Article contents
Synthesis of Carbon-Hydrotalcite Complex and Its Thermal Degradation Behavior
Published online by Cambridge University Press: 28 February 2024
Abstract
An abstract is not available for this content so a preview has been provided. Please use the Get access link above for information on how to access this content.
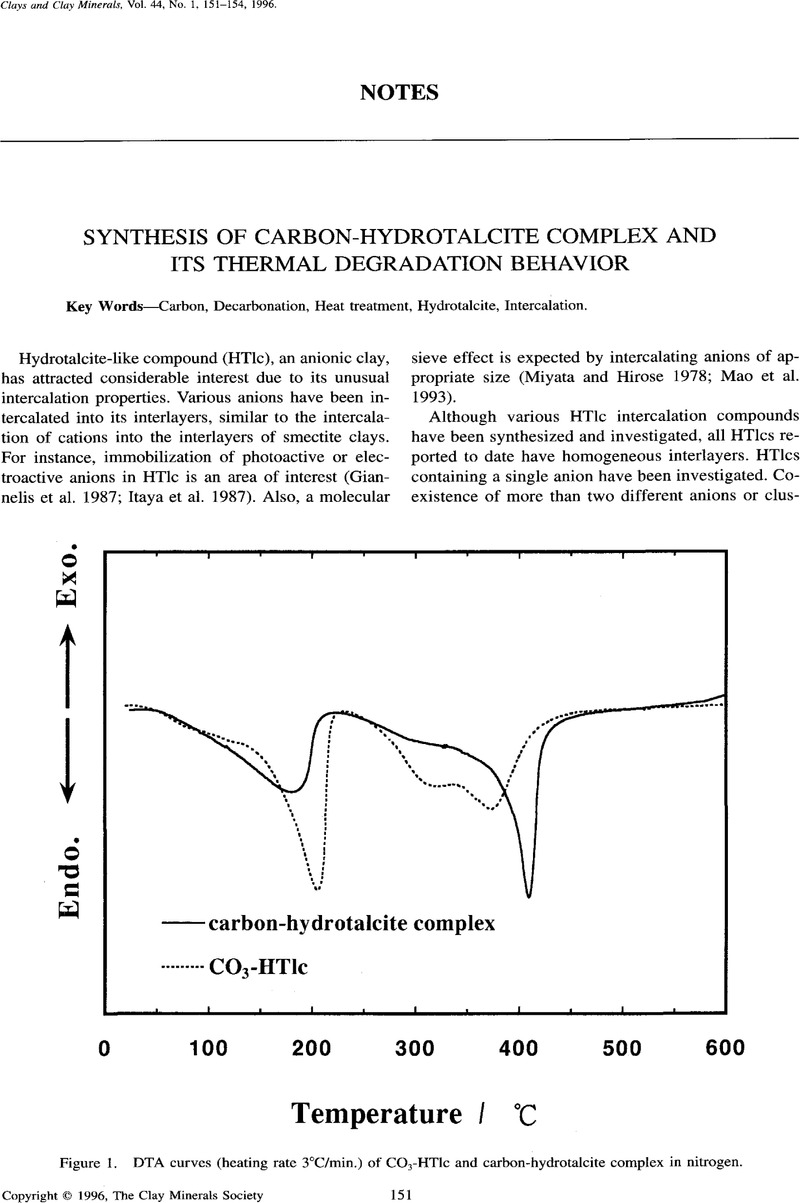
- Type
- Notes
- Information
- Copyright
- Copyright © 1996, The Clay Minerals Society
References
Giannelis, E.P., Nocera, D.G. and Pinnavaia, T.J.. 1987. Anionic photocatalysts supported in layered double hydroxides: Intercalation and photophysical properties of a ruthenium complex anion in synthetic hydrotalcite. Inorg Chem 26: 203–205.CrossRefGoogle Scholar
Hibino, T., Yamashita, Y., Kosuge, K. and Tsunashima, A.. 1995. Decarbonation behavior of Mg-Al-CO3 hydrotalcite-like compounds during heat treatment. Clays & Clay Miner 43: 427–432.CrossRefGoogle Scholar
Hudson, M.J., Carlino, S. and Apperley, D.C.. 1995. Thermal conversion of layered (Mg/Al) double hydroxide to the oxide. J Mater Chem 5: 323–329.CrossRefGoogle Scholar
Itaya, K., Chang, H.C. and Uchida, I.. 1987. Anion-exchanged hydrotalcite-like-clay-modified electrodes. Inorg Chem 26: 624–626.CrossRefGoogle Scholar
MacKenzie, K.J.D., Meinhold, R.H., Sherriff, B.L. and Xu, Z.. 1993. 27Al and 25Mg solid-state magic-angle spinning nuclear magnetic resonance study of hydrotalcite and its thermal decomposition sequence. J Mater Chem 3: 1263–1269.CrossRefGoogle Scholar
Mao, G., Tsuji, M. and Tamaura, Y.. 1993. Synthesis and CO2 adsorption features of a hydrotalcite-like compound of the Mg2+-Al3+-Fe(CN)64– system with high layer-charge density. Clays & Clay Miner 41: 731–737.CrossRefGoogle Scholar
Miyata, S. and Hirose, T.. 1978. Adsorption of N2, O2, CO2 and H2 on hydrotalcite-like system: Mg2+-Al+-(Fe(CN)6)4–. Clays & Clay Miner 26: 441–447.CrossRefGoogle Scholar
Oya, A., Mita, H., Tosaka, Y. and Otani, S.. 1990. Thermal degradation behavior of hydrotalcite/flavianic acid complex in nitrogen. Appl Clay Sci 5: 13–22.CrossRefGoogle Scholar
Oya, A., Omata, Y. and Otani, S.. 1985. Thermal degradation behavior of montmorillonite-α-naphthylamine complex under nitrogen. J Mater Sci 20: 255–260.CrossRefGoogle Scholar
Pesic, L., Salipurovic, S., Markovic, V., Vucelic, D., Kagunya, W. and Jones, W.. 1992. Thermal characteristics of a synthetic hydrotalcite-like material. J Mater Chem 2: 1069–1073.CrossRefGoogle Scholar
Rey, F., Fornés, V. and Rojo, J.M.. 1992. Thermal decomposition of hydrotalcites: An infrared and nuclear magnetic resonance spectroscopic study. J Chem Soc Faraday 88: 2233–2238.CrossRefGoogle Scholar
The following corrections have been issued for this article:
- 15
- Cited by
Linked content
Please note have been issued for this article.




