14 results
Double-swab 5% versus single-swab 10% iodophor for reducing methicillin-resistant Staphylococcus aureus with routine chlorhexidine bathing
- Lauren T. Heim, Loren G. Miller, Raveena D. Singh, James A. McKinnell, Tabitha D. Catuna, Marlene Estevez, Kaye D. Evans, Tom K. Tjoa, Gabrielle M. Gussin, Shaun D. Dahl, Linda Budy, Ellena M. Peterson, Susan S. Huang
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 12 / December 2022
- Published online by Cambridge University Press:
- 26 August 2021, pp. 1937-1939
- Print publication:
- December 2022
-
- Article
- Export citation
-
In a prospective cohort study, we compared a 2-swabs-per-nostril 5% iodophor regimen with a 1-swab-per-nostril 10% iodophor regimen on methicillin-resistant Staphylococcus aureus carriage in nursing-home residents. Compared with baseline, both single-swab and double-swab regimens resulted in an identical 40% reduction in nasal carriage and 60% reduction in any carriage, skin or nasal.
Results of a Multicenter Diagnostic Stewardship Collaborative to Optimize Blood Culture Use in Critically Ill Children
- Danielle Koontz, Charlotte Woods-Hill, Annie Voskertchian, Anping Xie, Marlene Miller, James Fackler, Elizabeth Colantuoni, Aaron Milstone, Ximin Li
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Group Name: Bright STAR Authorship Group
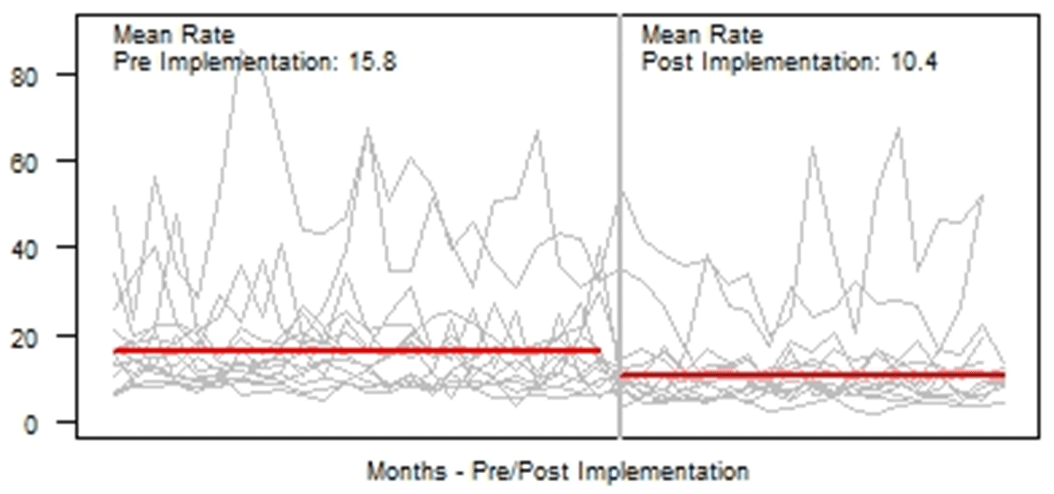
Background: Blood cultures are fundamental in the diagnosis and treatment of sepsis. Culture practices vary widely, and overuse can lead to false-positive results and unnecessary antibiotics. Our objective was to describe the implementation of a multisite quality improvement collaborative to reduce unnecessary blood cultures in pediatric intensive care unit (PICU) patients, and its 18-month impact on blood culture rates and safety metrics. Methods: In 2018, 14 PICUs joined the Blood Culture Improvement Guidelines and Diagnostic Stewardship for Antibiotic Reduction in Critically Ill Children (Bright STAR) Collaborative, designed to understand and improve blood culture practices in critically ill children. Guided by a centralized multidisciplinary study team, sites first reviewed existing evidence for safe reduction of unnecessary blood cultures and assessed local practices and barriers to change. Subsequently, local champions developed and implemented clinical decision-support tools informed by local patient needs to guide new blood-culture practices. The coordinating study team facilitated regular evaluations and discussions of project progress through monthly phone calls, site visits if requested by sites or the study team, and collaborative-wide teleconferences. The study team collected monthly blood culture rates and monitored for possible delays in obtaining blood cultures using a standardized review process as a safety balancing metric. We compared 24 months of baseline data to 18 months of postimplementation using a Poisson regression model accounting for the site-specific patient days and correlation of culture use within a site over time. Results: Across the 14 sites, before implementation, 41,768 blood cultures were collected over 259,701 PICU patient days. The mean preimplementation site-specific blood culture rate was 15.7 cultures per 100 patient days (rate range, 9.6–48.2 cultures per 100 patient days). After implementation, 22,397 blood cultures were collected over 208,171 PICU patient days. The mean postimplementation rate was 10.4 cultures per 100 patient days (rate range, 4.7–28.3 cultures per 100 patient days), which was 33.6% lower than the preimplementation (relative rate 0.66; 95% CI, 0.65–0.68 p <0.01). In 18 months post-implementation, sites reviewed 793 positive blood cultures, and identified only one suspected delay in culture collection possibly attributable to the site’s blood culture reduction program. Conclusions: Multidisciplinary quality improvement teams safely facilitated a 33.6% average reduction in blood culture use in critically ill children at 14 hospitals. Future collaborative work will determine the impact of blood culture diagnostic stewardship on antibiotic use and other important patient safety outcomes.
Funding: No
Disclosures: None
Figure 1.
Alcohol-impregnated caps and ambulatory central-line–associated bloodstream infections (CLABSIs): A randomized clinical trial
- Aaron M. Milstone, Carol Rosenberg, Gayane Yenokyan, Danielle W. Koontz, Marlene R. Miller, for the CCLIP Authorship Group
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 42 / Issue 4 / April 2021
- Published online by Cambridge University Press:
- 12 October 2020, pp. 431-439
- Print publication:
- April 2021
-
- Article
- Export citation
-
Objective:
To evaluate the effect of 70% isopropyl alcohol–impregnated central venous catheter caps on ambulatory central-line–associated bloodstream infections (CLABSIs) in pediatric hematology-oncology patients.
Design:This study was a 24-month, cluster-randomized, 2 period, crossover clinical trial.
Setting:The study was conducted in 15 pediatric healthcare institutions, including 16 pediatric hematology-oncology clinics.
Participants:All patients with an external central line followed at 1 of the 16 hematology-oncology clinics.
Intervention:Usual ambulatory central-line care per each institution using 70% isopropyl alcohol–impregnated caps at home compared to usual ambulatory central-line care in each institution without using 70% isopropyl alcohol–impregnated caps.
Results:Of the 16 participating clinics, 15 clinics completed both assignment periods. As assigned, there was no reduction in CLABSI incidence in clinics using 70% isopropyl alcohol–impregnated caps (1.23 per 1,000 days) compared with standard practices (1.38 per 1,000 days; adjusted incidence rate ratio [aIRR], 0.83; 95% CI, 0.63–1.11). In the per-protocol population, there was a reduction in positive blood culture incidence in clinics using 70% isopropyl alcohol-impregnated caps (1.51 per 1,000 days) compared with standard practices (1.88 per 1,000 days; aIRR, 0.72; 95% CI, 0.52–0.99). No adverse events were reported.
Conclusions:Isopropyl alcohol–impregnated central-line caps did not lead to a statistically significant reduction in CLABSI rates in ambulatory hematology-oncology patients. In the per-protocol analysis, there was a statistically significant decrease in positive blood cultures. Larger trials are needed to elucidate the impact of 70% isopropyl alcohol–impregnated caps in the ambulatory setting.
Registration:ClinicalTrials.gov; NCT02351258
Universal Decolonization Reduces MDRO Burden on High-Touch Objects in Nursing Home Resident Rooms and Common Areas
- Gabrielle M. Gussin, Raveena D. Singh, Raheeb Saavedra, Tabitha D. Catuna, Lauren Heim, Job Mendez, Ryan Franco, Marlene Estevez, Harold Custodio, Kaye D. Evans, Ellena M. Peterson, James A. McKinnell, Loren Miller, Susan Huang
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s54-s55
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: More than half of nursing home (NH) residents harbor a multidrug-resistant organism (MDRO), and MDRO contamination of the environment is common. Whether NH decolonization of residents reduces MDRO contamination remains unclear. The PROTECT trial was a cluster-randomized trial of decolonization versus routine care in 28 California NHs from April 2017 through December 2018. Decolonization involved chlorhexidine bathing plus nasal iodophor (Monday–Friday, every other week), and it reduced resident nares and skin MDRO colonization by 36%. Methods: We swabbed high-touch objects in resident rooms and common areas for MDROs before and after the 3-month decolonization phase-in (April–July 2017). Five high-touch objects (bedrail, call button and TV remote, doorknob, light switch, and bathroom handles) were swabbed in 3 resident rooms per NH based on care needs (Alzheimer’s disease and related dementias (ADRD), ie, total care; ADRD, ambulatory care; and short stay). Five high-touch objects were also swabbed in the common area (nursing station, table, chair, railing, and drinking fountain). Swabs were processed for methicillin-resistant S. aureus (MRSA), vancomycin-resistant Enterococcus (VRE), extended-spectrum β-lactamase (ESBL) producing Enterobacteriaceae, and carbapenem-resistant Enterobacteriaceae (CRE). We used generalized linear mixed models to assess the impact of decolonization on MDRO environmental contamination when clustering by NH and room and adjusting for room type and object because unclustered and unadjusted results are likely to be inaccurate. Results: A high proportion of rooms were contaminated with any MDRO in control NHs: 43 of 56 (77%) in the baseline period and 46 of 56 (82%) in the intervention period. In contrast, decolonization NHs had similar baseline contamination (45 of 56, 80%) but lower intervention MDRO contamination (29 of 48, 60%). When evaluating the intervention impact using multivariable models, decolonization was associated with significantly less room contamination for any MDRO (OR, 0.25; 95% CI, 0.06–0.96; P = .04) and MRSA (OR, 0.16; 95% CI, 0.05–0.55; P = .004) but nonsignificant reductions in VRE contamination (OR, 0.86; 95% CI, 0.23–3.13) and ESBL contamination (OR, 0.13; 95% CI, 0.01–1.62). CRE was not modeled due to rare counts (2 rooms total). In addition, room type was important, with common areas associated with 5-fold, 9-fold, and 3-fold higher contamination with any MDRO, MRSA, and VRE, respectively, compared with short-stay rooms. Conclusions: The high burden of MDROs in NHs calls for universal prevention strategies that can protect all residents. Although decolonization was associated with an 84% reduction in odds of MRSA contamination of inanimate room objects, significant reductions in VRE or ESBL contamination were not seen, possibly due to the lower proportion of baseline contamination due to these organisms. Multimodal strategies are needed to address high levels of MDRO contamination in NHs.
Funding: None
Disclosures: Gabrielle Gussin: Stryker (Sage Products): Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Clorox: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Medline: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Xttrium: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes.
Impact of Roommates on MDRO Spread in Nursing Homes
- Gabrielle M. Gussin, Ken Kleinman, Raveena D. Singh, Raheeb Saavedra, Lauren Heim, Marlene Estevez, Tabitha D. Catuna, Eunjung Lee, Avy Osalvo, Kaye D. Evans, Julie A. Shimabukuro, James A. McKinnell, Loren Miller, Cassiana E. Bittencourt, Ellena M. Peterson, Susan Huang
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s66-s67
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Addressing the high burden of multidrug-resistant organisms (MDROs) in nursing homes is a public health priority. High interfacility transmission may be attributed to inadequate infection prevention practices, shared living spaces, and frequent care needs. We assessed the contribution of roommates to the likelihood of MDRO carriage in nursing homes. Methods: We performed a secondary analysis of the SHIELD OC (Shared Healthcare Intervention to Eliminate Life-threatening Dissemination of MDROs in Orange County, CA) Project, a CDC-funded regional decolonization intervention to reduce MDROs among 38 regional facilities (18 nursing homes, 3 long-term acute-care hospitals, and 17 hospitals). Decolonization in participating nursing homes involved routine chlorhexidine bathing plus nasal iodophor (Monday through Friday, twice daily every other week) from April 2017 through July 2019. MDRO point-prevalence assessments involving all residents at 16 nursing homes conducted at the end of the intervention period were used to determine whether having a roommate was associated with MDRO carriage. Nares, bilateral axilla/groin, and perirectal swabs were processed for methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococcus (VRE), extended-spectrum β-lactamase (ESBL)–producing Enterobacteriaceae, and carbapenem-resistant Enterobacteriaceae (CRE). Generalized linear mixed models assessed the impact of maximum room occupancy on MDRO prevalence when clustering by room and hallway, and adjusting for the following factors: nursing home facility, age, gender, length-of-stay at time of swabbing, bedbound status, known MDRO history, and presence of urinary or gastrointestinal devices. CRE models were not run due to low counts. Results: During the intervention phase, 1,451 residents were sampled across 16 nursing homes. Overall MDRO prevalence was 49%. In multivariable models, we detected a significant increasing association of maximum room occupants and MDRO carriage for MRSA but not other MDROs. For MRSA, the adjusted odds ratios for quadruple-, triple-, and double-occupancy rooms were 3.5, 3.6, and 2.8, respectively, compared to residents in single rooms (P = .013). For VRE, these adjusted odds ratios were 0.3, 0.3, and 0.4, respectively, compared to residents in single rooms (P = NS). For ESBL, the adjusted odds ratios were 0.9, 1.1, and 1.5, respectively, compared to residents in single rooms (P = nonsignificant). Conclusions: Nursing home residents in shared rooms were more likely to harbor MRSA, suggesting MRSA transmission between roommates. Although decolonization was previously shown to reduce MDRO prevalence by 22% in SHIELD nursing homes, this strategy did not appear to prevent all MRSA transmission between roommates. Additional efforts involving high adherence hand hygiene, environmental cleaning, and judicious use of contact precautions are likely needed to reduce transmission between roommates in nursing homes.
Funding: None
Disclosures: Gabrielle M. Gussin, Stryker (Sage Products): Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Clorox: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Medline: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Xttrium: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes.
Decreased Hospitalizations and Costs From Infection in Sixteen Nursing Homes in the SHIELD OC Regional Decolonization Initiative
- Gabrielle M. Gussin, James A. McKinnell, Raveena D. Singh, Ken Kleinman, Amherst Loren Miller, Raheeb Saavedra, Lauren Heim, Marlene Estevez, Tabitha D. Catuna, Eunjung Lee, Thomas Tjoa, Rachel Slayton, Nimalie Stone, John Jernigan, Matthew Zahn, Lynn Janssen, Shruti K Gohil, Philip Alan Robinson, Steven Park, Robert Weinstein, Mary Hayden, Cassiana E. Bittencourt, Ellena M. Peterson, Susan Huang
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s7-s8
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Distinguished Oral
Background: Shared Healthcare Intervention to Eliminate Life-threatening Dissemination of MDROs in Orange County, California (SHIELD OC) was a CDC-funded regional decolonization intervention from April 2017 through July 2019 involving 38 hospitals, nursing homes (NHs), and long-term acute-care hospitals (LTACHs) to reduce MDROs. Decolonization in NH and LTACHs consisted of universal antiseptic bathing with chlorhexidine (CHG) for routine bathing and showering plus nasal iodophor decolonization (Monday through Friday, twice daily every other week). Hospitals used universal CHG in ICUs and provided daily CHG and nasal iodophor to patients in contact precautions. We sought to evaluate whether decolonization reduced hospitalization and associated healthcare costs due to infections among residents of NHs participating in SHIELD compared to nonparticipating NHs. Methods: Medicaid insurer data covering NH residents in Orange County were used to calculate hospitalization rates due to a primary diagnosis of infection (counts per member quarter), hospital bed days/member-quarter, and expenditures/member quarter from the fourth quarter of 2015 to the second quarter of 2019. We used a time-series design and a segmented regression analysis to evaluate changes attributable to the SHIELD OC intervention among participating and nonparticipating NHs. Results: Across the SHIELD OC intervention period, intervention NHs experienced a 44% decrease in hospitalization rates, a 43% decrease in hospital bed days, and a 53% decrease in Medicaid expenditures when comparing the last quarter of the intervention to the baseline period (Fig. 1). These data translated to a significant downward slope, with a reduction of 4% per quarter in hospital admissions due to infection (P < .001), a reduction of 7% per quarter in hospitalization days due to infection (P < .001), and a reduction of 9% per quarter in Medicaid expenditures (P = .019) per NH resident. Conclusions: The universal CHG bathing and nasal decolonization intervention adopted by NHs in the SHIELD OC collaborative resulted in large, meaningful reductions in hospitalization events, hospitalization days, and healthcare expenditures among Medicaid-insured NH residents. The findings led CalOptima, the Medicaid provider in Orange County, California, to launch an NH incentive program that provides dedicated training and covers the cost of CHG and nasal iodophor for OC NHs that enroll.
Funding: None
Disclosures: Gabrielle M. Gussin, University of California, Irvine, Stryker (Sage Products): Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Clorox: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Medline: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Xttrium: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes.
Bright STAR Collaborative Consensus Guidelines for Blood Culture Use in Critically Ill Children
- Charlotte Woods-Hill, Danielle Koontz, Annie Voskertchian, Marlene Miller, James Fackler, Judy Shea, Aaron Michael Milstone
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s22-s23
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Blood cultures are essential diagnostic tools used to identify bloodstream infections and to guide antimicrobial therapy. However, collecting cultures without clear indications or that do not inform management can lead to false-positive results and unnecessary use of antibiotics. Blood culture practices vary significantly in critically ill children. Our objective was to create a consensus guideline focusing on when to safely avoid blood cultures in pediatric intensive care unit (PICU) patients. Methods: A panel of multidisciplinary experts, many participating in the Blood Culture Improvement Guidelines and Diagnostic Stewardship for Antibiotic Reduction in Critically Ill Children (Bright STAR) Collaborative, engaged in a 2-part modified Delphi process. Round 1 consisted of a preparatory literature summary and an electronic survey sent to subject matter experts (SMEs). In the survey, SMEs rated a series of recommendations about when to avoid blood cultures on a 5-point Likert scale, 1 being the lowest score and 5 being the highest score. Consensus was achieved for each recommendation if 75% of respondents chose a score of 4 or 5, and these were included in the final guideline. Any recommendations that did not meet these a priori criteria for consensus were set aside for discussion during the in-person expert panel review (round 2). An outside expert in consensus methodology facilitated round 2. After a review of the survey results and comments from round 1 and group discussion, the SMEs voted on these recommendations in real time. Voting was blinded. Participants included Bright STAR site leads, national content experts, and representatives from relevant national societies. Results: We received 29 completed surveys from 34 invited participants for an 85% response rate. Of the 27 round 1 recommendations, 18 met predetermined criteria for consensus. Round 2 included 26 in-person voting participants who (1) discussed and modified the 9 recommendations that had not met round 1 consensus, and (2) modified for clarity or condensed from multiple into single recommendations the 18 recommendations that had met the round 1 consensus. The final document contains 19 recommendations that provide guidance on how to safely improve blood culture use in PICU patients (Table 1). Also, 8 recommendations discussed did not reach consensus for inclusion. Conclusions: Using a modified Delphi process, we created consensus recommendations on when to avoid blood cultures and prevent overuse in critically ill children. These guidelines are a critical step in disseminating diagnostic stewardship and reducing unnecessary testing on a wider scale.
Funding: Agency for Healthcare Research and Quality, R18 HS025642-01, 9/2017 – 9/2020 (Aaron Milstone, PI)
Disclosures: None
A Prospective, Holistic, Multicenter Approach to Tracking and Understanding Bloodstream Infections in Pediatric Hematology-Oncology Patients
- Aditya H. Gaur, David G. Bundy, Eric J. Werner, Jeffrey D. Hord, Marlene R. Miller, Li Tang, John P. Lawlor, Amy L. Billett, Children’s Hospital Association Childhood Cancer & Blood Disorders Network (CCBDN)
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 38 / Issue 6 / June 2017
- Published online by Cambridge University Press:
- 12 April 2017, pp. 690-696
- Print publication:
- June 2017
-
- Article
-
- You have access Access
- HTML
- Export citation
-
OBJECTIVE
To assess the burden of bloodstream infections (BSIs) among pediatric hematology-oncology (PHO) inpatients, to propose a comprehensive, all-BSI tracking approach, and to discuss how such an approach helps better inform within-center and across-center differences in CLABSI rate
DESIGNProspective cohort study
SETTINGUS multicenter, quality-improvement, BSI prevention network
PARTICIPANTSPHO centers across the United States who agreed to follow a standardized central-line–maintenance care bundle and track all BSI events and central-line days every month.
METHODSInfections were categorized as CLABSI (stratified by mucosal barrier injury–related, laboratory-confirmed BSI [MBI-LCBI] versus non–MBI-LCBI) and secondary BSI, using National Healthcare Safety Network (NHSN) definitions. Single positive blood cultures (SPBCs) with NHSN defined common commensals were also tracked.
RESULTSBetween 2013 and 2015, 34 PHO centers reported 1,110 BSIs. Among them, 708 (63.8%) were CLABSIs, 170 (15.3%) were secondary BSIs, and 232 (20.9%) were SPBCs. Most SPBCs (75%) occurred in patients with profound neutropenia; 22% of SPBCs were viridans group streptococci. Among the CLABSIs, 51% were MBI-LCBI. Excluding SPBCs, CLABSI rates were higher (88% vs 77%) and secondary BSI rates were lower (12% vs 23%) after the NHSN updated the definition of secondary BSI (P<.001). Preliminary analyses showed across-center differences in CLABSI versus secondary BSI and between SPBC and CLABSI versus non-CLABSI rates.
CONCLUSIONSTracking all BSIs, not just CLABSIs in PHO patients, is a patient-centered, clinically relevant approach that could help better assess across-center and within-center differences in infection rates, including CLABSI. This approach enables informed decision making by healthcare providers, payors, and the public.
Infect Control Hosp Epidemiol 2017;38:690–696
Prevalence of and Factors Associated With Multidrug Resistant Organism (MDRO) Colonization in 3 Nursing Homes
- James A. McKinnell, Loren G. Miller, Raveena Singh, Ken Kleinman, Ellena M. Peterson, Kaye D. Evans, Tabitha D. Dutciuc, Lauren Heim, Adrijana Gombosev, Marlene Estevez, Bryn Launer, Tom Tjoa, Steven Tam, Michael A. Bolaris, Susan S. Huang
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 37 / Issue 12 / December 2016
- Published online by Cambridge University Press:
- 27 September 2016, pp. 1485-1488
- Print publication:
- December 2016
-
- Article
- Export citation
-
Nursing home residents are at risk for acquiring and transmitting MDROs. A serial point-prevalence study of 605 residents in 3 facilities using random sampling found MDRO colonization in 45% of residents: methicillin-resistant Staphylococcus aureus (MRSA, 26%); extended-spectrum β-lactamase–producing Enterobacteriaceae (ESBL, 17%); vancomycin-resistant Enterococcus spp. (VRE, 16%); carbapenem-resistant Enterobacteriaceae (CRE, 1%). MDRO colonization was associated with history of MDRO, care needs, incontinence, and catheters.
Infect Control Hosp Epidemiol 2016;1485–1488
Contributors
-
- By Mitchell Aboulafia, Frederick Adams, Marilyn McCord Adams, Robert M. Adams, Laird Addis, James W. Allard, David Allison, William P. Alston, Karl Ameriks, C. Anthony Anderson, David Leech Anderson, Lanier Anderson, Roger Ariew, David Armstrong, Denis G. Arnold, E. J. Ashworth, Margaret Atherton, Robin Attfield, Bruce Aune, Edward Wilson Averill, Jody Azzouni, Kent Bach, Andrew Bailey, Lynne Rudder Baker, Thomas R. Baldwin, Jon Barwise, George Bealer, William Bechtel, Lawrence C. Becker, Mark A. Bedau, Ernst Behler, José A. Benardete, Ermanno Bencivenga, Jan Berg, Michael Bergmann, Robert L. Bernasconi, Sven Bernecker, Bernard Berofsky, Rod Bertolet, Charles J. Beyer, Christian Beyer, Joseph Bien, Joseph Bien, Peg Birmingham, Ivan Boh, James Bohman, Daniel Bonevac, Laurence BonJour, William J. Bouwsma, Raymond D. Bradley, Myles Brand, Richard B. Brandt, Michael E. Bratman, Stephen E. Braude, Daniel Breazeale, Angela Breitenbach, Jason Bridges, David O. Brink, Gordon G. Brittan, Justin Broackes, Dan W. Brock, Aaron Bronfman, Jeffrey E. Brower, Bartosz Brozek, Anthony Brueckner, Jeffrey Bub, Lara Buchak, Otavio Bueno, Ann E. Bumpus, Robert W. Burch, John Burgess, Arthur W. Burks, Panayot Butchvarov, Robert E. Butts, Marina Bykova, Patrick Byrne, David Carr, Noël Carroll, Edward S. Casey, Victor Caston, Victor Caston, Albert Casullo, Robert L. Causey, Alan K. L. Chan, Ruth Chang, Deen K. Chatterjee, Andrew Chignell, Roderick M. Chisholm, Kelly J. Clark, E. J. Coffman, Robin Collins, Brian P. Copenhaver, John Corcoran, John Cottingham, Roger Crisp, Frederick J. Crosson, Antonio S. Cua, Phillip D. Cummins, Martin Curd, Adam Cureton, Andrew Cutrofello, Stephen Darwall, Paul Sheldon Davies, Wayne A. Davis, Timothy Joseph Day, Claudio de Almeida, Mario De Caro, Mario De Caro, John Deigh, C. F. Delaney, Daniel C. Dennett, Michael R. DePaul, Michael Detlefsen, Daniel Trent Devereux, Philip E. Devine, John M. Dillon, Martin C. Dillon, Robert DiSalle, Mary Domski, Alan Donagan, Paul Draper, Fred Dretske, Mircea Dumitru, Wilhelm Dupré, Gerald Dworkin, John Earman, Ellery Eells, Catherine Z. Elgin, Berent Enç, Ronald P. Endicott, Edward Erwin, John Etchemendy, C. Stephen Evans, Susan L. Feagin, Solomon Feferman, Richard Feldman, Arthur Fine, Maurice A. Finocchiaro, William FitzPatrick, Richard E. Flathman, Gvozden Flego, Richard Foley, Graeme Forbes, Rainer Forst, Malcolm R. Forster, Daniel Fouke, Patrick Francken, Samuel Freeman, Elizabeth Fricker, Miranda Fricker, Michael Friedman, Michael Fuerstein, Richard A. Fumerton, Alan Gabbey, Pieranna Garavaso, Daniel Garber, Jorge L. A. Garcia, Robert K. Garcia, Don Garrett, Philip Gasper, Gerald Gaus, Berys Gaut, Bernard Gert, Roger F. Gibson, Cody Gilmore, Carl Ginet, Alan H. Goldman, Alvin I. Goldman, Alfonso Gömez-Lobo, Lenn E. Goodman, Robert M. Gordon, Stefan Gosepath, Jorge J. E. Gracia, Daniel W. Graham, George A. Graham, Peter J. Graham, Richard E. Grandy, I. Grattan-Guinness, John Greco, Philip T. Grier, Nicholas Griffin, Nicholas Griffin, David A. Griffiths, Paul J. Griffiths, Stephen R. Grimm, Charles L. Griswold, Charles B. Guignon, Pete A. Y. Gunter, Dimitri Gutas, Gary Gutting, Paul Guyer, Kwame Gyekye, Oscar A. Haac, Raul Hakli, Raul Hakli, Michael Hallett, Edward C. Halper, Jean Hampton, R. James Hankinson, K. R. Hanley, Russell Hardin, Robert M. Harnish, William Harper, David Harrah, Kevin Hart, Ali Hasan, William Hasker, John Haugeland, Roger Hausheer, William Heald, Peter Heath, Richard Heck, John F. Heil, Vincent F. Hendricks, Stephen Hetherington, Francis Heylighen, Kathleen Marie Higgins, Risto Hilpinen, Harold T. Hodes, Joshua Hoffman, Alan Holland, Robert L. Holmes, Richard Holton, Brad W. Hooker, Terence E. Horgan, Tamara Horowitz, Paul Horwich, Vittorio Hösle, Paul Hoβfeld, Daniel Howard-Snyder, Frances Howard-Snyder, Anne Hudson, Deal W. Hudson, Carl A. Huffman, David L. Hull, Patricia Huntington, Thomas Hurka, Paul Hurley, Rosalind Hursthouse, Guillermo Hurtado, Ronald E. Hustwit, Sarah Hutton, Jonathan Jenkins Ichikawa, Harry A. Ide, David Ingram, Philip J. Ivanhoe, Alfred L. Ivry, Frank Jackson, Dale Jacquette, Joseph Jedwab, Richard Jeffrey, David Alan Johnson, Edward Johnson, Mark D. Jordan, Richard Joyce, Hwa Yol Jung, Robert Hillary Kane, Tomis Kapitan, Jacquelyn Ann K. Kegley, James A. Keller, Ralph Kennedy, Sergei Khoruzhii, Jaegwon Kim, Yersu Kim, Nathan L. King, Patricia Kitcher, Peter D. Klein, E. D. Klemke, Virginia Klenk, George L. Kline, Christian Klotz, Simo Knuuttila, Joseph J. Kockelmans, Konstantin Kolenda, Sebastian Tomasz Kołodziejczyk, Isaac Kramnick, Richard Kraut, Fred Kroon, Manfred Kuehn, Steven T. Kuhn, Henry E. Kyburg, John Lachs, Jennifer Lackey, Stephen E. Lahey, Andrea Lavazza, Thomas H. Leahey, Joo Heung Lee, Keith Lehrer, Dorothy Leland, Noah M. Lemos, Ernest LePore, Sarah-Jane Leslie, Isaac Levi, Andrew Levine, Alan E. Lewis, Daniel E. Little, Shu-hsien Liu, Shu-hsien Liu, Alan K. L. Chan, Brian Loar, Lawrence B. Lombard, John Longeway, Dominic McIver Lopes, Michael J. Loux, E. J. Lowe, Steven Luper, Eugene C. Luschei, William G. Lycan, David Lyons, David Macarthur, Danielle Macbeth, Scott MacDonald, Jacob L. Mackey, Louis H. Mackey, Penelope Mackie, Edward H. Madden, Penelope Maddy, G. B. Madison, Bernd Magnus, Pekka Mäkelä, Rudolf A. Makkreel, David Manley, William E. Mann (W.E.M.), Vladimir Marchenkov, Peter Markie, Jean-Pierre Marquis, Ausonio Marras, Mike W. Martin, A. P. Martinich, William L. McBride, David McCabe, Storrs McCall, Hugh J. McCann, Robert N. McCauley, John J. McDermott, Sarah McGrath, Ralph McInerny, Daniel J. McKaughan, Thomas McKay, Michael McKinsey, Brian P. McLaughlin, Ernan McMullin, Anthonie Meijers, Jack W. Meiland, William Jason Melanson, Alfred R. Mele, Joseph R. Mendola, Christopher Menzel, Michael J. Meyer, Christian B. Miller, David W. Miller, Peter Millican, Robert N. Minor, Phillip Mitsis, James A. Montmarquet, Michael S. Moore, Tim Moore, Benjamin Morison, Donald R. Morrison, Stephen J. Morse, Paul K. Moser, Alexander P. D. Mourelatos, Ian Mueller, James Bernard Murphy, Mark C. Murphy, Steven Nadler, Jan Narveson, Alan Nelson, Jerome Neu, Samuel Newlands, Kai Nielsen, Ilkka Niiniluoto, Carlos G. Noreña, Calvin G. Normore, David Fate Norton, Nikolaj Nottelmann, Donald Nute, David S. Oderberg, Steve Odin, Michael O’Rourke, Willard G. Oxtoby, Heinz Paetzold, George S. Pappas, Anthony J. Parel, Lydia Patton, R. P. Peerenboom, Francis Jeffry Pelletier, Adriaan T. Peperzak, Derk Pereboom, Jaroslav Peregrin, Glen Pettigrove, Philip Pettit, Edmund L. Pincoffs, Andrew Pinsent, Robert B. Pippin, Alvin Plantinga, Louis P. Pojman, Richard H. Popkin, John F. Post, Carl J. Posy, William J. Prior, Richard Purtill, Michael Quante, Philip L. Quinn, Philip L. Quinn, Elizabeth S. Radcliffe, Diana Raffman, Gerard Raulet, Stephen L. Read, Andrews Reath, Andrew Reisner, Nicholas Rescher, Henry S. Richardson, Robert C. Richardson, Thomas Ricketts, Wayne D. Riggs, Mark Roberts, Robert C. Roberts, Luke Robinson, Alexander Rosenberg, Gary Rosenkranz, Bernice Glatzer Rosenthal, Adina L. Roskies, William L. Rowe, T. M. Rudavsky, Michael Ruse, Bruce Russell, Lilly-Marlene Russow, Dan Ryder, R. M. Sainsbury, Joseph Salerno, Nathan Salmon, Wesley C. Salmon, Constantine Sandis, David H. Sanford, Marco Santambrogio, David Sapire, Ruth A. Saunders, Geoffrey Sayre-McCord, Charles Sayward, James P. Scanlan, Richard Schacht, Tamar Schapiro, Frederick F. Schmitt, Jerome B. Schneewind, Calvin O. Schrag, Alan D. Schrift, George F. Schumm, Jean-Loup Seban, David N. Sedley, Kenneth Seeskin, Krister Segerberg, Charlene Haddock Seigfried, Dennis M. Senchuk, James F. Sennett, William Lad Sessions, Stewart Shapiro, Tommie Shelby, Donald W. Sherburne, Christopher Shields, Roger A. Shiner, Sydney Shoemaker, Robert K. Shope, Kwong-loi Shun, Wilfried Sieg, A. John Simmons, Robert L. Simon, Marcus G. Singer, Georgette Sinkler, Walter Sinnott-Armstrong, Matti T. Sintonen, Lawrence Sklar, Brian Skyrms, Robert C. Sleigh, Michael Anthony Slote, Hans Sluga, Barry Smith, Michael Smith, Robin Smith, Robert Sokolowski, Robert C. Solomon, Marta Soniewicka, Philip Soper, Ernest Sosa, Nicholas Southwood, Paul Vincent Spade, T. L. S. Sprigge, Eric O. Springsted, George J. Stack, Rebecca Stangl, Jason Stanley, Florian Steinberger, Sören Stenlund, Christopher Stephens, James P. Sterba, Josef Stern, Matthias Steup, M. A. Stewart, Leopold Stubenberg, Edith Dudley Sulla, Frederick Suppe, Jere Paul Surber, David George Sussman, Sigrún Svavarsdóttir, Zeno G. Swijtink, Richard Swinburne, Charles C. Taliaferro, Robert B. Talisse, John Tasioulas, Paul Teller, Larry S. Temkin, Mark Textor, H. S. Thayer, Peter Thielke, Alan Thomas, Amie L. Thomasson, Katherine Thomson-Jones, Joshua C. Thurow, Vzalerie Tiberius, Terrence N. Tice, Paul Tidman, Mark C. Timmons, William Tolhurst, James E. Tomberlin, Rosemarie Tong, Lawrence Torcello, Kelly Trogdon, J. D. Trout, Robert E. Tully, Raimo Tuomela, John Turri, Martin M. Tweedale, Thomas Uebel, Jennifer Uleman, James Van Cleve, Harry van der Linden, Peter van Inwagen, Bryan W. Van Norden, René van Woudenberg, Donald Phillip Verene, Samantha Vice, Thomas Vinci, Donald Wayne Viney, Barbara Von Eckardt, Peter B. M. Vranas, Steven J. Wagner, William J. Wainwright, Paul E. Walker, Robert E. Wall, Craig Walton, Douglas Walton, Eric Watkins, Richard A. Watson, Michael V. Wedin, Rudolph H. Weingartner, Paul Weirich, Paul J. Weithman, Carl Wellman, Howard Wettstein, Samuel C. Wheeler, Stephen A. White, Jennifer Whiting, Edward R. Wierenga, Michael Williams, Fred Wilson, W. Kent Wilson, Kenneth P. Winkler, John F. Wippel, Jan Woleński, Allan B. Wolter, Nicholas P. Wolterstorff, Rega Wood, W. Jay Wood, Paul Woodruff, Alison Wylie, Gideon Yaffe, Takashi Yagisawa, Yutaka Yamamoto, Keith E. Yandell, Xiaomei Yang, Dean Zimmerman, Günter Zoller, Catherine Zuckert, Michael Zuckert, Jack A. Zupko (J.A.Z.)
- Edited by Robert Audi, University of Notre Dame, Indiana
-
- Book:
- The Cambridge Dictionary of Philosophy
- Published online:
- 05 August 2015
- Print publication:
- 27 April 2015, pp ix-xxx
-
- Chapter
- Export citation
Evaluating Application of the National Healthcare Safety Network Central Line—Associated Bloodstream Infection Surveillance Definition: A Survey of Pediatric Intensive Care and Hematology/Oncology Units
- Aditya H. Gaur, Marlene R. Miller, Cuilan Gao, Carol Rosenberg, Gloria C. Morrell, Susan E. Coffin, W. Charles Huskins
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 34 / Issue 7 / July 2013
- Published online by Cambridge University Press:
- 02 January 2015, pp. 663-670
- Print publication:
- July 2013
-
- Article
- Export citation
-
Objective.
To evaluate the application of the National Healthcare Safety Network (NHSN) central line-associated bloodstream infection (CLABSI) definition in pediatric intensive care units (PICUs) and pediatric hematology/oncology units (PHOUs) participating in a multicenter quality improvement collaborative to reduce CLABSIs; to identify sources of variability in the application of the definition.
Design.Online survey using 18 standardized case scenarios. Each described a positive blood culture in a patient and required a yes-or-no answer to the question “Is this a CLABSI?” NHSN staff responses were the reference standard.
Setting.Sixty-five US PICUs and PHOUs.
Participants.Staff who routinely adjudicate CLABSIs using NHSN definitions.
Results.Sixty responses were received from 58 (89%) of 65 institutions; 78% of respondents were infection preventionists, infection control officers, or infectious disease physicians. Responses matched those of NHSN staff for 78% of questions. The mean (SE) percentage of concurring answers did not differ for scenarios evaluating application of 1 of the 3 criteria (“known pathogen,” 78% [1.7%]; “skin contaminant, >1 year of age,” 76% [SE, 2.5%]; “skin contaminant, ≤1 year of age,” 81% [3.8%]; P = .3 ). The mean percentage of concurring answers was lower for scenarios requiring respondents to determine whether a CLABSI was present or incubating on admission (64% [4.6%]; P = .017) or to distinguish between primary and secondary bacteremia (65% [2.5%]; P = .021).
Conclusions.The accuracy of application of the CLABSI definition was suboptimal. Efforts to reduce variability in identifying CLABSIs that are present or incubating on admission and in distinguishing primary from secondary bloodstream infection are needed.
Surveillance of Hospital-Acquired Central Line–Associated Bloodstream Infections in Pediatric Hematology-Oncology Patients Lessons Learned, Challenges Ahead
- Aditya H. Gaur, David G. Bundy, Cuilan Gao, Eric J. Werner, Amy L. Billett, Jeff D. Hord, Jane D. Siegel, David Dickens, Cindi Winkle, Marlene R. Miller, Children's Hospital Association Hematology-Oncology Quality Transformation Collaborative Project
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 34 / Issue 3 / March 2013
- Published online by Cambridge University Press:
- 02 January 2015, pp. 315-320
- Print publication:
- March 2013
-
- Article
- Export citation
-
Across 36 US pediatric oncology centers, 576 central line-associated bloodstream infections (CLABSIs) were reported over a 21-month period. Most infections occurred in those with leukemia and/or profound neutropenia. The contribution of viridans streptococci infections was striking. Study findings depict the contemporary epidemiology of CLABSIs in hospitalized pediatric cancer patients.
Epidemiology of Central Line-Associated Bloodstream Infections in the Pediatric Intensive Care Unit
- Matthew F. Niedner, W. Charles Huskins, Elizabeth Colantuoni, John Muschelli, J. Mitchell Harris II, Tom B. Rice, Richard J. Brilli, Marlene R. Miller
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 32 / Issue 12 / December 2011
- Published online by Cambridge University Press:
- 02 January 2015, pp. 1200-1208
- Print publication:
- December 2011
-
- Article
- Export citation
-
Objective.
Describe central line-associated bloodstream infection (CLA-BSI) epidemiology in pediatric intensive care units (PICUs).
Design.Descriptive study (29 PICUs); cohort study (18 PICUs).
Setting.PICUs in a national improvement collaborative.
Patients/Participants.Patients admitted October 2006 to December 2007 with 1 or more central lines.
Methods.CLA-BSIs were prospectively identified using the National Healthcare Safety Network definition and then readjudicated using the revised 2008 definition. Risk factors for CLA-BSI were examined using age-adjusted, time-varying Cox proportional hazards models.
Results.In the descriptive study, the CLA-BSI incidence was 3.1/1,000 central line-days; readjudication with the revised definition resulted in a 17% decrease. In the cohort study, the readjudicated incidence was 2.0/1,000 central line-days. Ninety-nine percent of patients were CLA-BSI-free through day 7, after which the daily risk of CLA-BSI doubled to 0.27% per day. Compared with patients with respiratory diagnoses (most prevalent category), CLA-BSI risk was higher in patients with gastrointestinal diagnoses (hazard ratio [HR], 2.7 [95% confidence interval {CI}, 1.43-5.16]; P<.002) and oncologic diagnoses (HR, 2.6 [CI, 1.06-6.45]; P=.037). Among all patients, including those with more than 1 central line, CLA-BSI risk was lower among patients with a central line inserted in the jugular vein (HR, 0.43 [CI, 0.30-0.95]; P<.03).
Conclusions.The 2008 CLA-BSI definition change decreased the measured incidence. The daily CLA-BSI risk was very low in patients during the first 7 days of catheterization but doubled thereafter. The risk of CLA-BSI was lower in patients with lines inserted in the jugular vein and higher in patients with gastrointestinal and oncologic diagnoses. These patients are target populations for additional study and intervention.
Increased Catheter-Related Bloodstream Infection Rates After the Introduction of a New Mechanical Valve Intravenous Access Port
- Lisa L. Maragakis, Karen L. Bradley, Xiaoyan Song, Claire Beers, Marlene R. Miller, Sara E. Cosgrove, Trish M. Perl
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 27 / Issue 1 / January 2006
- Published online by Cambridge University Press:
- 21 June 2016, pp. 67-70
- Print publication:
- January 2006
-
- Article
- Export citation
-
The technology of intravenous catheter access ports has evolved from open ports covered by removable caps to more-sophisticated, closed versions containing mechanical valves. We report a significant increase in catheter-related bloodstream infections after the introduction of a new needle-free positive-pressure mechanical valve intravenous access port at our institution.




