3 results
Rapid improvements in MADRS with zuranolone in major depressive disorder and postpartum depression: results from the LANDSCAPE/NEST clinical development programmes
- A. H. Clayton, K. M. Deligiannidis, J. A. Ramos-Quiroga, R. Lasser, A. J. Sankoh, B. Leclair, M. Kotecha, J. Doherty
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S93-S94
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Rapid-acting therapies remain an unmet need in the treatment of major depressive disorder (MDD) and postpartum depression (PPD). Zuranolone (ZRN) is being evaluated as a once-daily, oral, 14-day treatment for adult patients with MDD and PPD.
ObjectivesTo evaluate the efficacy (assessed by Montgomery–Åsberg Depression Rating Scale [MADRS]) and safety of ZRN versus placebo across clinical studies with MDD and PPD.
MethodsIn 5 completed Phase 2/3 placebo-controlled randomised studies of once-daily ZRN 30 or 50 mg in adults with MDD or PPD, improvement in depressive symptoms was assessed at Day 15 (end of 14-day treatment) by change from baseline in MADRS total score and the percentage of patients achieving MADRS response (≥ 50% improvement from baseline in total score) and remission (total score ≤ 10). Safety was assessed throughout.
ResultsPatients in the ZRN arm achieved improvements in depressive symptoms, as assessed by MADRS. Improvements in MADRS total score at Day 15 were observed in all 5 studies and were nominally significant (p < 0.05) versus placebo in 4 studies (Fig. 1). Percentage of patients achieving response and/or remission in the ZRN arm was numerically greater than placebo in all MDD studies and significantly greater than placebo in the PPD studies (Fig. 2 and 3). ZRN was generally well tolerated with consistent safety and tolerability profiles across studies. The most common treatment-emergent adverse events (≥5 % in ZRN treatment arm) were headache, somnolence, dizziness, nausea, sedation, diarrhea, upper respiratory tract infection, fatigue, and COVID-19.
Image:

Image 2:
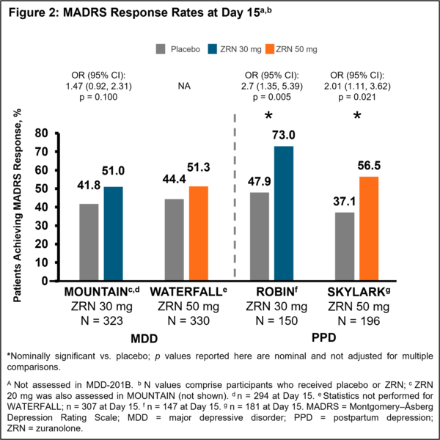
Image 3:
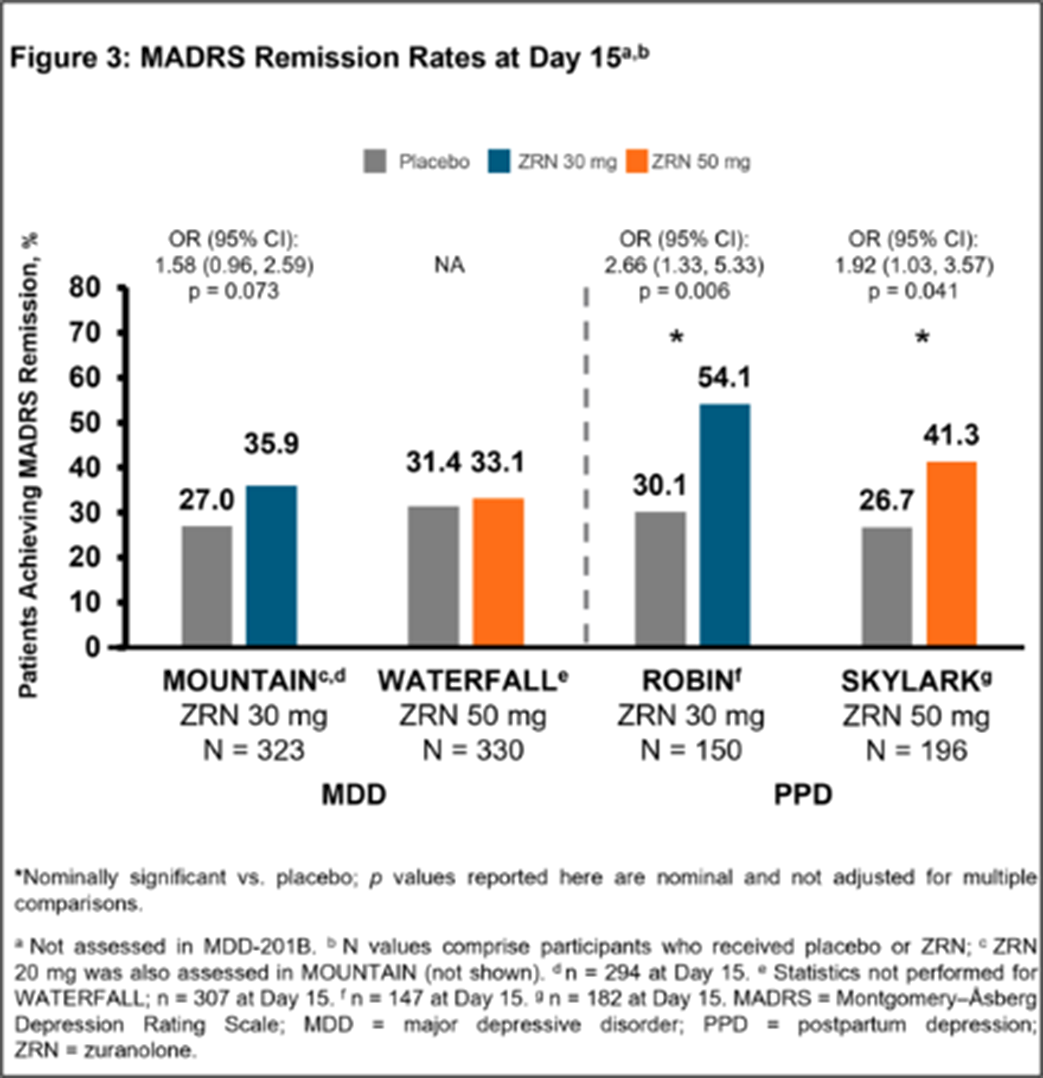 Conclusions
ConclusionsIn the 5 completed clinical studies, rapid improvement in depressive symptoms assessed by MADRS was observed across studies of adults with MDD and PPD who received a 14-day treatment of once-daily ZRN. In all studies, ZRN was generally well tolerated. These data support further development of ZRN as a potential oral, rapid-acting treatment for patients with MDD or PPD.
FundingThe MDD-201B, MOUNTAIN, and ROBIN studies were sponsored by Sage Therapeutics, Inc.; the WATERFALL and SKYLARK studies were sponsored by Sage Therapeutics, Inc. and Biogen Inc. Medical writing and editorial support were provided by MediTech Media, Ltd, and funded by Sage Therapeutics, Inc. and Biogen Inc.
Disclosure of InterestA. Clayton Shareolder of: Royalties from Ballantine Books/Random House, the Changes in Sexual Functioning Questionnaire, and Guilford Publications; and restricted stock in Euthymics, Mediflix LLC, and S1 Biopharma., Grant / Research support from: Daré Bioscience, Janssen, Otsuka, Praxis Precision Medicines, Relmada Therapeutics, Inc., and Sage Therapeutics, Inc, Consultant of: AbbVie, Inc., Brii Biosciences, Inc., Fabre-Kramer, Janssen Research & Development, LLC, Mind Cure Health, Ovoca Bio plc, Praxis Precision Medicines, PureTech Health, Reunion Neuroscience (formerly Field Trip Health) S1 Biopharma, Sage Therapeutics, Inc., Takeda/Lundbeck, Vella Bioscience, Inc., and WCG MedAvante-ProPhase, K. Deligiannidis Shareolder of: Royalties from an NIH employee invention outside of the submitted work. , Grant / Research support from: Received grants from from NIH and Vorso Corporation. Grants awarded to Zucker Hillside Hospital/Feinstein Institutes for Medical Research during the conduct of the brexanolone injection and zuranolone clinical trials (Sage Therapeutics), Consultant of: Sage Therapeutics, Inc., Brii Biosciences, Inc., and GH Research Ireland Limited, J. A. Ramos-Quiroga Grant / Research support from: The Department of Mental Health chaired by him received unrestricted educational and research support from the following companies in the last 3 years: Janssen-Cilag, Shire, Oryzon, Roche, Psious, and Rubió. Dr Ramos-QuirogaReceived travel awards (air tickets + hotel) for taking part in psychiatric meetings from Janssen-Cilag, Rubió, Shire, Takeda, Shionogi, Bial, and Medice., Consultant of: Was on the speaker’s bureau and/or acted as consultant for Janssen-Cilag, Novartis, Shire, Takeda, Bial, Shionogi, Sincrolab, Novartis, BMS, Medice, Technofarma, Rubió and Raffo in the last 3 years., Speakers bureau of: Was on the speaker’s bureau and/or acted as consultant for Janssen-Cilag, Novartis, Shire, Takeda, Bial, Shionogi, Sincrolab, Novartis, BMS, Medice, Technofarma, Rubió and Raffo in the last 3 years., R. Lasser Shareolder of: May hold stock and/or stock options of Sage Therapeutics, Inc. , Employee of: Employee of Sage Therapeutics, Inc., A. Sankoh Shareolder of: May hold stock and/or stock options of Sage Therapeutics, Inc., Employee of: Employee of Sage Therapeutics, Inc. , B. Leclair Shareolder of: May hold stock of Biogen Inc., Employee of: Employee of Biogen Inc., M. Kotecha Shareolder of: May hold stock of Biogen Inc., Employee of: Employee of Biogen Inc., J. Doherty Shareolder of: May hold stock and/or stock options of Sage Therapeutics, Inc., Employee of: Employee of Sage Therapeutics, Inc.
Clinical Efficacy of a 2-Week Treatment Course of Zuranolone for the Treatment of Major Depressive Disorder and Postpartum Depression: Outcomes From the Clinical Development Program
- A. Clayton, A.J. Cutler, K.M. Deligiannidis, R. Lasser, A.J. Sankoh, J. Doherty, M. Kotecha
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, pp. S97-S98
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Antidepressants that offer a rapid onset of action without requiring chronic use are greatly needed in both major depressive disorder (MDD) and postpartum depression (PPD). Zuranolone is an investigational, oral, neuroactive steroid and GABAA receptor positive allosteric modulator in clinical development as a 2-week treatment course for MDD and PPD.
ObjectivesTo present the efficacy and safety of zuranolone vs placebo in Phase 2 and 3 trials.
MethodsIn the studies presented (Table 1), improvements in depressive symptoms were assessed by least-squares mean (LSM) using a mixed-effects model for repeated measures on the change from baseline (CFB) at Day 15 in the 17-item Hamilton Rating Scale for Depression total score (HAMD-17; primary endpoint for all trials) and the Montgomery–Åsberg Depression Rating Scale (MADRS; secondary endpoint) following a 14-day treatment course of once-daily zuranolone.
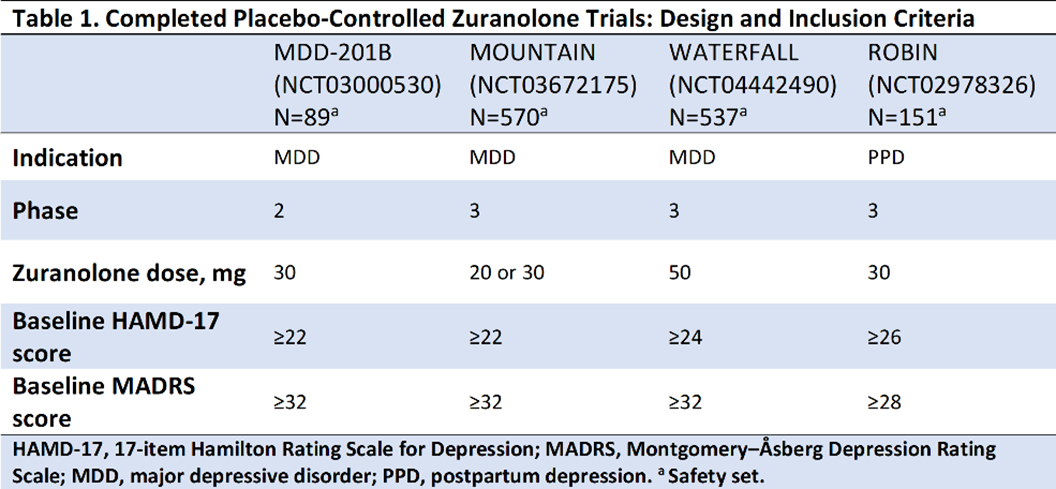 Results
ResultsCompared with placebo, zuranolone treatment led to rapid improvements in depressive symptoms across clinical trials, with significant improvements (LSM treatment difference [SE] in CFB) in HAMD-17 and MADRS scores at Day 15 in 3 of the 4 trials (Table 2). Common treatment-emergent adverse events (≥5% in zuranolone treatment arms) were headache, somnolence, dizziness, nausea, sedation, diarrhea, upper respiratory tract infection, and fatigue (Table 3). No incidences of loss of consciousness or excessive sedation were observed.
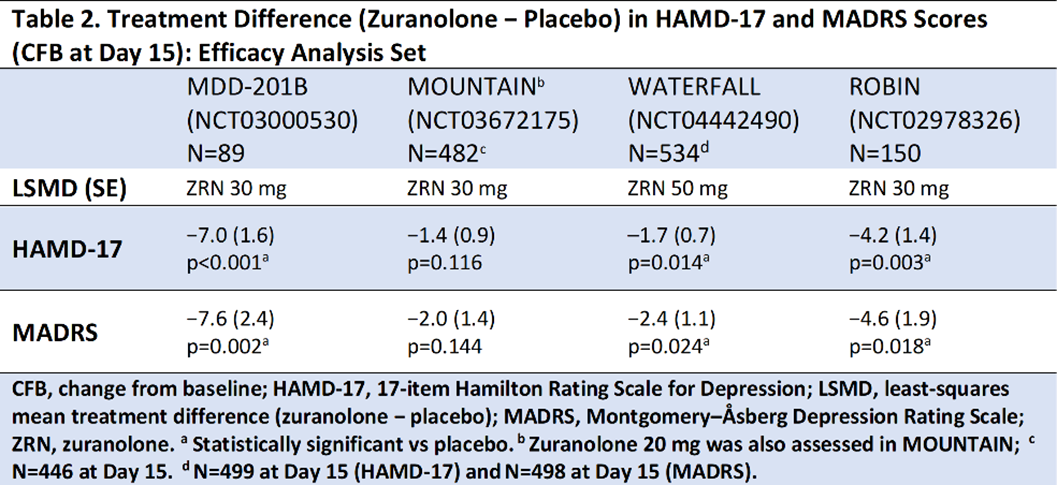
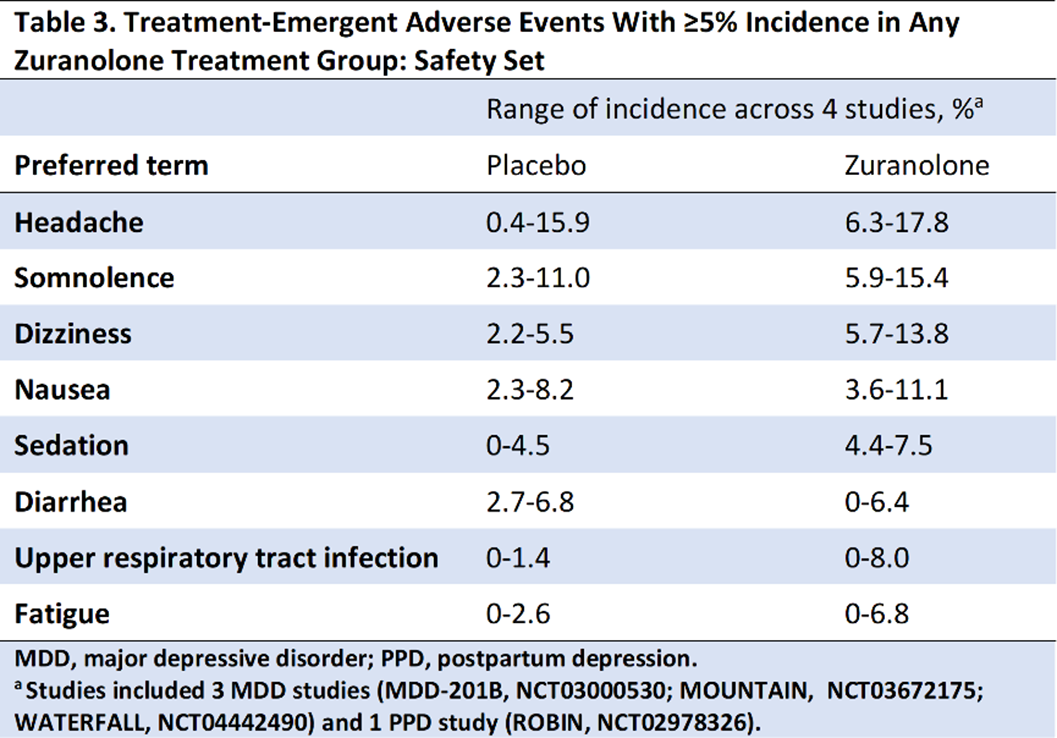 Conclusions
ConclusionsAcross the completed studies in the zuranolone clinical trial program, patients receiving zuranolone consistently experienced improvement in depressive symptoms following a 2-week treatment course. Treatment with zuranolone was generally well tolerated with a consistent safety and tolerability profile.
DisclosureThe MDD-201B, MOUNTAIN, and ROBIN studies were sponsored by Sage Therapeutics, Inc; the WATERFALL study was sponsored by Sage Therapeutics, Inc, and Biogen. Medical writing and editorial support were provided by MediTech Media, Ltd, and funded by Biogen.
Consortium for the lifespan examination of ADHD registry (Clear): An update on methodology and recruitment
- R. Lasser, C. Eksteen, M. Brod, L. Politza, M. Dauphin, P. Asherson, M. Huss, S. Tetali
-
- Journal:
- European Psychiatry / Volume 26 / Issue S2 / March 2011
- Published online by Cambridge University Press:
- 16 April 2020, p. 279
-
- Article
-
- You have access Access
- Export citation
-
Introduction
ADHD is often unrecognised and untreated, particularly in Europe. There is a need for improved understanding of ADHD in adults and its impact on patients across the lifespan. Registries allow for multiple real-world comparisons based on assessment of patients that are typically excluded from clinical studies. Data describing the cost, burden and consequences of ADHD are essential to inform clinicians, regulatory agencies, payers and patients about the condition.
ObjectivesTo address the gaps in our current understanding of the care and cost of treating adult patients with ADHD by documenting real-life experiences.
MethodsA prospective, longitudinal, observational study of adults > 18 years with ADHD, designed to follow 2500 patients in the USA, UK, Germany, The Netherlands and Canada for a minimum of 5 years.
In year one, patients will be asked questions regarding:
the impact of childhood diagnosis and/or treatment on adult ADHD, long- and short-term treatment patterns and their consequences, differences between ADHD treatments, the relationship between disease severity and compliance with treatment, increased tendency to risk behaviour/substance abuse, costs.
In subsequent years, topics may be expanded to include the transition into adulthood and healthcare access for young adults.
ResultsTen sites are currently recruiting, with 45 patients enrolled up to 7 October 2010. Challenges associated with initiating a global registry in North America and Europe will be discussed.
ConclusionData generated from CLEAR will help improve the global understanding of the longitudinal impact of ADHD.
Supported by Shire Development, Inc.









