4 results
Crystal structure and formula revision of deliensite, Fe[(UO2)2(SO4)2(OH)2](H2O)7
- J. Plášil, J. Hauser, V. Petříček, N. Meisser, S. J. Mills, R. Škoda, K. Fejfarová, J. Čejka, J. Sejkora, J. Hloušek, J.-M. Johannet, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 7 / December 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2837-2860
-
- Article
- Export citation
-
The crystal structure of deliensite, Fe[(UO2)2(SO4)2(OH)2](H2O)7, was solved by direct methods and refined to R1 = 6.24% for 5211 unique observed reflections [Iobs > 3σ(I)], on a crystal that was found to consist of rotational and inversion (merohedral) twins, from Jeroným mine, Abertamy in the Czech Republic. The presence of four twin domains was taken into account in the refinement. The structure is orthorhombic, space group Pnn2, with unit-cell parameters a = 15.8514(9), b = 16.2478(7), c = 6.8943(3) Å, V = 1775.6(1) Å3 and Z = 4. The crystal structure of deliensite contains uranyl-sulfate sheets with a phosphuranylite topology, consisting of dimers of edge-sharing uranyl pentagonal bipyramids linked by corner-sharing with sulfate tetrahedra. The sheets lie in the (100) plane and are decorated by [Fe2+O(H2O)5] octahedra; two weakly bonded H2O molecules are present in the interlayer. The [Fe2+O(H2O)5] octahedron is linked directly to the sheet via the uranyl oxygen atom. Adjacent sheets are linked by hydrogen bonds only. The sheet topology and geometrical isomerism is discussed and a comparison of the composition obtained from electron-probe microanalysis, powder-diffraction data, Raman and infrared spectra of deliensite samples from Mas d'Alary, Lodève, France; L'Ecarpière mine, Gétigné, France; and several localities at Jáchymov, Western Bohemia, Czech Republic is made.
Švenekite, Ca[AsO2(OH)2]2, a new mineral from Jáchymov, Czech Republic
- P. Ondruš, R. Skála, J. Plášil, J. Sejkora, F. Veselovský, J. Čejka, A. Kallistová, J. Hloušek, K. Fejfarová, R. Škoda, M. Dušek, A. Gabašová, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 6 / August 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2711-2724
-
- Article
- Export citation
-
Švenekite (IMA 99-007), Ca[AsO2(OH)2]2, is a rare supergene arsenate mineral occurring in the Geschieber vein, Jáchymov ore district, Western Bohemia, Czech Republic. It grows directly on the granite rocks and occurs isolated from other arsenate minerals otherwise common in Jáchymov. Švenekite usually forms clear transparent coatings composed of indistinct radiating to rosette-shaped aggregates up to 3 mm across. They are composed of thin lens- or bladed-shaped crystals, usually 100 – 150 μm long. Švenekite is transparent to translucent and has a white streak and a vitreous lustre; it does not fluoresce under ultraviolet light. Cleavage is very good on {010}. The Mohs hardness is ∼2. Švenekite is biaxial, non-pleochroic. The refractive indices are α' = 1.602(2), γ' = 1.658(2). The empirical formula of švenekite (based on As + P + S = 2 a.p.f.u., an average of 10 spot analyses) is (Ca1.00Mg0.01)Σ1.01[AsO2(OH)2]1.96[PO2(OH)2]0.03(SO4)0.01. The simplified formula is Ca[AsO2(OH)2]2 and requires CaO 17.42, As2O571.39, H2O 11.19, total 100.00 wt.%. Raman and infrared spectroscopy exhibit dominance of O – H vibrations and vibration modes of distorted tetrahedral AsO2(OH)2 units. Švenekite is triclinic, space group P
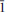 , with a = 8.5606(5), b = 7.6926(6), c = 5.7206(4) Å, α = 92.605(6), β = 109.9002(6), γ = 109.9017(6)º, and V = 327.48(4) Å3, Z = 2, Dcalc = 3.26 g·cm–3. The a:b:c ratio is 0.7436:1:1.1082 (for single-crystal data). The six strongest diffraction peaks in the X-ray powder diffraction pattern are [d (Å)/I(%)/(hkl)]: 3.968(33)(2
, with a = 8.5606(5), b = 7.6926(6), c = 5.7206(4) Å, α = 92.605(6), β = 109.9002(6), γ = 109.9017(6)º, and V = 327.48(4) Å3, Z = 2, Dcalc = 3.26 g·cm–3. The a:b:c ratio is 0.7436:1:1.1082 (for single-crystal data). The six strongest diffraction peaks in the X-ray powder diffraction pattern are [d (Å)/I(%)/(hkl)]: 3.968(33)(2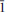 0); 3.766(35)(2
0); 3.766(35)(2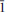
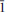 ); 3.697(49)(101); 3.554(100)(020); 3.259(33)(2
); 3.697(49)(101); 3.554(100)(020); 3.259(33)(2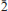 0); 3.097(49)(1
0); 3.097(49)(1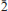 1). The crystal structure of švenekite was refined from single-crystal X-ray diffraction data to R1 = 0.0250 based on 1309 unique observed, and to wR2 = 0.0588, for all 1588 unique reflections (with GOFall = 1.20). The structure of švenekite consists of sheets of corner-sharing CaO8 polyhedra and AsO2 OH2 groups, stacked parallel to (001). Adjacent sheets are linked by hydrogen bonds. The švenekite structure possesses very short symmetrical hydrogen bonds (with the D–H lengths ∼1.22 Å). The mineral is named to honour Jaroslav Švenek, the former curator of the mineralogical collection of the National Museum in Prague, Czech Republic.
1). The crystal structure of švenekite was refined from single-crystal X-ray diffraction data to R1 = 0.0250 based on 1309 unique observed, and to wR2 = 0.0588, for all 1588 unique reflections (with GOFall = 1.20). The structure of švenekite consists of sheets of corner-sharing CaO8 polyhedra and AsO2 OH2 groups, stacked parallel to (001). Adjacent sheets are linked by hydrogen bonds. The švenekite structure possesses very short symmetrical hydrogen bonds (with the D–H lengths ∼1.22 Å). The mineral is named to honour Jaroslav Švenek, the former curator of the mineralogical collection of the National Museum in Prague, Czech Republic.
Hydroniumjarosite, (H3O)+Fe3(SO4)2(OH)6, from Cerros Pintados, Chile: Single-crystal X-ray diffraction and vibrational spectroscopic study
- J. Plášil, R. Škoda, K. Fejfarová, J. Čejka, A. V. Kasatkin, M. Dušek, D. Talla, L. Lapčák, V. Machovič, M. Dini
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 3 / June 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 535-547
-
- Article
- Export citation
-
The natural hydroniumjarosite sample from Cerros Pintados (Chile) was investigated by electron microprobe, single-crystal X-ray diffraction and vibrational spectroscopy (Infrared and Raman). The chemical composition of studied specimens (wt.%, mean of seven analyses) obtained from electron microprobe (in wt.%): Na2O 1.30, K2O 0.23, CaO 0.04, Fe2O3 50.49, Al2O3 0.37, SiO2 0.33, SO3 33.88, H2O (calculated on the basis of Σ(OH–+H3O+) deduced from the charge balance) 13.32, total 99.98, corresponds to the empirical formula (H3O)0.77+(Na0.20K0.02)∑0.22(Fe2.95Al0.03)∑2.98 (OH)6.12[(SO4)1.97(SiO4)0.03]∑2.00 (calculated on the basis of S + Si = 2 a.p.f.u. (atoms per formula unit)). The studied hydroniumjarosite is trigonal, with space group R
 m, with a = 7.3408(2), c = 17.0451(6) Å and V = 795.46(4) Å3. The refined structure architecture is consistent with known jarosite-series minerals, including synthetic hydroniumjarosite. However, in the current study the presence of H3O+ is well documented in difference Fourier maps, where characteristic positive difference Fourier maxima, with apparent trigonal symmetry, were localized in the vicinity of the O4 atom in the channel-voids of the structure. The structure of natural hydroniumjarosite, including the H atoms, was refined to R1 = 0.0166 for 2113 unique observed reflections, with Iobs>3σ(I). The present structure model, which includes the position of the H atom within the hydronium ion, is discussed with regard to the vibration spectroscopy results and earlier published density-functional theory (DFT) calculations for the alunite-like structure containing H3O+.
m, with a = 7.3408(2), c = 17.0451(6) Å and V = 795.46(4) Å3. The refined structure architecture is consistent with known jarosite-series minerals, including synthetic hydroniumjarosite. However, in the current study the presence of H3O+ is well documented in difference Fourier maps, where characteristic positive difference Fourier maxima, with apparent trigonal symmetry, were localized in the vicinity of the O4 atom in the channel-voids of the structure. The structure of natural hydroniumjarosite, including the H atoms, was refined to R1 = 0.0166 for 2113 unique observed reflections, with Iobs>3σ(I). The present structure model, which includes the position of the H atom within the hydronium ion, is discussed with regard to the vibration spectroscopy results and earlier published density-functional theory (DFT) calculations for the alunite-like structure containing H3O+.
Leydetite, Fe(UO2)(SO4)2(H2O)11, a new uranyl sulfate mineral from Mas d’Alary, Lodève, France
- J. Plášil, A. V. Kasatkin, R. Škoda, M. Novák, A. Kallistová, M. Dušek, R. Skála, K. Fejfarová, J. Čejka, N. Meisser, H. Goethals, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 4 / June 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 429-441
-
- Article
- Export citation
-
Leydetite, monoclinic Fe(UO2)(SO4)2(H2O)11(IMA 2012–065), is a new supergene uranyl sulfate from Mas d'Alary, Lodève, Hérault, France. It forms yellow to greenish, tabular, transparent to translucent crystals up to 2 mm in size. Crystals have a vitreous lustre. Leydetite has a perfect cleavage on (001). The streak is yellowish white. Mohs hardness is ∼2. The mineral does not fluoresce under long- or short-wavelength UV radiation. Leydetite is colourless in transmitted light, non-pleochroic, biaxial, with α = 1.513(2), γ = 1.522(2) (further optical properties could not be measured). The measured chemical composition of leydetite, FeO 9.28, MgO 0.37, Al2O30.26, CuO 0.14, UO340.19, SO321.91, SiO20.18, H2O 27.67, total 100 wt.%, leads to the empirical formula (based on 21 O a.p.f.u.), (Fe0.93Mg0.07Al0.04Cu0.01)Σ1.05(U1.01O2)(S1.96Si0.02)Σ1.98O8(H2O)11. Leydetite is monoclinic, space group C2/c, with a = 11.3203(3), b = 7.7293(2), c = 21.8145(8) Å, β = 102.402(3)°, V = 1864.18(10) Å3, Z = 4, and Dcalc = 2.55 g cm–3. The six strongest reflections in the X-ray powder diffraction pattern are [dobs in Å (I) (hkl)]: 10.625 (100) (002), 6.277 (1) (
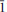 11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.0224 for 5211 observed reflections with [I > 3σ(I)]. Leydetite possesses a sheet structure based upon the protasite anion topology. The sheet consists of UO7 bipyramids, which share four of their equatorial vertices with SO4 tetrahedra. Each SO4 tetrahedron, in turn, shares two of its vertices with UO7 bipyramids. The remaining unshared equatorial vertex of the bipyramid is occupied by H2O, which extends hydrogen bonds within the sheet to one of a free vertex of the SO4 tetrahedron. Sheets are stacked perpendicular to the c direction. In the interlayer, Fe2+ ions and H2O groups link to the sheets on either side via a network of hydrogen bonds. Leydetite is isostructural with the synthetic compound Mg(UO2)(SO4)2(H2O)11. The name of the new mineral honours Jean Claude Leydet (born 1961), an amateur mineralogist from Brest (France), who discovered the new mineral.
11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.0224 for 5211 observed reflections with [I > 3σ(I)]. Leydetite possesses a sheet structure based upon the protasite anion topology. The sheet consists of UO7 bipyramids, which share four of their equatorial vertices with SO4 tetrahedra. Each SO4 tetrahedron, in turn, shares two of its vertices with UO7 bipyramids. The remaining unshared equatorial vertex of the bipyramid is occupied by H2O, which extends hydrogen bonds within the sheet to one of a free vertex of the SO4 tetrahedron. Sheets are stacked perpendicular to the c direction. In the interlayer, Fe2+ ions and H2O groups link to the sheets on either side via a network of hydrogen bonds. Leydetite is isostructural with the synthetic compound Mg(UO2)(SO4)2(H2O)11. The name of the new mineral honours Jean Claude Leydet (born 1961), an amateur mineralogist from Brest (France), who discovered the new mineral.




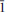 , with
, with 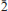 0); 3.097(49)(1
0); 3.097(49)(1
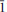 11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to
11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to