Volume 52 - February 2004
Research Article
Parallel intergrowths in cronstedtite-1T: Implications for structure refinement
- Slavomil Ďurovič, Jiří Hybler, Toshihiro Kogure
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 613-621
-
- Article
- Export citation
-
Cronstedtite is a member of the kaolin-serpentine group. It yields a wealth of more or less disordered polytypes. The crystals of polytype 1T (space group P31m, a = 5.512, c = 7.106 Å) contain, within coherently scattering blocks, variable concentrations of stacking faults so that domains of the basic 3D periodic structure can be shifted by 1/3 (a2 − a1) or 1/3(a1 − a2). These so-called OD parallel intergrowths have been confirmed by high-resolution transmission electron microscopy. The effect manifests itself in the diffraction pattern so that reflections with h-k = 3n — the family reflections — are always sharp, whereas remaining reflections — the characteristic polytype reflections — may be smeared out parallel to c*. The intensities of the latter are thus underestimated during diffractometer measurements. An analysis of such multiple OD intergrowths reveals that the moduli of structure factors for all characteristic (i.e. non-family) polytype reflections are reduced relative to those calculated for the non-intergrown basic structure, by a common factor. This fact usually leads to the appearance of ghost peaks in Fourier maps and to their erroneous interpretation. The structure of the basic model can, however, be refined much better if two scale factors are assigned to the family and non-family reflections, respectively.
Crystal-Chemical Changes in an Oxidative Weathering Front in a Georgia Kaolin Deposit
- Paul A. Schroeder, Robert J. Pruett, Nathan D. Melear
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 211-220
-
- Article
- Export citation
-
The Eocene Huber Formation, exposed in the CFI Hall mine south of Irwinton, in Wilkinson County, Georgia, displays colored zones that are a consequence of an oxidative weathering front. These zones vary from very light gray (gray) to pinkish white (pink) to very pale yellow (cream) (Munsell N8, 5YR 8/2 and 2.5Y 8/2, respectively) with increasing degree of oxidation. A representative sample from each zone was collected and analyzed for its chemical, crystallographic and Raman spectroscopic properties. The comparison of these genetically related samples allowed for a quantitatively accurate assessment of anatase’s contribution to the total TiO2 content. All samples contain ∼1.5 wt.% TiO2. Progressing from gray to pink to cream, anatase contents range from half to nearly all the TiO2. The a lattice parameter for anatase is constant in all three kaolins (3.7916 Å), suggesting a constant 4.6 mol.% isomorphous substitution of Fe for Ti.
Phase characterization and mass-balance considerations of the gray, pink and cream kaolins indicate that ilmenite and pyrite are precursor Ti- and Fe-bearing phases in the gray kaolin that undergo oxidation. Pseudorutile is a proposed intermediate phase resulting from the anodic corrosion of ilmenite. Hematite, goethite and anatase are the dominant end-products via dissolution-precipitation from the reactants pyrite and pseudorutile. The removal of Fe sulfides and organic matter and addition of hematite and goethite causes kaolin colors to change from gray to pink. Oxygen diffusing from groundwater in permeable overlying and underlying formations facilitates a process that probably involves aerobic and anaerobic bacteria that utilize Fe from pyrite, hematite and goethite. We postulate that the end result is the mobilization of Fe by siderophores and a kaolin color change from pink to cream.
Very low-temperature alteration of sideromelane in hyaloclastites and hyalotuffs from Kilauea and Mauna Kea volcanoes: Implications for the mechanism of palagonite formation
- Ahmed Drief, Peter Schiffman
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 622-634
-
- Article
- Export citation
-
Three petrographically distinct styles of altered glasses in two hyaloclastites and one hyalotuff were studied. The texture and chemistry of these samples were investigated using electron probe microanalysis, scanning electron microscopy and transmission electron microscopy in order to understand better the mechanism by which alteration of sideromelane and formation of palagonite occurred in these samples. The results show that clay minerals (primarily smectites) are present in three different microenvironments: (1) coating the surfaces of glass and crystals or vesicle walls; (2) as a relatively heterogeneous, but well crystallized, replacement product (i.e. reddened smectite grain replacement or RSGR) of glass or; (3) as a relatively homogeneous, amorphous to poorly crystalline replacement product (i.e. palagonite). Both the grain size and composition of these smectite-like materials vary considerably.
Crystalline smectites occur in both hyaloclastites and have an intermediate composition between the two end-members nontronite and saponite. This composition could correspond to a mechanical intergrowth and/or an interstratification of two different smectites: one dioctahedral (i.e. nontronite) and one trioctahedral (i.e. saponite or stevensite) or simply to a true di-trioctahedral smectite. The coating smectite appears to have precipitated by a paragenetically-early, dissolution-precipitation mechanism prior to the formation of the RSGR. The high Ti content found in RSGR is attributable to an amorphous Ti-rich material which is intergrown with smectite and which behaves as a sink for immobile elements and those not included in smectite.
Palagonite from both hyaloclastites and hyalotuff is poorly to non-crystalline and more aluminous than the coating smectites. Palagonite from the hyalotuff has an Fe-rich montmorillonite-like composition. The TEM images show a 30–50 nm thick leached layer formed by selective (non-stoichiometric) dissolution that takes place in the fracture domain. The hydration and replacement of glass during the palgonitization process is accompanied by the loss of Fe, Mg and Ca with a concomitant gain of Al. Both palagonites (from hyaloclastites and hyalotuff) show smilar textural and chemical characteristics.
Illite-Smectite Mixed-Layer Minerals in Felsic Volcaniclastic Rocks from Drill Cores, Kakkonda, Japan
- Atsuyuki Inoue, Alain Meunier, Daniel Beaufort
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 66-84
-
- Article
- Export citation
-
Crystallization processes of the illite-smectite (I-S) mixed-layer mineral series during alteration of felsic vitric materials in volcaniclastic sediments through two drill holes (IT-2 and IT-8) near the Kakkonda active geothermal system, Japan, were examined by optical microscopy, scanning and transmission electron microscopy (SEM and TEM), electron microprobe analysis, X-ray diffraction (XRD), and oxygen isotope analysis. Temperatures measured through the drill holes increased nearly linearly with depth up to 317°C at the bottom (1700 m) of IT-8. Homogenization temperature measurements of fluid inclusions indicated that the alteration occurred at temperatures similar to the present temperatures. In selected volcaniclastic rocks, excluding andesitic rocks and black shales, clay minerals occurred as glass replacements and pore fillings as seen under SEM and optical microscopy, and exhibited predominantly euhedral hexagonal and elongated forms under TEM, implying that they precipitated in situ through hydrolytic reactions of glass and fluid. Based on XRD examination, I-S minerals showed a sigmoidal variation in illite layer percentage (%I) in the range of ∼150 to 220°C and R0 I-S minerals with intermediate %I between 20 and 40% rarely occurred (where R is the Reichweite parameter). The chemical composition also showed a specific variation with depth. Intermediate clays including smectite and I-S minerals are enriched in Al compared to those reported previously from hydrothermal alteration of almost equivalent parent rocks. The oxygen isotope data indicated that the reacting solution was percolating groundwater in the shallow levels and with fossil seawater in the deeper levels. Furthermore, calculating the fluid/rock (W/R) ratio from the isotope variations revealed that the alteration occurred at a nearly constant W/R ratio condition irrespective of %I. Consequently, the observed specific variations in structure and chemical composition of I-S minerals reflect the compositional variations of fluid participating in the crystallization at given temperatures under the conditions of a given original rock and constant W/R ratio. High pH and Na-rich solutions generated by progressive hydrolytic reactions between felsic glass and groundwater favored the precipitation of Al-rich smectite up to ∼150°C and was followed by precipitation of an aluminous R1 I-S mineral with few intermediate R0 I-S minerals at higher temperatures. The crystallization obeys Ostwald’s step rule behavior of smectite illitization processes under a high geothermal gradient.
Formation and Properties of a Continuous Crystallinity Series of Synthetic Ferrihydrites (2- to 6-line) and their Relation to FeOOH Forms
- Udo Schwertmann, Josef Friedl, Andreas Kyek
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 221-226
-
- Article
- Export citation
-
Ferrihydrite is a poorly crystalline Fe oxide of which 2-(XRD)line and 6-line varieties are commonly used in experiments, although species with intermediate numbers of peaks have been found in nature. To simulate nature, we synthesized two continuous series of ferrihydrites with between 2 and 6–7 peaks at room temperature in two different ways: (1) by varying the rate of hydrolysis of an Fe(NO3)3 solution (HR series); and (2) by oxidizing an FeCl2 solution containing up to 73 mmol Si/L (Si series), both at pH 7. Mössbauer spectra at 4.2 K showed that the ferrihydrites of the HR series had a constant magnetic hyperfine field (Bhf) at 4.2 K of 48.8 T whereas Bhf in the Si series dropped from 49.4 to 46.7 T as the Si content of the ferrihydrites increased from 0 to 74.7 g/kg of Si. Temperature scans between 4 and 170 K illustrate that the magnetic order breaks down at a temperature which is lower the higher the hydrolysis rate and the Si concentration in the ferrihydrite.
Mixed-layer kaolinite-smectite minerals in a red-black soil sequence from basalt in Sardinia (Italy)
- Simona Vingiani, Dominique Righi, Sabine Petit, Fabio Terribile
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 473-483
-
- Article
- Export citation
-
Clay minerals from soils of a red-black soil complex developed from basaltic parent material in Sardinia are formed along a short toposequence (200 m). At the foot of the sequence, a clay-rich, black Vertisol forms, whereas at the summit, the soil is a dark reddish-brown Inceptisol. X-ray diffraction, infrared spectroscopy (FTIR), cation exchange capacity (CEC) and permanent and variable charges analyses were used, and the data show that clay minerals varied according to soil horizon and topographic position of the soil. Clay minerals in the Inceptisol are dominated by kaolinite and mixed-layer kaolinitesmectite (K-S, K:S >0.5), whereas the Vertisol contains smectites and K-S with K:S proportions <0.5. In the Vertisol, the proportion of kaolinitic layers in the K-S increases from the C horizon (K:S ∼0.35–0.40) to the Ap horizon (K:S ∼0.40–0.45). This soil clay-mineral distribution, in relation to topography, is similar to that reported for other (kaolinitic) red-black (smectitic) soil associations in subtropical and tropical areas. The sequence forms by downward drainage on summits and slopes, and buildup of ions in ‘lows’ produces smectites. Fourier transform infrared spectra indicate that two types of smectite are formed in the C horizon of the Vertisol; one is more ferric (Fe-beidellite, nontronite), the other more aluminous. Mineralogical evolution in the soil profile (from C to Ap horizon) shows a decreasing proportion of ferric smectite layers (compared to the more aluminous smectite layers). This would indicate that ferric smectite layers are preferentially transformed (or dissolved) to give kaolinite layers, with Fe precipitating as oxides and/or oxy-hydroxides or retained partly in kaolinite layers. Because the surface properties of clay minerals are related to mineralogy, the CEC (33–41 cmol kg−1) in the brown Inceptisol is ∼50% pH-dependent charge while in the Vertisol up to ∼75% of the CEC (48–61 cmol kg−1) comes from accessible permanent charges.
Effects of Pyrogallol on Al13 Tridecamer Formation And Humification
- G. S. R. Krishnamurti, M. K. Wang, P. M. Huang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 734-741
-
- Article
- Export citation
-
The Al13 polycation is the predominant hydroxy-Al species in partially neutralized solutions. However, the existence of the Al13 species and the factors governing its formation in terrestrial environments still remain obscure. The objective of this study was to investigate the influence of pyrogallol, a common polyphenol in soils, on the formation of Al13 tridecamer. Hydroxy-Al solutions with an OH/Al molar ratio of 2.2 (pH 4.53) at pyrogallol/Al molar ratios of 0, 0.01, 0.05, 0.1 and 0.5 were prepared and mixed with 0.5 M sodium sulfate to form aluminum sulfate precipitation products. The solid-state 27Al nuclear magnetic resonance (NMR) spectra of the precipitates show that the pyrogallol perturbed the formation of Al13 tridecamer species as indicated by the decrease in the intensity of resonance peak, observed at 62.5 ppm, with increase in the pyrogallol/Al molar ratio. The crystallization of the precipitated Al sulfates was also hampered by pyrogallol, resulting in the formation of X-ray non-crystalline products at a pyrogallol/Al molar ratio of 0.50. The absorbance at 465 and 650 nm of the hydroxy-Al-pyrogallol solutions, the C coprecipitated, the electron spin resonance and 13C CPMAS-NMR spectra of the precipitates indicate the concomitant enhanced abiotic humification of pyrogallol by the X-ray amorphous Al species.
Adsorption of Poly(Vinyl Alcohol) on Montmorillonite
- Silvia G. de Bussetti, Eladio A. Ferreiro
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 334-340
-
- Article
- Export citation
-
The adsorption of poly(vinyl alcohol) (PVA) on montmorillonites saturated with calcium (Ca-Mt) and sodium (Na-Mt) as a function of the pH value and PVA concentration in aqueous solution was studied. Owing to the binding effect of the bivalent cation, the adsorption of PVA on Ca-Mt decreases as the pH of the suspension increases, whereas adsorption on Na-Mt is unaffected by the suspension pH. The adsorption maximum of PVA (pH 6) on Ca-Mt was 151.2 mg of PVA g−1 of clay, which is considerably lower than that on Na-Mt (496.2 mg g−1). These adsorption data coincide with the basal spacings obtained for the clays: 1.72 and 2.26 nm for Ca-Mt and Na-Mt, respectively. Sodium permits a greater separation between the clay laminae than calcium, but in both clays the presence of the polymer gives rise to a material in which PVA is intercalated between the laminae and is also adsorbed on the external surface. Adsorption is a slow process and is irreversible in both clays.
Correction
The Kübler Index in Late Diagenetic to Low-Grade Metamorphic Pelites: A Critical Comparison of Data From 10 Å and 5 Å Peaks
- Stefano Battaglia, Leonardo Leoni, Franco Sartori
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 85-105
-
- Article
- Export citation
-
A set of 99 samples covering the whole range of low- and very low-grade metamorphic conditions has been used to compare Kübler index (KI) values measured on the illite 10 Å reflection (KI10 Å) with those obtained from the 5 Å reflection (KI5 Å). Evaluation of peak widths have been carried out both graphically on the recorded peak profiles from chart-strip X-ray diffraction (XRD) patterns and with the WINFIT computer program (Krumm, 1996) on fitted and decomposed profiles. All the measurements were performed both on air-dried and glycolated preparations. The data collected show that in the rock samples where illite is associated with significant amounts of I-S interstratified minerals, and/or K/Na intermediate micas, paragonite and other interfering phases, full width at half-maximum (FWHM) measurements on the 5 Å peak in fitted and decomposed XRD profiles from glycolated mounts give more reliable KI values than those obtained from the 10 Å peak. This is because of the easier and more complete de-summation of the illite second reflection from the contributions of all the interfering phases. In each sample population in which the rocks have the same metamorphic grade, KI5 Å values from fitted and decomposed profiles show much lower scattering in comparison with KI10 Å values and appear consistent with the values measured in lithologies without interfering phases.
The relationship between KI10 Å and KI5 Å appears very close to a 1:1 linear relationship; nevertheless, the conversion from KI5 Å to KI10 Å through the appropriate equation (KI10 Å = KI5 Å × 0.965 + 0.023°2θ) is recommended in order to avoid a small, but systematic, error.
In the range from late diagenesis to middle anchizone, where several phases may give diffraction effects interfering with illite peaks, the proposed procedure (i.e. FWHM measurements on the 5 Å peak in fitted and decomposed XRD profiles from glycolated mounts) seems to allow better estimates of the metamorphic grade than those obtained through traditional KI measurements.
Research Article
Dehydration and rehydration of palygorskite and the influence of water on the nanopores
- Wenxing Kuang, Glenn A. Facey, Christian Detellier
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 635-642
-
- Article
- Export citation
-
The dehydration and rehydration processes of the clay mineral palygorskite (PFl-1) were studied by textural analysis, thermogravimetric analysis connected with mass spectrometry (TGA-MS), and 29Si and 1H solid-state NMR techniques. The TGA-MS results clearly reveal weight losses at maxima of 70°C, 190°C, 430°C and 860°C. PFl-1 is characterized by a micropore area of 93 m2/g, corresponding to a micropore volume of 47 mm3/g. These values are also obtained for the sample heated up to 200°C for 20 h. Further heating at 300°C produces a collapse of the structure, as shown by the almost complete loss of microporosity.
The 29Si NMR spectra of palygorskite show two main resonances at −92.0 and −97.5 ppm, attributed to one of the two pairs of equivalent Si nuclei in the basal plane. A minor resonance at −84.3 ppm is attributed to Q2(Si-OH) Si nuclei. The resonance at −92.0 ppm is assigned to the central Si position, while the resonance at −97.5 ppm is assigned to the edge Si sites. It is confirmed bysolid-state 29Si and 1H NMR that nearly complete rehydration is achieved by exposing palygorskite samples that have been partially dehydrated at 150°C and 300°C, to D2O or water vapor at room temperature. When the rehydration is accomplished with D2O, the atoms are disordered across all the protons sites.
Characteristics of a Mg-palygorskite in Miocene rocks, Madrid Basin (Spain)
- E. García-Romero, M. Suárez Barrios, M. A. Bustillo Revuelta
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 484-494
-
- Article
- Export citation
-
Palygorskite in Miocene mudstones, palustrine limestones and calcretes from the Esquivias locality (Madrid Basin, Spain) has been analyzed by X-ray diffraction, infrared spectroscopy, scanning electron microscopy, transmission electron microscopy and analytical electron microscopy to determine its characteristics and chemical composition. Other palygorskites from the literature are used as references. The mean structural formula obtained from the analysis of isolated particles is (Si7.87Al0.13)O20(
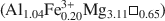 )(OH)2(OH2)4(Ca0.02K0.05Na0.08). This palygorskite has the largest Mg content reported in the literature, and it seems that, chemically, it fills the ‘compositional gap’ existing between sepiolite and palygorskite. Infrared spectroscopy reveals the absence of trioctahedral Mg and therefore the possibility of the existence of magnesic clusters in the ribbons is discounted. An homogeneous distribution of the octahedral cations (Al, Fe3+ and Mg) along the ribbons is proposed.
)(OH)2(OH2)4(Ca0.02K0.05Na0.08). This palygorskite has the largest Mg content reported in the literature, and it seems that, chemically, it fills the ‘compositional gap’ existing between sepiolite and palygorskite. Infrared spectroscopy reveals the absence of trioctahedral Mg and therefore the possibility of the existence of magnesic clusters in the ribbons is discounted. An homogeneous distribution of the octahedral cations (Al, Fe3+ and Mg) along the ribbons is proposed.
Adsorption and Photo-reactivity of Bensulfuron-Methyl On Homoionic Clays
- Youbin Si, Shenqiang Wang, Dongmei Zhou, Huaiman Chen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 742-748
-
- Article
- Export citation
-
The adsorption and photolysis of the herbicide bensulfuron-methyl [2-(4, 6-dimethoxypyrimidm-2-carbamoylsulfamoyl)-o-toluic acid methyl ester] on homoionic Na+-, Ca2+- and Fe3+-montmorillonite and kaolinite clays were studied. The Freundlich adsorption coefficient, Kf, measured from isotherms on clays followed the order Na+ < Ca2+ < Fe3+. Montmorillonite showed a greater adsorptive capacity than kaolinite. Analysis of Fourier transform infrared spectra of bensulfuron-methyl adsorbed on clay suggested probable bonding interactions between bensulfuron-methyl and homoionic clays. The photolysis rate of herbicide adsorbed on homoionic clay surfaces was quite slow to its free state and decreased in the order Na+ > Ca2+ > Fe3+, indicating that adsorption may have prevented photolysis.
Sorption of Isoxaflutole and DKN on Organoclays
- Maria Jose Carrizosa, Pamela J. Rice, William C. Koskinen, Ignacio Carrizosa, Maria Carmen del Hermosin
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 341-349
-
- Article
- Export citation
-
Sorption of the herbicide isoxaflutole and its main degradate, diketonitrile (DKN), to natural clays, SAz-1, SWy-2 and SHCa-1, and the organoclay derivatives (octadecylammonium (ODA) and hexadecyltrimethylammonium (HDTMA)) of these clays was investigated. Isoxaflutole hydrolysis to DKN was too rapid in aqueous solutions with organoclays to characterize sorption. No measurable DKN sorption was observed for the natural clays. Sorption of DKN was greater on organoclays with an interlayer paraffin-like complex that were prepared from the high-charge SAz-1 clay than on organoclays with a bilayer or monolayer interlayer complex prepared using lower-charge SWy-2 or SHCa-1 clays. Desorption isotherms indicated that sorption was irreversible. For SAz-1 with HDTMA at ∼100% of the clay CEC, the d001 values suggest that DKN enters the interlamellar space of the organoclay and dissociates into the anion. The DKN anion forms a very stable chelate complex with the residual cations and/or partially-coordinated structural cations. This strong interaction supports the irreversibility of the sorptive process.
Ultramafic-Rock-Hosted Vein Sepiolite Occurrences in the Ankara Ophiolitic Mélange, Central Anatolia, Turkey
- Hüseyin Yalçin, Ömer Bozkaya
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 227-239
-
- Article
- Export citation
-
A 2 m thick brecciated zone containing magnesian minerals is present at the contact of tectonites and cumulates. Tectonites below this zone comprise serpentinized orthopyroxenite and serpentinite. An alteration zone with vein-type bedding comprises four different levels; from bottom to top they are: (1) green-brown serpentinite with dolomite (0.9 m), (2) light greenish-white dolomite with serpentine (0.5 m), (3) white dolomite with sepiolite (0.4 m), and (4) greenish-white dolomite with smectite-chlorite (0.2 m). The first level has a mineral association of serpentine + dolomite ± calcite ± aragonite, the second level consists of dolomite + serpentine ± calcite or dolomite + magnesite + serpentine, the third level comprises dolomite + sepiolite, and the fourth level is made up of dolomite + chlorite + smectite + serpentine. Dolomite, the main mineral of the alteration zone, occurs as coarse crystals (microsparitic-sparitic) in fractures and as small crystals (microsparitic-micritic) in the matrix, which includes serpentine and gabbro relics. Sepiolite developed at the edges and surfaces of dolomite and as fibrous forms in voids. Cumulate rocks above this zone comprise uralitic gabbros. The occurrences of magnesian minerals developed in three stages: the first stage was the serpentinization of olivine; the second stage was the dissolution of serpentine by groundwater and/or meteoric water containing carbon dioxide; and the last stage was the synthesis of neoformed minerals.
Dickite and Other Kaolin Polymorphs from an Al-rich Clay Deposit Formed in Volcanic Tuff, Southeastern Korea
- Chang Oh Choo, Soo Jin Kim
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 749-759
-
- Article
- Export citation
-
Kaolins from an Al-rich clay deposit formed in volcanic tuff, southeastern Korea contain predominant dickite and quartz with accessory diaspore, pyrophyllite, alunite, illite, sulfides and calcite. Intergrowth of blocky crystals with thin ones within stacks suggests that blocky crystals gradually replaced thin ones within the same stacks. Numerous dickite plates with pseudo-hexagonal outline are placed in rows eventually forming a U-shaped stack and a vermiform stack. Individual crystals within the stack are of pseudo-hexagonal form and vermiform, accompanying slips across basal planes. The euhedral dickite with blocky habit has basal planes (001) and edge planes (110) perpendicular to each other, and it shows pseudo-hexagonal crystallites elongated in one direction. Fourier transform infrared spectra at OH-stretching and far-infrared regions show that the dickitic mass contains a small proportion of kaolinite or nacrite, indicating interstratification or at least mixtures of kaolin polytypes. The formation of mixtures of various polytypes that are of kaolinite-dickite, dickite and mixtures of dickite-nacrite could be attributed to hydrothermal processes ranging over a broad spectrum of temperature and fluid chemistry.
Conformation of Surfactant Molecules in the Interlayer of Montmorillonite Studied by 13C MAS NMR
- Hongping He, Ray L. Frost, Feng Deng, Jianxi Zhu, Xiaoyan Wen, Peng Yuan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 350-356
-
- Article
- Export citation
-
The ordering conformation of surfactant molecules in intercalated montmorillonite prepared at various concentrations was investigated by 13C MAS NMR. The 13C MAS NMR study demonstrates the coexistence of ordered and disordered chain conformations. Two main resonance peaks are associated with the backbone alkyl chains: the resonance at 33 ppm corresponds to the ordered conformation (all-trans), and the resonance at 30 ppm corresponds to the disordered conformation (mixture of trans and gauche). Deconvolution of 13C MAS NMR spectra indicates that the ordering conformation of surfactant molecules within the gallery of montmorillonite depends very much on their orientation and packing density. When amine chains are oriented parallel to the silicate layers, the amount of all-trans conformer decreases with the increase of amine concentration. However, the amount of all-trans conformer increases with the increase of amine concentration when amine chains radiate from the silicate layers. Furthermore, 13C MAS NMR spectra show that the intercalated surfactant molecules in the clay minerals never attained the complete liquidlike or solidlike behavior.
Mineralogical and chemical characterization of sepiolite occurrences at Karapinar (Konya Basin, Turkey)
- N. Karakaya, M. Çelik Karakaya, A. Temel, Ş. Küpeli, C. Tunoğlu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 495-509
-
- Article
- Export citation
-
The Konya region in central Anatolia is covered by Pliocene-Late Pleistocene sediments and volcanites related to the sediments NNW of Karapınar, Turkey. In the area, the Upper Miocene-Quaternary Üzecek Dağı and Karacadağ volcanites are generally of the same age and formed from magmas of similar composition. The Karapınar formation is brown to whitish-beige, partly fossiliferous and consists of limestone, marl, claystone and, locally, sandy layers. Silica-rich lenses, nodules and layers are observed in the upper strata which locally contain sepiolite-rich layers. The mineralogical composition of sepiolite samples taken from the area was determined by powder X-ray diffractometry, while the abundance of major-element oxides was measured by X-ray fluorescence spectrometry. The crystallographic and morphological properties of samples were determined by means of scanning electron microscopy and energy dispersive spectroscopy. Samples were taken from three sections and from random locations. Mineral assemblages in the same stratigraphic position are generally similar in the three sections, while the thickness of the individual beds varies between the sections. Dolomite and calcite are the main carbonate minerals in the sections. Sepiolite occurs primarily with dolomite and, locally, dolomite and calcite, and less commonly with just calcite. Generally, quartz, feldspar and mica are found, especially in the upper parts of the sections where tuff is abundant. CaO and MgO dominate the major-element oxides. The CaO content is between 1 and 30% while MgO is 3–21%. Al2O3 and SiO2 are generally higher in the sepiolitic and tuffitic layers. Al2O3 is <3% and SiO2 is between 15–18% in the sepiolitic layers. The average structural formula of sepiolite was calculated as: (Mg7.00Al0.44Fe0.18)(Si11.71Al0.29)O30 (OH)4(OH2)4Ca0.13K0.09Na0.01. Sepiolite occurs as fibers and dolomite as subhedral or euhedral crystals. It is considered that sepiolite was formed either by conversion of dolomite or by direct precipitation from solution under alkaline and saline conditions in the Karapınar paleolake. The paleolake was saturated with respect to Mg, Ca and Si derived from groundwater that percolated along fracture systems.
Synthesis of Birnessite from the Oxidation of Mn2+ by O2 in Alkali Medium: Effects of Synthesis Conditions
- Xiong Han Feng, Fan Liu, Wen Feng Tan, Xiang Wen Liu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 240-250
-
- Article
- Export citation
-
An O2 oxidation and freeze-dry procedure has been used to synthesize birnessite through the oxidation of Mn2+ in alkali media. The effects of O2 flow rate, the fluxion velocity of reaction suspension, the reaction temperature, pretreatment the reaction solutions with N2, and the hydration conditions on the purity of birnessite, the crystallinity, the ion-exchange properties, and the phase transformation of intermediate phases were examined. Buserite with a 1 nm basal spacing, produced after the oxidation, is transformed to 0.7 nm Na birnessite by complete freeze drying. Increasing the fluxion velocity of the reaction suspension and the O2 flow rate facilitated oxidation of Mn(OH)2. Prephase I (a phase related to hausmannite, γ-Mn3O4 (Luo and Suib, 1997; Luo et al., 1998), and feitknechtite (β-MnOOH) were formed as intermediates during the synthesis. Mechanical stirring was used to change the fluxion velocity of the reactive suspension. When the speed of stirring and the O2 flow rate were raised to 250 rpm and 3.0 L/min, respectively, or 450 rpm and 2.0 L/min, respectively, birnessite was the only phase in the final product. Irrespective of temperature in other reactions, pure birnessite was synthesized as long as the temperature during the initial mixing of the reaction solutions was maintained below 10°C. Increasing the reaction temperature led to a larger crystal size, better crystallinity and lower surface area. The pretreatment of solutions with N2 or O2 had little effect on the synthesis; synthesized birnessites had the same purity (100%) as, and similar crystallinity to, that of the no-pretreatment control. Dehydration of the buserite by freeze drying and heating at 60°C did not affect the production of birnessite; however, the latter caused partial loss of ion-exchange capacity with Mg2+. The pathways of the birnessite formation in this study might be:
(1) Mn(OH)2 (amorphous) → feitknechtite → buserite → birnessite, and
(2) Mn(OH)2 (amorphous) → prephase I → feitknechtite → buserite → birnessite
Mn(OH)2 existed in an X-ray amorphous state, not in the form of ‘pyrochroite’, during the synthesis.
The adopted conditions for synthesis of pure birnessite were NaOH to Mn molar ratio of 13.7, O2 flow rate of 2 L/min and oxidation for 5 h during vigorous stirring at 450 rpm at room temperature. The birnessite synthesized had a hexagonal platy morphology with good crystallinity, an average composition of Na0.25MnO2.07.0.66H2O, and a surface area of 38 m2/g.
Formation of Nontronite from Oxidative Dissolution of Pyrite Disseminated in Precambrian Felsic Metavolcanics of the Southern Iberian Massif (Spain)
- J. C. Fernández-Caliani, E. Crespo, M. Rodas, J. F. Barrenechea, F. J. Luque
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 106-114
-
- Article
- Export citation
-
This paper describes a rare occurrence of nontronite associated with sulfide-bearing felsic metavolcanics, providing evidence of colloidal deposition in open spaces as result of a low-temperature water-rock interaction. Microbotryoidal masses of green nontronite with impurities of kaolinite, illite, barite, amorphous silica and iron oxyhydroxides are found as vein and cavity fillings in deeply kaolinized rhyolites and rhyolitic tuffs of Precambrian age, at Oliva de Merida in SW Spain. Clay mineral characterization has been carried out by X-ray diffraction, infrared spectroscopy, thermal analysis, analytical electron microscopy and stable isotope (oxygen and hydrogen) analysis. Nontronite was formed under low-temperature alteration conditions, from a continuous sequence of reactions and aqueous solution compositions, involving two basic processes that acted in concert: oxidative dissolution of pyrite and hydrolysis of K-feldspar. After acidity neutralization, dissolved silica released by incongruent dissolution of K-feldspar reacted with ferric sulfate derived from pyrite oxidation to form nontronite under oxidizing conditions, in the presence of relatively warm meteoric water.
Interactions of two sulfonylurea herbicides with organoclays
- Maria Jose Carrizosa, Maria del Carmen Hermosin, William C. Koskinen, Juan Cornejo
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 643-649
-
- Article
- Export citation
-
The sorption of two sulfonylurea herbicides (SU), metsulfuron methyl and nicosulfuron, on pure clays and organoclays was investigated. Three clays (Arizona smectite, SAz-1, Wyoming smectite, SWy-2, and hectorite, SHCa-1), were treated with amounts of octadecylammonium (ODA) or dioctadecyldimethylammonium (DODMA) cations equal to ∼50 and 100% of the clays’ cation exchange capacity (CEC). Sorption isotherms were fitted to the Freundlich equation. While no measurable sorption was found on the pure clays (Kf = 0), organoclays prepared using both primary and quaternary amines were effective as SU sorbents. The metsulfuron methyl Kf values ranged between 196 and 1498 µmol1−1/n kg−1 L1/n, and Kf values for nicosulfuron, which were lower than those of metsulfuron methyl, ranged from 35 to 198 umol1−1/n kg−1 L1/n. As shown by sorption coefficients, Kd and KOC, SWy-2 treated with DODMA at ∼100% of the CEC was the most effective sorbent for metsulfuron, Kd = 684 L kg−1 and KOC = 2138 L kg−1. For nicosulfuron the most effective sorbent was SAz-1 with ODA at ∼ 50% of the CEC (Kd = 147 L kg−1 and KOC = 1233 L kg−1). In contrast to other weak-acid herbicides, such as phenoxy and picolinic acids, no clear relationships were found between sorption and layer charge, organic carbon content, and basal spacing of the organoclays for both sulfonylurea herbicides. Sorption of both herbicides on organoclays was assumed to involve hydrophobic and polar interactions for which the availability of interlayer room between organocations was a very important factor.