3 results
Using Best-Worst Scaling to assess preferences for online psychological interventions to decrease cannabis use in young adults with psychosis
- O. Tatar, A. Abdel-Baki, H. Bakouni, A. Bahremand, T. Lecomte, J. Côté, D. Crockford, S. L’Heureux, C. Ouellet-Plamondon, M.-A. Roy, P. G. Tibbo, M. Villeneuve, D. Jutras-Aswad
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S528-S529
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
In individuals with first episode psychosis (FEP) and cannabis use disorder (CUD), reducing cannabis use is associated with improved clinical outcomes. Access to evidence-based psychological interventions to decrease cannabis use in FEP clinics is highly variable; E-mental health interventions may help to address this gap. Development of E-interventions for CUD in individuals with FEP is in its incipient phases.
ObjectivesTo assess preferences for online psychological interventions aiming at decreasing or stopping cannabis use in young adults with psychosis and CUD.
MethodsIndividuals aged 18 to 35 years old with psychosis and CUD were recruited from seven FEP intervention programs in Canada and responded to an electronic survey between January 2020-July 2022. We used the Case 2 Best Worst Scaling methodology that is grounded in the trade-off utility concept to collect and analyse data. Participants selected the best or worst option for each of the nine questions corresponding to three distinct domains. For each domain we used conditional logistic regression and marginal models (i.e., three models in total) to estimate preferences for attributes (e.g., duration, frequency of online intervention sessions) and attribute levels (e.g., 15 minutes, every day).
ResultsParticipants (N=104) showed higher preferences for the following attributes: duration of online sessions; mode of receiving the intervention; method of feedback delivery and the frequency of feedback from clinicians (Table 1). Attribute-level analyses showed higher preferences for participating once a week in short (15 minutes) online interventions (Figure 1). Participants valued the autonomy offered by online interventions which aligns with their preference for completing the intervention outside the clinic and only require assistance once a week (Figure 2). Participants’ preferences were higher for receiving feedback related to cannabis consumption both from the application and clinicians at a frequency of once a week from clinicians (Figure 3).
Table 1. Preferences for Attributes. Results of conditional logistic regression Attributes Domains OR 95% CI for OR Duration session A 1.62 1.45; 1.82 Frequency sessions 0.98 0.87; 1.09 Duration intervention ref Preferred mode of receiving the intervention B 1.63 1.46; 1.83 Preferred location for participating 1.07 0.96; 1.20 Frequency of assistance from the clinician ref Preference for the feedback delivery method C 1.21 1.08; 1.36 Frequency of feedback from the treating clinician 1.14 1.02; 1.28 Frequency of feedback from the application ref Note: In boldface significant odds ratios (OR) and confidence intervals (CI)
Image:
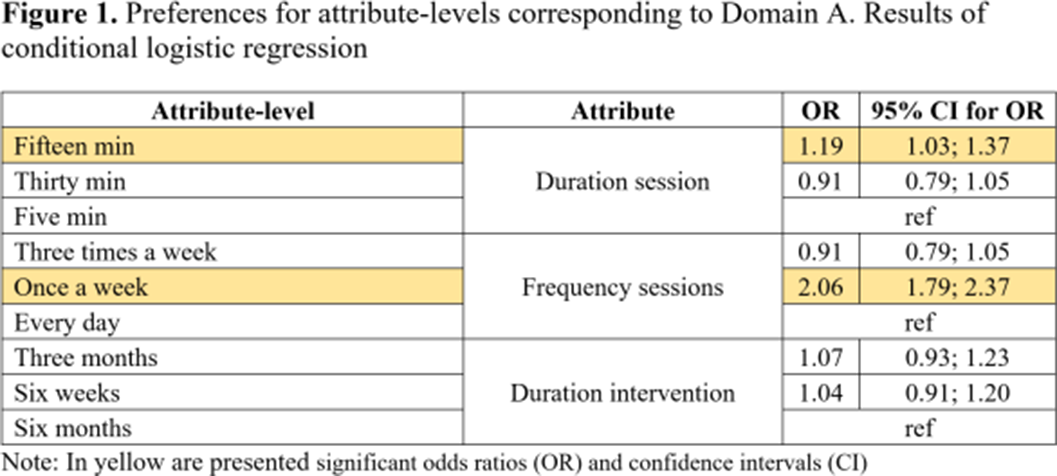
Image 2:
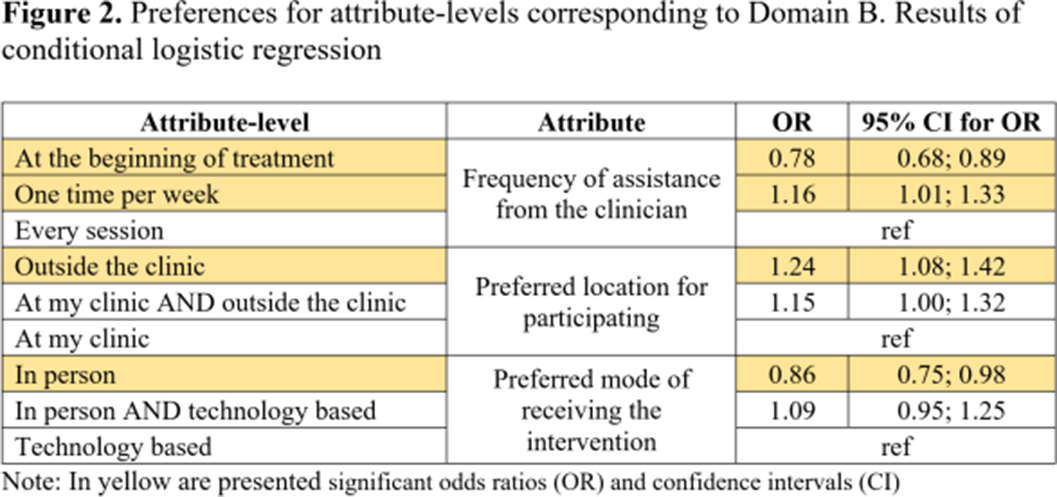
Image 3:
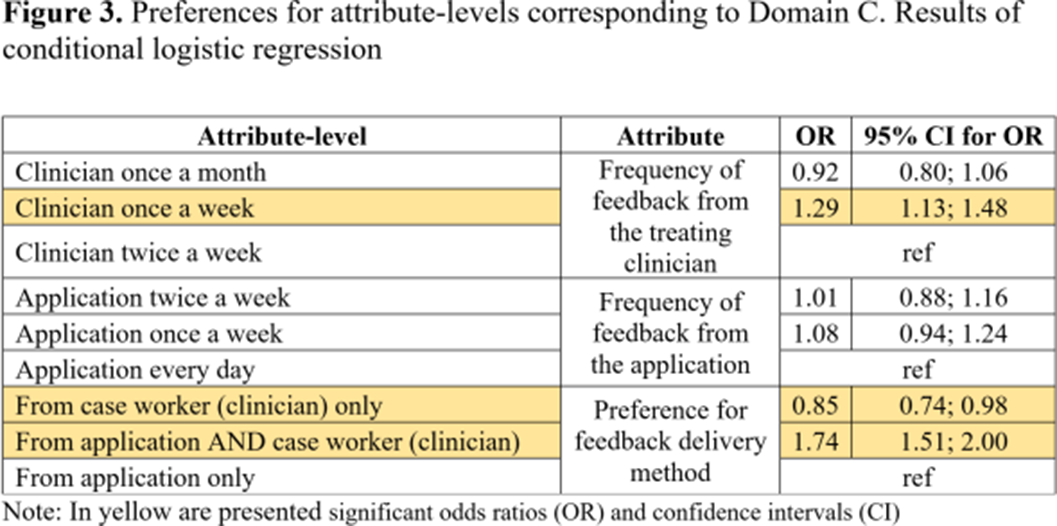 Conclusions
ConclusionsUsing advanced methodologies to assess preferences, our results can inform the development of highly acceptable E-Mental health interventions for decreasing cannabis use in individuals with CUD and FEP.
Disclosure of InterestNone Declared
A National Spinal Muscular Atrophy Registry for Real-World Evidence
- Victoria L. Hodgkinson, Maryam Oskoui, Joshua Lounsberry, Saïd M’Dahoma, Emily Butler, Craig Campbell, Alex MacKenzie, Hugh J. McMillan, Louise Simard, Jiri Vajsar, Bernard Brais, Kristine M. Chapman, Nicolas Chrestian, Meghan Crone, Peter Dobrowolski, Susan Dojeiji, James J. Dowling, Nicolas Dupré, Angela Genge, Hernan Gonorazky, Simona Hasal, Aaron Izenberg, Wendy Johnston, Edward Leung, Hanns Lochmüller, Jean K. Mah, Alier Marerro, Rami Massie, Laura McAdam, Anna McCormick, Michel Melanson, Michelle M. Mezei, Cam-Tu E. Nguyen, Colleen O’Connell, Erin K. O’Ferrall, Gerald Pfeffer, Cecile Phan, Stephanie Plamondon, Chantal Poulin, Xavier Rodrigue, Kerri L. Schellenberg, Kathy Selby, Jordan Sheriko, Christen Shoesmith, Garth Smith, Monique Taillon, Sean Taylor, Jodi Warman Chardon, Scott Worley, Lawrence Korngut
-
- Journal:
- Canadian Journal of Neurological Sciences / Volume 47 / Issue 6 / November 2020
- Published online by Cambridge University Press:
- 04 June 2020, pp. 810-815
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background:
Spinal muscular atrophy (SMA) is a devastating rare disease that affects individuals regardless of ethnicity, gender, and age. The first-approved disease-modifying therapy for SMA, nusinursen, was approved by Health Canada, as well as by American and European regulatory agencies following positive clinical trial outcomes. The trials were conducted in a narrow pediatric population defined by age, severity, and genotype. Broad approval of therapy necessitates close follow-up of potential rare adverse events and effectiveness in the larger real-world population.
Methods:The Canadian Neuromuscular Disease Registry (CNDR) undertook an iterative multi-stakeholder process to expand the existing SMA dataset to capture items relevant to patient outcomes in a post-marketing environment. The CNDR SMA expanded registry is a longitudinal, prospective, observational study of patients with SMA in Canada designed to evaluate the safety and effectiveness of novel therapies and provide practical information unattainable in trials.
Results:The consensus expanded dataset includes items that address therapy effectiveness and safety and is collected in a multicenter, prospective, observational study, including SMA patients regardless of therapeutic status. The expanded dataset is aligned with global datasets to facilitate collaboration. Additionally, consensus dataset development aimed to standardize appropriate outcome measures across the network and broader Canadian community. Prospective outcome studies, data use, and analyses are independent of the funding partner.
Conclusion:Prospective outcome data collected will provide results on safety and effectiveness in a post-therapy approval era. These data are essential to inform improvements in care and access to therapy for all SMA patients.
Differential illness intrusiveness associated with sleep-promoting medications
- GM Devins, M Flanigan, JAE Fleming, R Morehouse, A Moscovitch, J Plamondon, L Reinish, CM Shapiro
-
- Journal:
- European Psychiatry / Volume 10 / Issue S3 / 1995
- Published online by Cambridge University Press:
- 16 April 2020, pp. 153s-159s
-
- Article
- Export citation
-
Differences in daytime sleepiness, lifestyle disruptions, and emotional distress were compared across nine groups taking sleep-promoting substances. Groups included individuals taking zopiclone (n = 274), amitriptyline (n = 107), lorazepam (n = 258), oxazepam (n= 141), diphenhydramine HCl (n = 99), triazolam(n = 137), long acting benzodiazepines (n = 120), temazepam (n = 176), and miscellaneous other medications (n = 286). Data were gathered by self-report, using standardized instruments in a mail-back questionnaire procedure. Respondents included the first 1,598 participants enrolled in a Canadian multicentre project, including six sites attached to academic psychiatric units. Results indicated that quality of life effects differed across groups in both daytime sleepiness and lifestyle disruptions (illness intrusiveness), but not in depressive symptoms. Daytime sleepiness was significantly higher among people taking diphenhydramine HCl as compared to temazepam, zopiclone, lorazepam, and oxazepam. Illness intrusiveness was significantly higher among patients taking amitriptyline as compared to those taking triazolam, oxazepam, long-acting benzodiazepines, and zopiclone. It may be useful to inform patients of differences in psychosocial outcomes when prescribing hypnotic medications.






