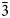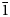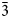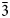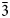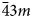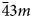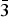7 results
Enricofrancoite, KNaCaSi4O10, a new Ca–K–Na silicate from Somma–Vesuvius volcano, southern Italy
- Giuseppina Balassone, Taras L. Panikorovskii, Annamaria Pellino, Ayya V. Bazai, Vladimir N. Bocharov, Olga F. Goychuk, Evgenia Yu. Avdontseva, Victor N. Yakovenchuk, Sergey V. Krivovichev, Carmela Petti, Piergiulio Cappelletti, Nicola Mondillo, Anna Moliterni, Angela Altomare, Francesco Izzo
-
- Journal:
- Mineralogical Magazine , FirstView
- Published online by Cambridge University Press:
- 26 February 2024, pp. 1-11
-
- Article
- Export citation
-
Enricofrancoite (IMA2023–002), ideally KNaCaSi4O10, is a new litidionite-group member found as the product of high-temperature alteration of hosting silicates with the enrichment by Cu-bearing fluids at the rock–fumaroles interface related to the 1872 eruption of Somma–Vesuvius volcano, southern Italy. It occurs as euhedral and platy crystals or crusts together with litidionite, tridymite, wollastonite and Al- and Fe-bearing diopside, kamenevite, perovskite, rutile, Ti-rich magnetite and colourless Si-glass. Single crystals of enricofrancoite are transparent colourless or light blue with a vitreous lustre. Mohs hardness is 5.5. Dmeas is 2.63(3) g/cm3 and Dcalc is 2.63 g/cm3. The mineral is optically biaxial (−), α = 1.542(5), β = 1.567(5),γ = 1.575(5); 2V(meas) = 60(2)° and 2Vcalc = 58°. The mean chemical composition (wt.%, electron-microprobe data) is: SiO2 64.81, Al2O3 0.03, TiO2 0.08, FeO 0.07, MgO 1.71, CaO 10.64, CuO 2.22, Na2O 8.56, K2O 11.41, total 99.94. The empirical formula based on 10 O apfu is: K0.90Na1.03(Ca0.71Mg0.16Cu0.10)Σ0.97Si4.02O10. The Raman spectrum contains bands at 133, 248, 265, 290, 335, 400, 438, 510, 600, 690 and 1120 cm–1 and the wavenumbers of the IR absorption bands are: 424, 470, 492, 530, 600, 630, 690, 750, 788, 970, 1040 and 1160 cm–1. The eight strongest lines of the powder X-ray diffraction pattern are [d, Å (I, %) hkl]: 6.75 (42) 01$\bar{1}$
 , 3.65 (20) 11$\bar{2}$
, 3.65 (20) 11$\bar{2}$ , 3.370 (100) 02$\bar{2}$
, 3.370 (100) 02$\bar{2}$ , 3.210 (52) 102, 3.051 (18) 111, 3.033 (25) 2$\bar{1}\bar{2}$
, 3.210 (52) 102, 3.051 (18) 111, 3.033 (25) 2$\bar{1}\bar{2}$ , 2.834 (22) 02$\bar{3}$
, 2.834 (22) 02$\bar{3}$ and 2.411 (72) 03$\bar{2}$
and 2.411 (72) 03$\bar{2}$ . Enricofrancoite is triclinic, space group P$\bar{1}$
. Enricofrancoite is triclinic, space group P$\bar{1}$ , unit-cell parameters refined from the single-crystal data are a = 7.0155(4) Å, b = 8.0721(4) Å, c = 10.0275(4) Å, α = 104.420(4)°, β = 99.764(4)°, γ = 115.126(5)° and V = 472.74(5) Å3. The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.035 on the basis of 2078 independent reflections with Fo > 4σ(Fo). Enricofrancoite is an H2O-free analogue of calcinaksite with 5-coordinated Ca2+ at the M site.
, unit-cell parameters refined from the single-crystal data are a = 7.0155(4) Å, b = 8.0721(4) Å, c = 10.0275(4) Å, α = 104.420(4)°, β = 99.764(4)°, γ = 115.126(5)° and V = 472.74(5) Å3. The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.035 on the basis of 2078 independent reflections with Fo > 4σ(Fo). Enricofrancoite is an H2O-free analogue of calcinaksite with 5-coordinated Ca2+ at the M site.
Dmitryvarlamovite, Ti2(Fe3+Nb)O8, a new columbite-supergroup mineral related to the wolframite group
- Oksana V. Udoratina, Taras L. Panikorovskii, Nikita V. Chukanov, Mikhail V. Voronin, Vladimir P. Lutoev, Atali A. Agakhanov, Sergey I. Isaenko
-
- Journal:
- Mineralogical Magazine / Volume 88 / Issue 2 / April 2024
- Published online by Cambridge University Press:
- 01 February 2024, pp. 147-154
-
- Article
- Export citation
-
The new columbite-supergroup mineral dmitryvarlamovite, ideally Ti2(Fe3+Nb)O8, was discovered in weathered alkaline metasomatic assemblages formed after late Riphaean sedimentary carbonate rocks of the Verkhne-Shchugorskoe deposit, Middle Timan Mts., Russia. The associated minerals are columbite-(Fe), pyrochlore-group minerals, monazite-(Ce), xenotime-(Y), baryte, pyrite, drugmanite and plumbogummite. Dmitryvarlamovite occurs as isolated anhedral equant grains up to 0.5 mm across. The colour of dmitryvarlamovite is black, the streak is black and the lustre is submetallic. The new mineral is brittle, with the mean VHN hardness of 753 kg mm–2 corresponding to the Mohs’ hardness of 6. No cleavage is observed. The fracture is conchoidal. The calculated density is 4.891 g⋅cm–3. In reflected light, dmitryvarlamovite is light grey; no pleochroism is observed. The reflectance values (Rmin, % / Rmax, % / λ, nm) are: 19.8/20.3/470, 18.3/18.9/546, 17.8/18.5/589 and 17.3/17.8/650. The chemical composition is (electron microprobe data, with iron divided into Fe2O3 and FeO based on the charge balance, wt.%): MnO 0.11, FeO 1.51, V2O3 0.89, Cr2O3 0.28, Fe2O3 19.26, TiO2 37.72, Nb2O5 40.08, total 99.85. The IR and Raman spectra indicate the absence of H-, C- and N-bearing groups. The empirical formula is (Fe2+0.08V3+0.05Cr3+0.01Fe3+0.92Ti1.79Nb1.15)Σ4.00O8. The crystal structure was determined using single-crystal X-ray diffraction data and refined to R = 0.048. Dmitryvarlamovite is orthorhombic, space group P21212, a = 4.9825(6), b = 4.6268(4), c = 5.5952(6) Å and V = 5.5952(6) Å3 (Z = 1). The structure is related to those of wolframite-group minerals but differs in the scheme of cation ordering. The crystal-chemical formula derived based on the structural data is (Ti0.57Nb0.21Fe3+0.15Fe2+0.04V0.02Cr0.01)2(Nb0.36Ti0.33Fe3+0.31)2O8. The strongest lines of the powder X-ray diffraction pattern [d, Å (I, %) (hkl)] are: 3.58 (40) (011), 2.911 (100) (111), 2.809 (40) (002), 2.497 (38) (020), 2.447 (29) (103), 1.7363 (32) (103) and 1.7047 (29) (220). Dmitryvarlamovite is named after Dmitry Anatol'evich Varlamov (b. 1965).
Structural and chemical complexity of minerals: an update
- Sergey V. Krivovichev, Vladimir G. Krivovichev, Robert M. Hazen, Sergey M. Aksenov, Margarita S. Avdontceva, Alexander M. Banaru, Liudmila A. Gorelova, Rezeda M. Ismagilova, Ilya V. Kornyakov, Ivan V. Kuporev, Shaunna M. Morrison, Taras L. Panikorovskii, Galina L. Starova
-
- Journal:
- Mineralogical Magazine / Volume 86 / Issue 2 / April 2022
- Published online by Cambridge University Press:
- 04 April 2022, pp. 183-204
-
- Article
-
- You have access Access
- HTML
- Export citation
-
The complexities of chemical composition and crystal structure are fundamental characteristics of minerals that have high relevance to the understanding of their stability, occurrence and evolution. This review summarises recent developments in the field of mineral complexity and outlines possible directions for its future elaboration. The database of structural and chemical complexity parameters of minerals is updated by H-correction of structures with unknown H positions and the inclusion of new data. The revised average complexity values (arithmetic means) for all minerals are 3.54(2) bits/atom and 345(10) bits/cell (based upon 4443 structure reports). The distributions of atomic information amounts, chemIG and strIG, versus the number of mineral species fit the normal modes, whereas the distributions of total complexities, chemIG,total and strIG,total, along with numbers of atoms per formula and per unit cell are log normal. The three most complex mineral species known today are ewingite, morrisonite and ilmajokite, all either discovered or structurally characterised within the last five years. The most important complexity-generating mechanisms in minerals are: (1) the presence of isolated large clusters; (2) the presence of large clusters linked together to form three-dimensional frameworks; (3) formation of complex three-dimensional modular frameworks; (4) formation of complex modular layers; (5) high hydration state in salts with complex heteropolyhedral units; and (6) formation of ordered superstructures of relatively simple structure types. The relations between symmetry and complexity are considered. The analysis of temporal dynamics of mineralogical discoveries since 1875 with the step of 25 years show the increasing chemical and structural complexities of human knowledge of the mineral kingdom in the history of mineralogy. In the Earth's history, both diversity and complexity of minerals experience dramatic increases associated with the formation of Earth's continental crust, initiation of plate tectonics and the Great Oxidation event.
The complex mechanism of Ti4+ incorporation into litidionite from the Somma–Vesuvius volcano, Italy
- Giuseppina Balassone, Taras L. Panikorovskii, Annamaria Pellino, Ayya V. Bazai, Vladimir N. Bocharov, Sergey V. Krivovichev, Carmela Petti, Piergiulio Cappelletti, Nicola Mondillo
-
- Journal:
- Mineralogical Magazine / Volume 86 / Issue 2 / April 2022
- Published online by Cambridge University Press:
- 16 February 2022, pp. 222-233
-
- Article
- Export citation
-
For this study, the rare Cu-bearing silicate fumarolic assemblages from the Somma–Vesuvius volcano, Italy, characterised by the rare mineral litidionite, CuKNaSi4O10, were investigated. We report new data about Cu- and Ti-bearing phases found in these mineralisations, in which Ti-bearing litidionite occurs together with kamenevite, perovskite and rutile. Ti-bearing litidionite appears on the latest stages of partial crystallisation of Ti-bearing silica glass. Incorporation of Ti4+ into the litidionite crystal structure was investigated in detail. The Raman spectra of Ti-bearing litidionite contains an intense band at 597 cm−1 related to anti-symmetric bending vibrations of Si‒O bonds or overlapping stretching vibrations of Ti‒O bonds. The bands in the range 350‒500 cm−1 correspond to symmetric bending vibrations of O‒Si‒O bonds and overlapping stretching vibrations of Ti‒O bonds. The crystal structure of Ti-litidionite has been refined in the P$\bar{1}$
 space group, a = 6.9699(7), b = 7.9953(10), c = 9.8227(10) Å, α = 105.186(9), β = 99.458(8) and γ = 114.489(10) to R1 = 0.064 for 1726 unique observed reflections. The refinement of the site-occupation factors confirmed the presence of Ti at a five-coordinated M site. The mean bond distance of 2.125 Å for the M site agrees with its refined occupancy (Ti0.32Cu0.30Ca0.29Fe0.09)1.00. The incorporation of Ti into the litidionite structure is accompanied by the complex heteropolyhedral substitution according to the scheme VTi4+ + VII–VIII□ + IVAl3+ ↔ VCu2+ + VII-VIII(Na,K)+ + IVSi4+. Two possible configurations for the phase with maximal TiO2 content (12.06 wt.% or 0.56 Ti apfu) CuTiK□Na2Si7AlO20 (Z = 1) or CuTiK2Na□Si7AlO20 (Z = 1) have been proposed.
space group, a = 6.9699(7), b = 7.9953(10), c = 9.8227(10) Å, α = 105.186(9), β = 99.458(8) and γ = 114.489(10) to R1 = 0.064 for 1726 unique observed reflections. The refinement of the site-occupation factors confirmed the presence of Ti at a five-coordinated M site. The mean bond distance of 2.125 Å for the M site agrees with its refined occupancy (Ti0.32Cu0.30Ca0.29Fe0.09)1.00. The incorporation of Ti into the litidionite structure is accompanied by the complex heteropolyhedral substitution according to the scheme VTi4+ + VII–VIII□ + IVAl3+ ↔ VCu2+ + VII-VIII(Na,K)+ + IVSi4+. Two possible configurations for the phase with maximal TiO2 content (12.06 wt.% or 0.56 Ti apfu) CuTiK□Na2Si7AlO20 (Z = 1) or CuTiK2Na□Si7AlO20 (Z = 1) have been proposed.
Trigonal variation in the garnet supergroup: the crystal structure of nikmelnikovite, Ca12Fe2+Fe3+3Al3(SiO4)6(OH)20, from Kovdor massif, Kola Peninsula, Russia
- Sergey V. Krivovichev, Taras L. Panikorovskii, Victor N. Yakovenchuk, Ekaterina A. Selivanova, Gregory Yu. Ivanyuk
-
- Journal:
- Mineralogical Magazine / Volume 85 / Issue 4 / August 2021
- Published online by Cambridge University Press:
- 28 June 2021, pp. 620-626
-
- Article
- Export citation
-
The crystal structure of nikmelnikovite, Ca12Fe2+Fe3+3Al3(SiO4)6(OH)20, a new member of the garnet supergroup from Kovdor massif, Kola Peninsula, Russia (R$\bar{3}$
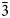 , a = 17.2072(6), c = 10.5689(4) Å, V = 2710.1(2) Å3 and Z = 3) has been refined to R1 = 0.046 on the basis of 1184 unique observed reflections. Nikmelnikovite is the first mineral species in the garnet supergroup that has a trigonal (rhombohedral) symmetry. The relationship between its unit cell and the pseudocubic (ideal garnet) unit cell can be described by the transformation matrix [1$\bar{1}$
, a = 17.2072(6), c = 10.5689(4) Å, V = 2710.1(2) Å3 and Z = 3) has been refined to R1 = 0.046 on the basis of 1184 unique observed reflections. Nikmelnikovite is the first mineral species in the garnet supergroup that has a trigonal (rhombohedral) symmetry. The relationship between its unit cell and the pseudocubic (ideal garnet) unit cell can be described by the transformation matrix [1$\bar{1}$ 0 | 01$\bar{1}$
0 | 01$\bar{1}$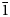 | ½½½]. The crystal-chemical relations between the ideal Ia$\bar{3}$
| ½½½]. The crystal-chemical relations between the ideal Ia$\bar{3}$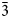 d garnet and the nikmelnikovite structure type can be described by the following series of imaginary modifications: (1) the symmetry is lowered according to the Ia$\bar{3}$
d garnet and the nikmelnikovite structure type can be described by the following series of imaginary modifications: (1) the symmetry is lowered according to the Ia$\bar{3}$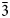 d → R$\bar{3}$
d → R$\bar{3}$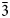 group–subgroup relationship; (2) the cation sites are split according to the following sequences: X → {X1, X2}; Y → {Y1, Y2, Y3, Y4}; Z → {Z1, Z2}; (3) the X sites remain fully occupied by Ca; (4) each Y site is occupied predominantly by a distinct chemical species: Y1 → Al (Al site), Y2 → Fe2+ (Fe1 site), Y3 → Fe3+ (Fe2 site), Y4 → vacancy (Mn site); (5) one of the Z sites (Z1) is occupied by Si, whereas the other site (Z2) is predominantly vacant. The crystal-chemical formula that takes into account the transition between the archetype and the nikmelnikovite structure type can be described as X{Ca12}Y[Fe2+Al4Fe3+2□]Z(Si6□6)O24(OH)20□4. The structural complexity of nikmelnikovite (4.529 bit/atom and 434.431 bit/cell, after H-correction) is higher than those for andradite, grossular and katoite, which is typical for low-temperature minerals formed after primary minerals with simpler structures.
group–subgroup relationship; (2) the cation sites are split according to the following sequences: X → {X1, X2}; Y → {Y1, Y2, Y3, Y4}; Z → {Z1, Z2}; (3) the X sites remain fully occupied by Ca; (4) each Y site is occupied predominantly by a distinct chemical species: Y1 → Al (Al site), Y2 → Fe2+ (Fe1 site), Y3 → Fe3+ (Fe2 site), Y4 → vacancy (Mn site); (5) one of the Z sites (Z1) is occupied by Si, whereas the other site (Z2) is predominantly vacant. The crystal-chemical formula that takes into account the transition between the archetype and the nikmelnikovite structure type can be described as X{Ca12}Y[Fe2+Al4Fe3+2□]Z(Si6□6)O24(OH)20□4. The structural complexity of nikmelnikovite (4.529 bit/atom and 434.431 bit/cell, after H-correction) is higher than those for andradite, grossular and katoite, which is typical for low-temperature minerals formed after primary minerals with simpler structures.
Crystal chemistry of ivanyukite-group minerals, A3–xH1+x[Ti4O4(SiO4)3](H2O)n (A = Na, K, Cu), (n = 6–9, x = 0–2): crystal structures, ion-exchange, chemical evolution
- Taras L. Panikorovskii, Victor N. Yakovenchuk, Nataliya Yu. Yanicheva, Yakov A. Pakhomovsky, Vladimir V. Shilovskikh, Vladimir N. Bocharov, Sergey V. Krivovichev
-
- Journal:
- Mineralogical Magazine / Volume 85 / Issue 4 / August 2021
- Published online by Cambridge University Press:
- 07 June 2021, pp. 607-619
-
- Article
- Export citation
-
Microporous slicates with the pharmacosiderite structure and the general formula A3–xH1+x[Ti4O4(SiO4)3](H2O)n (A = Na, K, Cu), (n = 6–9, x = 0–2) are outstanding in their ion-exchange properties. The ivanyukite mineral group consists of three species, one of which has two polymorphs. The minerals forming a progressive series: ivanyukite-Na-T → ivanyukite-Na-C → ivanyukite-K → Cu-rich ivanyukite-K → ivanyukite-Cu, have been studied by single-crystal X-ray diffraction, electron microprobe analysis and Raman spectroscopy. The microporous heteropolyhedral framework of the ivanyukite-group minerals is based on cubane-like [Ti4O4]8+ clusters that share common corners with SiO4 tetrahedra to form wide three-dimensional channels suitable for the migration of Na+, K+ and Cu2+ ions. Ivanyukite-Na-T that has a R3m symmetry loses Na+ in aqueous solutions via the substitution Na+ + O2‒ ↔ □ + OH‒, which allows for the migration of K+ ions and transformation of initial structure into the cubic (P$\bar{4}3m$
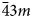 ) ivanyukite-Na-C polymorph or into ivanyukite-K, when most of Na is lost. Natural ivanyukite-Na-C is shown to contain domains of both R3m (subordinate) and P$\bar{4}3m$
) ivanyukite-Na-C polymorph or into ivanyukite-K, when most of Na is lost. Natural ivanyukite-Na-C is shown to contain domains of both R3m (subordinate) and P$\bar{4}3m$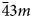 (dominant) symmetry with the chemical composition determining the stability and dominance of cubic or trigonal forms. Incorporation of Cu into the crystal structure ivanyukite-K via the substitution K+ + OH− ↔ Cu2+ + O2− in aqueous solutions results in the formation of ivanyukite-Cu. Post-crystallisation processes (such as exchange of Na+, K+, Cu2+, and/or hydration/dehydration of primary phases) are widespread in hyperagpaitic rocks of the Kola alkaline massif and the respective mineral transformations contribute to the diversity of mineral species.
(dominant) symmetry with the chemical composition determining the stability and dominance of cubic or trigonal forms. Incorporation of Cu into the crystal structure ivanyukite-K via the substitution K+ + OH− ↔ Cu2+ + O2− in aqueous solutions results in the formation of ivanyukite-Cu. Post-crystallisation processes (such as exchange of Na+, K+, Cu2+, and/or hydration/dehydration of primary phases) are widespread in hyperagpaitic rocks of the Kola alkaline massif and the respective mineral transformations contribute to the diversity of mineral species.
Hydroxynatropyrochlore, (Na,Сa,Ce)2Nb2O6(OH), a new member of the pyrochlore group from the Kovdor phoscorite–carbonatite pipe, Kola Peninsula, Russia
- Gregory Yu. Ivanyuk, Victor N. Yakovenchuk, Taras L. Panikorovskii, Nataliya Konoplyova, Yakov A. Pakhomovsky, Ayya V. Bazai, Vladimir N. Bocharov, Sergey V. Krivovichev
-
- Journal:
- Mineralogical Magazine / Volume 83 / Issue 1 / February 2019
- Published online by Cambridge University Press:
- 15 May 2018, pp. 107-113
-
- Article
- Export citation
-
Hydroxynatropyrochlore, (Na,Сa,Ce)2Nb2O6(OH), is a new Na–Nb–OH-dominant member of the pyrochlore supergroup from the Kovdor phoscorite–carbonatite pipe, Kola Peninsula, Russia. It is cubic, Fd
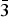 $\bar{3}$m, a = 10.3211(3) Å, V = 1099.46(8) Å3 and Z = 8 (from powder diffraction data) or a = 10.3276(5) Å, V = 1101.5(2) Å3 and Z = 8 (from single-crystal diffraction data). Hydroxynatropyrochlore is a characteristic accessory mineral of the low-carbonate phoscorite in the contact zone of the phoscorite–carbonatite pipe with host foidolite as well as in the carbonate-rich phoscorite and carbonatite of the pipe axial zone. It usually forms zonal cubic or cubooctahedral crystals (up to 0.5 mm in diameter) with irregularly shaped relics of amorphous U–Ta-rich hydroxykenopyrochlore inside. Characteristic associated minerals include rock-forming calcite, dolomite, forsterite, hydroxylapatite, magnetite and phlogopite, accessory baddeleyite, baryte, barytocalcite, chalcopyrite, chamosite–clinochlore, galena, gladiusite, juonniite, ilmenite, magnesite, pyrite, pyrrhotite, quintinite, spinel, strontianite, valleriite and zirconolite. Hydroxynatropyrochlore is pale brown, with an adamantine to greasy lustre and a white streak. The cleavage is average on {111} and the fracture is conchoidal. Mohs hardness is ~5. In transmitted light, the mineral is light brown, isotropic and n = 2.10(5) (λ = 589 nm). The calculated and measured densities are 4.77 and 4.60(5) g cm−3, respectively. The mean chemical composition determined by electron microprobe is: F 0.05, Na2O 7.97, CaO 10.38, TiO2 4.71, FeO 0.42, Nb2O5 56.44, Ce2O3 3.56, Ta2O5 4.73, ThO2 5.73, UO2 3.66, total 97.65 wt.%. The empirical formula calculated on the basis of Nb + Ta + Ti = 2 apfu is (Na1.02Ca0.73Ce0.09Th0.09 U0.05
$\bar{3}$m, a = 10.3211(3) Å, V = 1099.46(8) Å3 and Z = 8 (from powder diffraction data) or a = 10.3276(5) Å, V = 1101.5(2) Å3 and Z = 8 (from single-crystal diffraction data). Hydroxynatropyrochlore is a characteristic accessory mineral of the low-carbonate phoscorite in the contact zone of the phoscorite–carbonatite pipe with host foidolite as well as in the carbonate-rich phoscorite and carbonatite of the pipe axial zone. It usually forms zonal cubic or cubooctahedral crystals (up to 0.5 mm in diameter) with irregularly shaped relics of amorphous U–Ta-rich hydroxykenopyrochlore inside. Characteristic associated minerals include rock-forming calcite, dolomite, forsterite, hydroxylapatite, magnetite and phlogopite, accessory baddeleyite, baryte, barytocalcite, chalcopyrite, chamosite–clinochlore, galena, gladiusite, juonniite, ilmenite, magnesite, pyrite, pyrrhotite, quintinite, spinel, strontianite, valleriite and zirconolite. Hydroxynatropyrochlore is pale brown, with an adamantine to greasy lustre and a white streak. The cleavage is average on {111} and the fracture is conchoidal. Mohs hardness is ~5. In transmitted light, the mineral is light brown, isotropic and n = 2.10(5) (λ = 589 nm). The calculated and measured densities are 4.77 and 4.60(5) g cm−3, respectively. The mean chemical composition determined by electron microprobe is: F 0.05, Na2O 7.97, CaO 10.38, TiO2 4.71, FeO 0.42, Nb2O5 56.44, Ce2O3 3.56, Ta2O5 4.73, ThO2 5.73, UO2 3.66, total 97.65 wt.%. The empirical formula calculated on the basis of Nb + Ta + Ti = 2 apfu is (Na1.02Ca0.73Ce0.09Th0.09 U0.05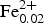 ${\rm Fe}_{{\rm 0}{\rm. 02}}^{2 +} $)Σ2.00(Nb1.68Ti0.23Ta0.09)Σ2.00O6.03(OH1.04F0.01)Σ1.05. The simplified formula is (Na,Ca,Ce)2Nb2O6(OH). The mineral dissolves slowly in hot HCl. The strongest X-ray powder-diffraction lines [listed as (d in Å)(I)(hkl)] are as follows: 5.96(47)(111), 3.110(30)(311), 2.580(100)(222), 2.368(19)(400), 1.9875(6)(333), 1.8257(25)(440) and 1.5561(14)(622). The crystal structure of hydroxynatropyrochlore was refined to R1 = 0.026 on the basis of 80 unique observed reflections. The mineral belongs to the pyrochlore structure type A2B2O6Y1 with octahedral framework of corner-sharing BO6 octahedra with A cations and OH groups in the interstices. The Raman spectrum of hydroxynatropyrochlore contains characteristic bands of the lattice, BO6, B–O and O–H vibrations and no characteristic bands of the H2O vibrations. Within the Kovdor phoscorite–carbonatite pipe, hydroxynatropyrochlore is the latest hydrothermal mineral of the pyrochlore supergroup, which forms external rims around grains of earlier U-rich hydroxykenopyrochlore and separated crystals in voids of dolomite carbonatite veins. The mineral is named in accordance with the pyrochlore supergroup nomenclature.
${\rm Fe}_{{\rm 0}{\rm. 02}}^{2 +} $)Σ2.00(Nb1.68Ti0.23Ta0.09)Σ2.00O6.03(OH1.04F0.01)Σ1.05. The simplified formula is (Na,Ca,Ce)2Nb2O6(OH). The mineral dissolves slowly in hot HCl. The strongest X-ray powder-diffraction lines [listed as (d in Å)(I)(hkl)] are as follows: 5.96(47)(111), 3.110(30)(311), 2.580(100)(222), 2.368(19)(400), 1.9875(6)(333), 1.8257(25)(440) and 1.5561(14)(622). The crystal structure of hydroxynatropyrochlore was refined to R1 = 0.026 on the basis of 80 unique observed reflections. The mineral belongs to the pyrochlore structure type A2B2O6Y1 with octahedral framework of corner-sharing BO6 octahedra with A cations and OH groups in the interstices. The Raman spectrum of hydroxynatropyrochlore contains characteristic bands of the lattice, BO6, B–O and O–H vibrations and no characteristic bands of the H2O vibrations. Within the Kovdor phoscorite–carbonatite pipe, hydroxynatropyrochlore is the latest hydrothermal mineral of the pyrochlore supergroup, which forms external rims around grains of earlier U-rich hydroxykenopyrochlore and separated crystals in voids of dolomite carbonatite veins. The mineral is named in accordance with the pyrochlore supergroup nomenclature.