The omega-3 index was defined as the percentage of EPA and DHA in a total of twenty-six specific fatty acids in erythrocytes in 2004(Reference Harris and von Schacky1). An integral part of the definition was the highly standardised analytical procedure(Reference Harris and von Schacky1,Reference Harris, von Schacky, Park and McNamara2) . As discussed in more detail elsewhere, every step of the analytical method impacts substantially on the result(Reference Harris, von Schacky, Park and McNamara2). Moreover, parameters such as time and vigour of shaking the sample during lipid extraction, or time and agent used for transmethylation impacted on results(Reference Harris, von Schacky, Park and McNamara2). Nevertheless, it was possible to standardise the analytical method in three geographically distinct laboratories, with regular proficiency testing demonstrating the fruitfulness of the efforts(Reference Harris, von Schacky, Park and McNamara2).
Many laboratories around the world have their own methods to analyse erythrocyte fatty acids, and some are using these methods to produce results they call omega-3 index. However, quantitatively, the results differ from the results obtained with the standardised method, i.e. are either higher or lower(Reference Harris, von Schacky, Park and McNamara2). This is problematic if the target range of 8–11 % of the standardised method is used as a reference. This can lead to over supplementation (overshooting the target range, potential risk of bleeding) or under supplementation (not reaching the target value, increased risk for total mortality and other untoward clinical events)(Reference Harris, Tintle and Etherton3–Reference Bhatt, Steg and Miller7). Either way, non-standardised measurements create serious ethical issues; standardising the omega-3 index methodology is needed. Currently, the standardised analytical method can be identified by the trademark HS-Omega-3 Index®. As with all other laboratory parameters, standardisation of the omega-3 index is one of the prerequisites for this parameter entering clinical medicine.
The present review of scientific publications highlights developments in the past two years.
Omega-3 index and human life
Using the standardised method to analyse erythrocytes, and more than 20 000 samples from Omegametrix clinical routine determinations, no sample was found with an omega-3 index <2 % (Fig. 1). The same was true for tens of thousands of samples from the Omegametrix two sister laboratories called Omegaquant in Sioux Falls, SD, USA and Seoul, South Korea (data not shown). Thus, human life without a defined minimum of EPA and DHA in erythrocytes remains to be found, i.e. minimum levels of EPA and DHA seem to be needed for human life. This puts the discussion, as to whether EPA and DHA can be considered drugs, to rest. Life without drugs is possible, whereas life without EPA and DHA is not.
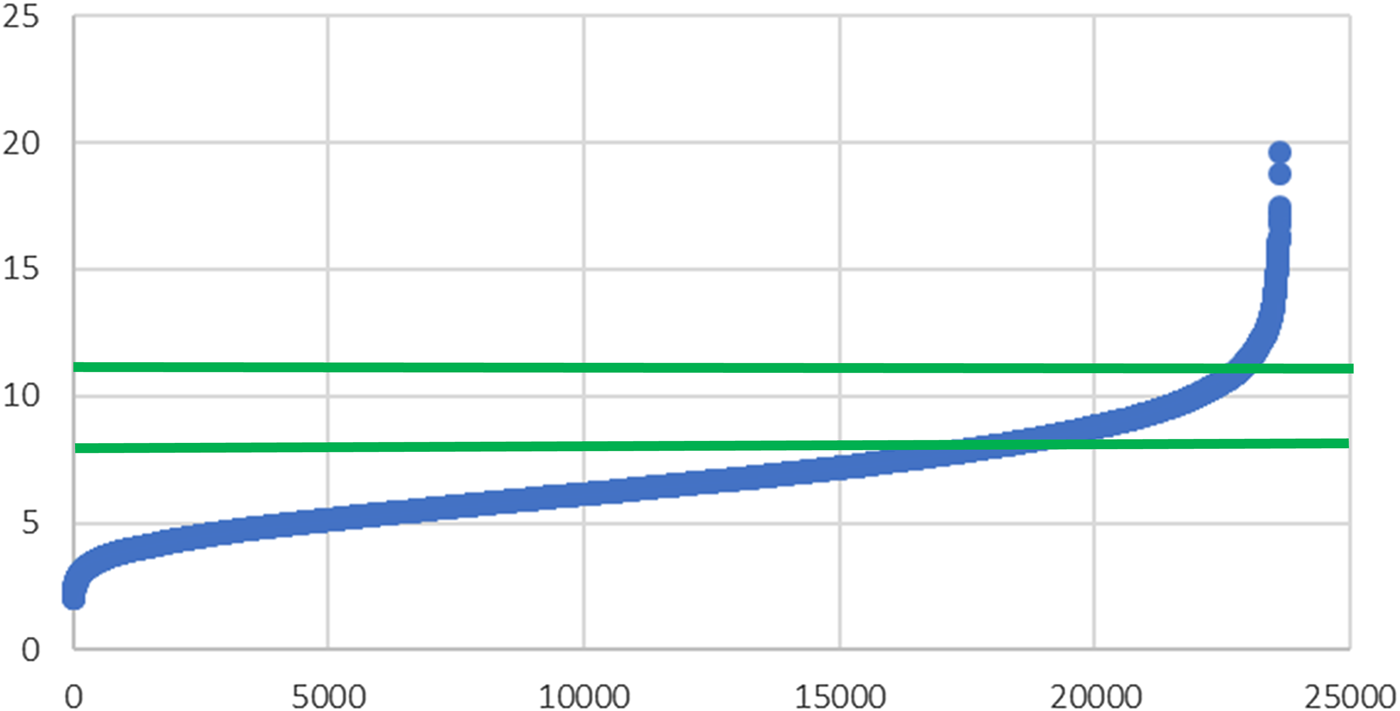
Fig. 1. Omega-3 index (y-axis) in 23 615 erythrocyte samples from Europe (x-axis), as determined in the clinical routine of Omegametrix(Reference von Schacky66).
How the human body maintains these minimum levels is unclear. Individuals, such as vegans or vegetarians, ingest minimal amounts EPA and DHA (e.g. in one study the mean was 1⋅5 (sd 3⋅5) mg/d EPA and 2⋅8 (sd 10⋅1) mg DHA/d according to dietary recall), yet in these individuals, the mean omega-3 index found was 3⋅7 (sd 1⋅0) %(Reference Sarter, Kelsey and Schwartz8). In vegans or vegetarians, the omega-3 index did not correlate with the intake of α-linolenic acid(Reference Sarter, Kelsey and Schwartz8), which was also true for omnivores in a country, such as Canada, with high intake of α-linolenic containing flaxseed(Reference Langlois and Ratnayake9). As a general rule, no level is solely controlled by the influx, i.e. amounts going into the pool, but also by other factors, such as distribution volume (pool size), and efflux, i.e. amounts leaving the pool. The pool size for EPA and DHA in human subjects has not been determined, and what factors regulate the catabolism of EPA and DHA have not yet been well defined in quantitative terms. It is not yet clear whether small amounts of α-linolenic acid are metabolised to EPA or DHA, or whether catabolism of EPA and DHA is shut off to maintain a minimum omega-3 index(Reference Plourde and Cunnane10,Reference Metherel and Bazinet11) . Correspondingly, EPA and DHA might be essential because life without a minimum omega-3 index is not possible. Alternatively, this may not be the case if a minimum omega-3 index arises from α-linoleic acid metabolism to EPA and/or DHA. The question of essentiality cannot be resolved by simply analysing the levels of fatty acids, but only by detailed metabolic studies in appropriate populations.
Issues in trial design
Frequently, clinical research organisations are hired to conduct a multi-centre trial. By default, clinical research organisations ask trial participants to ingest their trial medication in the morning, which was also the case in many, but not all large trials with clinical endpoints in the omega-3 field(Reference von Schacky12,Reference Rice, Bernasconi and Maki13) . Trial medication in such trials frequently was one capsule containing 1 g fat containing 860 mg EPA and DHA(Reference von Schacky12,Reference Rice, Bernasconi and Maki13) . In many countries, breakfast is a low-fat meal, if eaten at all. Bioavailability of EPA and DHA is minimal with a low-fat meal, since fat absorption requires a number of steps that are activated by a high-fat, but not by a low-fat meal(Reference Davidson, Johnson and Rooney14,Reference Schuchardt and Hahn15) . In other words, the bioavailability of EPA and DHA was minimised by asking participants to ingest EPA and DHA in the morning.
Moreover, in all trials so far, participants were recruited irrespective of their baseline omega-3 index(Reference von Schacky12,Reference Rice, Bernasconi and Maki13) . Patients with congestive heart failure with reduced ejection fraction or major depression are characterised by a low omega-3 index, but in many other health issues, such as coronary artery disease, this is not necessarily the case(Reference von Schacky12,Reference Rice, Bernasconi and Maki13,Reference Berliner, Mattern and Wellige16,Reference Baghai, Varallo-Bedarida and Born17) . In addition, uptake of EPA and DHA has high inter-individual variability, e.g. by a factor 13 in one trial(Reference Köhler, Bittner and Löw18). The consequence of both inhomogeneous baseline levels and high inter-individual variability of uptake was that on-trial levels of EPA and DHA largely overlapped among placebo and verum groups(Reference Muhlhausler, Gibson and Yelland19). Since clinical events correlate with the levels rather than with the intake of omega-3s, this made it impossible to discern an effect of the intervention with EPA and DHA(Reference von Schacky12,Reference Rice, Bernasconi and Maki13) . The issues mentioned here are discussed in more detail elsewhere(Reference von Schacky12,Reference Rice, Bernasconi and Maki13) . In the future, recruitment of trial participants will have to depend on a low baseline omega-3 index (room for improvement needed), and doses of EPA and DHA will have to be individualised, in order to separate the levels in the placebo and verum groups. Positive trial results in populations characterised by a low omega-3 index, such as patients with major depression or congestive heart failure with reduced ejection fraction, support this approach, although the dose was not individualised in the pertinent trials(Reference Grosso, Pajak and Marventano20,Reference Tavazzi, Maggioni and Marchioli21) . Individualising the dose is likely to increase the effect, and thus reduce the sample size, as well as reduce the untoward effects(Reference von Schacky12). The latter is supported by the results of trials using high doses of EPA and/or DHA, as in the JELIS or REDUCE-It trials(Reference Yokoyama, Origasa and Matsuzaki6,Reference Bhatt, Steg and Miller7) . In both cases, the cost of a positive trial result was a small increase in bleeding episodes (0⋅1 %/year)(Reference Yokoyama, Origasa and Matsuzaki6,Reference Bhatt, Steg and Miller7) .
Unfortunately, perception of the effectiveness of EPA and DHA in clinical medicine is frequently shaped by the results of pertinent Cochrane-Analyses. Cochrane-Analyses include trials based on standardised Cochrane-criteria designed for drug trials. In areas in which EPA and DHA have been tested in randomised controlled trials, Cochrane-Analyses tend to demonstrate no or only small effects(Reference Abdelhamid, Brown and Brainard22). This is because Cochrane-criteria do not allow trials with the design issues just discussed to be excluded.
Cardiovascular effects
The magnitude of the potential effects of EPA and DHA can be estimated by considering the results of epidemiologic studies, e.g. the Framingham study, using the original omega-3 index. With an omega-3 index >6⋅8 %, total mortality was 65 % of total mortality with an omega-3 index <4⋅2 %(Reference Harris, Tintle and Etherton3), reflecting very similar results from earlier studies(Reference Kleber, Delgado and Lorkowski4,Reference Harris, Luo and Pottala5) . The respective numbers for total CVD (63 %), total CHD (59 %) and total stroke (47 %) were similar(Reference Harris, Tintle and Etherton3). Interestingly, simultaneously measured cholesterol levels were no risk for any of the events mentioned(Reference Harris, Tintle and Etherton3). The risk for peripheral artery disease was also substantially lower with a higher omega-3 index than with a lower omega-3 index(Reference Ramirez, Zahner and Spaulding23). When dietary intake was assessed, associations with the health issues mentioned were substantially smaller(Reference Micha, Peñalvo and Cudhea24,Reference Chowdhury, Warnakula and Kunutsor25,Reference Chowdhury, Stevens and Gorman26) . Together with the positive results from large intervention trials, the numbers mentioned support the use of the omega-3 index to maximise the benefit and minimise the risk of EPA and DHA in the prevention of mortality and CVD. In keeping, the American Heart Association has issued scientific advisories recommending EPA and DHA for the secondary prevention of CVD but refrained from mentioning a dose(Reference Siscovick, Barringer and Fretts27,Reference Rimm, Appel and Chiuve28) .
Low levels of EPA and DHA precede the development of congestive heart failure, and a low omega-3 index can be found in these patients(Reference Berliner, Mattern and Wellige16,Reference Mozaffarian, Lemaitre and King29,Reference Harris, Masson and Barlera30) . A large intervention trial with approximately 860 mg EPA and DHA daily in patients with congestive heart failure with reduced ejection fraction increased the omega-3 index in the verum group from 4⋅75 (sd 1⋅68) to 6⋅73 (sd 1⋅93) % after 3 months, whereas it remained constant about 4⋅77 (sd 1⋅60) in the placebo group(Reference Harris, Masson and Barlera30). The target range was not reached, but, since omega-3 index levels in the verum group were somewhat separated from omega-3 index levels in the placebo group, the primary endpoint, a combination of total mortality and hospitalisations, was significantly reduced in the verum group(Reference Tavazzi, Maggioni and Marchioli21).
An association between the intake of fish and blood pressure was not found(Reference Yang, Shi and Li31). However, the circulating levels of EPA and DHA were inversely related to blood pressure(Reference Yang, Shi and Li31). In a representative assessment of the entire population of Liechtenstein, the omega-3 index correlated inversely with blood pressure (both systolic and diastolic), whether assessed over 24 h, during daytime or night, or conventionally in the office. The inverse correlation was independent of age and sex and other confounders such as BMI, smoking status, glycated haemoglobin A1c, educational status, fruit and vegetable consumption, physical activity and others(Reference Filipovic, Aeschbacher and Reiner32). Endothelial function improved in intervention trials, also depending on the omega-3 index(Reference Skulas-Ray, Kris-Etherton and Harris33,Reference Wang, Liang and Wang34) . Meta-analyses of intervention trials demonstrated that EPA and DHA lower blood pressure(Reference Miller, Van Elswyk and Alexander35).
Brain and the omega-3 index
As mentioned, in the Framingham study, an omega-3 index of 6⋅8 % was associated with a 53 % lower risk for total stroke than an omega-3 index of 4⋅2 %(Reference Harris, Tintle and Etherton3). In the Reduce-It trial, in the verum group, stroke occurred in ninety-eight patients, whereas it occurred in 134 patients in the placebo group, a 28 % reduction (hazard ratio 0⋅72, 95 % CI 55, 0⋅93, P = 0⋅01)(Reference Bhatt, Steg and Miller7). Therefore, n-3 fatty acids must be considered a powerful possibility for preventing the stroke, a catastrophic event for brain structure and thus brain function.
In an epidemiologic study in children with a mean age of 4, the omega-3 index correlated with complex brain functions, such as executive function, in this case assessed as the dimensional change card sort task(Reference Adjepong, Yakah and Harris36), extending similar findings from study populations aged 31, 67 and 78 years(Reference Dretsch, Johnston and Bradley37–Reference Bigornia, Scott and Harris40). Recent intervention trials confirmed that improvements in complex brain functions correlate with omega-3 index, when measured(Reference Witte, Kerti and Hermannstädter41,Reference Külzow, Witte and Kerti42) .
Pregnancy and lactation
Premature birth before week 34 of pregnancy is more likely with low levels than with high levels of EPA and DHA in plasma or erythrocytes, with the risk for preterm birth correlating inversely with those levels(Reference Olsen, Halldorsson and Thorne-Lyman43,Reference Hoge, Donneau and Dardenne44) . Another publication found post-partum depression to strongly depend on the omega-3 index, i.e. with a higher omega-3 index, post-partum depression was much less likely than with a lower omega-3 index(Reference Hoge, Tabar and Donneau45). Unfortunately, however, the findings are not directly comparable to the results obtained with the original omega-3 index, since different analytical procedures were used(Reference Hoge, Donneau and Dardenne44,Reference Hoge, Tabar and Donneau45) .
In 2018, a large Cochrane Meta-Analysis clearly found premature birth to be reduced both before weeks 34 and 37, gestational age to be prolonged by 1⋅67 days (95 % CI 0⋅95, 2⋅39), based on forty-one randomised controlled trials in 12 517 pregnant women, a lower incidence of low birth weight 15⋅6 v. 14 %; relative risk 0⋅90 (95 % CI 0⋅82, 0⋅99), based on fifteen randomised controlled trials in 8 449 pregnant women, and relative risk for perinatal death of the child to be 0⋅75 (95 % CI 0⋅54, 1⋅03), based on ten randomised controlled trials in 7 416 pregnant women, and more benefits for mother and child (all comparisons relative to placebo or control)(Reference Middleton, Gomersall and Gould46). In the practical conclusion, the authors mentioned: A universal supplementation can make sense, although, with better knowledge, it should be aimed for women benefitting the most(Reference Middleton, Gomersall and Gould46).
Already in 2016, the omega-3 index had been assessed in an almost representative manner in pregnant and lactating women in Germany and was found to be between 2⋅49 and 11⋅10 % regardless of timepoint (pregnancy v. lactation) and whether the woman supplemented EPA and DHA(Reference Gellert, Schuchardt and Hahn47). Taken together, in this author's opinion, the data to date make it necessary to determine the omega-3 index before or early in pregnancy, later in pregnancy and in lactation to make a targeted supplementation feasible, with a target range of 8–11 % for the omega-3 index.
Omega-3 index in sports medicine
In 2014, we had observed that 106 German world class athletes had a mean omega-3 index of 4⋅97 (sd 1⋅19) %, with only one athlete in the target range(Reference von Schacky, Haslbauer and Kemper48). More recently, similar data were found in 404 US national division I college football athletes with a mean omega-3 index of 4⋅4 (sd 0⋅8) %, again only one athlete in the target range(Reference Anzalone, Carbuhn and Jones49). Interestingly, the more energy was burned throughout the season, the lower the omega-3 index became(Reference Davinelli, Corbi and Righetti50). This has important implications for athletes, since a low omega-3 index predisposes to delayed-onset muscle soreness, and increasing the omega-3 index results in less creatine kinase release from muscle, less inflammatory reaction (measured as muscle swelling and pro-inflammatory cytokines in plasma) and less loss of function(Reference Kim and Lee51,Reference Ochi and Tsuchiya52) . Joint pain was reduced with a higher omega-3 index, at least in dogs(Reference Burri, Wyse and Gray53). Joint stability, e.g. in the case of the shoulder rotator cuff, was higher with a higher omega-3 index(Reference Hudek, von Schacky and Passow54). In an intervention trial, it had been demonstrated that brain damage, assessed as neurofilament light (the pertinent biomarker), can be reduced with high doses of EPA and DHA in professional players of American football(Reference Oliver, Jones and Kirk55). Jointly, these findings point towards higher membrane stability with a higher omega-3 index. Together with the aspects of cognition that have been improved by EPA and DHA in intervention trials in athletes, such as reaction time and efficiency(Reference Guzmán, Esteve and Pablos56), current data indicate that an omega-3 index in the target range may be an important asset for the athlete. This is supported by the fact that athletes are at an increased risk for major depression, suicide and sudden cardiac death, all in keeping with a low omega-3 index, and all seriously impacting on an athlete's life(Reference von Schacky, Haslbauer and Kemper48).
Other recent findings
Conventionally, SFA are considered as a homogenous group with an ominous effect on life expectancy. With the determination of the original omega-3 index comes the measurement of six individual SFA in erythrocytes(Reference Harris and von Schacky1). It was thus determined that palmitic acid (C16:0) was associated with increased total mortality in a 10-year epidemiologic study of 3259 participants, while the other erythrocyte SFA investigated (C14:0, C18:0, C20:0, C22:0, C24:0) had no association with total mortality(Reference Kleber, Delgado and Dawczynski57). In a meta-analysis, higher levels of palmitic acid were associated with a higher risk for developing type 2 diabetes(Reference Huang, Lin and Aris58). In a single study, this was also true for gestational diabetes(Reference Zhu, Tsai and Sun59). Cell membrane levels of palmitic acid are partly derived from ingested palmitic acid, and partly produced endogenously, e.g. in reaction to energetic surplus(Reference Carta, Murru and Banni60). Together with other findings of individual structure, metabolism, biologic effects and impacts on life expectancy, our findings question the conventional group-wise nomenclature of fatty acids (e.g. SFA, PUFA and others), as well as the conventional recommendation to avoid all SFA. Whether high levels of palmitic acid might be a biomarker for metabolically untoward body fat, such as excess hepatic fat, remains to be investigated, which is also true for the potential effects of actively reducing levels of palmitic acid.
The findings just discussed were mirrored in an epidemiologic study in children with a median age of 11 years, with palmitic acid correlating positively with waist circumference, TAG, fasting insulin and fatty liver index, against the background of a low omega-3 index (4⋅7 (sd 0⋅8) %)(Reference Bonafini, Tagetti and Gaudino61). Interestingly, the levels of arachidonic acid correlated inversely with the parameters mentioned(Reference Bonafini, Tagetti and Gaudino61). According to a recent meta-analysis, high levels of EPA and DHA in plasma or erythrocytes were associated with a low risk for the metabolic syndrome(Reference Jang and Park62). Thus, the omega-3 index makes it possible to investigate the relations between metabolism and non-n-3 fatty acids in more detail.
In patients with chronic fatigue syndrome/myalgic encephalomyelitis, a low mean omega-3 index was found(Reference Castro-Marreroa, Zaragozáa and Domingo63). Chronic fatigue syndrome/myalgic encephalomyelitis is a debilitating chronic medical condition without a known aetiology, no clinically established diagnostic test and no effective pharmacologic treatment(Reference Castro-Marreroa, Zaragozáa and Domingo63). Our findings add chronic fatigue syndrome/myalgic encephalomyelitis to the list of chronic inflammatory diseases where EPA and DHA should be tested in intervention trials for potential therapeutic value(Reference Castro-Marreroa, Zaragozáa and Domingo63).
Some recent intervention trials with EPA and DHA had neutral results. In one trial, it became clear by measuring the omega-3 index that this was due to participants' non-compliance(Reference van der Wurff, von Schacky and Bergeland64). In another trial, effects on cognitive parameters were smaller than expected within the 16-week trial period, demonstrating the difficulties of such trials(Reference Schättin, Baier and Mai65).
Conclusion
In the years 2018 and 2019, a perspective on EPA and DHA levels has proven useful to generate new knowledge on fatty acids, such as there is no human life with an omega-3 index ≤2 % in erythrocytes. Deficits in EPA and DHA, i.e. an omega-3 index below the target range of 8–11 %, have been found in many countries and populations. A deficit in EPA and DHA is associated with increased total mortality, fatal and non-fatal cardiovascular events, stroke, impaired cognition, premature birth, perinatal mortality and other health issues(Reference Harris, Tintle and Etherton3–Reference Harris, Luo and Pottala5,Reference Sarter, Kelsey and Schwartz8,Reference Langlois and Ratnayake9,Reference Berliner, Mattern and Wellige16,Reference Baghai, Varallo-Bedarida and Born17,Reference Harris, Masson and Barlera30,Reference Filipovic, Aeschbacher and Reiner32,Reference Dretsch, Johnston and Bradley37–Reference Lukaschek, von Schacky and Kruse39,Reference Hoge, Tabar and Donneau45,Reference Gellert, Schuchardt and Hahn47,Reference von Schacky, Haslbauer and Kemper48,Reference Hudek, von Schacky and Passow54,Reference Castro-Marreroa, Zaragozáa and Domingo63,Reference van der Wurff, von Schacky and Bergeland64) . A deficit in EPA and DHA in the population studied is required for positive results of a randomised controlled intervention trial. Reducing a deficit in EPA and DHA prolongs life, and reduces fatal and non-fatal cardiovascular events, strokes, premature birth, perinatal mortality and other health issues. In a trial, this can only be detected if the levels in the verum group differ sufficiently from the levels in the placebo or control group. Increasing the omega-3 index into the target range of 8–11 % with individualised doses of toxin-free sources for EPA and DHA is tolerable and safe.
Financial Support
The present work was not supported by any funding agency or by industry.
Conflict of Interest
C. v. S. operates Omegametrix, a laboratory for fatty acid analyses. In the past 3 years, C. v. S. received honoraria for speaking and consulting from BASF/Pronova, EPAX, Huntsworth Medical, Abbott, DSM, Marine Ingredients and Norsan.
Authorship
The author had sole responsibility for all aspects of preparation of this paper.




