Volume 50 - June 2002
Research Article
Smectite-Type Clay Minerals as Nanomaterials
- Robert A. Schoonheydt
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 411-420
-
- Article
- Export citation
-
Dilute aqueous smectite suspensions are well suited to convert into functional nanofilms. The functionalization is performed by ion exchange of amphiphilic molecules carrying the desired functionality. A functionalized clay mineral nanofilm is obtained by the organization of the molecules at the clay surface and by the organization of the elementary clay platelets in the film. Two techniques are presented: self-assembling and Langmuir-Blodgett (LB). Self-assembling is a very simple experiment, but the organizational order of the clay mineral particles is low. Nevertheless, films with nonlinear optical properties which are able to generate frequency-doubled light have been produced. The drawback of disorder of the clay platelets in the film can be largely overcome with the LB technique. Well organized films are obtained. In the presence of amphiphilic ruthenium(II)-bipyridine complexes, nonlinear optical properties are measured. In the presence of two dyes, oxacarbocyanine and rhodamine B, simultaneously adsorbed, efficient energy transfer between the donor (oxacarbocyanine) and the acceptor (rhodamine B) is measured. Fundamental research into the organization of molecules at clay mineral platelets is necessary to optimize the materials for specific applications.
Hydrothermal Synthesis of Zn-Smectites
- Shoji Higashi, Kazuhiko Miki, Sridhar Komarneni
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 299-305
-
- Article
- Export citation
-
Two varieties of Zn-smectite were synthesized hydrothermally: sauconite, with an ideal composition of Na0.4Zn3(Si3.6Al0.4)O10(OH)2·nH2O; and a Zn equivalent of hectorite, with an ideal composition of Na0.4 (Li0.4Zn2.6)Si4O10(OH)2·nH2O (referred to here as Zn-hectorite). For comparison, hydrothermal synthesis of the related trioctahedral smectites of hectorite, Na0.4(Li0.4Mg2.6)Si4O10(OH)2·nH2O and hectorites containing Cu, Co or Ni in the octahedral sheets instead of Mg were also attempted. The results showed that sauconite, Zn-hectorite and hectorite could be synthesized in the temperature range 100–125°C but hectorites containing Cu, Co or Ni in the octahedral sheet, under the same conditions or even at a temperature of 150°C, could not.
Weathering of Chlorite: I. Reactions and Products in Microsystems Controlled by the Primary Mineral
- Mehrooz F. Aspandiar, Richard A. Eggleton
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 685-698
-
- Article
- Export citation
-
The weathering of chlorite in hydrothermally-altered basalt was studied with XRD, TEM and electron microprobe to determine the type and orientation of secondary minerals. Optical examination indicated chlorite assemblages to have altered in two distinct microsites: one microsite near micro-fissures traversing the regolith units, and the other away from the continuous passages. In this paper, weathering mechanisms and products of chlorite present in microsites distant from the micro-fissures are reported. In all the regolith units the original chlorite grain remained intact and was pseudomorphed by secondary products. In the saprock, chlorite altered to corrensite with possible random interstratifications of chlorite and corrensite and corrensite and vermiculite. In the saprolite, corrensite altered to vermiculite. Parallelism of two axes of the products with the host indicated topotactic alteration. In the fine saprolite, vermiculite was found to alter to kaolinite via a randomly interstratified kaolinite-vermiculite stage with a high proportion of kaolinite. Goethite crystallized in between packets of kaolinite, vermiculite and kaolinite-vermiculite. Though the disruption of the crystal structure of vermiculite is necessary in its alteration to kaolinite, the reaction was such as to maintain parallelism of the c axis. The alteration of chlorite to vermiculite was characterized by the loss of Mg and Fe and minor Al, all ions considered to be lost from the brucite-like sheet of chlorite. The Fe released during the alteration of vermiculite to kaolinite is likely to have migrated to micropores to form goethite. The presence of interstratifications of the end-members of layer silicates involved in the reaction sequence suggests that interstratifications are common during layer silicate weathering in environments where space is limited and consequently solution and ionic transport passages are restrictive.
Origin of Allophane and Retardation of Pebble Weathering in Quaternary Marine Terrace Deposits
- Gi Young Jeong, Jin Han Bae, Chang Sik Cheong
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 145-156
-
- Article
- Export citation
-
Quaternary marine terrace deposits consisting of gravels interbedded with thin sandy gravel layers have been subjected to subaerial weathering. Restricted to the sandy gravel layers, allophane gel either replaced bytownite sands to form a pseudomorph or coated the pebbles. The allophane has an average Al/Si atomic ratio of 1.5 with 45% H2O. The sandy gravels were originally rich in bytownite (av. An86) sands derived from underlying Tertiary basaltic lapilli tuff. The highly soluble and aluminous bytownite favored the formation of allophane. In the sandy gravel layers, pebbles coated with allophane gel were almost fresh whereas those in the gravel layers were highly weathered to form halloysite-rich clays. Allophane gels acted as a somewhat impermeable geochemical barrier impeding a mineral-water reaction in the bytownite-rich sandy gravel layers and thus significantly retarding pebble weathering, while prolonged weathering in the gravel layers resulted in the severe decomposition of pebbles. Bytownite protected the pebbles against weathering, implying that minor soluble minerals might be one of the factors in the natural variation of the weathering rates of rocks and sediments.
Diffusion of H2O and I− in Expandable Mica and Montmorillonite Gels: Contribution of Bound H2O
- Yoshito Nakashima
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-10
-
- Article
- Export citation
-
Self-diffusion coefficients of H2O molecules in water-rich gels of Na-rich expandable mica synthesized using natural talc were measured by pulsed-gradient spin-echo 1H nuclear magnetic resonance (NMR), and the dependence on mica fraction (0.00–43.8 wt.%) and temperature (30.0–60.9°C) was examined. On the basis of the NMR results, the self-diffusion coefficient of H2O, Dwater, in the gel can be expressed by ln(Dwater/D0water) = 1.64[exp( −0.0588w) - 1], where D0water is the self-diffusivity of bulk water at temperature and w is the weight fraction of the mica (wt.%). The activation energy of H2O diffusivity in mica gel is nearly equal to that in bulk water. These findings indicate that the normalized diffusivity, Dwater/D0water, is independent of temperature. The diffusivity of I−, Diodine, in the gels was examined by X-ray computed tomography (CT) at 22°C, and the influence of the mica fraction (0.00–24.8 wt.%) was studied to determine the contribution of bound H2O. The X-ray CT results show that the normalized I− diffusivity, Diodine/D0iodine, obeys the above-mentioned phenomenological curve where D0iodine is the I− diffusivity in bulk water. I− is non-sorbing, and thus its diffusion is restricted only by the geometrical complexity of the pore structure of gels. Therefore, the effect of bound H2O molecules on average H2O diffusivity is negligible for w <24.8 wt.%. Diffusivity is governed by free or unbound H2O molecules diffusing in the geometrically complex and tortuous pore structure of mica-mineral grains. This is a result of the large population of unbound H2O far from the grain surface compared to the small population of bound H2O near the grain surface. The diffusion of I− ions in montmorillonite gels was examined by X-ray CT for w <16.7 wt.% montmorillonite. The normalized iodine diffusivity, (Diodine/D0iodine) obtained is in reasonably close agreement with the literature data for the normalized diffusivity of H2O and is similar to the master curve of expandable mica. Therefore, bound H2O molecules near negatively charged clay surfaces do not play a major role in the H2O diffusivity for water-rich montmorillonite gels.
Micro-Structure and Hydraulic Conductivity of Simulated Sand-Bentonite Mixtures
- Tarek Abichou, Craig H. Benson, Tuncer B. Edil
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 537-545
-
- Article
- Export citation
-
This paper describes the relationship between the micro-structure and hydraulic conductivity of simulated sand-bentonite mixtures (SSBMs) prepared with powdered and granular bentonite. Glass beads were used to simulate sand grains because of their superior optical properties. The micro-structure of SSBMs was observed using optical micrography and scanning electron microscopy. For mixtures prepared with powdered bentonite, the indications are that bentonite coats the particles. As the bentonite content increases, the thickness of bentonite coating increases and reduces the area available for flow. For mixtures containing granular bentonite, the dry bentonite granules occupy the space between the particles and then swell to fill the void space. As the bentonite content increases, the number of granules increases, leading to more void spaces being filled with bentonite. At higher bentonite content (>8%), flow paths devoid of bentonite are unlikely, and the hydraulic conductivity appears to be controlled by the hydraulic conductivity of bentonite. The changes in micro-structure that were observed are consistent with the decrease in hydraulic conductivity that occurs with increasing bentonite content. However, the relationship between hydraulic conductivity and bentonite content differs depending on whether a mixture contains powdered or granular bentonite.
Benzene Vapor Sorption by Organobentonites From Ambient Air
- Lizhong Zhu, Yuhong Su
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 421-427
-
- Article
- Export citation
-
A number of organobentonites were synthesized by exchanging the metal ions in bentonite with the cationic surfactant, cetyltrimethylammonium bromide. Samples of natural bentonite and organobentonites were analyzed for their organic carbon contents, examined by X-ray diffraction (XRD) for interlayer spacings, and viewed using a scanning electron microscope (SEM) for surface morphology. The sorption isotherm of benzene vapor from ambient air (relative humidity (RH) = 45 ± 5%) on natural bentonite was nonlinear; however, the isotherms of benzene from ambient air onto organobentonites were virtually linear over a large range of relative vapor concentrations. The sorption capacities of air-dried organobentonites were far greater than that of the natural bentonite. For air-dried organobentonites, the sorption coefficients correlated positively with the sample organic carbon contents and negatively with the sample BET-N2 surface areas. The heats of benzene vapor sorption onto air-dried organobentonites were less exothermic than the heat of benzene-vapor condensation. These findings suggest that benzene vapor sorption by air-dried organobentonites occurs essentially by vapor partition into the sample organic-matter fractions. This offers a potential application of organobentonites for the removal of organic vapors from flue gases and for assessing the efficiency of vapor removal.
Weathering of Chlorite: II. Reactions and Products in Microsystems Controlled by Solution Avenues
- Mehrooz F. Aspandiar, Richard A. Eggleton
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 699-709
-
- Article
- Export citation
-
Weathering of chlorite adjacent to macro- and micro-passages (fissures) in regolith units has been studied to determine the alteration mechanisms and products and compare them to those that prevail at a distance from the open passages. The micro-fissures in the saprolith units evolve as a function of weathering with an increase in micro-fissure density and their infilling with ferriargillans corresponding to an increase in weathering intensity. In the saprock and saprolite units, goethite-rich bands with surrounding reddish haloes invade the chlorite adjoining the micro-fissures. In the reddish haloes surrounding the goethite bands and fissures, the phyllosilicates alter to an intimate mixture of goethite, kaolinite, feroxyhyte and halloysite with the possible nanoscale presence of other fine-grained iron aluminosilicates such as hisingerite and smectite. In the fine saprolite, the weathering products after original chlorite adjoining the micro-fissures bearing ferriargillans, alter directly to ferriargillan products — kaolinite and goethite. The lack of orientation of the products with the parent phyllosilicates indicates the operation of a dissolution-precipitat ion mechanism which is in contrast to the topotactic alteration mechanism functioning at a distance within the phyllosilicate grain assemblage. The differences in alteration mechanisms and products of chlorite weathering in different microsites suggest the rate of weathering of chlorite can differ in microsites within individual regolith units. The presence of fine-grained metastable products in the form of feroxyhyte and halloysite adjoining the fissures suggests an Ostwald Step Rule sequence during alteration of phyllosilicates with rapid oxidation of Fe and the presence of Si in the microsite considered the main factor favoring fine-grained metastable products.
Transmission Electron Microscopy Study of Smectite Illitization During Hydrothermal Alteration of a Rhyolitic Hyaloclastite From Ponza, Italy
- Blanca Bauluz, Donald R. Peacor, Robert F. Ylagan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 157-173
-
- Article
- Export citation
-
Dioctahedral phyllosilicates from an altered rhyolitic hyaloclastite located at Ponza Island, Italy, were studied by scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The samples are from a sequence previously characterized by X-ray diffraction (XRD) methods, indicating that a complete range of illitization accompanies alteration. Backscattered electron (BSE) images, obtained from ion-milled samples, show that samples partly retain the original textures since clay minerals pseudomorph lapilli fragments and preserve vesicular texture. The lowest-grade sample studied contains obsidian clasts partially replaced by smectite. As the alteration grade increases, illitization proceeds with formation of interstratified illite-smectite (I-S), zeolites, illitic phases, feldspars and quartz. The most altered sample contains illite, mica and quartz. Lattice-fringe images show that following the formation of smectite, illitization takes place through the formation of (R=1) I-S, highly illitic I-S and illite with mica; (R=1) I-S is the only ordered interstratified I-S. The BSE and TEM images of Ponza samples show irregular cavities filled with euhedral dioctahedral clay minerals and the zeolite mordenite, providing direct evidence for neocrystallization from a fluid. Chemical compositions by analytical electron microscopy (AEM) support the sequence described. Selected area electron diffraction (SAED) patterns indicate the predominance of 1 Md polytypism both in I-S and illitic phases, and the coexistence in the more altered samples of 1Md illite and a 2-layer mica polytype (probably 2M1), without the intermediate 1M polytype generally assumed to exist in prograde sequences. Previous XRD studies indicated progressive change from cis-vacant, turbostratically stacked smectite, to interstratified cis- and trans-vacant, 1Md I-S, to trans-vacant, 1M illite, and then to 2M1 illite in Ponza Island samples. We observed a clear correlation between the chemical compositions as determined by AEM and the proportion of cis-vacant determined by XRD, suggesting that the octahedral cation distributions change in the studied samples with increasing degree of illitization.
Infrared Evidence of Dioctahedral-Trioctahedral Site Occupancy in Palygorskite
- A. Chahi, S. Petit, A. Decarreau
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 306-313
-
- Article
- Export citation
-
A Mg-rich palygorskite sample from phosphorite deposits of Ganntour (Morocco) with the structural formula
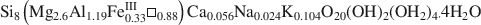 , was studied by FTIR spectroscopy. In both OH-stretching and OH-bending regions, there is evidence of dioctahedral Al2□OH, AlFe□OH and trioctahedral Mg3OH features, leading to a di-trioctahedral crystallochemical model of octahedral site occupancies in ribbons of Ganntour palygorskite.
, was studied by FTIR spectroscopy. In both OH-stretching and OH-bending regions, there is evidence of dioctahedral Al2□OH, AlFe□OH and trioctahedral Mg3OH features, leading to a di-trioctahedral crystallochemical model of octahedral site occupancies in ribbons of Ganntour palygorskite.This model, established through the IR spectroscopy study of a Mg-rich palygorskite, seems to be appropriate for many other palygorskites with lower Mg content in the octahedral sheet.
Monolayer to Bilayer Transitional Arrangements of Hexadecyltrimethylammonium Cations on Na-Montmorillonite
- James L. Bonczek, W. G. Harris, Peter Nkedi-Kizza
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 11-17
-
- Article
- Export citation
-
A low-charge Na-montmorillonite (SWy-2) was exchanged with hexadecyltrimethyl-ammonium (HDTMA) at levels equal to 20, 40, 60, 70, 80, 90, 100, 150 and 200% of the cation exchange capacity (819 mmol(+)/kg) to determine the nature of adsorption and the ionic composition of the clay interlayers. In contrast with earlier work with smaller aliphatic cations, which suggested random interstratification of interlayers occupied by either organic or metallic cations, there was no evidence of cation segregation into homogeneous interlayers. Instead, X-ray analysis indicated that the organic cations assumed two dominant configurations which were roughly equivalent in prevalence at ∼70% coverage of the CEC. Below 70% exchange the organocations existed predominantly in heterogeneous monolayers with Na+, attaining basal spacings of between 1.41 and 1.44 nm which were sensitive to changes in relative humidity. Relative humidity effects indicated that Na+ and HDTMA occupied functionally discrete domains within the interlayer as shown by the free interaction of water and a neutral organic solute, naphthalene, with Na+ and HDTMA, respectively. At greater levels of HDTMA exchange (up to 100% of the CEC), the organocations assumed a predominantly bilayer configuration. Transition to a fully-developed bilayer indicated by a 1.77 nm d-spacing at 100% coverage was gradual, suggesting some interstratification of the monolayers and bilayer configurations between 70 and 100% exchange. Sorption of naphthalene to the organoclays within this range of coverage was well correlated with clay organic carbon content, consistent with relatively unimpeded interlayer access of neutral organic molecules.
Hydraulic Performance of Untreated and Polymer-Treated Bentonite in Inorganic Landfill Leachates
- Alaa K. Ashmawy, Darwish El-Hajji, Nestor Sotelo, Naim Muhammad
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 546-552
-
- Article
- Export citation
-
Short- and long-term exposure to inorganic solutions can cause significant degradation of the hydraulic properties of bentonite clay used in geosynthetic clay liners (GCLs). In particular, the increase in hydraulic conductivity due to cation exchange when Na-montmorillonite is subjected to leachates rich in Ca and Mg has caused problems in incinerator ash landfill liners located in wet environments, where large quantities of leachates are generated. Experimental results are presented to evaluate the immediate change in hydraulic conductivity of seven types of GCL clays upon permeation with leachate generated from three ash landfills. The composition of the ash, which is a by-product of the incineration of municipal solid waste (MSW), in turn influences the composition of the resulting leachate. Falling head permeability tests were performed on flexible-wall permeameter specimens, with back-pressure saturation. Chemical analysis shows that the three leachate products contain high, medium, and low concentration Ca and Mg cations. The clay component of GCL materials tested in this study consists of regular and polymer-treated bentonite. Polymer treatment is believed to render the clay non-reactive to many organic and inorganic chemicals. The results of this study indicate that: (1) polymer treatment is generally more beneficial if the clay is first saturated with water and not directly with the leachate; (2) high swell potential of the bentonite is more advantageous than polymer treatment, especially when low hydraulic conductivity is required in the short term and if the clay is pre-hydrated. Experiment setup and special specimen preparation procedures are also discussed.
Intercalation of 8-Hydroxyquinoline into Al-Smectites by Solid-Solid Reactions
- Nithima Khaorapapong, Kazuyuki Kuroda, Makoto Ogawa
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 428-434
-
- Article
- Export citation
-
Tris(8-hydroxyquinoline)aluminum (III) complexes (Alq3), one of the molecules studied most widely for organic light-emitting devices, were formed in the interlayer spaces of smectites by solid-solid reactions between Al-smectites (Al-montmorillonite and Al-synthetic saponite (Sumecton)) and 8-hydroxyquinoline (8Hq) at room temperature. The intercalation of 8-hydroxyquinoline molecules into Al-smectites was demonstrated by powder XRD, FTIR, DTA, TG, TG-MS, and chemical analysis. The coordination of the ligand to the interlayer Al cations was proved by FTIR, UV-Vis, and photoluminescence spectroscopies. The luminescence intensity of Alq3-Sumecton was much greater than that of Alq3-montmorillonite, and this was ascribed to the very small amount of quenching impurities in Sumecton.
Biogenic Saponite From an Active Submarine Hot Spring, Iceland
- A. Geptner, H. Kristmannsdóttir, J. Kristjansson, V. Marteinsson
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 174-185
-
- Article
- Export citation
-
A study of the mineralogy, chemical composition and structure of poorly-crystalline saponite precipitated from a submarine hot spring in Eyjafjordur, northern Iceland is reported. Special emphasis was placed on the microstructures of the minerals and a possible connection with biological activity during their precipitation. The microstructures of the minerals were found to be very similar to specific clay minerals precipitated from geothermal vents in oceanic rift zones. The composition of the minerals was, however, found to be similar to magnesium silicate scales formed in geothermal installations in Iceland where geothermal waters were mixed with cold fresh waters. High contents of organic substances were found in the clay mineral samples as compared to geothermal precipitates from other localities. Microstructural features of the layer silicates in one of the samples suggest that a gelatinous substance was a precursor of the saponite clay. The organic matter content appears to be greater when the precipitates are more crystalline.
In Situ Characterization and Differentiation of Kaolinites in Lateritic Weathering Profiles Using Infrared Microspectroscopy
- Anicet Beauvais, Jacques Bertaux
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 314-330
-
- Article
- Export citation
-
Lateritic weathering kaolinites have been characterized in situ and differentiated for the first time by means of infrared microspectroscopy (IRMS). Four classical OH-stretching bands have been observed in the Fourier transform infrared (FTIR) spectra, at 3695, 3668, 3650 and 3620 cm−1, denoted ν1, ν2, ν3 and ν4, respectively, plus a band at 3595 cm−1 associated with the octahedral substitution of Fe3+ for Al3+. Infrared microspectroscopy of thin-sections of lateritic weathering profiles provides useful information on the types of kaolinite present in different horizons of the profile. The spectra obtained from large well-ordered kaolinite crystals look like those obtained by diffuse reflectance in that, compared with the KBr disk spectra of <2 µm powders, bands at 3668 and 3650 cm−1 are enhanced, and the strong absorption of KBr disks at 3695 cm−1 is replaced by a broad weaker band from 3700–3680 cm−1. In laterites, these large well-ordered kaolinites often exhibit a band at 3595 cm−1 indicative of significant Fe3+ substitution for Al3+ in the structure. The IR microspectra obtained from regions of small, more poorly-ordered kaolinites do not differ so markedly from that of KBr disks. All show enhanced absorption around 3650 cm−1 compared with well-ordered kaolinites, indicating that the disorder is due, at least in part, to domains of dickite-like and/or nacrite-like stacking in their structure. The 3595 cm−1 band is always weaker than that of the well-ordered kaolinite in the same profile. The IRMS data from well-characterized reference kaolinites show that the ratio Aν2/Aν3 is a pertinent IR order index for kaolinites. The larger this index, the larger is the area of the 3595 cm−1 band, and the larger and the more ordered is the kaolinite sample. It is suggested that the diversity of FTIR spectra observed reflects intergrowths of kaolinite-dickite polymorphs, or at least mixtures of high- and low-defect kaolinites which are frequently encountered in the lateritic geosphere rather than pure kaolinitic phases. The largest kaolinites having secondary crystallized in voids are the most ordered and the most ferruginous and have been considered as useful mineralogical tracers of the recent evolution of old lateritic terrains.
Water Vapor Sorption Behavior of Smectite-Kaolinite Mixtures
- William J. Likos, Ning Lu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 553-561
-
- Article
- Export citation
-
An experimental program was conducted to investigate the water-vapor sorption characteristics of smectite and kaolinite mixtures. End-member smectite and kaolinite were slurry-mixed together at mass-controlled ratios corresponding to 0%, 20%, 50%, 70%, 80%, 90% and 100% smectite. Vapor desorption isotherms for the mixtures were measured at 24°C for relative humidity (RH) ranging from ∼95% to 0%.
Results show that the amount of water adsorbed by the clay mixtures at a given RH increases systematically with increasing smectite content. Derivative analysis of the sorption isotherms shows evidence of transitions between the two-, one- and zero-layer hydrate-states for the smectite-rich mixtures. The transitions become less apparent as the smectite content decreases. Monolayer coverage, specific surface area, and heat of adsorption were estimated from the isotherms using BET theory. It is shown that monolayer coverage and specific surface for the clay mixtures can be reasonably approximated by weighted averaging of the end-member clay properties. General methodologies are presented for predicting the sorption behavior (i.e. soil-water characteristics) and effective specific surface area from measurements of the end-member sorption isotherms.
Smectite Quasicrystals in Aqueous Solutions as a Function of Cationic Surfactant Concentration
- Satoru Kuwaharada, Hiroshi Tateyama, Satoshi Nishimura, Hideharu Hirosue
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 18-24
-
- Article
- Export citation
-
Quasicrystals of synthetic fluoromagnesian smectite (FMS) in dodecyltrimethylammonium chloride (DTAC) solutions were investigated as a function of the DTAC concentration by wet type X-ray diffraction (XRD) ζ potential measurements, and dispersion and coagulation (DC) tests. The FMS had an electronegative potential and it was dispersed randomly in an aqueous suspension without DTAC. When the DTAC concentration was 0.002 mol/dm3, FMS tactoids started to develop a structure in which the layer thickness was 1.85 nm. At the isoelectric point, the DTAC concentration was 0.012 mol/dm3 and the FMS tactoid formed a regular stacked structure with a 2.25 nm layer thickness. As the ζ potential of FMS changed from negative to positive, the DC test and XRD measurement showed that the FMS association gradually changed from coagulation to dispersion, which indicates that the formation of the bilayer of surfactants on the surface of FMS produces a repulsion between DTA+ adsorbed on the silicate layer of the FMS surfaces. When the equilibrium concentration of DTAC in solution exceeded the critical micelle concentration (CMC), the ζ potential of FMS became greater than the previous values. The XRD analysis of this suspension showed that there were two kinds of rational FMS stackings; one has a layer thickness of 3.20 nm, and the other has a layer thickness of 5.45 nm due to an interstratified structure composed of 3.20 and 2.25 nm layers. The interstratified structure was confirmed by the calculated XRD profiles.
Zeolitization of a Phonolitic Ash Flow by Groundwater in the Laach Volcanic Area, Eifel, Germany
- Franz Bernhard, Ulrike Barth-Wirsching
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 710-725
-
- Article
- Export citation
-
Field and experimental studies were performed to understand the formation conditions of the Nettetal zeolite deposit, Laach volcanic area, Germany. This deposit shows pronounced small- (cm) and large-scale (tens of meters) variations of zeolitization, despite the same phonolitic precursor glass throughout the occurrence. Zeolitization of the pyroclastic ash flow is restricted to three distinct layers that are 0.15 to 10 m thick and separated by fresh ash. The glassy matrix is altered to chabazite, phillipsite, analcime and K-feldspar in various combinations, whereas the pumice clasts are altered predominantly to chabazite. Mass changes during zeolite formation appear to be small, and Ca enrichment in chabazite and phillipsite may have occurred after their formation by cation exchange.
The zeolites and zeolite assemblages observed in the Nettetal deposit were experimentally reproduced by reacting the phonolitic glass at 100–200°C with distilled water and 0.01 M alkaline solutions as well as with varying solid/liquid ratios and grain-sizes. Chabazite and phillipsite represented metastable transition phases with respect to analcime and K-feldspar. A high solid/liquid ratio accelerated the conversion of glass to zeolites.
None of the classic models of zeolite formation is fully applicable to the Nettetal deposit. The most probable environment for zeolitization in this deposit is the stagnant fringe water zone immediately above the groundwater table. In this zone, representing a relatively closed system, favorable solution compositions for zeolite formation could have been developed rather quickly by glass-water interaction, which is not possible within the more thoroughly flushed deeper parts of the groundwater system. The three distinct zeolite layers are probably the result of temporarily changing groundwater levels.
Lepidocrocite in Hydrothermal Sediments of the Atlantis II and Thetis Deeps, Red Sea
- Nurit Taitel Goldman, Christian Bender Koch, Arieh Singer
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 186-197
-
- Article
- Export citation
-
Lepidocrocite (γ-FeOOH) formation in the hydrothermal brines of the Thetis and Atlantis II Deeps in the Red Sea results in markedly different crystals (size and shape). The only foreign element associated with the crystals is Si and analyses of samples from the two deeps yielded average Si/Fe (molar) ratios of 0.03 and 0.11, respectively. The Si/Fe ratio does not affect formation of a perfect lattice along [010]. Direct observations of crystal morphology as well as X-ray diffraction patterns, Mössbauer and infrared spectra, all indicate that the Atlantis II Deep lepidocrocite is less crystalline than the Thetis Deep lepidocrocite. In one sample a poly-disperse size distribution was resolved indicating a fine-scale variation in precipitation conditions. Infrared spectroscopy suggests that the Si is adsorbed on the lepidocrocite surfaces, probably also forming polymers, as both Fe-O-Si and Si-O-Si bonds can be detected. The formation of the Atlantis II Deep lepidocrocite is due to fast oxidation of Fe2+. The blanket-like layer of lepidocrocite in Atlantis II and Thetis Deeps lepidocrocite was probably formed as a result of precipitation during an abrupt oxidation event of the brine, triggered by down-welling of a condensed oxidized brine, which originated in the northern part of the Red Sea. A difference in Si concentrations determined the different crystal properties of the lepidocrocite formed in the two deeps.
Expansion of Smectite by Hexadecyltrimethylammonium
- Seung Yeop Lee, Soo Jin Kim
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 435-445
-
- Article
- Export citation
-
The adsorption of hexadecyltrimethylammonium (HDTMA) in smectite was studied by adsorption isotherms, X-ray diffraction (XRD) and high-resolution transmission electron microscopy (HRTEM). Smectites that had reacted for 48 h with HDTMA cations equivalent to 0.2–3.0 times the cation exchange capacity (CEC) were converted to HDTMA-exchanged smectites with various d-spacings. Study of HDTMA-smectites by HRTEM suggests that the HDTMA adsorption results in interlayer expansion with various d-spacings and irregular wavy layer structures. We believe that HDTMA loading beyond the CEC of smectite affects the structure of clay by the additional adsorption of HDTMA-Br− via hydrophobic bonding. Surfactant orientation probably depends on the quantity of surfactant in the interlayer. Our TEM study shows that the structure of the adsorbed HDTMA layer in the interlayers of smectite depends on the charge distribution and chemical composition of smectite.