The aim of this work is the investigation of the metal-hydride transformation in magnesium (Mg) nanoparticles both as a function of particle size and in response to surface functionalization by clusters of transition metals (TM): Pd, Ni, Ti.
Mg nanoparticles were synthesized by the inert-gas condensation technique, which yields single crystals with six-fold symmetry whose average size can be controlled by tuning the inert gas pressure. After the synthesis the nanoparticles were passivated by slow exposure to oxygen, obtaining a core-shell morphology where a metallic core is coated by a MgO shell of about 5 nm thickness.
The material structure was investigated by Transmission Electron Microscopy (TEM), also in High Resolution (HRTEM) mode, and by X-Ray Diffraction (XRD). The sorption kinetics were analysed by a volumetric Sievert apparatus, which also allowed for a determination of the activation energies.
Small nanoparticles (≈35 nm) display interesting kinetics with gravimetric capacity of 4.5 wt.% at saturation, limited by the oxide fraction. Hydride formation proceeds by one-dimensional growth controlled by diffusion through the hydride, while the reverse transformation to metal involves interface-controlled three-dimensional growth of nuclei formed at constant rate.
On the contrary, large nanoparticles (≈450 nm) exhibit very low reactivity due to reduced probability of hydrogen dissociation/recombination and nucleation at the particle surface. For this reason, large nanoparticles were surface-decorated by TM through in situ evaporation in the inert-gas condensation chamber. This procedure results in clusters of 3-4 nm located over a portion of the MgO shell, as shown by XRD and HRTEM on Pd-decorated sample. This treatment results in dramatically improved hydrogen sorption behavior. In fact, previously inert nanoparticles now exhibit of up to 5.6 wt.%.
Real-time diffraction studies using Synchrotron Radiation were carried out during hydrogen desorption on the Pd-decorated nanoparticles. We clearly show that a Mg-Pd intermetallic phase is formed after the first heating treatment and takes active part in the transformation.



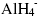 complex into the vacancy in another
complex into the vacancy in another