Introduction
Alteplase (tissue plasminogen activator) has been a proven treatment for acute ischaemic stroke since 1995.1, 2 Additionally, clinicians and health systems appreciate that the effectiveness of this treatment is highly time-dependent,Reference Saver3, Reference Emberson, Lees and Lyden4 which has resulted in calls to reduce treatment times, or door-to-needle (DTN) times, at hospitals to a median of 45 minutes by the US Target Stroke Initiative5, Reference Fonarow, Smith and Saver6 and SITS WATCH,7 and to a median of 30 minutes by Canadian neurologistsReference Kamal, Benavente and Boyle8 and the Canadian Best Practice Recommendation for hyper-acute stroke care.Reference Casaubon, Boulanger and Blacquiere9 Numerous hospitals have reported reductions in their DTN times from their local quality improvement initiatives.Reference Meretoja, Strbian and Mustanoja10–Reference Kamal, Smith, Jeerakathil and Hill14 However, most of these reductions have been achieved in large tertiary centres that are well resourced with stroke neurologists, neurology trainees, round the clock access to imaging technicians and radiologists, and pre-existing streamlined processes in the Emergency Department (ED). There are unique failures and vulnerabilities in the DTN process for community hospitals when compared to academic hospitals, and there are more total number of failures at community hospitals compared to academic centres, which makes reducing the DTN time at community hospitals more difficult.Reference Prabhakaran, Khorzad, Brown, Nannicelli, Khare and Holl15 A single-centre study in the Netherlands reported reducing median DTN times to 25 minutes from 60 minutes at a community hospital; however, this hospital has neurology residents in-house and their stroke volumes are large enough that the hospital would not be considered a community hospital in Canada.Reference Van Schaik, Van der Veen, Van den Berg-Vos, Weinstein and Bosboom16 There have also been two abstracts published which show reductions of DTN at community hospitals; however, their final DTN numbers are well above the 30- to 45-minute medians targets, as one hospital reduced their times from 77 minutes to 70 minutesReference Linzey, Shilling-brewer, Thibodeaux, Cory, Berger and Neil17 and the other reduced the DTN time from 65 minutes to 50 minutes.Reference Funk18 Furthermore, results from the SITS registry suggests that smaller hospitals are seeing little or no improvement in their DTN times.Reference Strbian, Ahmed and Wahlgren19, Reference Smith20 We will present the DTN results from a community hospital through their participation in a provincial quality improvement initiative. The overall objective of this initiative was to reduce the median DTN time across the entire province to a median of 30 minutes.
Methods
Red Deer Regional Hospital Centre (RDRHC) participated in a provincial initiative aimed at improving DTN times across the province called Quality Improvement and Clinical Research (QuICR) Alberta Stroke Program’s DTN initiative (by receiving funding from Alberta Innovates’ CRIO Team grant).21 The provincial initiative used the Institute of Healthcare Improvement’s Breakthrough Series Collaborative model to improve DTN times across the province.22 The Improvement Collaborative provided participating hospitals with a change package detailing known interventions for reducing DTN times,Reference Ford, Williams and Spencer13, Reference Funk18, Reference Strbian, Ahmed and Wahlgren19 and the hospitals used Plan-Do-Study-Act cycles to customize the interventions for their site. The change package was developed based on existing literature on changes that result in improved DTN time,Reference Fonarow, Smith and Saver6, Reference Kamal, Benavente and Boyle8, Reference Meretoja, Strbian and Mustanoja10–Reference Busby, Owada and Dhungana12, Reference Kamal, Smith, Jeerakathil and Hill14 and it included additional interventions outside of existing literature. These interventions included having Emergency Medical Service (EMS) place IV line en route to hospital, swarming the patient upon arrival to obtain history and to assess patient and giving the treatment team a small reward for each fast treatment case.
The RDRHC is a mid-sized referral community hospital with 370 beds, and it serves the city of Red Deer (population = 100,000) in the Canadian province of Alberta. It is a primary stroke centre within Alberta’s stroke system, serving a total stroke catchment population of 150,000 people with an average of 400 annual stroke admissions. As a primary stroke centre, this hospital provides imaging and thrombolysis to acute stroke patients. It is staffed by four to five community neurologists and locum neurologists without neurology residents or fellows. All neurologists participate in stroke treatment, and they provide 24/7 coverage of hyper-acute strokes arriving in the ED. The RDRHC was able to provide consistent 24/7 coverage by 2015; before this, there were occasional times without neurology coverage for hyper-acute stroke patients arriving in the ED. The neurologists are fee-for-service and bill a general consultation fee. The neurologists and radiologists may interpret imaging through joint decision-making, based on their sub-specialty, as there is one stroke neurologist and two neuro-radiologists on staff, but they are not always readily available. The emergency physician is involved in the initial rapid assessment of patient stability upon arrival in the ED and the neurologist assumes responsibility of the patient once the patient is deemed to be stable. The RDRHC have CT and CTA technologists in-house 24/7, and they have radiologist in-house from 8 a.m. to 11 p.m. every day with on-call service outside of these hours.
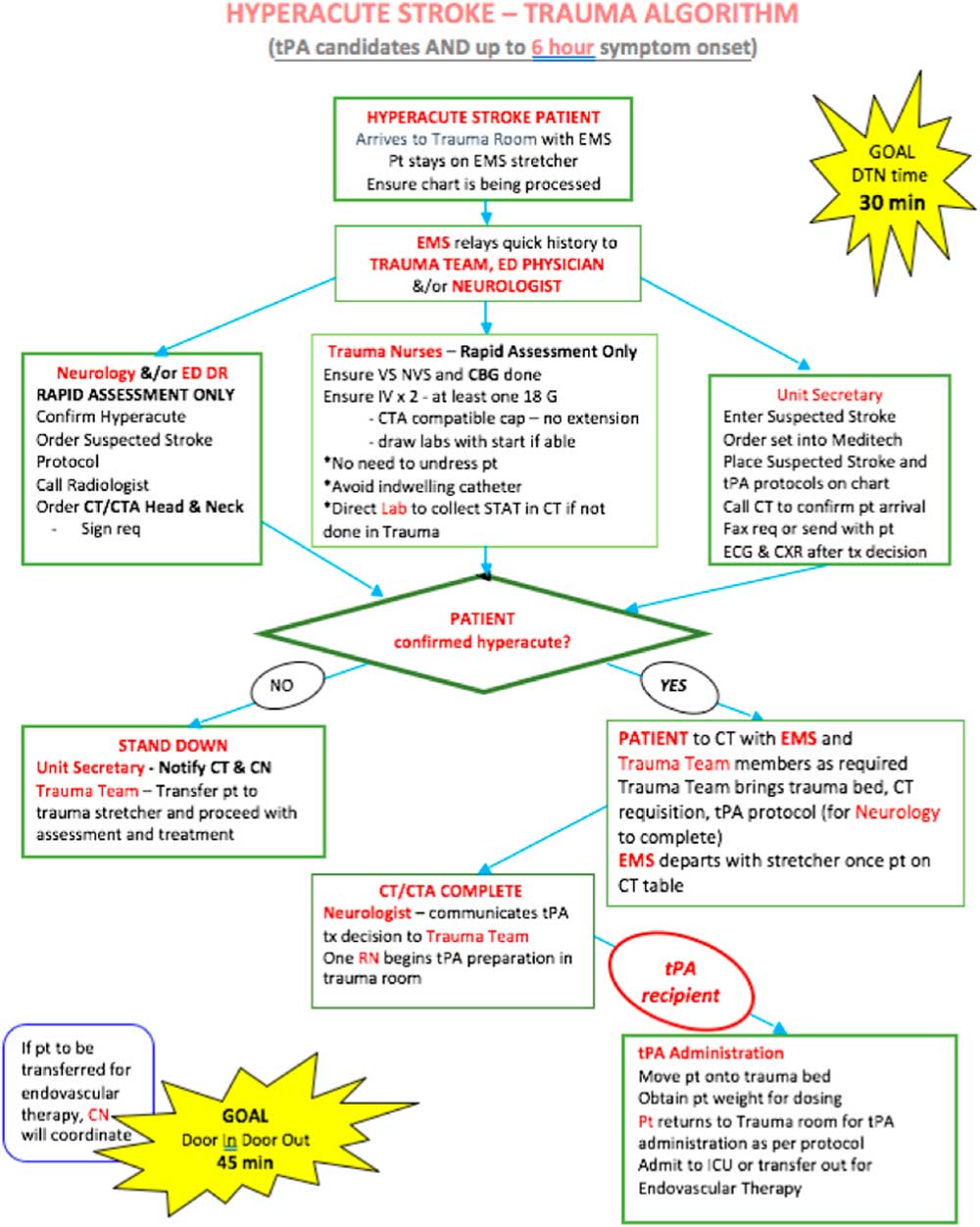
The QuICR DTN initiative started in July 2015 with a site visit from QuICR DTN experts and ended at the end of September 2016. The pre-hospital process was improved. Before the arrival of the stroke patient, paramedics identify a STAT stroke patient through the provincial stroke screen, and they alert the hospital of an incoming STAT stroke patient through a secure cell phone line, where they relay identifiable patient information. A process was implemented where the triage nurse alerts the neurologist, CT technician and ED of an arriving acute stroke patient when they are arriving by ambulance, or immediately alerts them that an acute stroke patient has arrived at the ED by a private vehicle. Since RDRHC’s participation in the QuICR DTN initiative, it has implemented a streamlined acute stroke algorithm (Figure 1). This algorithm included a team-based approach by the entire care team, who “swarms” the patient immediately upon arrival. The algorithm provides clearly defined roles for each team member. The team works in parallel to ensure that the patient is stable and confirm an acute stroke diagnosis. The patient is then taken to the CT scanner on the EMS stretcher. Once an acute ischaemic stroke diagnosis is confirmed and alteplase treatment ordered, the treatment is initiated in the CT scanner or the trauma bay. Treatment can be initiated in either the trauma bay or in the CT scanner depending on whether there are delays in preparing the alteplase for administration; however, the trauma bay and CT scanner are < 1 minute from each other, so there are not huge delays for those patients who are moved to the trauma bay to initiate treatment. These changes used three key strategies for reducing DTN time: (1) taking the patient to the CT scanner on the EMS stretcher;Reference Kamal, Holodinsky and Stephenson11, Reference Kamal, Smith, Jeerakathil and Hill14 (2) administering alteplase in the CT scanner or imaging area and (3) single-call activation of the stroke team for an incoming (when arriving by ambulance) or in ED acute stroke patient.Reference Kamal, Holodinsky and Stephenson11, Reference Kamal, Smith, Jeerakathil and Hill14, Reference Xian, Xu and Lytle23, Reference Xian, Smith and Zhao24 The team that undertook these changes was an interdisciplinary team made up of a community neurologist, stroke coordinator, emergency nurse, paramedic, diagnostic imaging technician and a hospital administrator. Data feedback is provided centrally by QuICR and internally at the hospital by their stroke coordinator: QuICR provides provincial medians for DTN times; QuICR also provides specific DTN times for all treated patients in the province ranked from fastest to slowest; RDRHC posts their DTN times monthly in the ED; their DTN times are presented at monthly staff meetings on the stroke unit and RDRHC reports their DTN times and treatment volumes every 2 months at their Stroke QI Committee meetings.
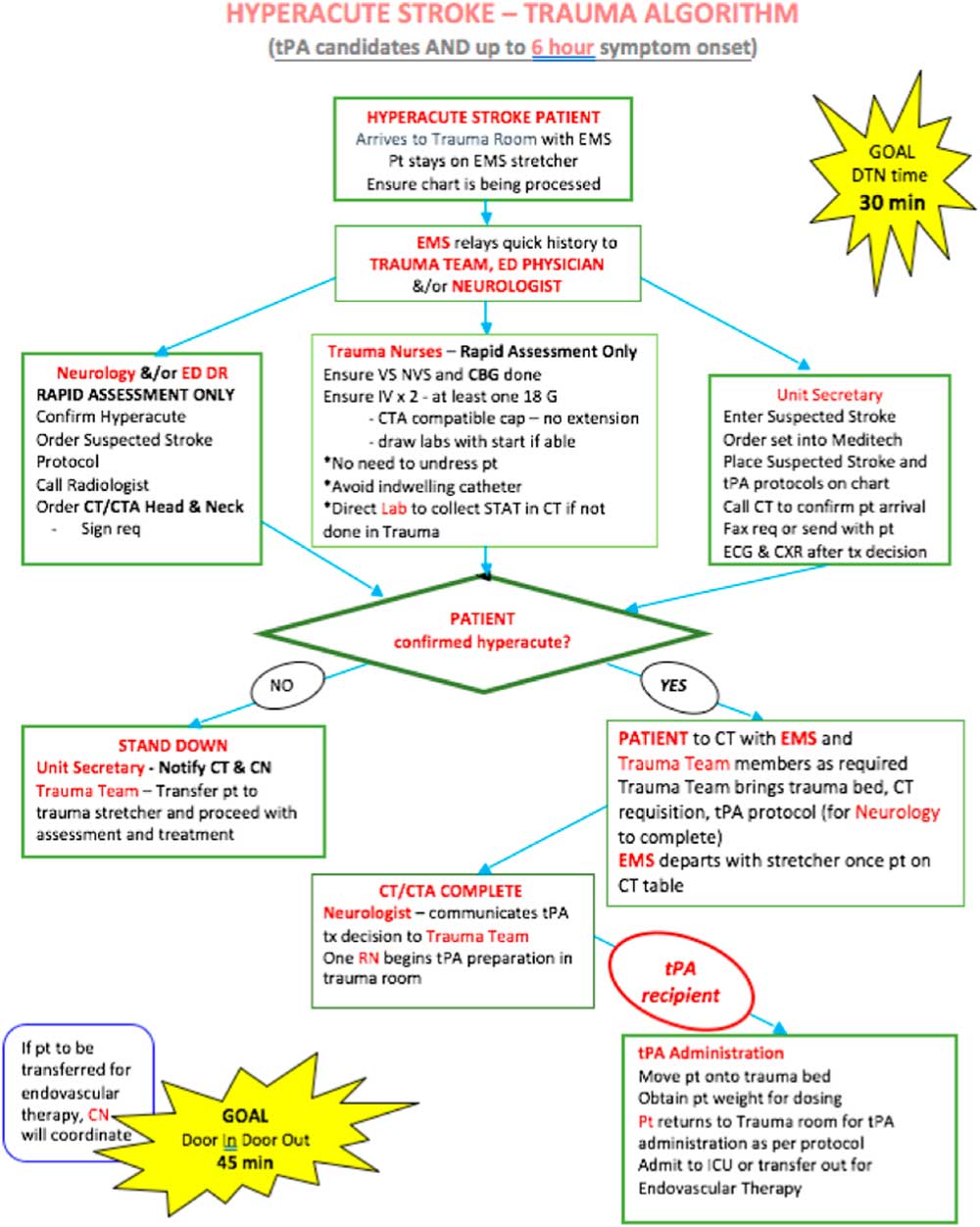
Figure 1 Red Deer Community Hospital’s algorithm for acute ischaemic stroke treatment. DTN = door-to-needle; EMS = Emergency Medical Service; CT = computed tomography, CN = charge nurse; pt = patient; ICU = intensive care unit; tPA = tissue plasminogen activator
The RDRHC has been collecting and submitting DTN data to provincial groups since July 2007. From July 2007 to June 2015, data including patient demographics, stroke data and time metrics for all patients that received alteplase were submitted to the Alberta Provincial Stroke Strategy (APSS) as part of a provincial initiative to set up an acute stroke system of care; data were collected by a stroke coordinator at the RDRHC, entered into a paper form and then scanned to a central group to be entered into the APSS registry.Reference Jeerakathil, Burridge, Thompson, Fang and Hill25 Since July 2015, data have been entered into the QuICR registry by the RDRHC stroke coordinator (ES), which is a provincial registry that captures patient demographics, stroke data and time metrics for all patients that received alteplase and/or endovascular therapy in Alberta. Additionally, RDRHC has been collecting data on all patients that have received alteplase locally at their hospital. Data from the APSS registry and the QuICR registry were checked against the internal hospital records to ensure that all alteplase treated patients were captured in this study. Ethics board approval was received from the University of Calgary’s Conjoint Research Ethics Board for the QuICR registry and quality improvement of DTN times across the province.
Door-to-needle times and volumes were analysed by trends and comparison between the pre-implementation and post-implementation phases of RDRHC’s process changes as described above. The trend analysis included annual trends of median DTN times and the number of patients receiving thrombolysis from 2007 to 2017. The results were visualized using Tableau (version 10.2, Seattle, WA, USA). The DTN time comparison between the pre- and post-intervention periods was conducted using a Mann-Whitney U-test because DTN data have a non-normal distribution (right-skewed). This comparison was conducted using STATA (version 13, College Station, TX, USA). The analysis included all patients that received alteplase with a community onset stroke, and no patients were excluded based on system or patient factors. Strokes that occurred in RDRHC inpatients (hospital onset stroke patients) were excluded.
Results
There were 289 patients thrombolysed at RDRHC from July 2007 to December 2017. There were 165 patients thrombolysed before the implementation of the acute stroke algorithms (the intervention) from July 2007 to June 2015. There were 104 patients thrombolysed after the intervention from January 2016 to December 2017. There were 20 patients thrombolysed during the intervention phase from July 2015 to December 2015.
The annual trend of median DTN time with the 25–75th percentile band and the annual trend of the number of patients treated are shown in Figure 2. The median DTN time dropped from 88 minutes (70–107 minutes) in 2008 to 30 minutes (22–41 minutes) in 2017. During this same period, the number of patients per year receiving treatment increased from 9 in 2008 to 63 in 2017.
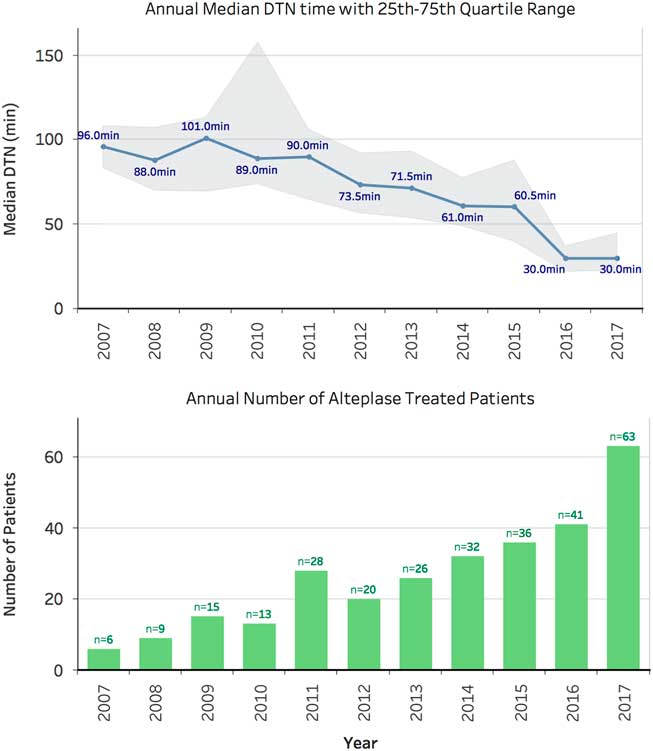
Figure 2 Annual trend of the total number of alteplase treated patients (bottom) and median door-to-needle time in minutes with the 25th and 75th quartile range shown (top). Total n = 289 DTN = door-to-needle time
In comparing the pre- and post-intervention phases, the median DTN time dropped from 77 minutes (60–103 minutes) to 30 minutes (22–42 minutes). This resulted in a highly significant difference between the two periods (p < 0.001). The variability in DTN time also reduced by 50% with the interquartile range (IQR) dropping from 43 minutes in the pre-intervention period to 20 minutes in the post-intervention period. Figure 3 shows a box-whisker plot of the pre- and post-intervention periods.
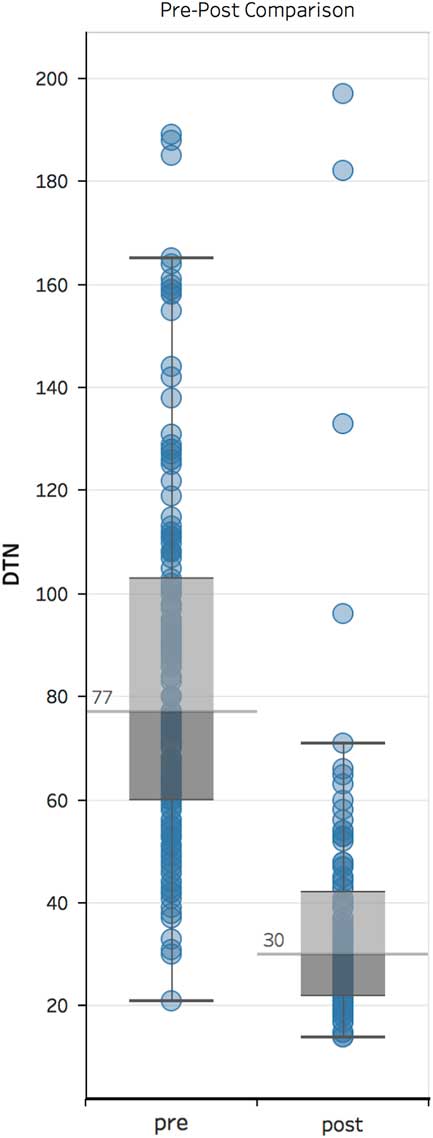
Figure 3 Box-whisker plot of Red Deer Community Hospital comparing DTN times in the pre- and post-intervention periods. DTN = door-to-needle
Interpretation
The successes at RDRHC provide evidence that community hospitals can achieve median DTN times of 30 minutes or less, and the goal set by the Canadian Best Practice Recommendations is possible at similar hospitals.Reference Casaubon, Boulanger and Blacquiere9 Pre-intervention annual median DTN time times at RDRHC were consistently around 90 minutes with a broad IQR of 40 minutes or more. After the quality improvement interventions, RDRHC was able to achieve a median DTN time of 30 minutes with an IQR of around 20 minutes despite being a community hospital staffed with community neurologists.
In parallel, a remarkable increase in the annual number of patients that received treatment with alteplase occurred. The trend shows an increase from 9 to 29 patients being treated annually in the pre-intervention phase to 63 patients being treated in 2017. This increase could have been due to several factors. The focus on fast treatment and the implementation of the acute stroke algorithm could have allowed the care team to become more familiar with the evidence and treatment process. Second, the care team potentially saw improved patient outcomes through faster treatment. This same effect was seen in Helsinki in the seminal DTN improvement study.Reference Meretoja, Strbian and Mustanoja10 Additionally, there have been several national efforts such as the updated Canadian Best Practice Recommendations for hyper-acute stroke that have illuminated the importance of fast thrombolysis treatment, which likely also increased awareness and likely the number of treated cases.Reference Casaubon, Boulanger and Blacquiere9 There are also some critical learnings from these improvements at RDRHC, where there is continued interest to further understand and improve their hyper-acute stroke process; for example, there is an off-shoot study being undertaken by a pharmacist student to determine if having a pharmacist present during the process reduces DTN times, as a pharmacist is present in some STAT stroke cases. Additionally, the stroke team always review their cases and iterate their processes to ensure that all patients are treated in a timely manner.
Acute stroke cases were ascertained through the electronic system where all STAT stroke patients are identified. Although we have endeavoured to include all alteplase treated patients presenting acutely to hospital, we did not conduct an exhaustive chart review of all stroke patients admitted to RDRHC. There is a possibility that some patients were excluded from this study that received alteplase. The intervention period was defined by specific dates, but the nature of quality improvement interventions is that they are not “turned on” on a specific date, and it is possible that some improvements were being made during the period we defined as pre-intervention. As the trend shows, there were some improvements made during the pre-intervention period. This may have resulted in overestimation of the magnitude of the intervention. Additionally, the patient population between the pre- and post-intervention periods may have been different, which could have an effect on the difference in DTN times. The DTN data that was presented was not adjusted for stroke severity, sex, age and co-morbidities. Additionally, we have not reviewed outcomes and adverse events; although existing research shows that faster treatment results in better outcomes with no difference in adverse events,Reference Emberson, Lees and Lyden4, Reference Fonarow, Smith and Saver6 we have not confirmed this for these patients.
A critical piece to any quality improvement effort is to ensure that the improvements are maintained. These results show that RDRHC was able to sustain their improvement for more than 1 year after the provincial initiative ended. They were able to achieve this by the following: continued data feedback and data review in internal meetings; internal championing on an ongoing basis of fast DTN times especially by the physician leadership; informing staff about outcomes for specific treated patients and an engaged stroke coordinator was able to act as a liaison for various staff to learn more about DTN process and performance. These sustainment efforts are continuing to-date.
In conclusion, community hospitals staffed with community neurologists can achieve median DTN times of 30 minutes or less. We cannot confirm if similar results are possible in community hospitals that do not have in-house community neurologists. Supported by a provincial DTN improvement initiative, RDRHC was able to reduce the DTN times from a median of 77 minutes to 30 minutes by implementing the following changes: early activation of neurologists and the care team, the care team meeting the patient immediately upon arrival, parallel processes, keeping the patient on the EMS stretcher to the CT scanner and administering alteplase in the CT or imaging area.
Disclosures
Noreen Kamal reports grants from Alberta Innovates, during the conduct of the study.
Michael D. Hill owns stock in Calgary Scientific Incorporated, a company that focusses on medical imaging software; is a director of the Canadian Neurological Sciences Federation, a not-for-profit group; and has received grant support from Alberta Innovates Health Solutions, CIHR, Heart & Stroke Foundation of Canada and National Institutes of Neurological Disorders and Stroke.
Elaine Shand, Robert Swanson, Thomas Jeerakathil, Jennifer Bestard, Oje Imoukhuede, Irvin Heinrichs, Jackie Bakker, Carol Stoyberg, Laura Fowler, Sandy Duckett, Scott Holsworth, Balraj Mann and Shelley Valaire have nothing to disclose.
Statement of Authorship
Noreem Kamal: writing, data analysis, manuscript review, central study/QI leadership. Elaine Shand: manuscript review, study design, intervention. Robert Swanson: manuscript review, intervention. Michael D. Hill: manuscript review, central study/QI leadership. Thomas Jeerakathil: manuscript review, central study/QI leadership. Oje Imoukheude: manuscript review, intervention. Irvin Heinrichs: manuscript review, intervention. Jackie Bakker: manuscript review, intervention. Carol Stoyberg: manuscript review, intervention. Laura Fowler: manuscript review, intervention. Sandy Duckett: manuscript review, intervention. Scott Holsworth: manuscript review, intervention. Balraj Mann: manuscript review, central study/QI leadership. Shelley Valaire: manuscript review, central study/QI leadership. Jennifer Bestard: manuscript review, study design, intervention.