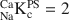Volume 52 - February 2004
Research Article
Interpretation of infrared spectra of dioctahedral smectites in the region of OH-stretching vibrations
- Bella B. Zviagina, Douglas K. McCarty, Jan Środoń, Victor A. Drits
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 399-410
-
- Article
- Export citation
-
Dioctahedral smectite samples of a wide range of compositions (beidellites, montmorillonites, nontronites, Fe-rich montmorillonites and Al-rich nontronites) were studied by infrared (IR) spectroscopy. A special sample-preparation technique was used to eliminate the contribution of molecular water. The OH-stretching regions of the spectra were decomposed and curve-fitted, and the individual OH-stretching bands were assigned to all the possible types of OH-bonded cation pairs that involve Al, Mg and Fe. The integrated optical densities of the OH bands were assumed to be proportional to the contents of the specific types of OH-linked cation pairs with the absorption coefficients being the same for all individual OH bands. Good agreement between the samples’ octahedral cation compositions calculated from the IR data and those given by crystal-chemical formulae was obtained for a representative collection of samples in terms of a unique set of individual OH-band positions that vary within narrow wavenumber intervals. This has allowed us to minimize the ambiguity in spectra decomposition caused by the poor resolution of smectite spectra and confirmed the validity of the resulting band identification.
The bands associated with specific OH-bonded cation pairs in the spectra of smectites are, on the whole, shifted to greater wavenumbers with respect to the corresponding bands in micas. In addition to OH bands that refer to the smectite structure, AlOHAl and AlOHFe bands of the pyrophyllite structural fragments were identified. The band-position variation ranges overlap in a few cases (AlOHFe and MgOHMg; AlOHAl of smectite and AlOHFe of pyrophyllite-like component).
Unambiguous interpretation of the OH-stretching vibrations was found to be possible only for smectite samples with known chemical compositions, so that IR data cannot be used for quantitative determination of octahedral cation composition of mixtures of dioctahedral 2:1 phyllosilicates. In the case of the studied monomineral smectites with known chemical compositions, IR data provided information on the short-range order/disorder in the distribution of octahedral cations along cation-OH-cation directions. This information can be employed, in conjunction with the data of other spectroscopic and diffraction techniques, in the analysis of short-range octahedral cation distribution.
Improved Constraints on Sedimentary Environments of Palygorskite Deposits of the Hawthorne Formation, Southern Georgia, from a Detailed Study of a Core
- Mark P. S. Krekeler
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 253-262
-
- Article
- Export citation
-
The sedimentology and mineralogy of a 2.5 m core from a palygorskite deposit of the Miocene Hawthorne Formation, southern Georgia is described. The lithology involves laminated clay-rich sediment composed of ∼90% clay and 10% sand, with six clay-pebble layers present. Sand to pebble-size clasts of phosphate material are common throughout the core. The sand laminations are probably flood-related and the clay-pebble layers are storm deposits, with the pebbles being derived locally from subaerial environments. Phosphate clasts are reworked bone material.
The sands are quartz-rich and are subarkosic in composition with average quartz counts of 86.50% and average total feldspar counts of 11.50%. Heavy minerals observed include orthopyroxene, clinopyroxene, amphibole, zircon, rutile, garnet, tourmaline, kyanite, muscovite, biotite, spinels and opaques. Palygorskite fibers dominate the clay-size fraction of the samples and comprise ∼80–90% of sample material with smectite comprising the remainder. Hydroxylapatite comprises ∼3% of sediment volume and occurs as individual euhedral hexagonal crystals and as clusters of crystals.
Investigation of this core suggests that the palygorskite deposit represents a dynamic system with regular flooding and storm deposition being common. Mineral composition of sands may be useful for stratigraphic correlation of palygorskite deposits in the Apalachicola Embayment. This study supports the general environmental interpretations of previous workers for the palygorskite deposits of southern Georgia, but provides greater detail.
Hydraulic Conductivity and Cation Exchange in Non-prehydrated And Prehydrated Bentonite Permeated with Weak Inorganic Salt Solutions
- Ho Young Jo, Craig H. Benson, Tuncer B. Edil
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 661-679
-
- Article
- Export citation
-
Sets of replicate hydraulic conductivity tests were conducted using 100 mM KCl and 20 and 40 mM CaCl2 solutions to evaluate how changes in hydraulic conductivity are related to changes in the exchange complex and physical properties (water content and free swell) of prehydrated and non-prehydrated bentonite used for geosynthetic clay liners (GCLs). Essentially no change in hydraulic conductivity and water content (or void ratio) occurred during tests with the 100 mM KCl solution even though K+ was replacing Na+ on the exchange complex. In contrast, for the CaCl2 solutions (20 mM and 40 mM), the hydraulic conductivity increased and the free swell and water content decreased as exchange of Ca2+ for Na+ occurred. Faster changes in hydraulic conductivity and the exchange complex occurred in the tests with the 40 mM CaCl2 solution and the non-prehydrated bentonite (i.e. the hydraulic conductivity changed more rapidly when exchange occurred more rapidly). Even though exchange of Ca2+ for Na+ was essentially complete at the end of testing, the hydraulic conductivity obtained with the 20 mM CaCl2 solution was less and the water content greater than obtained with the 40 mM CaCl2 solution (2.6 × 10−8 cm/s vs. 6.7 × 10−8 cm/s, 122% vs. 111%, and 3.2 vs. 2.9). Similarly, the prehydrated bentonite had lower hydraulic conductivity (1.9 × 10−8 cm/s vs. 6.7 × 10−8 cm/s) and greater water content (167% vs. 111%) than the non-prehydrated bentonite at the end of testing, even though Ca-for-Na exchange was essentially complete.
Characterizing Clay Mineral Suspensions using Acoustic and Electroacoustic Spectroscopy — A Review
- Marianne Guerin, John C. Seaman
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 145-157
-
- Article
- Export citation
-
Recently, significant advances have been made in the theory and application of acoustic and electroacoustic spectroscopies for measuring the particle-size distribution (PSD) and zeta potential (ζ potential) of colloidal suspensions, respectively. These techniques extend or replace other techniques, such as light-scattering methods, particularly in concentrated suspensions. In this review, we summarize acoustic and electroacoustic theory and published results on clay mineral suspensions, detail theoretical constraints, and indicate potential applications for the study of environmentally significant clay mineral suspensions. Using commercially available instrumentation and suspension concentrations up to 45 vol.%, acoustic spectroscopy can characterize particle sizes from 10 nm to 10 µm, or greater. Electroacoustic spectroscopy can determine the ζ potential of a suspension with a precision and accuracy in the mV range. Despite the clear potential for their use in environmental settings, to date, acoustic methods have been used mainly on clay mineral colloids with industrial application, typically combined with similar measurements such as isoelectric point (IEP) determined from shear yield stress or ζ potential from electrophoretic mobility measurements. Potential applications in environmentally relevant suspension concentrations are significant, as PSD and ζ potential are important factors influencing the transport of mineral colloids and associated contaminants through porous media. Applications include determining the effects of suspension concentration, surfactants, electrolyte strength, pH and solution composition on soil clay properties and colloidal interactions, and determining changes in PSD, aggregation and ζ potential due to adsorption or variations in the clay mineralogy.
Clay minerals in the Meuse-Haute Marne underground laboratory (France): Possible influence of organic matter on clay mineral evolution
- Francis Claret, Boris A. Sakharov, Victor A. Drits, Bruce Velde, Alain Meunier, Lise Griffault, Bruno Lanson
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 515-532
-
- Article
- Export citation
-
A clay-rich Callovo-Oxfordian sedimentary formation was selected in the eastern Paris Basin (MHM site) to host an underground laboratory dedicated to the assessment of nuclear waste-disposal feasibility in deep geological formations. As described initially, this formation shows a mineralogical transition from an illite-smectite (I–S) mixed-layered mineral (MLM), which is essentially smectitic and randomly interstratified (R = 0) in the top part of the series to a more illitic, ordered (R ⩾ 1) I–S in its deeper part.
This description has been challenged by using the multi-specimen method developed by Drits et al. (1997a) and Sakharov et al. (1999). It is shown that all samples contain a physical mixture of an unusually (?) illitic (∼65% I) randomly interstratified I-Exp (illite-expandable MLM) and of a discrete smectite, in addition to discrete illite, kaolinite and chlorite. Structural parameters of the different clay phases vary little throughout the series. According to the proposed model, the mineralogical transition corresponds to the disappearance of smectite with increasing burial depth.
Comparison with clay minerals from formations of similar age (Oxfordian-Toarcian) throughout the Paris Basin shows that the clay mineralogy in the deeper part of the series originates from a smectite-to-illite transition resulting from a low-temperature burial diagenesis. The anomalous lack of evolution of clay minerals in the upper part of the series is thought to be related to specific interactions between organic matter and clay minerals.
Alteration of Kaolinite to Cancrinite and Sodalite by Simulated Hanford Tank Waste and its Impact on Cesium Retention
- Hongting Zhao, Youjun Deng, James B. Harsh, Markus Flury, Jeffrey S. Boyle
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-13
-
- Article
- Export citation
-
Caustic nuclear wastes have leaked from tanks at the US Department of Energy’s Hanford site in Washington State (USA) causing hundreds of thousands of gallons of waste fluids to migrate into the underlying sediments. In this study, four simulant tank waste (STW) solutions, which are high in NaOH (1.4 and 2.8 mol/kg), NaNO3 (3.7 mol/kg) and NaAlO2 (0.125 and 0.25 mol/kg), were prepared and reacted with reference kaolinite KGa-1 and KGa-2 at 50 and 80°C for up to 2 months. The structure and morphology of the resulting products were characterized using X-ray diffraction, scanning electron microscopy, and Fourier transform infrared spectroscopy. The products were also examined for cation exchange and Cs+ sorption as a function of ionic strength and types of cations in the background solutions. Cancrinite and sodalite were the only new minerals observed in all of the conditions tested in this experiment. Two major chemical processes were involved in the reactions: dissolution of kaolinite and precipitation of cancrinite and sodalite. Increasing NaOH concentration and temperature, and decreasing NaAlO2 concentration increased the transformation rate. Both cancrinite and sodalite appeared stable thermodynamically under the experimental conditions. The newly formed feldspathoids were vulnerable to acid attack and pronounced dissolution occurred at pH below 5.5. Cancrinite and sodalite can incorporate NaNO3 ion pairs in their cages or channels. Sodium in cancrinite and sodalite was readily exchangeable by K+, but less easily by Cs+ or Ca2+. The feldspathoid products sorb nearly an order of magnitude more Cs+ than the unaltered kaolinite. The Cs adsorption is reduced by competing cations in the background solutions. At low ionic strength (0.01 M NaNO3 or 0.005 M Ca(NO3)2), Ca2+ was more competitive than Na+. When the concentration of the background solution was increased 10 times, Na+ was more competitive than Ca2+.
Electrical conductivity spectra of smectites as influenced by saturating cation and humidity
- Sally D. Logsdon, David A. Laird
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 411-420
-
- Article
- Export citation
-
Electrical conductivity is an important soil property related to salinity, and is often used for delineating other soil properties. The purpose of this study was to examine the influence of smectite properties on the complex electrical conductivity spectra of hydrated smectitic clays. Four smectites were saturated with Ca, Mg, Na or K and equilibrated at four relative humidities ranging from 56 to 99%. X-ray diffraction was used to determine fractions of the various smectite layer hydrates (0 to 4 layers of interlayer water molecules) in each sample. A vector network analyzer was used to determine the real component of the complex electrical conductivity spectra (σ′) for frequencies (f) ranging from 300 kHz to 3 GHz. Values of the dc electrical conductivity(σ0), the frequency where the slope changes in the spectra (fr), and the slope at the high-frequency end of the spectra (n) were determined by fitting σ′ to σ′(f) = σ0(1 + f/fr)n. Both σ0 and fr increased with the total amount of water, the amount of interlayer water, and, for saturating cations in the order K < Mg < Ca < Na. The opposite trends were observed for n. The values of these parameters were influenced by the type of smectite, but the trends were not consistent for the effect of layer charge. The results indicate that interlayer water in smectites contributes to the electrical conductivity of hydrated smectites, and that polarization of water by local electrical fields has a substantial influence on the complex electrical conductivity spectra of smectites. The accuracy of salinity estimates for soils and sediments that are based on conductivity measurements maybe adversely affected unless the effects of hydrated clays on electrical conductivity are considered.
A Microtexture Study of Palygorskite-Rich Sediments from the Hawthorne Formation, Southern Georgia, by Transmission Electron Microscopy and Atomic Force Microscopy
- Mark P. S. Krekeler, Stephen Guggenheim, John Rakovan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 263-274
-
- Article
- Export citation
-
A microtexture analysis by TEM and AFM of palygorskite deposits from the Hawthorne Formation, southern Georgia is given. Palygorskite is the dominant mineral comprising an average of 65–70% of the sample volume with smaller volumes of smectite, illite and kaolinite. Morphologic observations indicate that the palygorskite formed in an unconfined environment, such as in the water column or in open-pore space. Some palygorskite textures appear to be secondary growths filling voids. An unusual texture is observed where smectite or illite-smectite (Reichweite, R = 0) form epitaxially on detrital illite and kaolinite particles. Oxides of Fe and Ti are common, and authigenic cassiterite is present but rare. Apatite is a common trace mineral in these sediments and occurs in a variety of textures. Apatite occurs as clusters which are believed to be small fecal pellets. These clusters have been partially dissolved and recrystallized and the crystals in the clusters are 50–100 µm in diameter. Other apatite crystals occur either as single crystals or in clusters that are not associated with fecal pellets.
The textural data of this study suggest that there was an evolving and complex mineralogical and geochemical system during and after deposition of the palygorskite deposits in the Hawthorne. The epitaxial overgrowths of smectite on detrital illite and kaolinite particles indicate an intermittent stratified water column occurring in the system. Freshwater was introduced into the system from the northeast of the Apalachicola embayment and overrode more saline water in the southwest portion of the embayment. The results of this study are consistent with previous environmental interpretations and provide additional details.
Acoustic and Electroacoustic Characterization of Variable-Charge Mineral Suspensions
- Marianne Guerin, John C. Seaman, Charlotte Lehmann, Arthur Jurgenson
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 158-170
-
- Article
- Export citation
-
Acoustic and electroacoustic measurements of particle-size distribution (PSD) and zeta potential (ζ potential), respectively, were used to obtain in situ measures of the effects of suspension concentration and pH on interactions between mixed-charge clays and clay minerals from a highly weathered sediment. Measurements were obtained in concentrated suspensions as a function of weight fraction and as a function of pH during titrations. Standard dispersion and centrifugation methods were used to obtain a comparative measure of PSD. Thermogravimetric analysis and X-ray diffraction patterns were used to obtain semi-quantitative and descriptive analyses, respectively, of the sediment, which is composed of Fe oxide minerals, kaolinite, gibbsite, quartz, crandallite, chlorite and traces of other clay minerals. Acoustic measurements showed that the PSD of the clay fraction varied with suspension concentration, and electroacoustic measurements showed the ‘bulk’ ζ potential increased in absolute value as the suspension concentration decreased. Titration results were also sensitive to suspension concentration. Acoustic measurements indicated that the suspensions became unstable at ∼pH 7.5–8.0, as the attenuation spectra changed character near this pH and the calculated PSD shifted to a larger particle size. This pH value is near the points of zero charge of goethite and gibbsite, as verified by titrations on mineral standards. The results confirm the central role oxide minerals play in regulating clay mineral interactions in highly weathered sediments, and indicate that the average ζ potential of a suspension may be a poor indicator of controls on suspension stability.
Influence of the Outer Surface Layers of Crystals on the X-Ray Diffraction Intensity of Basal Reflections
- Boris A. Sakharov, Alain Plançon, Bruno Lanson, Victor A. Drits
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 680-692
-
- Article
- Export citation
-
This study presents a mathematical formalism describing diffraction effects from periodic and mixed-layer minerals in which the outer surface layers of crystals possibly differ from layers forming the core of the crystals. The X-ray diffraction (XRD) patterns calculated for structure models of chlorite and irregular chlorite-smectites terminated on both sides of the crystals by either brucite-like sheets or 2:1 layers show the strong influence that different outer surface layers have on the distribution of basal reflection intensities. Simulation of the experimental XRD patterns from two chlorite samples having different Fe contents shows that in these two samples the chlorite crystals were terminated by brucite-like sheets on both sides. In contrast, crystals in a corrensite sample were terminated by water molecules and exchangeable cations. The nature of diffraction effects due to outer surface layers is discussed.
Smectite-to-illite alteration in salt-bearing bentonites (The East Slovak Basin)
- M. Honty, P. Uhlík, V. Šucha, M. Čaplovičová, J. Franců, N. Clauer, A. Biroň
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 533-551
-
- Article
- Export citation
-
The effect of a saline environment on illitization in volcanoclastic rocks is examined in deep boreholes in the East Slovak Basin. Based on X-ray diffraction analysis, it is concluded that illite-smectite (I–S) expandability is always less in the salt-bearing bentonites (SBB) than in the salt-free bentonites (SFB) for a given depth interval. These two lithologies can be distinguished easily by water-leachate chemistry. Within the depth interval 2100–2500 m, the expandability in SBB varies within the range 25–10% expandable with R1 and R3 ordering in SBB and 68–35% expandable with R0 ordering in SFB. In two shallow SBB samples the expandability is close to that of SFB, suggesting that salinity alone does not enhance the illitization; but salinity may enhance it when combined with higher burial temperature. Vitrinite reflectance and Tmax of RockEval pyrolysis measured in adjacent shales confirm that the increased illitization in SBB is not due to heating and/or erosion. The model of burial and thermal history calibrated by organic maturity suggests that the same thermal history produces two different expandabilities in the two lithologies (SBB and SFB). Particle thickness measurements and K-Ar data were used to deduce the crystal growth mechanism of illitization in SBB. Whereas surface-controlled growth is typical for SFB, simultaneous nucleation and growth played a more important role in the case of SBB. The effect of a salty environment on the illitization is not yet fully understood and may have severe consequences for the utilization of bentonites as engineering barriers in radioactive waste disposal sites if salt formations used as host rocks are taken into account.
Behavior of Smectite in Strong Salt Brines under Conditions Relevant to the Disposal of Low- to Medium-Grade Nuclear Waste
- Heiko Hofmann, Andreas Bauer, Laurence N. Warr
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 14-24
-
- Article
- Export citation
-
Two industrial bentonites, IBECO SEAL-80 and TIXOTON TE, have been proposed as potential backfill material in the German Asse salt dome, a test field for the disposal of low- to medium-grade active nuclear waste. Considering the unlikely but possible case of a barrier breakdown with infiltration of a highly concentrated salt brine, the physicochemical stability and material behavior of these bentonites in a saturated salt brine (predominantly MgCl2) at 25°C were studied over the time period of 150 days. The results show that no mineral transformations occurred throughout the duration of the experiments and minor dissolution was only active during the first days. Some chemical properties, namely sorption capability and swelling, were reduced during contact with the salt brine, but could be reversed by removing the salt after treatment. Despite restriction of the CEC in the presence of salt solution, interlayer cation exchange reactions are still active in this environment. The long-term chemical stability of smectite in salt brine is predicted under these low-temperature conditions, but the increased permeability during aggregate formation could lead to physical breakdown of the backfill component.
Heterogeneous mixed-layer clays from the Cretaceous Greensand, Isle of Wight, southern England
- Douglas K. McCarty, Victor A. Drits, Boris Sakharov, Bella B. Zviagina, Alastair Ruffell, Grant Wach
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 552-575
-
- Article
- Export citation
-
The sea-cliffs of the Isle of Wight were deposited during a period of overall sea-level rise starting in the Barremian (Lower Cretaceous) and continuing into the Aptian and Albian. They consist of fluvial, coastal and lagoonal sediments including greensands and clays. Numerous episodes of erosion, deposition and faunal colonization reflect condensation and abandonment of surfaces with firmgrounds and hardgrounds. This study focused mainly on shallow marine cycles where variations in clay mineralogy would not be expected, because overall system composition, sediment source, and thermal history are similar for all the samples in the studied section. Instead we found a wide variety of clay assemblages even in single samples within a 200 m interval.
In this interval, distinct clay mineral assemblages were found and can be described as consisting of Al-rich, Fe-richand intermediate Fe and Al compositions withrespect to 2:1 and 1:1 layers in mixed-layer arrangements. Nearly pure glauconite-nontronite clays exist in the <2 µm fraction only when the bulk rock is free of K- and plagioclase feldspar. Conditions favorable to glauconite-nontronite formation are interpreted to result from a hiatus in volcanoclastic sedimentation, thus providing a stable substrate for glauconitization.
The Fe-bearing mixed-layer clay assemblages consist of glauconite, nontronite and berthierine-like layers in various proportions with several mixed-layer clays often coexisting in the same sample. In different samples, Al-richand Fe-Mg-rich mixed-layer clays are similar in their content and distribution of 1:1 and 2:1 layers. This suggests that the original clay assemblages were similar and later diagenesis affected certain horizons resulting in substitution of Al by Fe + Mg while preserving the original layer structure and arrangement.
Structural formulae for the berthierine-like phase and berthierine-like layers in these mixed-layer clays show their layer cation composition is intermediate between odinite and standard berthierine. The total sum of octahedral cations varies from 5.26 to 5.55 whereas the amount of Fe2+ cations varies from 2.12 to 2.22 per O10(OH)8. A feature of the berthierine-like phase as well as of berthierine-like layers is that they are di-trioctahedral and Fe2+ and Fe3+ are the prevalent cations. Moreover, in these berthierine-like components, the amount of Fe2+ is greater than that of Mg (in contrast to odinite) and Fe3+ cations prevail over Al (in contrast to berthierine). The presence of authigenic ferrous Fe clays and the relationship between glauconite-nontronite and bulk mineralogy has implications for sedimentological processes and geochemical conditions during and shortly after deposition.
DIFFaX Simulations of Polytypism and Disorder in Hydrotalcite
- Grace S. Thomas, Michael Rajamathi, P. Vishnu Kamath
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 693-699
-
- Article
- Export citation
-
DIFFaX simulations show that the 3R1 and 3R2 polytypes of hydrotalcites (HT) have distinctly different X-ray powder diffraction (XRPD) patterns. The HT samples obtained by coprecipitation as well as those subjected to a subsequent hydrothermal treatment exhibit non-uniform broadening of lines in their XRPD patterns as well as anomalous variation in the intensities of the basal reflections. The reflections appearing in the mid-2θ region (30–50°; 3–1.8 Å) are excessively broadened and do not correspond to either of the 3R polytypes. This broadening is shown to be due to a stacking of layers randomly rotated about the c crystallographic axis by n × 120° (n = 1,2). The intensities of the basal reflections vary due to the departure of the layer composition from the nominal value, essentially due to an increase in the intercalated water content.
Effects of Some Water Repellents on the Structure and Water Sorption of Smectite
- Sae Jung Chang, Soo Jin Kim, Kideok Kwon
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 25-30
-
- Article
- Export citation
-
Waterproofing treatment is important for the preservation of smectite-rich rocks because of their shrinking and swelling properties. The effects of water repellents on the smectite structure and the water sorption of untreated and treated smectites were studied using X-ray diffraction. The hydrophobic Wacker BS 290 (around 100% silane/siloxane) does not prevent water from sorbing on the interlayer surface of smectite because the hydrophobic silane/siloxane is not intercalated into the interlayer space, but adsorbed onto the external surface of smectite. However, the hydrophilic Wacker BS 1001 (water-soluble emulsified silane/siloxane) prevents water from sorbing onto the interlayer surface beyond 33.3 wt.% because the hydrophobic part of the intercalated surfactant has a poor affinity for water. These results imply that water repellent containing a surfactant is suitable for smectite-rich rocks, but the expansion of smectite by intercalation of the surfactant is likely to influence volume change in smectite-rich rocks. This study notes that smectite within a rock can be one of the most important factors influencing the effectiveness of waterproof treatment. This study proposes that an intensive preliminary examination should be performed before applying water repellents to rocks, and furthermore, encourages development of new chemical reagents suitable for protecting smectite-rich rocks from moisture.
Progressive Stages in the Formation of Kaolin Minerals of Different Morphologies in the Weathering of Plagioclase
- Dimitris Papoulis, Panayota Tsolis-Katagas, Christos Katagas
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 275-286
-
- Article
- Export citation
-
Halloysite and kaolinite occur in gneissic weathering profiles developed under a temperate climate in the Leucogia area of NE Greece. X-ray diffraction analyses of samples from three profiles of partially weathered gneisses have revealed that kaolinite becomes more abundant than halloysite with increasing intensity of the decomposition processes. To resolve the genetic relationship between halloysite and kaolinite, microtextures were examined by scanning electron microscopy using polished thin-sections and freshly exposed fracture surfaces, and their chemical compositions were determined using an analytical scanning electron microscope fitted with energy dispersive and wavelength dispersive spectrometers. Different morphological forms of halloysite and kaolinite were detected within the same rock mass. In the earliest stage of weathering, spheroidal aggregates consisting of microcrystalline halloysite are formed on the plagioclase surface. With progressive weathering, spheroidal halloysite converts to tubular halloysite. As weathering advances, tubular halloysite converts to platy halloysite, which in turn converts to kaolinite. Halloysite and kaolinite may coexist in the upper parts of the profiles. Electron microprobe analyses of spheroidal, tubular and platy halloysite and kaolinite show that the chemical composition of the various forms indicate a progressive Fe enrichment and Al depletion with advancing kaolinization, from VIFe0.08VIAl3.70 a.p.f.u. in spheroidal halloysite through VIFe0.29VIAl3.50 in platy halloysite to VIFe0.53VIAl3.32 in newly formed kaolinite. Final-stage, book-type kaolinite approaches its ideal chemical composition (VIFe0.04VIAl3.95). The mineralogy and composition of the kaolin minerals and the development of the various morphological forms of halloysite and kaolinite in different parts of the profiles are attributed to the chemistry of the ambient solutions. However, other physical and microenvironmental conditions (e.g. time and space available) also seem to have strong influence on the precipitation processes producing the morphological variations.
Cation exchange capacity measurements on illite using the sodium and cesium isotope dilution technique: Effects of the index cation, electrolyte concentration and competition: Modeling
- Bart Baeyens, Michael H. Bradbury
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 421-431
-
- Article
- Export citation
-
The isotope dilution technique using Na and Cs as index cations was used to determine the cation exchange capacity (CEC) of illite du Puy as a function of background electrolyte composition. The work showed, in accord with previous studies, that the CEC values were in the order Cs-CEC > Na-CEC. Sodium is commonly chosen as the index cation in CEC determinations using the isotope dilution method. The experimentally measured Na-CEC values for Na-illite increased from ∼75 to ∼200 meq kg−1 for NaClO4 concentrations in the range 5.6 × 10−4 to 1.25 × 10−2 M. Cesium CEC determinations showed a much less pronounced trend over a CsNO3 concentration range from 10−3 to 10−2 M. A reference Cs-CEC value of 225 meq kg−1 was chosen. Careful chemical analyses of the supernatant solutions revealed that Ca and Mg at the (sub)μmolar level were present in all the determinations, despite the extensive conditioning procedures used. Competition between (Ca + Mg) and Na for the exchange sites was put forward as an explanation for the variation of Na-CEC values. This hypothesis was confirmed in a series of single (45Ca) and double (45Ca plus 22Na) labeling experiments. Calcium-sodium selectivity coefficients (
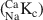 ) were calculated from the experimental data for NaClO4 concentrations from 5.6 × 10−4 to 0.1 M and exhibited a variation from 1.6 to 14.3. A two-site cation exchange model was developed with site capacities and
) were calculated from the experimental data for NaClO4 concentrations from 5.6 × 10−4 to 0.1 M and exhibited a variation from 1.6 to 14.3. A two-site cation exchange model was developed with site capacities and 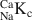 values for each site: planar site capacity =180 meq kg−1,
values for each site: planar site capacity =180 meq kg−1, 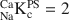 ; type II site capacity = 45 meq kg−1,
; type II site capacity = 45 meq kg−1,  . The model was able to predict the Na and Ca occupancies in the Na-CEC experiments over the whole range of NaClO4 concentrations. It is recommended that Cs should be used instead of Na as the index cation for determining the CEC of illite.
. The model was able to predict the Na and Ca occupancies in the Na-CEC experiments over the whole range of NaClO4 concentrations. It is recommended that Cs should be used instead of Na as the index cation for determining the CEC of illite.
Improved Thermal Stability of Organically Modified Layered Silicates
- Rick D. Davis, Jeffrey W. Gilman, Thomas E. Sutto, John H. Callahan, Paul C. Trulove, Hugh C. De Long
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 171-179
-
- Article
- Export citation
-
Bromide-containing impurities were found to decrease the thermal stability of quaternary alkyl ammonium-modified layered silicates. Improved purification procedures completely removed bromide and led to a 20°C to >100°C increase in organic modified layered silicate thermal stability. Using mass spectrometry and thermal and electrochemical analysis, N,N-dimethyl-N,N-dioctadecyl quaternary ammonium-modified montmorillonite and fluorinated synthetic mica were found to degrade primarily through elimination and nucleophilic attack by these anions. The nature of residual bromides was identified and quantified, and the efficiency of removing these anions was found to be solvent dependent; sequential extraction, first ethanol then tetrahydrofuran, gave the best results. This exhaustive extraction method represents a viable alternative to the use of expensive, more thermally stable oniumion treatments for layered silicates.
An In situ Time-Resolved XRD-PSD Investigation into Na-Montmorillonite Interlayer and Particle Rearrangement during Dehydration
- James Wilson, Javier Cuadros, Gordon Cressey
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 180-191
-
- Article
- Export citation
-
X-ray diffraction with a position-sensitive detector (XRD-PSD) was used to make a time-resolved study of the dynamics of deposition and dehydration of Na-montmorillonite crystallites on flat substrates from deionized water suspensions. The static PSD geometry and simultaneous counting procedure allowed the acquisition of high-resolution data on the dynamics of interlayer and interparticle arrangements during dehydration. Three experimental datasets of Na-smectite dehydration are presented, each one representing different initial sample states (suspension, slurry and re-wetted thin film). The computer program NEWMOD was used to simulate one of the three datasets (dehydration of a smectite suspension) and thus obtain the apparent changes in relative proportions of different 00l values as smectite crystallites formed and dehydrated. Two types of diffracting domains formed: water-dispersed ‘packets’ of 1–2 smectite layers gaining long-range order in the c axis direction as water was lost to evaporation, and smectite layers deposited as hydrated crystallites with variable interlayer water contents. The experimental patterns show the rapid step-wise transition of Na-montmorillonite layers from d values of ∼55 to 18.5, 15.4 and 12.5 Å, with variations that depended upon how the hydrated smectite sample was prepared. The simulations show that there was a wide range of d values whose frequency distribution changed as dehydration proceeded and that transient d values occurred between the peaks observed experimentally. The data obtained in this study illustrate that XRD-PSD instruments have great potential in providing detailed data on the rapid kinetics of interlayer reorganization.
Mass Changes during Adsorption of Metal Bipyridyl Complexes by Clay Films Monitored with an Electrochemical Quartz Crystal Microbalance
- Laisheng Sun, Gilles Villemure
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 31-39
-
- Article
- Export citation
-
The adsorption of [M(bpy)3]2+ ions (M = Ru or Os) by clay films immersed in pure water and in electrolyte solutions was investigated by electrochemical quartz crystal microbalance (EQCM), UV-visible spectroscopy and powder X-ray diffraction. In water, the adsorption of the cations resulted in a decrease in the mass of the films. This decrease in mass is attributed to the expulsion of some 50 water molecules from the clay interlayer spaces for each cation adsorbed. Water is lost to make room for the large metal complex cations in the interlayer spaces, and because of decreases in the volume of the interlayer spaces during adsorption of the cations. In 0.05 M NaCl or 0.05 M Na2SO4, UV-visible measurements show a rapid initial adsorption of the cations by ion exchange, followed by a slower additional adsorption of the cations above the clay’s CEC, presumably as ion pairs with the electrolyte counter ions. The EQCM show initial reductions in the mass of the films that were two to three times larger in pure water. These initial mass decreases were followed by smaller mass ‘re-increases’ at longer times that were not observed in water. The larger initial mass losses are attributed to the loss of more water from the clay interlayer spaces. In 0.5 M Na2SO4 or 1.0 M NaCl, adsorption of the cations never exceeded the clay’s CEC. The initial decreases in mass upon addition of the cations all but disappeared, leaving only the smaller positive mass changes at longer times.