.91, total 99.04. The empirical formula based on 10 O atoms per formula unit is K0.98B5.005O7(OH)2⋅H2O. Yarzhemskiite is monoclinic, P21/c, a = 9.47340(18), b = 7.52030(16), c = 11.4205(2) Å, β = 97.3002(17)°, V = 807.03(3) Å3 and Z = 4. The strongest reflections of the powder XRD pattern [d,Å(I,%)(hkl)] are: 9.39(86)(100), 4.696(41)(200), 3.296(18)(
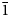 $\bar{1}$13), 3.130(19)(022, 300), 2.935(42)(220), 2.898(100)(
$\bar{1}$13), 3.130(19)(022, 300), 2.935(42)(220), 2.898(100)(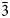 $\bar{3}$02,
$\bar{3}$02, 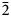 $\bar{2}$21, 310), 2.832(56)(004) and 1.867(18)(
$\bar{2}$21, 310), 2.832(56)(004) and 1.867(18)(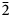 $\bar{2}$25). The crystal structure was solved based on single-crystal X-ray diffraction data, R1 = 3.36%. The structure contains infinite chains built by boron-centred polyhedra. The basic structural unit of the chain is a double ring B5O7(OH)2 consisting of one BO4 tetrahedron and four BO3 triangles. K+ cations centre ten-fold polyhedra which form, together with the borate chains [B5O7(OH)2]–∝, layers linked with each other only via H bonds. The mineral is named in honour of the Russian geologist, petrologist and mineralogist Yakov Yakovlevich Yarzhemskii (1901–?), a specialist in petrology of evaporite rocks and mineralogy and genesis of boron deposits related to evaporites.
$\bar{2}$25). The crystal structure was solved based on single-crystal X-ray diffraction data, R1 = 3.36%. The structure contains infinite chains built by boron-centred polyhedra. The basic structural unit of the chain is a double ring B5O7(OH)2 consisting of one BO4 tetrahedron and four BO3 triangles. K+ cations centre ten-fold polyhedra which form, together with the borate chains [B5O7(OH)2]–∝, layers linked with each other only via H bonds. The mineral is named in honour of the Russian geologist, petrologist and mineralogist Yakov Yakovlevich Yarzhemskii (1901–?), a specialist in petrology of evaporite rocks and mineralogy and genesis of boron deposits related to evaporites.
Crystal chemistry of halurgite, Mg4[B8O13(OH)2]2·7H2O, a microporous heterophylloborate mineral
- Igor V. Pekov, Natalia V. Zubkova, Dmitry A. Ksenofontov, Nikita V. Chukanov, Oksana V. Korotchenkova, Ilya I. Chaikovskiy, Vasiliy O. Yapaskurt, Sergey N. Britvin, Dmitry Yu. Pushcharovsky
-
- Journal:
- Mineralogical Magazine / Volume 83 / Issue 5 / October 2019
- Published online by Cambridge University Press:
- 31 May 2019, pp. 723-732
-
- Article
- Export citation
-
Three samples of halurgite were re-examined: two from the Chelkar salt dome in the North Caspian Region, Western Kazakhstan (the type locality and including the type specimen), and one from a new locality in the Satimola salt dome located in the same region. The crystal structure of halurgite has been solved for the first time on the specimen from Chelkar with the empirical formula Mg3.94[B8.03O13.03(OH)1.97]2·7.16H2O; refinement began with single-crystal X-ray diffraction data and was subsequently refined on a powder sample using the Rietveld method (Rp = 0.0232, Rwp = 0.0354 and Robs = 0.0558). The idealised crystal chemical formula of halurgite is Mg4[B8O13(OH)2]2·7H2O. The mineral is monoclinic, P2/c, a = 13.201(2), b = 7.5622(10), c = 13.185(2) Å, β = 91.834(14)°, V = 1315.6(4) Å3 and Z = 2. The crystal structure is unique. Eight B polyhedra form a fundamental building block [B8O16(OH)2], which is a six-membered borate ring (built by two pairs of B tetrahedra and two B triangles) with two additional triangular BO2(OH) groups. Each [B8O16(OH)2] ring is linked to six adjacent analogous rings to form a [B8O13(OH)2]∞ layer. These layers are connected via MgO6 and Mg(OH)2(H2O)4 octahedra into a microporous heteropolyhedral pseudo-framework. The crystal structure of halurgite can also be described in terms of an approach developed for heterophyllosilicates containing three-layer HOH modules, where HOH refers to an octahedral layer O sandwiched between two heteropolyhedral layers H. In halurgite the HOH module consists of two heteropolyhedral (BO3 triangles + BO4 tetrahedra) borate H layers [B8O13(OH)2]∞ and a central interrupted O layer composed of MgO6 octahedra, whereas a more voluminous Mg(OH)2(H2O)4 octahedral complex and additional H2O molecules are located between HOH modules. Halurgite and four related synthetic H-free borates M2Cd3B16O28 and M2Ca3B16O28 (M = Rb or Cs) can be considered microporous heterophylloborates.
Redefinition of satimolite
- Igor V. Pekov, Natalia V. Zubkova, Dmitry A. Ksenofontov, Nikita V. Chukanov, Vasiliy O. Yapaskurt, Oksana V. Korotchenkova, Ilya I. Chaikovskiy, Vladimir M. Bocharov, Sergey N. Britvin, Dmitry Yu. Pushcharovsky
-
- Journal:
- Mineralogical Magazine / Volume 82 / Issue 5 / October 2018
- Published online by Cambridge University Press:
- 28 February 2018, pp. 1033-1047
-
- Article
- Export citation
-
The borate mineral satimolite, which was first described in 1969 and remained poorly-studied until now, has been re-investigated (electron microprobe analysis, single-crystal and powder X-ray diffraction studies, crystal-structure determination, infrared spectroscopy) and redefined based on the novel data obtained for the holotype material from the Satimola salt dome and a recently found sample from the Chelkar salt dome, both in North Caspian Region, Western Kazakhstan. The revised idealized formula of satimolite is KNa2(Al5Mg2)[B12O18(OH)12](OH)6Cl4·4H2O (Z = 3). The mineral is trigonal, space group R
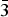 $\bar{3}$m, unit-cell parameters are: a = 15.1431(8), c = 14.4558(14) Å and V = 2870.8(4) Å3 (Satimola) and a = 15.1406(4), c = 14.3794(9) Å and V = 2854.7(2) Å3 (Chelkar). The crystal system and unit-cell parameters are quite different from those reported previously. The crystal structure of the sample from Chelkar was solved based on single-crystal data (direct methods, R = 0.0814) and the structure of the holotype from Satimola was refined on a powder sample by the Rietveld method (Rp = 0.0563, Rwp = 0.0761 and Rall = 0.0667). The structure of satimolite is unique for minerals. It contains 12-membered borate rings [B12O18(OH)12] in which BO3 triangles alternate with BO2(OH)2 tetrahedra sharing common vertices, and octahedral clusters [M7O6(OH)18] with M = Al5Mg2 in the ideal case, with sharing of corners between rings and clusters to form a three-dimensional heteropolyhedral framework. Each borate ring is connected with six octahedral clusters: three under the ring and three over the ring. Large ellipsoidal cages in the framework host Na and K cations, Cl anions and H2O molecules.
$\bar{3}$m, unit-cell parameters are: a = 15.1431(8), c = 14.4558(14) Å and V = 2870.8(4) Å3 (Satimola) and a = 15.1406(4), c = 14.3794(9) Å and V = 2854.7(2) Å3 (Chelkar). The crystal system and unit-cell parameters are quite different from those reported previously. The crystal structure of the sample from Chelkar was solved based on single-crystal data (direct methods, R = 0.0814) and the structure of the holotype from Satimola was refined on a powder sample by the Rietveld method (Rp = 0.0563, Rwp = 0.0761 and Rall = 0.0667). The structure of satimolite is unique for minerals. It contains 12-membered borate rings [B12O18(OH)12] in which BO3 triangles alternate with BO2(OH)2 tetrahedra sharing common vertices, and octahedral clusters [M7O6(OH)18] with M = Al5Mg2 in the ideal case, with sharing of corners between rings and clusters to form a three-dimensional heteropolyhedral framework. Each borate ring is connected with six octahedral clusters: three under the ring and three over the ring. Large ellipsoidal cages in the framework host Na and K cations, Cl anions and H2O molecules.