Nucleation in Atomic, Molecular, and Colloidal Systems
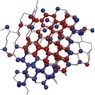
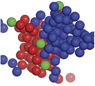
Nucleation in atomic, molecular, and colloidal systems. Nucleation is the first step in the formation of many materials. The simple picture of nucleation, inherent in classical nucleation theory, that has prevailed for more than a century does not account for complex multistep nucleation pathways observed in recent years in experiments and simulations. This issue illustrates and describes the many complex nucleation pathways seen across a range of material systems. The cover shows an artistic rendition of nucleation from a supersaturated calcium carbonate solution. The structures of the initial clusters that form as predicted by molecular dynamics simulations are depicted in full atomistic detail in the foreground and in red in the background. The simulations further predict that the clusters are dense liquid droplets that must later dehydrate to form the solid phase. Results like these call into question the century-old view of how crystals nucleate from solution. Artwork by Adam Wallace. See the technical theme that begins on page 357.
Nucleation in Atomic, Molecular, and Colloidal Systems
Introduction
Nucleation in atomic, molecular, and colloidal systems
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 357-360
-
- Article
-
- You have access
- HTML
- Export citation
Other
Meet Our Authors
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 361-362
-
- Article
-
- You have access
- HTML
- Export citation
Nucleation in Atomic, Molecular, and Colloidal Systems
Research Article
What do crystals nucleate on? What is the microscopic mechanism? How can we model nucleation?
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 363-368
-
- Article
- Export citation
Nonclassical pathways of crystallization in colloidal systems
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 369-374
-
- Article
- Export citation
Shape change in crystallization of biological macromolecules
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 375-380
-
- Article
- Export citation
DNA-programmable particle superlattices: Assembly, phases, and dynamic control
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 381-387
-
- Article
- Export citation
Nucleation on surfaces and in confinement
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 388-392
-
- Article
- Export citation
Nucleation of open framework materials: Navigating the voids
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 393-398
-
- Article
- Export citation
Technical Feature
Research Article
Materials science with large-scale data and informatics: Unlocking new opportunities
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 399-409
-
- Article
-
- You have access
- HTML
- Export citation
News & Analysis
Materials News
Other
Energy Focus: Elastic carbon films for micro-supercapacitors fabricated by wafer-scale process
-
- Published online by Cambridge University Press:
- 04 May 2016, p. 351
-
- Article
-
- You have access
- HTML
- Export citation
Energy Focus: Lithium-ion intercalation visualized in situ with new atomic-scale imaging technique
-
- Published online by Cambridge University Press:
- 04 May 2016, p. 352
-
- Article
-
- You have access
- HTML
- Export citation
Glassy poly(lactide) toughened with diblock copolymer micelles
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 352-353
-
- Article
-
- You have access
- HTML
- Export citation
Energy Focus: Carbon nanotubes improve radiation resistance of aluminum
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 353-354
-
- Article
-
- You have access
- HTML
- Export citation
Science Policy
Other
US administration makes efforts to increase inclusiveness in STEM industries: www.whitehouse.gov/demo-day
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 355-356
-
- Article
-
- You have access
- HTML
- Export citation
Society News
News
Robert Cammarata of Johns Hopkins University remembered
-
- Published online by Cambridge University Press:
- 04 May 2016, p. 410
-
- Article
-
- You have access
- HTML
- Export citation
ABET proposes accreditation criteria revisions
-
- Published online by Cambridge University Press:
- 04 May 2016, p. 411
-
- Article
-
- You have access
- HTML
- Export citation
Features
Books
Other
Solar Energy: An Introduction Michael E. Mackay: Oxford University Press, 2015 336 pages, $98.50 (paperback $49.95, e-book $48.99) ISBN 978-0-19-965210-5
-
- Published online by Cambridge University Press:
- 04 May 2016, p. 412
-
- Article
-
- You have access
- HTML
- Export citation
Moore’s Law: The Life of Gordon Moore, Silicon Valley’s Quiet Revolutionary Arnold Thackray, David C. Brock, and Rachel Jones: Basic Books, 2015 530 pages, $35.00 ISBN 978-0-465-05564-7
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 412-413
-
- Article
-
- You have access
- HTML
- Export citation
Nanoparticles Raz Jelinek: De Gruyter, 2015 283 pages, $98.00 (e-book $98.00) ISBN 978-3-11-033002-1
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 413-414
-
- Article
-
- You have access
- HTML
- Export citation
Posterminaries
Article Commentary
THE LARGE PROBLEM OF WATER SHORTAGES
-
- Published online by Cambridge University Press:
- 04 May 2016, pp. 415-416
-
- Article
-
- You have access
- HTML
- Export citation