2 results
Changes in US long-term care facility antibiotic prescribing, 2013–2021
- Katryna Gouin, Stephen Creasy, Mary Beckerson, Marti Wdowicki, Lauri Hicks, Sarah Kabbani
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s108-s109
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antibiotic use (AU) data are needed to improve prescribing in long-term care facilities (LTCFs). CMS requires AU tracking in LTCFs (effective 2017). Although most LTCFs have limited resources for AU tracking, LTCFs contract with LTCF pharmacies to dispense, monitor, and review medications. The objective of our analysis was to report LTCF antibiotic prescribing and characterize temporal changes from 2013 to 2021. Methods: We estimated annual systemic AU rates using prescription dispenses and resident census data from PharMerica, a LTCF-pharmacy services provider that covers ~20% of LTCFs nationwide, although the number of LTCFs and residents serviced by PharMerica varied over time (Fig. 1). We included LTCFs with ≥4 months of antibiotic dispensing and 12 months of census data. We identified courses by collapsing the same drug dispensed to the same resident within 3 days of the preceding end date. Course duration was calculated as the difference between the end and dispense dates. We reported yearly AU rates as courses per 1,000 residents and days of therapy (DOT) per 1,000 resident days from 2013 to 2021. We compared AU rates (percentage change) and antibiotic courses by class and agent (absolute percent difference) between 2013 and 2021. Results: From 2013 to 2021, AU course rates reported as antibiotic courses per 1,000 residents decreased (percentage change, −28%), with a notable increase in 2020 (Fig. 1). However, the median course duration remained the same (Table 1). The AU decline was mostly driven by decreases in fluoroquinolone courses (absolute difference, −10%, most commonly levofloxacin) and macrolide courses (−2%, most commonly azithromycin) (Figs. 2 and 3). Increases in cephalosporin courses (absolute difference, +7%, most commonly cephalexin) and tetracycline courses (+5%, most commonly doxycycline) were also observed (Figs. 2 and 3). During this period, AU DOT rates reported as DOT per 1,000 resident days decreased (percentage change, −13%) (Table 1). Conclusions: The LTCF AU rates, especially for fluoroquinolones, have decreased in recent years with associated shifts in the distribution of antibiotic classes. This finding may be due to CMS stewardship requirements and increased awareness of adverse events, including the FDA fluoroquinolone warnings. The observed increase in 2020 could be secondary to changes in prescribing practices and resident population during the COVID-19 pandemic. Opportunities to improve prescribing in LTCFs include optimizing treatment duration and leveraging LTCF-pharmacy resources to provide stewardship expertise and support AU tracking and reporting.




Disclosures: None
Evaluation of Electronic Health Record and Long-Term Care Pharmacy Data for Tracking and Reporting Antibiotic Use in the United States
- Matthew Hudson, Katryna Gouin, Stanley Wang, Manjiri Kulkarni, Mary Beckerson, Laura Ditz, Stephen Creasy, Marti Wdowicki, Nancy Chi, Lauri Hicks, Sarah Kabbani
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s13-s14
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antibiotics are frequently prescribed in nursing homes, often inappropriately. Data sources are needed to facilitate measurement and reporting of antibiotic use to inform antibiotic stewardship efforts. Previous analyses have shown that the type of nursing-home stay, that is, short stay (<100 days), is a strong predictor of high antibiotic use compared to longer nursing-home stays. The study objective was to compare 2 different data sources, electronic health record (EHR) and long-term care (LTC) pharmacy data, for surveillance of antibiotic use and type of nursing-home stay. Methods: EHR and pharmacy data during 2017 were included from 1,933 and 1,348 US-based nursing homes, respectively. We compared data elements available in each data source for antibiotic use reporting. In each data set, we attempted to describe antibiotic use as the proportion of residents on an antibiotic, days-of-therapy (DOT) per 1,000 resident days (RD), and distribution of antibiotic course duration, overall and at the facility level. Facility proportion of short-stay and long-stay (>100 days) nursing-home residents were calculated using admission dates and census data in the EHR data set and a payor variable in the pharmacy data set (Figure 1). The 2 data sources also provided antibiotic characteristics, including antibiotic class, agent, and route of administration. The deidentified nature of facility data prevented direct comparison of antibiotic use measures between facilities. Results: The EHR and pharmacy data sets contained 381,382 and 326,713 residents, respectively (Table 1). Within the EHR, 51% of residents were prescribed an antibiotic in 2017, at a median rate of 77 DOT per 1,000 RD. In the LTC pharmacy, 46% of residents were prescribed an antibiotic at a median rate of 79 DOT per 1,000 RD (Table 1). Short-stay residents contributed a smaller proportion of total RDs in the EHR relative to the pharmacy cohort (21% vs 50%, respectively). Conclusions: Nursing-home antibiotic use data obtained from EHR and pharmacy vendors can be used for calculating antibiotic use measures, which is important for antibiotic use reporting and facility-level tracking to identify opportunities for improving prescribing practices and provide facility-level benchmarks. Further validation of both data sources in the same facilities is needed to compare antibiotic use rates and to determine the most appropriate proxy for type of nursing-home stay for facility-level risk adjustment of antibiotic use rates.
Funding: No
Disclosures: None
Figure 1.
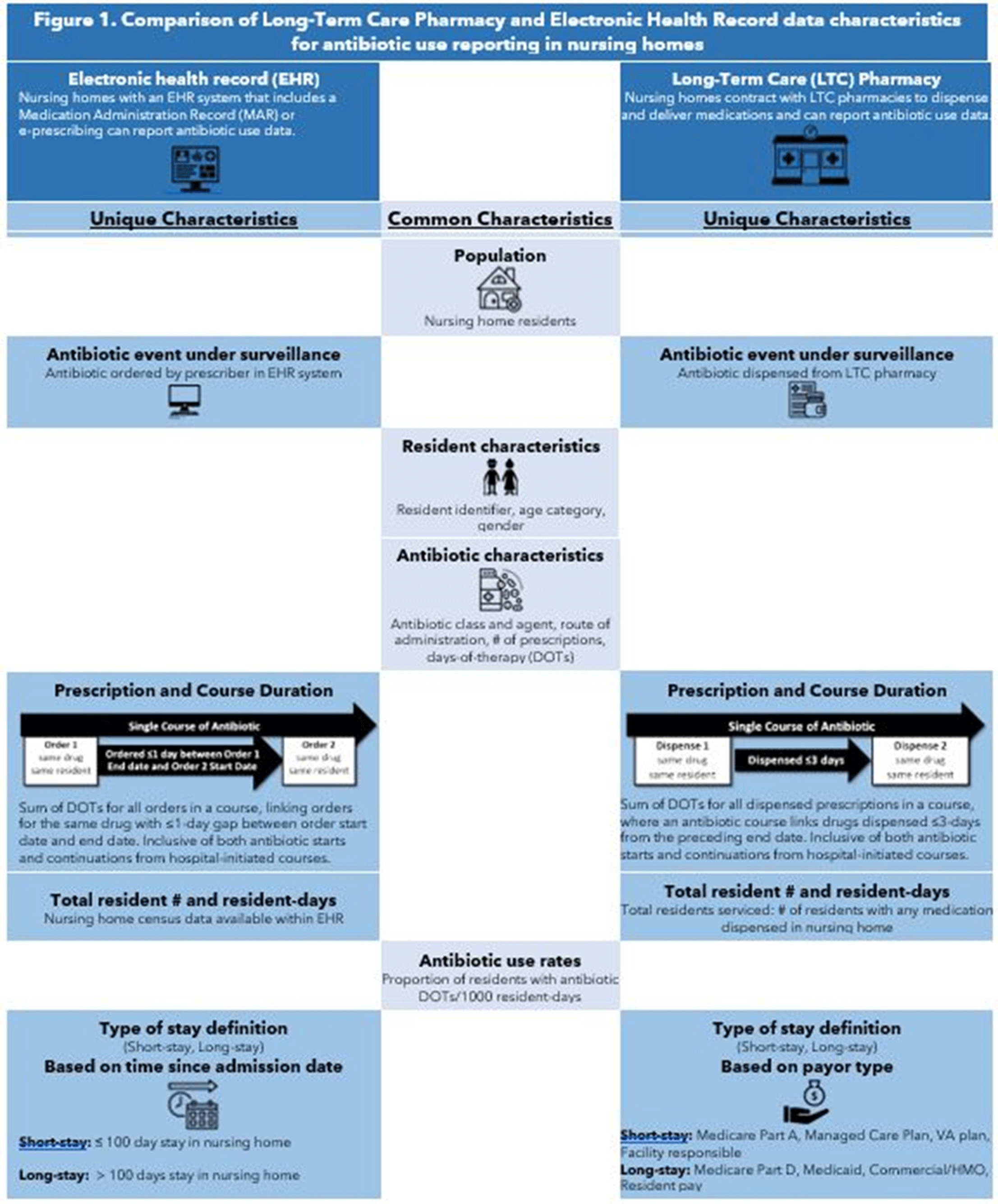
Table 1. 









