17 results
Identifying the relationship between hospital rurality and antibiotic overuse
- Hannah Hardin, Valerie Vaughn, Andrea White, Jennifer Horowitz, Elizabeth McLaughlin, Julia Szymczak, Lindsay Petty, Anurag Malani, Scott Flanders, Tejal Gandhi
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s34-s35
-
- Article
-
- You have access Access
- Open access
- Export citation
-
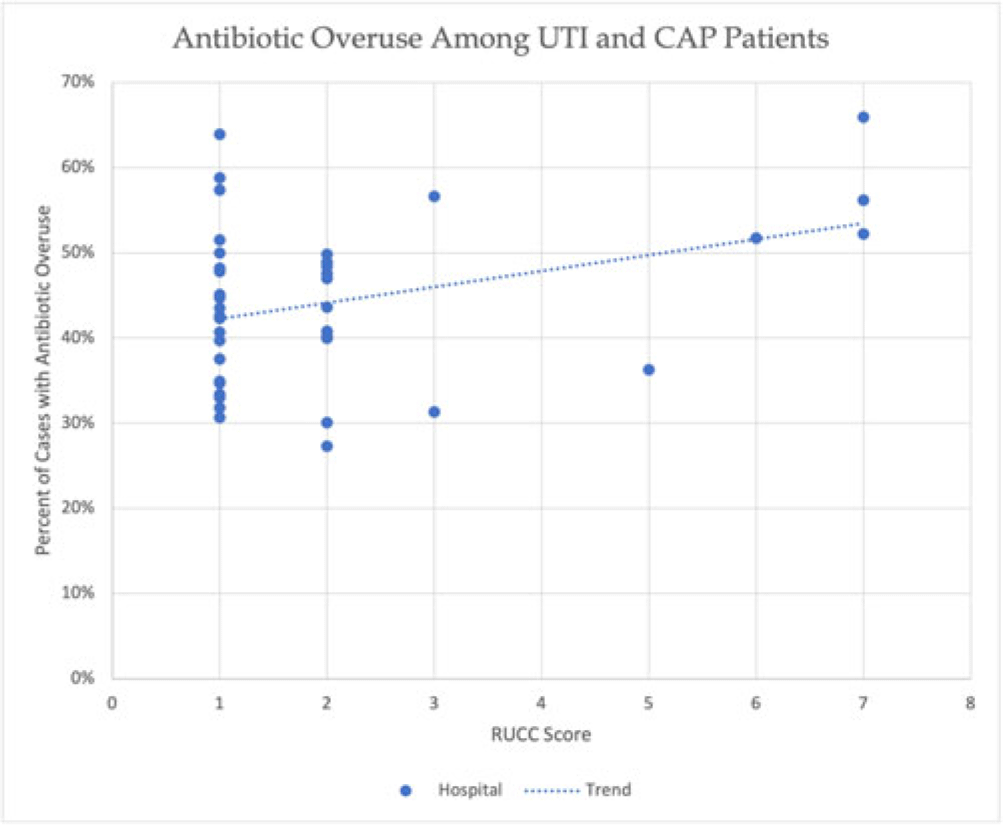
Background: Antibiotic overuse and the resulting patient outcomes span all hospitals. However, although antibiotic stewardship can improve antibiotic use, effective stewardship programs require expertise and an infrastructure that are not present in all hospitals. Rural hospitals have less access to resources, infectious disease expertise, and participation in academic research. Thus, we compared antibiotic overuse at discharge between rural and nonrural hospitals for patients diagnosed with community-associated pneumonia (CAP) or urinary tract infection (UTI)—the 2 most common hospital infections. Methods: To determine whether antibiotic overuse at discharge was higher among rural versus nonrural hospitals, we analyzed data from a 41-hospital prospective cohort of patients treated for CAP or UTI between July 1, 2017, and July 30, 2019, in Michigan. Antibiotic overuse was defined as treatment that was unnecessary (ie, patient did not have an infection), excessive (ie, duration >4 days for CAP), or included suboptimal fluoroquinolone use (ie, safer alternative available). Overuse was determined based on patient risk factors, symptoms, allergies, diagnostic results, and time to stability. Hospital rurality was defined using the Rural–Urban Continuum Codes (RUCC) score. We defined rural as a score ≥4 and very rural as a score of 7–9. We used t tests to compare the mean percentage of patients with antibiotic overuse at discharge between nonrural and rural (and very rural) hospitals. Results: Across 41 hospitals, we included 23,449 patients with CAP or UTI. There were 5 rural (and 3 very rural) hospitals with 2,039 (and 1,082) patients. Antibiotic overuse at discharge was present in 43.1% of patient cases in nonrural hospitals, 52.5% in rural hospitals (P = .04 vs nonrural) and 58.1% in very rural hospitals (P = .007 vs nonrural). Compared to nonrural hospitals, the mean percentage of cases with antibiotic overuse at discharge in rural hospitals was 9.4% higher (15.1% higher in very rural hospitals). Results were similar in a subgroup analysis of only patients with UTI (47.0% in rural vs 37.5% in nonrural, mean difference, 9.5%; P = .03) but were not statistically significant in patients with CAP (53.8% vs 48.0%, respectively; mean difference, 5.8%; P = 0.23). Conclusions: In this retrospective study, rural hospitals—especially very rural hospitals, had higher rates of antibiotic overuse at discharge than nonrural hospitals. Our findings suggest that antibiotic stewardship interventions tailored toward the unique differences in infrastructure, resources, and needs of rural hospitals are essential to community health.
Disclosures: None
Three-day antibiotic duration in patients with pneumonia: A sixty-eight–hospital cohort
- Valerie Vaughn, Lindsay Petty, David Ratz, Elizabeth McLaughlin, Tawny Czilok, Jennifer Horowitz, Anurag Malani, Danielle Osterholzer, Scott Flanders, Tejal Gandhi
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s22
-
- Article
-
- You have access Access
- Open access
- Export citation
-
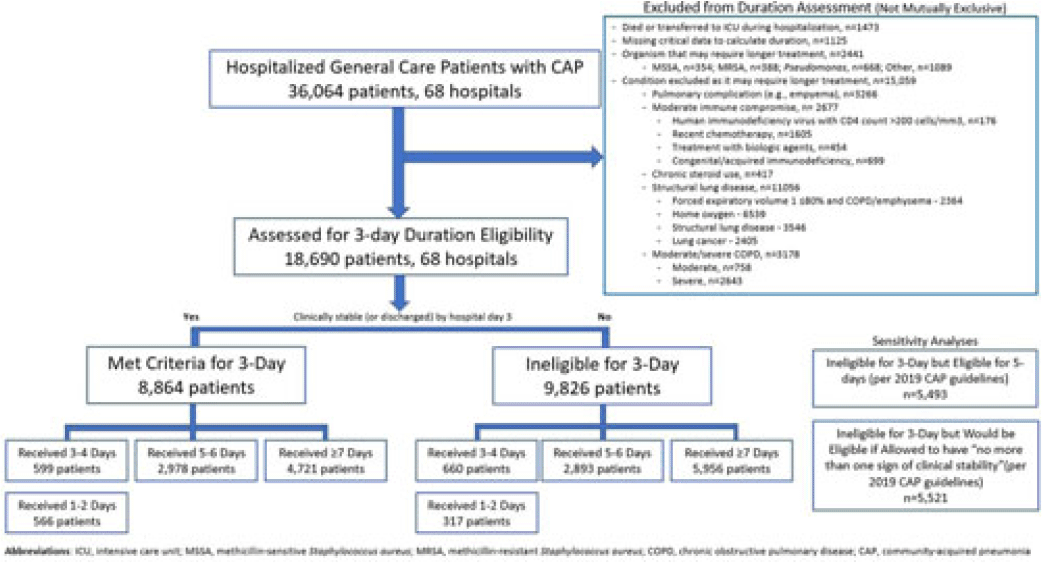
Background: Since 2019, community-acquired pneumonia (CAP) guidelines have recommended hospitalized patients be treated until clinical “stability and for no less than 5 days.” However, randomized trials have reported that, in patients who stabilize by hospital day 3, very short antibiotic durations (eg, 3 days) are noninferior to longer durations. How these trial results relate to real-world practice is unknown. Methods: Using a 68-hospital cohort study of hospitalized, general-care adults with CAP, we aimed to (1) quantify the percentage of patients who—according to trial criteria—qualify for a 3-day antibiotic duration, (2) quantify the percentage who actually received a 3-day duration, and (3) assess 30-day outcomes. Patients were considered to have CAP if they had a pneumonia discharge diagnosis and met clinical criteria for CAP. Patients with concomitant infections (including COVID-19), admission to intensive care, or severe immunocompromise were not included. Results: Between February 23, 2017, and August 3, 2022, 36,064 patients with CAP were included. Of those, 48.2% (9,826 of 36,064) were excluded due to a condition or organism ineligible for the 3-day treatment (Fig. 1). Of the 18,690 patients remaining, 52.6% (9,826) were unstable on day 3 and thus were ineligible for the 3-day treatment. Therefore, of all 36,064 patients, only 8,864 (24.6%) would be eligible under trial criteria for a 3-day treatment. Notably, 5,493 (55.9%) of 9,826 patients unstable on day 3 would be eligible for 5 days of treatment under national guidelines. In practice, use of 3–4-day treatment was rare, occurring in 599 (6.8%) of 8,864 patients eligible for a 3-day treatment versus 660 (6.7%) of 9,826 patients unstable on day 3 (P = .945). Use of 3–4-day treatment increased over time and comorbidities that could mimic CAP or a negative procalcitonin were more common in patients who received a 3–4-day treatment whereas specific symptoms of CAP were less common (Fig. 2). After adjustments, patients eligible for a 3-day duration who received a 3–4 day treatment versus a ≥5-day treatment had higher 30-day mortality (aOR, 1.87; 95% CI, 1.32–2.64) and readmission (aOR, 1.35; 95% CI, 1.17–1.56). Conclusions: Across 68 hospitals, <25% of patients hospitalized with CAP would be eligible for a 3-day antibiotic treatment. Though increasing over time, there was little use of 3–4-day treatments and, when prescribed, outcomes were worse, potentially due to CAP misdiagnosis. Given the small number of patients eligible for 3-day treatment, and the potential harm with too-short durations, it may be prudent to focus on increasing the use of 5-day treatments.
Disclosures: None
Prevalence of and risk factors for bacteremic UTIs in hospitalized adults without definitive signs or symptoms of UTI
- Sonali Advani, David Ratz, Jennifer Horowitz, Lindsay Petty, Kenneth Schmader, Tawny Czilok, Anurag Malani, Tejal Gandhi, Scott Flanders, Valerie Vaughn
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s27-s28
-
- Article
-
- You have access Access
- Open access
- Export citation
-
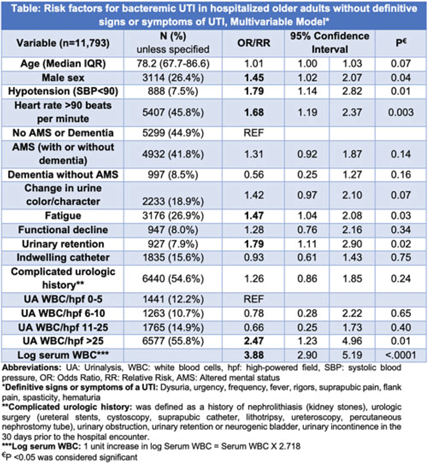
Background: IDSA guidelines recommend withholding treatment in patients with asymptomatic bacteriuria in the absence of systemic signs of infection. However, some patients with bacteriuria may not be able to express symptoms either due to presence of indwelling catheter, underlying complicated urologic anatomy, dementia, or altered mental status (AMS). Clinicians frequently treat bacteriuria in this population with antimicrobial therapy due to concern for sepsis. To determine treatment need, we aimed to review prevalence and risk factors for bacteremic urinary tract infection (UTI) in a cohort of hospitalized inpatients without definitive signs and symptoms of a UTI. Methods: This retrospective cohort study of inpatients with a positive urine culture who presented without definitive signs or symptoms of a UTI was conducted between July 1, 2017, and June 30, 2022, in 68 academic and community hospitals (Michigan Hospital Medicine Safety Consortium). Signs and symptoms were obtained from medical record review 3 days before and after urine-culture collection. Bacteremic UTI was defined as any positive blood culture growing at least 1 organism matching the urine culture. Risk factors for bacteremic UTI were assessed using multivariable logistic regression models with results expressed as odds ratios (ORs) for dichotomous variables and relative risks (RRs) for continuous variables. Results: Of 11,793 patients meeting study criteria, 73.6% were female with a median age of 78.2 years. Overall, 41.8% had AMS, 33.8% had dementia, 15.6% had an indwelling urinary catheter, and 54.6% had complicated urologic history (eg, urologic surgery). Of these, 166 patients (1.4%) developed bacteremic UTI. On adjusted analysis, male sex, hypotension, heart rate >90, urinary retention, fatigue, log of serum leukocytosis [1 log increase in serum WBC = 2.718 × serum white blood cell count (WBC)], and pyuria with >25 WBC per high-powered field (WBC/hpf) on urinalysis were associated with bacteremic UTI (Table). Older age, presence of an indwelling catheter, complicated urologic history, functional decline, AMS, dementia, and change in urine were not associated with higher odds for bacteremic UTI (Table). Of patients with AMS and no definitive signs or symptoms of a UTI, only 89 (1.8%) of 4,932 developed a bacteremic UTI. Conclusions: Bacteremic UTI is relatively rare in hospitalized inpatients presenting with bacteriuria without symptoms of UTI. Predictors of bacteremic UTI included male sex, hypotension, tachycardia, urinary retention, fatigue, serum leukocytosis, and higher levels of pyuria (>25 WBC/hpf) on urinalysis. Our findings provide stewards a framework to risk stratify inpatients of older age who present with positive urine cultures but without (or are unable to express) signs or symptoms of UTI.
Disclosures: None
Risk Factors and outcomes associated with inappropriate empiric broad-spectrum antibiotic use in hospitalized patients with community-acquired pneumonia
- Tejal Gandhi, Lindsay Petty, Valerie Vaughn, Anurag Malani, David Ratz, Tawny Czilok, Jennifer Horowitz, Elizabeth McLaughlin, Lisa Dumkow, Stephanie Burdick, Danielle Osterholzer, Mariam Younas, Steven Bernstein, Scott Flanders
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s31-s32
-
- Article
-
- You have access Access
- Open access
- Export citation
-
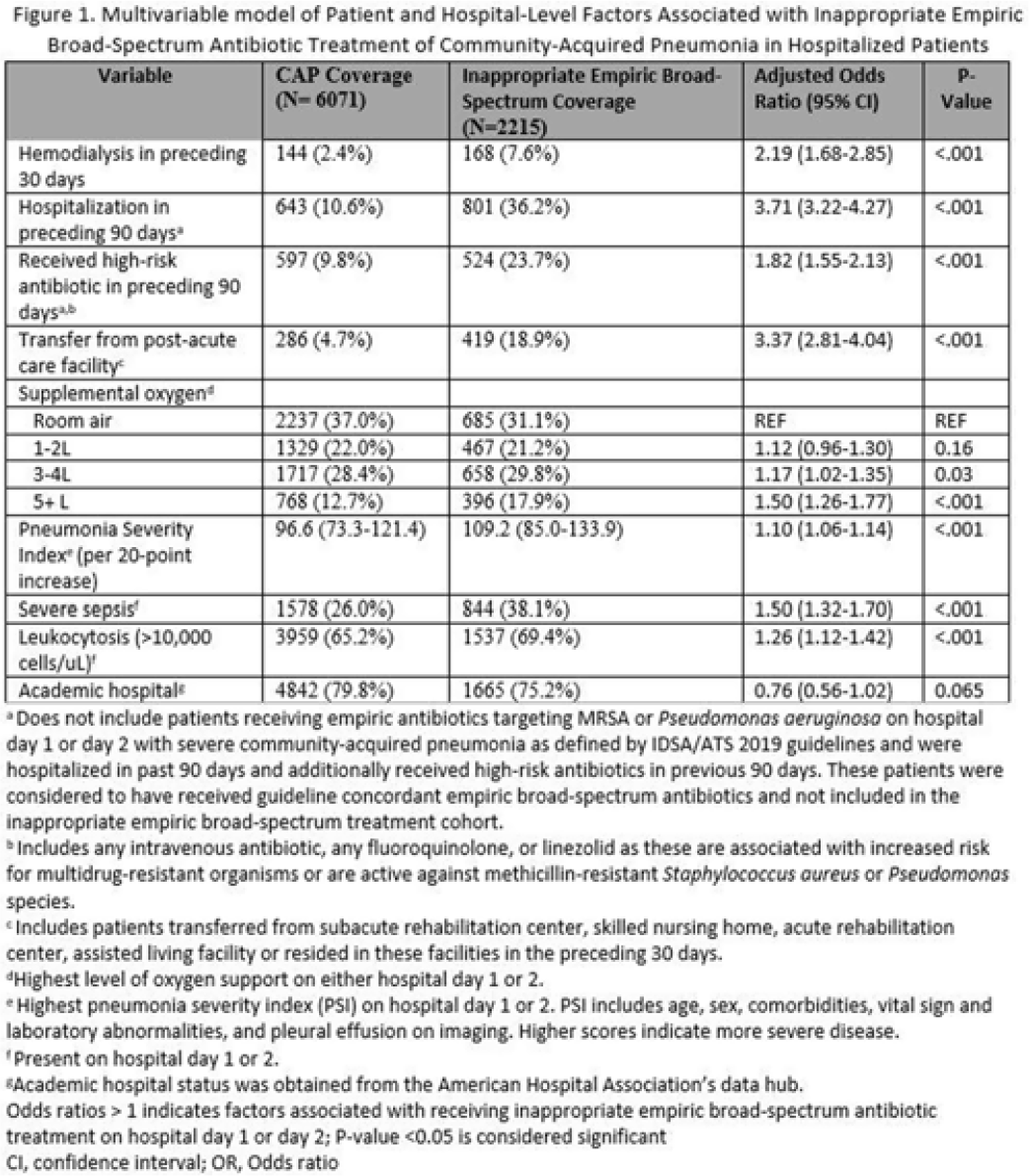
Background: Inappropriate broad-spectrum antibiotic use targeting methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa can result in increased adverse events, antibiotic resistance, and Clostridioides difficile infection. In 2019, revised ATS/IDSA community-acquired pneumonia (CAP) guidelines removed healthcare-associated pneumonia (HCAP) as a clinical entity and modified patient factors warranting empiric broad-spectrum antibiotic (BSA) use. As a result, most patients hospitalized with CAP should receive empiric antibiotics targeting standard CAP pathogens. Based on revised guidelines, we evaluated predictors and outcomes associated with inappropriate BSA use among hospitalized patients with CAP. Methods: Between November 2019 and July 2022, trained abstractors collected data on non-ICU adult medical patients admitted with CAP at 67 Michigan hospitals who received either an inappropriate empiric BSA on hospital day 1 or 2 or a standard CAP regimen. Inappropriate empiric BSA use was defined as use of an anti-MRSA or anti-pseudomonal antibiotic in a patient eligible for standard CAP coverage per IDSA guidelines. Patients with immune compromise, moderate or severe chronic obstructive pulmonary disease (COPD), pulmonary complication, or guideline-concordant treatment with BSA were excluded. Data collected included comorbidities, antibiotic use and hospitalizations in the preceding 90 days, cultures in the preceding year, signs or symptoms of pneumonia, hospital characteristics, and 30-day postdischarge patient outcomes. Data were collected through chart review and patient phone calls. Predictors of inappropriate empiric BSA were evaluated using logistic general estimating equation (GEE) models, accounting for hospital-level clustering. We assessed the effect of inappropriate empiric BSA (vs standard CAP therapy) on 30-day patient outcomes using logistic GEE models controlling for predictors associated with the outcome and probability of treatment. Results: Of 8,286 included patients with CAP, 2,215 (26.7%) were empirically treated with inappropriate BSA. The median BSA treatment was 3 days (IQR, 2.5). After adjustments, factors associated with inappropriate empiric BSA treatment included hospitalization or treatment with high-risk antibiotics in preceding 90 days, transfer from a postacute care facility, hemodialysis, support with ≥3 L supplemental oxygen, severe sepsis, leukocytosis, and higher pneumonia severity index (Fig. 1). After adjustments, patients with inappropriate empiric BSA treatment had higher readmissions 30 days after discharge, more transfers to the intensive care unit, more antibiotic-associated adverse events, and longer hospitalizations (Fig. 2). Conclusions: Patients hospitalized with CAP often received inappropriate BSA as empiric coverage, and this inappropriate antibiotic selection was associated with worse patient outcomes. To improve patient outcomes, stewardship efforts should focus on reducing inappropriate BSA use in patients hospitalized for CAP with historic HCAP risk factors or severe CAP without other guideline-directed indications for BSA.
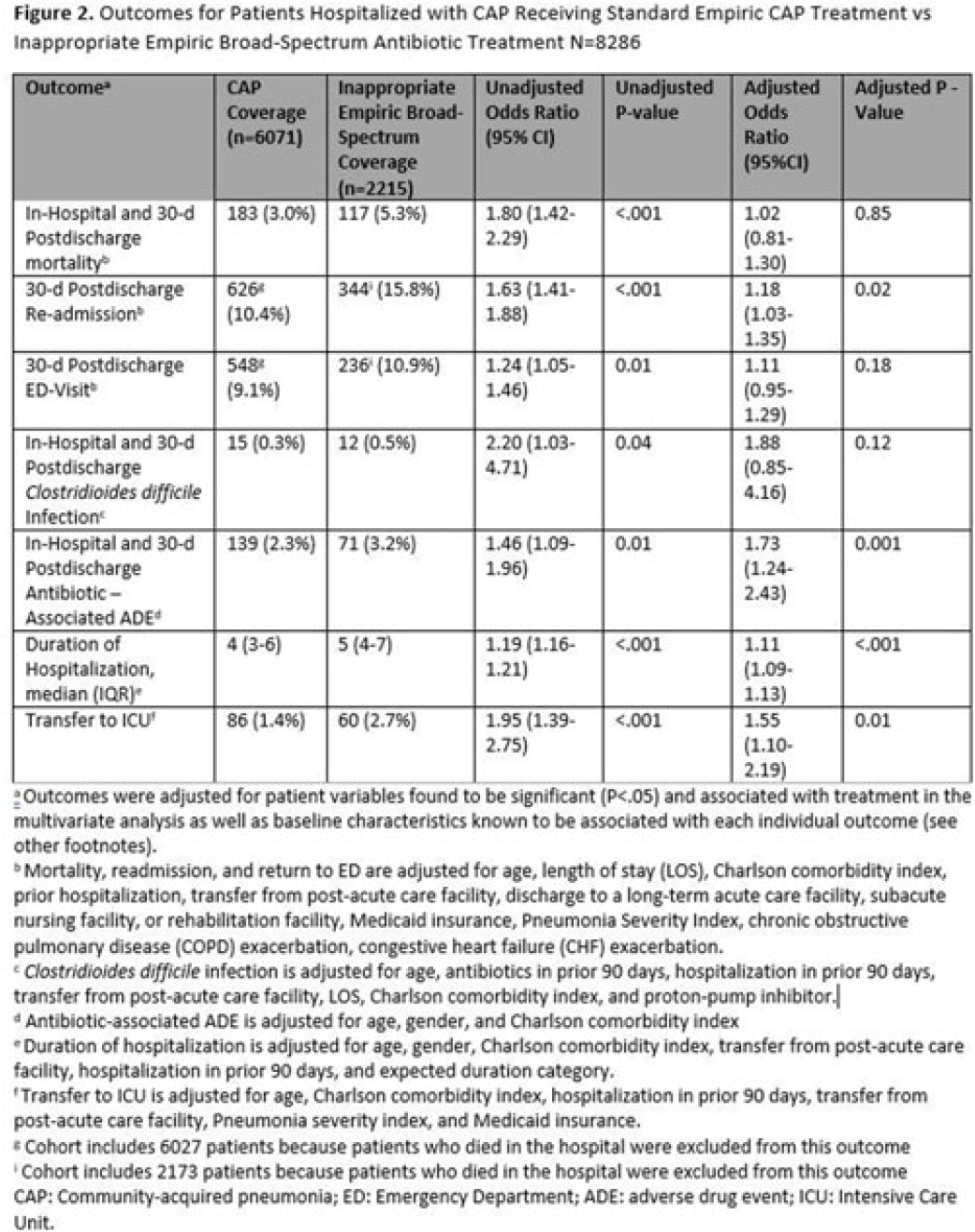
Financial support. H.M.S. initiative is underwritten by Blue Cross and Blue Shield of Michigan.
Disclosures: None
Pharmacist gender and physician acceptance of antibiotic stewardship recommendations: An analysis of the reducing overuse of antibiotics at discharge home intervention
- Part of
- Valerie M. Vaughn, Daniel L. Giesler, Daraoun Mashrah, Adamo Brancaccio, Katie Sandison, Emily S. Spivak, Julia E. Szymczak, Chaorong Wu, Jennifer K. Horowitz, Linda Bashaw, Adam L. Hersh
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 4 / April 2023
- Published online by Cambridge University Press:
- 07 June 2022, pp. 570-577
- Print publication:
- April 2023
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective:
To assess association of pharmacist gender with acceptance of antibiotic stewardship recommendations.
Design:A retrospective evaluation of the Reducing Overuse of Antibiotics at Discharge (ROAD) Home intervention.
Setting:The study was conducted from May to October 2019 in a single academic medical center.
Participants:The study included patients receiving antibiotics on a hospitalist service who were nearing discharge.
Methods:During the intervention, clinical pharmacists (none who had specialist postgraduate infectious disease residency training) reviewed patients on antibiotics and led an antibiotic timeout (ie, structured conversation) prior to discharge to improve discharge antibiotic prescribing. We assessed the association of pharmacist gender with acceptance of timeout recommendations by hospitalists using logistic regression controlling for patient characteristics.
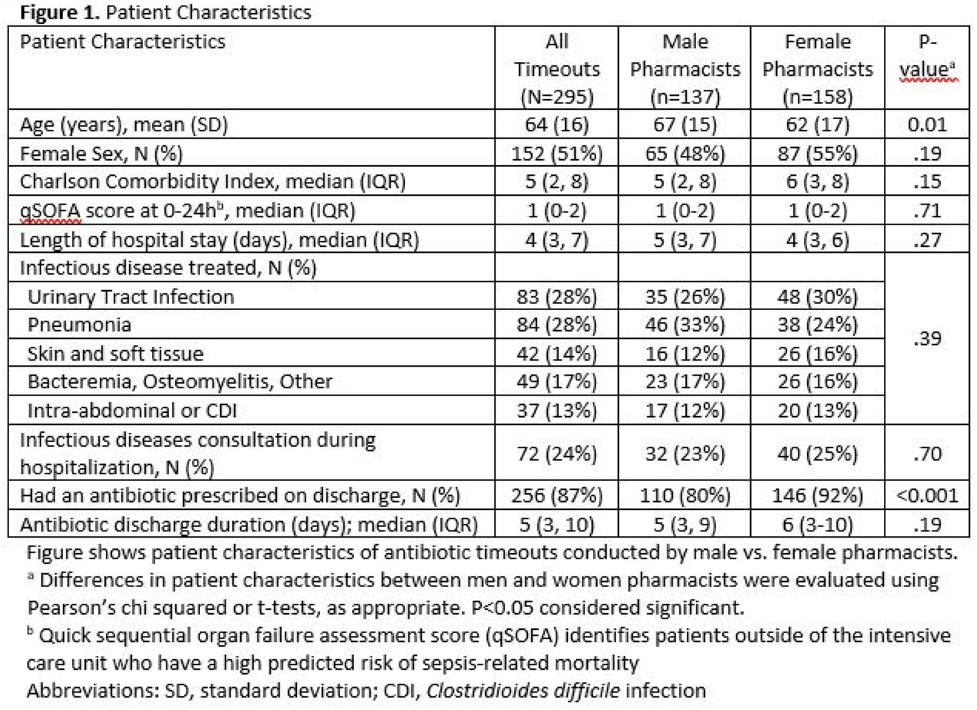
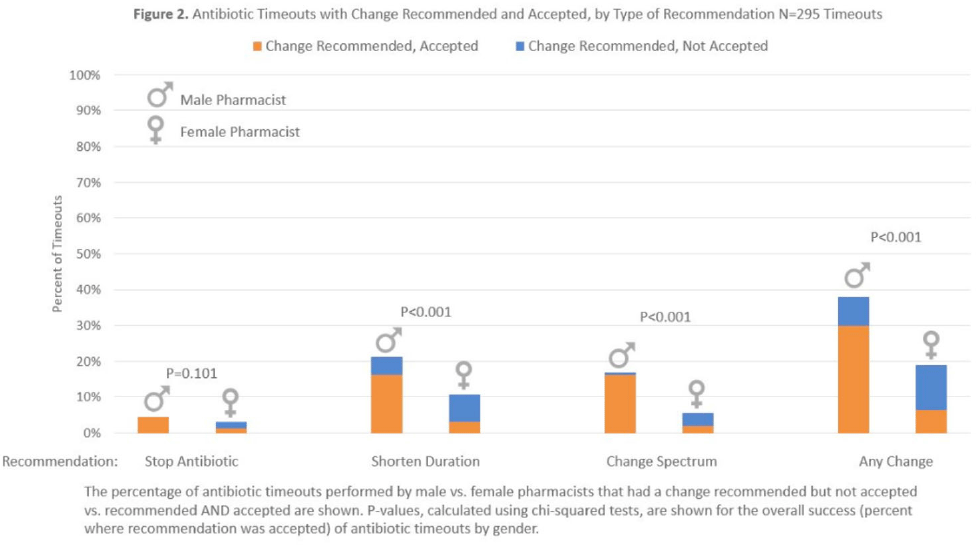
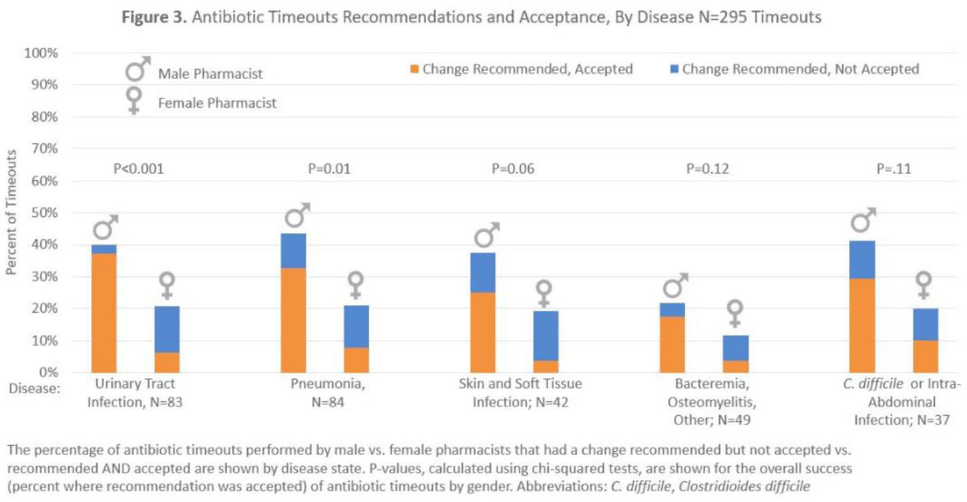
Results:Over 6 months, pharmacists conducted 295 timeouts: 158 timeouts (53.6%) were conducted by 12 women, 137 (46.4%) were conducted by 8 men. Pharmacists recommended an antibiotic change in 82 timeouts (27.8%), of which 51 (62.2%) were accepted. Compared to male pharmacists, female pharmacists were less likely to recommend a discharge antibiotic change: 30 (19.0%) of 158 versus 52 (38.0%) of 137 (P < .001). Female pharmacists were also less likely to have a recommendation accepted: 10 (33.3%) of 30 versus 41 (8.8%) of 52 (P < .001). Thus, timeouts conducted by female versus male pharmacists were less likely to result in an antibiotic change: 10 (6.3%) of 158 versus 41 (29.9%) of 137 (P < .001). After adjustments, pharmacist gender remained significantly associated with whether recommended changes were accepted (adjusted odds ratio [aOR], 0.10; 95%confidence interval [CI], 0.03–0.36 for female versus male pharmacists).
Conclusions:Antibiotic stewardship recommendations made by female clinical pharmacists were less likely to be accepted by hospitalists. Gender bias may play a role in the acceptance of clinical pharmacist recommendations, which could affect patient care and outcomes.
The effect of gender bias on acceptance of antibiotic stewardship recommendations by clinical pharmacists
- Valerie Vaughn, Daniel Giesler, Adamo Brancaccio, Daraoun Mashrah, Katie Sandison, Chaorong Wu, Jennifer Horowitz, Linda Bashaw, Adam Hersh
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s67-s68
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clinical pharmacists are a critical part of antibiotic stewardship. Stewardship often relies on relationships and persuasion, which may be affected by gender bias. Thus, we aimed to assess the association of sex with the acceptance of antibiotic stewardship recommendations. Methods: Between May and October 2019, medicine pharmacists at single hospital reviewed patients on antibiotics and–when a discharge was anticipated–led an antibiotic discussion (or “timeout”) prior to discharge. To explore differences in antibiotic timeout effectiveness by gender, we assessed the association of pharmacist sex with suggestion and acceptance of antibiotic changes using logistic regression controlling for patient characteristics. We also assessed whether hospitalist sex was associated with or moderated the effect of pharmacist sex on acceptance of timeout recommendations. Results: Between May 1, 2019, and October 31, 2019, pharmacists conducted 295 timeouts (patient characteristics in Fig. 1). Overall, 54% of timeouts were conducted by 12 female pharmacists and the remaining 46% were conducted by 8 male pharmacists. Overall, 82 (29%) of 295 timeouts resulted in a pharmacist recommending an antibiotic change, and male pharmacists were more likely to recommend a change: 52 (38%) of 137 versus 30 (19%) 158 (P Conclusions: In this discharge antibiotic intervention, timeouts conducted by women were less likely to result in an antibiotic change than those conducted by men. The difference in effectiveness resulted both from female pharmacists being less likely to recommend a change and from hospitalists being less likely to accept recommendations from a female pharmacist. These findings suggest that gender bias may play a role acceptance of antibiotic stewardship recommendations, which could affect antibiotic use, pharmacist job satisfaction, and patient outcomes.
Funding: None
Disclosures: None
Risk of bacterial bloodstream infection does not vary by central-line type during neutropenic periods in pediatric acute myeloid leukemia
- Caitlin W. Elgarten, William R. Otto, Luke Shenton, Madison T. Stein, Joseph Horowitz, Catherine Aftandilian, Staci D. Arnold, Kira O. Bona, Emi Caywood, Anderson B. Collier, M. Monica Gramatges, Meret Henry, Craig Lotterman, Kelly Maloney, Arunkumar J. Modi, Amir Mian, Rajen Mody, Elaine Morgan, Elizabeth A. Raetz, Anupam Verma, Naomi Winick, Jennifer J. Wilkes, Jennifer C. Yu, Richard Aplenc, Brian T. Fisher, Kelly D. Getz
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 2 / February 2023
- Published online by Cambridge University Press:
- 25 April 2022, pp. 222-229
- Print publication:
- February 2023
-
- Article
- Export citation
-
Background:
Bloodstream infections (BSIs) are a frequent cause of morbidity in patients with acute myeloid leukemia (AML), due in part to the presence of central venous access devices (CVADs) required to deliver therapy.
Objective:To determine the differential risk of bacterial BSI during neutropenia by CVAD type in pediatric patients with AML.
Methods:We performed a secondary analysis in a cohort of 560 pediatric patients (1,828 chemotherapy courses) receiving frontline AML chemotherapy at 17 US centers. The exposure was CVAD type at course start: tunneled externalized catheter (TEC), peripherally inserted central catheter (PICC), or totally implanted catheter (TIC). The primary outcome was course-specific incident bacterial BSI; secondary outcomes included mucosal barrier injury (MBI)-BSI and non-MBI BSI. Poisson regression was used to compute adjusted rate ratios comparing BSI occurrence during neutropenia by line type, controlling for demographic, clinical, and hospital-level characteristics.
Results:The rate of BSI did not differ by CVAD type: 11 BSIs per 1,000 neutropenic days for TECs, 13.7 for PICCs, and 10.7 for TICs. After adjustment, there was no statistically significant association between CVAD type and BSI: PICC incident rate ratio [IRR] = 1.00 (95% confidence interval [CI], 0.75–1.32) and TIC IRR = 0.83 (95% CI, 0.49–1.41) compared to TEC. When MBI and non-MBI were examined separately, results were similar.
Conclusions:In this large, multicenter cohort of pediatric AML patients, we found no difference in the rate of BSI during neutropenia by CVAD type. This may be due to a risk-profile for BSI that is unique to AML patients.
Duties, resources, and burnout of antibiotic stewards during the coronavirus disease 2019 (COVID-19) pandemic
- Valerie M. Vaughn, Guinn E. Dunn, Jennifer K. Horowitz, Elizabeth S. McLaughlin, Tejal N. Gandhi
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 05 November 2021, e39
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
Antibiotic overuse after discharge from medical short-stay units
- Nathaniel S. Soper, Abhinav J. Appukutty, David Paje, Lindsay A. Petty, Scott A. Flanders, Qisu Zhang, Jennifer K. Horowitz, Valerie M. Vaughn
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 24 August 2021, pp. 1689-1692
- Print publication:
- November 2022
-
- Article
- Export citation
-
Of 100 patients discharged from short-stay units (SSUs) with antibiotics, 47 had a skin and soft-tissue infection, 22 had pneumonia, and 21 had a urinary tract infection. Among all discharge antibiotic prescriptions, 78% involved antibiotic overuse, most commonly excess duration (54 of 100) and guideline discordant selection (44 of 100).
Do antimicrobial and antithrombogenic peripherally inserted central catheter (PICC) materials prevent catheter complications? An analysis of 42,562 hospitalized medical patients
- Amanda J. Ullman, Rebecca S. Paterson, Jessica A. Schults, Tricia M. Kleidon, Deanne August, Megan O’Malley, Jennifer Horowitz, Claire M. Rickard, David Paje, Vineet Chopra
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 4 / April 2022
- Published online by Cambridge University Press:
- 28 April 2021, pp. 427-434
- Print publication:
- April 2022
-
- Article
- Export citation
-
Objective:
To examine the effectiveness of antimicrobial and antithrombogenic materials incorporated into peripherally inserted central catheters (PICCs) to prevent bloodstream infection, thrombosis, and catheter occlusion.
Methods:Prospective cohort study involving 52 hospitals participating in the Michigan Hospital Medicine Safety Consortium. Sample included adult hospitalized medical patients who received a PICC between January 2013 and October 2019. Coated and impregnated catheters were identified by name, brand, and device marketing or regulatory materials. Multivariable Cox proportional hazards models with robust sandwich standard error estimates accounting for the clustered nature of data were used to identify factors associated with PICC complications in coated versus noncoated devices across general care, intensive care unit (ICU), and oncology patients. Results were expressed as hazard ratios (HRs) with corresponding 95% confidence intervals (CIs).
Results:Of 42,562 patients with a PICC, 39,806 (93.5%) were plain polyurethane, 2,263 (5.3%) incorporated antimicrobial materials, and 921 (2.2%) incorporated antithrombogenic materials. Most were inserted in general ward settings (n = 28,111, 66.0%), with 12, 078 (28.4%) and 1,407 (3.3%) placed in ICU and oncological settings, respectively. Within the entire cohort, 540 (1.3%) developed thrombosis, 745 (1.8%) developed bloodstream infection, and 4,090 (9.6%) developed catheter occlusion. Adjusting for known risk factors, antimicrobial PICCs were not associated with infection reduction (HR, 1.16; 95% CI, 0.82–1.64), and antithrombogenic PICCs were not associated with reduction in thrombosis and occlusion (HR, 1.15; 95% CI, 0.92–1.44). Results were consistent across populations and care settings.
Conclusions:Antimicrobial and antithrombogenic PICCs were not associated with a reduction in major catheter complications. Guidance aimed at informing use of these devices, balancing benefits against cost, appear necessary.
Influence of Infectious Disease Physician Approval on Appropriateness of PICC Use and Outcomes
- Valerie M Vaughn, Megan O’Malley, Scott A. Flanders, Tejal N. Gandhi, Lindsay A. Petty, Anurag Malani, Allison Weinmann, Jennifer Horowitz, Vineet Chopra
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s485-s487
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background:Peripherally inserted central catheters (PICCs) are frequently used to deliver intravenous (IV) antibiotic therapy after discharge from the hospital. Infectious disease (ID) physicians are often consulted prior to PICC placement, but whether their approval influences PICC appropriateness and complications is not known. Methods: Using data from the Michigan Hospital Medicine Safety Consortium (HMS) on PICCs placed in critically ill and hospitalized medical patients between January 1, 2015, and July 26, 2019, we examined the association between ID physician approval of PICC insertion for IV antibiotics and device appropriateness and outcomes. Appropriateness was defined according to the Michigan Appropriateness Guide for Intravenous Catheters (MAGIC) as a composite measure of (1) avoiding PICC use for durations ≤5 days; (2) using single-lumen instead of multilumen catheters; and (3) avoiding PICC use in patients with chronic kidney disease (eGFR>45 mL/min). The associations between ID approval of PICC use and odds of PICC-related complications (eg, deep vein thrombosis, central-line–associated bloodstream infection, and catheter occlusion) were also assessed. Multivariable models adjusting for patient severity of illness and hospital-level clustering were fit to both outcomes. Results were expressed as odds ratios (ORs) with corresponding 95% CIs. Results: Data from 36,594 patients who underwent PICC placement across 42 Michigan hospitals were included in the analysis. In total, 21,653 (55%) PICCs were placed for the indication of IV antibiotics; 14,935 (69%) of these had a documented ID consultation prior to placement, whereas 6,718 (31%) did not. Of the 14,935 PICCs with an ID consultation, 10,238 (69%) had ID approval documented prior to device placement (Fig. 1). Compared to no approval, PICCs approved by ID prior to insertion were more likely to be appropriate (OR, 3.51; 95% CI, 3.28–3.77; P < .001). Specifically, approval was associated with higher single-lumen use (OR, 5.13; 95% CI, 4.72–5.58; P < .001), less placement of PICCs with dwell times ≤ 5 days (OR, 0.29; 95% CI, 0.25–0.32; P < .001), and less frequent use in patients with chronic kidney disease (OR, 0.80; 95% CI, 0.73–0.87; P < .001). ID approval of PICCs prior to insertion was associated with a significantly lower odds of PICC-related complications (OR, 0.57; 95% CI, 0.51–0.64) (Table 1). Conclusions: ID approval of PICC use for IV antibiotic therapy in hospitalized patients was associated with greater appropriateness and fewer complications. Policies aimed at ensuring ID review prior to PICC use may help improve patient and device safety.
Funding: None
Disclosures:Valerie M. Vaughn reports contract research for Blue Cross and Blue Shield of Michigan, the Department of Veterans’ Affairs, the NIH, the SHEA, and the APIC. She also reports fees from the Gordon and Betty Moore Foundation Speaker’s Bureau, the CDC, the Pew Research Trust, Sepsis Alliance, and The Hospital and Health System Association of Pennsylvania.
Misdiagnosis of Urinary Tract Infection Linked to Misdiagnosis of Pneumonia: A Multihospital Cohort Study
- Valerie M. Vaughn, Ashwin Gupta, Lindsay A. Petty, Tejal N. Gandhi, Scott A. Flanders, Lakshmi Swaminathan, Lama Hsaiky, Lama Hsaiky, David Ratz, Jennifer Horowitz, Elizabeth McLaughlin, Vineet Chopra
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s488-s489
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Clinicians often diagnose bacterial infections such as urinary tract infection (UTI) and pneumonia in patients who are asymptomatic or have nonbacterial causes of their symptoms. Misdiagnosis of infection leads to unnecessary antibiotic use and potentially delays correct diagnoses. Interventions to improve diagnosis often focus on infections separately. However, if misdiagnosis is linked, broader interventions to improve diagnosis may be more effective. Thus, we assessed whether misdiagnosis of UTI and community-acquired pneumonia (CAP) was correlated. Methods: From July 2017 to July 2019, abstractors at 46 Michigan hospitals collected data on a sample of adult, non–intensive care, hospitalized patients with bacteriuria (positive urine culture) or who were treated for presumed CAP (discharge diagnosis plus antibiotics). Patients with concomitant bacterial infections were excluded. Using a previously described method,1,2 patients were assessed for UTI or CAP based on symptoms, signs, and laboratory or radiology findings. Misdiagnosis of UTI was defined as patients with asymptomatic bacteriuria (ASB) treated with antibiotics number of patients with bacteriuria Misdiagnosis of CAP was defined as patients treated for presumed CAP who did not have CAP number of patients treated for presumed CAP. Hospital-level correlation was assessed using Pearson’s correlation coefficient between misdiagnosis of UTI and CAP. For patients with prescriber data (N = 3,293), we also assessed emergency department (ED)-level correlation. Results: Of 11,914 patients with bacteriuria, 31.9% (N = 3,796) had ASB. Of those, 2,973 of 3,796 (78.3%) received antibiotics. Of 14,085 patients treated for CAP, 1,602 (11.4%) did not have CAP. Incidence of misdiagnosis varied by hospital: those with high rates of misdiagnosis of UTI were more likely to have high rates of misdiagnosis of CAP (Pearson’s correlation coefficient, 0.58; P ≤ .001) (Fig. 1). Of 2,137 patients misdiagnosed with UTI, 1,159 (54.2%) had antibiotic treatment started in the ED; of those, 942 (81.3%) remained on antibiotics on day 3 of hospitalization. Of 1,156 patients misdiagnosed with CAP, 871 (75.3%) had antibiotic therapy started in the ED, and 789 of these 871 patients (90.6%) were still on antibiotics on day 3 of hospitalization. Hospitals with high rates of UTI misdiagnosis in the ED were more likely to have high rates of CAP misdiagnosis in the ED (Pearson’s correlation coefficient, 0.33; P ≤ .001). Conclusions: Misdiagnosis of 2 unrelated infections was moderately correlated by hospital and weakly correlated by hospital ED. Potential causes include differences in organizational culture (eg, low tolerance for diagnostic uncertainty, emergency department culture), organizational initiatives (eg, sepsis, stewardship), or coordination between emergency and hospital medicine. Additionally, antibiotics initiated in the ED were typically continued following admission, potentially reflecting diagnosis momentum.
Funding: This work was supported by Blue Cross Blue Shield of Michigan and Blue Care Network.
Disclosures: Valerie M. Vaughn reports contract research for Blue Cross and Blue Shield of Michigan, the Department of Veterans’ Affairs, the NIH, the SHEA, and the APIC. She also reports fees from the Gordon and Betty Moore Foundation Speaker’s Bureau, the CDC, the Pew Research Trust, Sepsis Alliance, and The Hospital and Health System Association of Pennsylvania.
Home Health Service Provision After Hurricane Harvey
- Sue Anne Bell, Jennifer Horowitz, Theodore Iwashyna
-
- Journal:
- Disaster Medicine and Public Health Preparedness / Volume 14 / Issue 1 / February 2020
- Published online by Cambridge University Press:
- 20 June 2019, pp. 56-62
-
- Article
- Export citation
-
Objective:
To determine the extent of service disruption among home health agencies impacted by Hurricane Harvey.
Methods:Structured interviews with optional open-ended questions were conducted with home health agencies in and around Houston, Texas. A random sample of 277 agencies was selected and contacted via telephone during the study period, from February to May of 2018.
Results:Only 45% of 122 participating agencies indicated that their offices were open during Hurricane Harvey, and three-fourths reported that home visits were disrupted. The length of disruption varied: 7% reported a disruption of 1 day or less and 46% indicated a disruption of 1 week or longer. Disruption occurred even though nearly all (99%) of the agencies had—and close to all (92%) of them activated—an emergency preparedness plan.
Conclusions:Although most of the participating home health agencies activated their emergency preparedness plan, significant disruption in home health services occurred. While agencies are required to have clear, detailed plans in place, gaps in effective implementation of emergency preparedness plans remain.
Contributors
-
- By Mitchell Aboulafia, Frederick Adams, Marilyn McCord Adams, Robert M. Adams, Laird Addis, James W. Allard, David Allison, William P. Alston, Karl Ameriks, C. Anthony Anderson, David Leech Anderson, Lanier Anderson, Roger Ariew, David Armstrong, Denis G. Arnold, E. J. Ashworth, Margaret Atherton, Robin Attfield, Bruce Aune, Edward Wilson Averill, Jody Azzouni, Kent Bach, Andrew Bailey, Lynne Rudder Baker, Thomas R. Baldwin, Jon Barwise, George Bealer, William Bechtel, Lawrence C. Becker, Mark A. Bedau, Ernst Behler, José A. Benardete, Ermanno Bencivenga, Jan Berg, Michael Bergmann, Robert L. Bernasconi, Sven Bernecker, Bernard Berofsky, Rod Bertolet, Charles J. Beyer, Christian Beyer, Joseph Bien, Joseph Bien, Peg Birmingham, Ivan Boh, James Bohman, Daniel Bonevac, Laurence BonJour, William J. Bouwsma, Raymond D. Bradley, Myles Brand, Richard B. Brandt, Michael E. Bratman, Stephen E. Braude, Daniel Breazeale, Angela Breitenbach, Jason Bridges, David O. Brink, Gordon G. Brittan, Justin Broackes, Dan W. Brock, Aaron Bronfman, Jeffrey E. Brower, Bartosz Brozek, Anthony Brueckner, Jeffrey Bub, Lara Buchak, Otavio Bueno, Ann E. Bumpus, Robert W. Burch, John Burgess, Arthur W. Burks, Panayot Butchvarov, Robert E. Butts, Marina Bykova, Patrick Byrne, David Carr, Noël Carroll, Edward S. Casey, Victor Caston, Victor Caston, Albert Casullo, Robert L. Causey, Alan K. L. Chan, Ruth Chang, Deen K. Chatterjee, Andrew Chignell, Roderick M. Chisholm, Kelly J. Clark, E. J. Coffman, Robin Collins, Brian P. Copenhaver, John Corcoran, John Cottingham, Roger Crisp, Frederick J. Crosson, Antonio S. Cua, Phillip D. Cummins, Martin Curd, Adam Cureton, Andrew Cutrofello, Stephen Darwall, Paul Sheldon Davies, Wayne A. Davis, Timothy Joseph Day, Claudio de Almeida, Mario De Caro, Mario De Caro, John Deigh, C. F. Delaney, Daniel C. Dennett, Michael R. DePaul, Michael Detlefsen, Daniel Trent Devereux, Philip E. Devine, John M. Dillon, Martin C. Dillon, Robert DiSalle, Mary Domski, Alan Donagan, Paul Draper, Fred Dretske, Mircea Dumitru, Wilhelm Dupré, Gerald Dworkin, John Earman, Ellery Eells, Catherine Z. Elgin, Berent Enç, Ronald P. Endicott, Edward Erwin, John Etchemendy, C. Stephen Evans, Susan L. Feagin, Solomon Feferman, Richard Feldman, Arthur Fine, Maurice A. Finocchiaro, William FitzPatrick, Richard E. Flathman, Gvozden Flego, Richard Foley, Graeme Forbes, Rainer Forst, Malcolm R. Forster, Daniel Fouke, Patrick Francken, Samuel Freeman, Elizabeth Fricker, Miranda Fricker, Michael Friedman, Michael Fuerstein, Richard A. Fumerton, Alan Gabbey, Pieranna Garavaso, Daniel Garber, Jorge L. A. Garcia, Robert K. Garcia, Don Garrett, Philip Gasper, Gerald Gaus, Berys Gaut, Bernard Gert, Roger F. Gibson, Cody Gilmore, Carl Ginet, Alan H. Goldman, Alvin I. Goldman, Alfonso Gömez-Lobo, Lenn E. Goodman, Robert M. Gordon, Stefan Gosepath, Jorge J. E. Gracia, Daniel W. Graham, George A. Graham, Peter J. Graham, Richard E. Grandy, I. Grattan-Guinness, John Greco, Philip T. Grier, Nicholas Griffin, Nicholas Griffin, David A. Griffiths, Paul J. Griffiths, Stephen R. Grimm, Charles L. Griswold, Charles B. Guignon, Pete A. Y. Gunter, Dimitri Gutas, Gary Gutting, Paul Guyer, Kwame Gyekye, Oscar A. Haac, Raul Hakli, Raul Hakli, Michael Hallett, Edward C. Halper, Jean Hampton, R. James Hankinson, K. R. Hanley, Russell Hardin, Robert M. Harnish, William Harper, David Harrah, Kevin Hart, Ali Hasan, William Hasker, John Haugeland, Roger Hausheer, William Heald, Peter Heath, Richard Heck, John F. Heil, Vincent F. Hendricks, Stephen Hetherington, Francis Heylighen, Kathleen Marie Higgins, Risto Hilpinen, Harold T. Hodes, Joshua Hoffman, Alan Holland, Robert L. Holmes, Richard Holton, Brad W. Hooker, Terence E. Horgan, Tamara Horowitz, Paul Horwich, Vittorio Hösle, Paul Hoβfeld, Daniel Howard-Snyder, Frances Howard-Snyder, Anne Hudson, Deal W. Hudson, Carl A. Huffman, David L. Hull, Patricia Huntington, Thomas Hurka, Paul Hurley, Rosalind Hursthouse, Guillermo Hurtado, Ronald E. Hustwit, Sarah Hutton, Jonathan Jenkins Ichikawa, Harry A. Ide, David Ingram, Philip J. Ivanhoe, Alfred L. Ivry, Frank Jackson, Dale Jacquette, Joseph Jedwab, Richard Jeffrey, David Alan Johnson, Edward Johnson, Mark D. Jordan, Richard Joyce, Hwa Yol Jung, Robert Hillary Kane, Tomis Kapitan, Jacquelyn Ann K. Kegley, James A. Keller, Ralph Kennedy, Sergei Khoruzhii, Jaegwon Kim, Yersu Kim, Nathan L. King, Patricia Kitcher, Peter D. Klein, E. D. Klemke, Virginia Klenk, George L. Kline, Christian Klotz, Simo Knuuttila, Joseph J. Kockelmans, Konstantin Kolenda, Sebastian Tomasz Kołodziejczyk, Isaac Kramnick, Richard Kraut, Fred Kroon, Manfred Kuehn, Steven T. Kuhn, Henry E. Kyburg, John Lachs, Jennifer Lackey, Stephen E. Lahey, Andrea Lavazza, Thomas H. Leahey, Joo Heung Lee, Keith Lehrer, Dorothy Leland, Noah M. Lemos, Ernest LePore, Sarah-Jane Leslie, Isaac Levi, Andrew Levine, Alan E. Lewis, Daniel E. Little, Shu-hsien Liu, Shu-hsien Liu, Alan K. L. Chan, Brian Loar, Lawrence B. Lombard, John Longeway, Dominic McIver Lopes, Michael J. Loux, E. J. Lowe, Steven Luper, Eugene C. Luschei, William G. Lycan, David Lyons, David Macarthur, Danielle Macbeth, Scott MacDonald, Jacob L. Mackey, Louis H. Mackey, Penelope Mackie, Edward H. Madden, Penelope Maddy, G. B. Madison, Bernd Magnus, Pekka Mäkelä, Rudolf A. Makkreel, David Manley, William E. Mann (W.E.M.), Vladimir Marchenkov, Peter Markie, Jean-Pierre Marquis, Ausonio Marras, Mike W. Martin, A. P. Martinich, William L. McBride, David McCabe, Storrs McCall, Hugh J. McCann, Robert N. McCauley, John J. McDermott, Sarah McGrath, Ralph McInerny, Daniel J. McKaughan, Thomas McKay, Michael McKinsey, Brian P. McLaughlin, Ernan McMullin, Anthonie Meijers, Jack W. Meiland, William Jason Melanson, Alfred R. Mele, Joseph R. Mendola, Christopher Menzel, Michael J. Meyer, Christian B. Miller, David W. Miller, Peter Millican, Robert N. Minor, Phillip Mitsis, James A. Montmarquet, Michael S. Moore, Tim Moore, Benjamin Morison, Donald R. Morrison, Stephen J. Morse, Paul K. Moser, Alexander P. D. Mourelatos, Ian Mueller, James Bernard Murphy, Mark C. Murphy, Steven Nadler, Jan Narveson, Alan Nelson, Jerome Neu, Samuel Newlands, Kai Nielsen, Ilkka Niiniluoto, Carlos G. Noreña, Calvin G. Normore, David Fate Norton, Nikolaj Nottelmann, Donald Nute, David S. Oderberg, Steve Odin, Michael O’Rourke, Willard G. Oxtoby, Heinz Paetzold, George S. Pappas, Anthony J. Parel, Lydia Patton, R. P. Peerenboom, Francis Jeffry Pelletier, Adriaan T. Peperzak, Derk Pereboom, Jaroslav Peregrin, Glen Pettigrove, Philip Pettit, Edmund L. Pincoffs, Andrew Pinsent, Robert B. Pippin, Alvin Plantinga, Louis P. Pojman, Richard H. Popkin, John F. Post, Carl J. Posy, William J. Prior, Richard Purtill, Michael Quante, Philip L. Quinn, Philip L. Quinn, Elizabeth S. Radcliffe, Diana Raffman, Gerard Raulet, Stephen L. Read, Andrews Reath, Andrew Reisner, Nicholas Rescher, Henry S. Richardson, Robert C. Richardson, Thomas Ricketts, Wayne D. Riggs, Mark Roberts, Robert C. Roberts, Luke Robinson, Alexander Rosenberg, Gary Rosenkranz, Bernice Glatzer Rosenthal, Adina L. Roskies, William L. Rowe, T. M. Rudavsky, Michael Ruse, Bruce Russell, Lilly-Marlene Russow, Dan Ryder, R. M. Sainsbury, Joseph Salerno, Nathan Salmon, Wesley C. Salmon, Constantine Sandis, David H. Sanford, Marco Santambrogio, David Sapire, Ruth A. Saunders, Geoffrey Sayre-McCord, Charles Sayward, James P. Scanlan, Richard Schacht, Tamar Schapiro, Frederick F. Schmitt, Jerome B. Schneewind, Calvin O. Schrag, Alan D. Schrift, George F. Schumm, Jean-Loup Seban, David N. Sedley, Kenneth Seeskin, Krister Segerberg, Charlene Haddock Seigfried, Dennis M. Senchuk, James F. Sennett, William Lad Sessions, Stewart Shapiro, Tommie Shelby, Donald W. Sherburne, Christopher Shields, Roger A. Shiner, Sydney Shoemaker, Robert K. Shope, Kwong-loi Shun, Wilfried Sieg, A. John Simmons, Robert L. Simon, Marcus G. Singer, Georgette Sinkler, Walter Sinnott-Armstrong, Matti T. Sintonen, Lawrence Sklar, Brian Skyrms, Robert C. Sleigh, Michael Anthony Slote, Hans Sluga, Barry Smith, Michael Smith, Robin Smith, Robert Sokolowski, Robert C. Solomon, Marta Soniewicka, Philip Soper, Ernest Sosa, Nicholas Southwood, Paul Vincent Spade, T. L. S. Sprigge, Eric O. Springsted, George J. Stack, Rebecca Stangl, Jason Stanley, Florian Steinberger, Sören Stenlund, Christopher Stephens, James P. Sterba, Josef Stern, Matthias Steup, M. A. Stewart, Leopold Stubenberg, Edith Dudley Sulla, Frederick Suppe, Jere Paul Surber, David George Sussman, Sigrún Svavarsdóttir, Zeno G. Swijtink, Richard Swinburne, Charles C. Taliaferro, Robert B. Talisse, John Tasioulas, Paul Teller, Larry S. Temkin, Mark Textor, H. S. Thayer, Peter Thielke, Alan Thomas, Amie L. Thomasson, Katherine Thomson-Jones, Joshua C. Thurow, Vzalerie Tiberius, Terrence N. Tice, Paul Tidman, Mark C. Timmons, William Tolhurst, James E. Tomberlin, Rosemarie Tong, Lawrence Torcello, Kelly Trogdon, J. D. Trout, Robert E. Tully, Raimo Tuomela, John Turri, Martin M. Tweedale, Thomas Uebel, Jennifer Uleman, James Van Cleve, Harry van der Linden, Peter van Inwagen, Bryan W. Van Norden, René van Woudenberg, Donald Phillip Verene, Samantha Vice, Thomas Vinci, Donald Wayne Viney, Barbara Von Eckardt, Peter B. M. Vranas, Steven J. Wagner, William J. Wainwright, Paul E. Walker, Robert E. Wall, Craig Walton, Douglas Walton, Eric Watkins, Richard A. Watson, Michael V. Wedin, Rudolph H. Weingartner, Paul Weirich, Paul J. Weithman, Carl Wellman, Howard Wettstein, Samuel C. Wheeler, Stephen A. White, Jennifer Whiting, Edward R. Wierenga, Michael Williams, Fred Wilson, W. Kent Wilson, Kenneth P. Winkler, John F. Wippel, Jan Woleński, Allan B. Wolter, Nicholas P. Wolterstorff, Rega Wood, W. Jay Wood, Paul Woodruff, Alison Wylie, Gideon Yaffe, Takashi Yagisawa, Yutaka Yamamoto, Keith E. Yandell, Xiaomei Yang, Dean Zimmerman, Günter Zoller, Catherine Zuckert, Michael Zuckert, Jack A. Zupko (J.A.Z.)
- Edited by Robert Audi, University of Notre Dame, Indiana
-
- Book:
- The Cambridge Dictionary of Philosophy
- Published online:
- 05 August 2015
- Print publication:
- 27 April 2015, pp ix-xxx
-
- Chapter
- Export citation
Contributors
-
- By Rony A. Adam, Gloria Bachmann, Nichole M. Barker, Randall B. Barnes, John Bennett, Inbar Ben-Shachar, Jonathan S. Berek, Sarah L. Berga, Monica W. Best, Eric J. Bieber, Frank M. Biro, Shan Biscette, Anita K. Blanchard, Candace Brown, Ronald T. Burkman, Joseph Buscema, John E. Buster, Michael Byas-Smith, Sandra Ann Carson, Judy C. Chang, Annie N. Y. Cheung, Mindy S. Christianson, Karishma Circelli, Daniel L. Clarke-Pearson, Larry J. Copeland, Bryan D. Cowan, Navneet Dhillon, Michael P. Diamond, Conception Diaz-Arrastia, Nicole M. Donnellan, Michael L. Eisenberg, Eric Eisenhauer, Sebastian Faro, J. Stuart Ferriss, Lisa C. Flowers, Susan J. Freeman, Leda Gattoc, Claudine Marie Gayle, Timothy M. Geiger, Jennifer S. Gell, Alan N. Gordon, Victoria L. Green, Jon K. Hathaway, Enrique Hernandez, S. Paige Hertweck, Randall S. Hines, Ira R. Horowitz, Fred M. Howard, William W. Hurd, Fidan Israfilbayli, Denise J. Jamieson, Carolyn R. Jaslow, Erika B. Johnston-MacAnanny, Rohna M. Kearney, Namita Khanna, Caroline C. King, Jeremy A. King, Ira J. Kodner, Tamara Kolev, Athena P. Kourtis, S. Robert Kovac, Ertug Kovanci, William H. Kutteh, Eduardo Lara-Torre, Pallavi Latthe, Herschel W. Lawson, Ronald L. Levine, Frank W. Ling, Larry I. Lipshultz, Steven D. McCarus, Robert McLellan, Shruti Malik, Suketu M. Mansuria, Mohamed K. Mehasseb, Pamela J. Murray, Saloney Nazeer, Farr R. Nezhat, Hextan Y. S. Ngan, Gina M. Northington, Peggy A. Norton, Ruth M. O'Regan, Kristiina Parviainen, Resad P. Pasic, Tanja Pejovic, K. Ulrich Petry, Nancy A. Phillips, Ashish Pradhan, Elizabeth E. Puscheck, Suneetha Rachaneni, Devon M. Ramaeker, David B. Redwine, Robert L. Reid, Carla P. Roberts, Walter Romano, Peter G. Rose, Robert L. Rosenfield, Shon P. Rowan, Mack T. Ruffin, Janice M. Rymer, Evis Sala, Ritu Salani, Joseph S. Sanfilippo, Mahmood I. Shafi, Roger P. Smith, Meredith L. Snook, Thomas E. Snyder, Mary D. Stephenson, Thomas G. Stovall, Richard L. Sweet, Philip M. Toozs-Hobson, Togas Tulandi, Elizabeth R. Unger, Denise S. Uyar, Marion S. Verp, Rahi Victory, Tamara J. Vokes, Michelle J. Washington, Katharine O'Connell White, Paul E. Wise, Frank M. Wittmaack, Miya P. Yamamoto, Christine Yu, Howard A. Zacur
- Edited by Eric J. Bieber, Joseph S. Sanfilippo, University of Pittsburgh, Ira R. Horowitz, Emory University, Atlanta, Mahmood I. Shafi
-
- Book:
- Clinical Gynecology
- Published online:
- 05 April 2015
- Print publication:
- 23 April 2015, pp viii-xiv
-
- Chapter
- Export citation
Contributors
-
- By Yohance M. Allette, Christophe Altier, Charles E. Argoff, Nadine Attal, Paul J. Austin, Didier Bouhassira, Ian Carroll, Kristine M. Chapman, Stephen Coleman, Lynn Kerene Cooper, Michael R. Due, Mary-Ann Fitzcharles, Robyn Flynn, Andrea D. Furlan, Vishal Gupta, Maija Haanpää, Jennifer Hah, Steven H. Horowitz, John Hughes, Mark R. Hutchinson, Scott Jarvis, Maan Kattan, Manpreet Kaur, Bradley J. Kerr, Krishna Kumar, Yuen Hei Kwok, Wojciech Leppert, Liang Liu, Angela Mailis-Gagnon, Gila Moalem-Taylor, Dwight E. Moulin, Harsha Nagaraja, Dontese Nicholson, Lauren Nicotra, Anne Louise Oaklander, John Xavier Pereira, Syed Rizvi, Stephan A. Schug, Michael Serpell, Amanda Sherwin, Howard S. Smith, Peter A. Smith, Pam Squire, Peter A. Ste-Marie, Patrick L. Stemkowski, Nicole M. Sumracki, Cory Toth, Krista van Steeg, Jan H. Vranken, Bharati Vyawahare, Mark A. Ware, Linda R. Watkins, C. Peter N. Watson, Fletcher A. White
- Edited by Cory Toth, Dwight E. Moulin
-
- Book:
- Neuropathic Pain
- Published online:
- 05 December 2013
- Print publication:
- 07 November 2013, pp vii-x
-
- Chapter
- Export citation
Use of vitamin D supplements during infancy in an international feeding trial
- Eveliina Lehtonen, Anne Ormisson, Anita Nucci, David Cuthbertson, Susa Sorkio, Mila Hyytinen, Kirsi Alahuhta, Carol Berseth, Marja Salonen, Shayne Taback, Margaret Franciscus, Teba González-Frutos, Tuuli E Korhonen, Margaret L Lawson, Dorothy J Becker, Jeffrey P Krischer, Mikael Knip, Suvi M Virtanen, , Thomas Mandrup-Poulsen, Elias Arjas, Åke Lernmark, Barbara Schmidt, Jeffrey P. Krischer, Hans K. Åkerblom, Mila Hyytinen, Mikael Knip, Katriina Koski, Matti Koski, Eeva Pajakkala, Marja Salonen, David Cuthbertson, Jeffrey P. Krischer, Linda Shanker, Brenda Bradley, Hans-Michael Dosch, John Dupré, William Fraser, Margaret Lawson, Jeffrey L. Mahon, Mathew Sermer, Shayne P. Taback, Dorothy Becker, Margaret Franciscus, Anita Nucci, Jerry Palmer, Minna Pekkala, Suvi M. Virtanen, Jacki Catteau, Neville Howard, Patricia Crock, Maria Craig, Cheril L. Clarson, Lynda Bere, David Thompson, Daniel Metzger, Colleen Marshall, Jennifer Kwan, David K. Stephure, Daniele Pacaud, Wendy Schwarz, Rose Girgis, Marilyn Thompson, Shayne P. Taback, Daniel Catte, Margaret L. Lawson, Brenda Bradley, Denis Daneman, Mathew Sermer, Mary-Jean Martin, Valérie Morin, Lyne Frenette, Suzanne Ferland, Susan Sanderson, Kathy Heath, Céline Huot, Monique Gonthier, Maryse Thibeault, Laurent Legault, Diane Laforte, Elizabeth A. Cummings, Karen Scott, Tracey Bridger, Cheryl Crummell, Robyn Houlden, Adriana Breen, George Carson, Sheila Kelly, Koravangattu Sankaran, Marie Penner, Richard A. White, Nancy King, James Popkin, Laurie Robson, Eva Al Taji, Irena Aldhoon, Pavla Mendlova, Jan Vavrinec, Jan Vosahlo, Ludmila Brazdova, Jitrenka Venhacova, Petra Venhacova, Adam Cipra, Zdenka Tomsikova, Petra Krckova, Pavla Gogelova, Ülle Einberg, Mall-Anne Riikjärv, Anne Ormisson, Vallo Tillmann, Päivi Kleemola, Anna Parkkola, Heli Suomalainen, Anna-Liisa Järvenpää, Anu-Maaria Hämälainen, Hannu Haavisto, Sirpa Tenhola, Pentti Lautala, Pia Salonen, Susanna Aspholm, Heli Siljander, Carita Holm, Samuli Ylitalo, Raisa Lounamaa, Anja Nuuja, Timo Talvitie, Kaija Lindström, Hanna Huopio, Jouni Pesola, Riitta Veijola, Päivi Tapanainen, Abram Alar, Paavo Korpela, Marja-Liisa Käär, Taina Mustila, Ritva Virransalo, Päivi Nykänen, Bärbel Aschemeier, Thomas Danne, Olga Kordonouri, Dóra Krikovszky, László Madácsy, Yeganeh Manon Khazrai, Ernesto Maddaloni, Paolo Pozzilli, Carla Mannu, Marco Songini, Carine de Beaufort, Ulrike Schierloh, Jan Bruining, Margriet Bisschoff, Aleksander Basiak, Renata Wasikowa, Marta Ciechanowska, Grazyna Deja, Przemyslawa Jarosz-Chobot, Agnieszka Szadkowska, Katarzyna Cypryk, Malgorzata Zawodniak-Szalapska, Luis Castano, Teba Gonzalez Frutos, Mirentxu Oyarzabal, Manuel Serrano-Ríos, María Teresa Martínez-Larrad, Federico Gustavo Hawkins, Dolores Rodriguez Arnau, Johnny Ludvigsson, Malgorzata Smolinska Konefal, Ragnar Hanas, Bengt Lindblad, Nils-Osten Nilsson, Hans Fors, Maria Nordwall, Agne Lindh, Hans Edenwall, Jan Aman, Calle Johansson, Margrit Gadient, Eugen Schoenle, Dorothy Becker, Ashi Daftary, Margaret Franciscus, Carol Gilmour, Jerry Palmer, Rachel Taculad, Marilyn Tanner-Blasiar, Neil White, Uday Devaskar, Heather Horowitz, Lisa Rogers, Roxana Colon, Teresa Frazer, Jose Torres, Robin Goland, Ellen Greenberg, Maudene Nelson, Holly Schachner, Barney Softness, Jorma Ilonen, Massimo Trucco, Lynn Nichol, Erkki Savilahti, Taina Härkönen, Mikael Knip, Outi Vaarala, Kristiina Luopajärvi, Hans-Michael Dosch
-
- Journal:
- Public Health Nutrition / Volume 17 / Issue 4 / April 2014
- Published online by Cambridge University Press:
- 24 June 2013, pp. 810-822
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Objective
To examine the use of vitamin D supplements during infancy among the participants in an international infant feeding trial.
DesignLongitudinal study.
SettingInformation about vitamin D supplementation was collected through a validated FFQ at the age of 2 weeks and monthly between the ages of 1 month and 6 months.
SubjectsInfants (n 2159) with a biological family member affected by type 1 diabetes and with increased human leucocyte antigen-conferred susceptibility to type 1 diabetes from twelve European countries, the USA, Canada and Australia.
ResultsDaily use of vitamin D supplements was common during the first 6 months of life in Northern and Central Europe (>80 % of the infants), with somewhat lower rates observed in Southern Europe (>60 %). In Canada, vitamin D supplementation was more common among exclusively breast-fed than other infants (e.g. 71 % v. 44 % at 6 months of age). Less than 2 % of infants in the USA and Australia received any vitamin D supplementation. Higher gestational age, older maternal age and longer maternal education were study-wide associated with greater use of vitamin D supplements.
ConclusionsMost of the infants received vitamin D supplements during the first 6 months of life in the European countries, whereas in Canada only half and in the USA and Australia very few were given supplementation.











