Poster Presentation - Poster Presentation
Antibiotic Stewardship
Length of antibiotic therapy among adults aged ≥65 years hospitalized with uncomplicated community-acquired pneumonia, 2013-2020
- Natalie McCarthy, Hannah Wolford, Sophia Kazakova, James Baggs, Brandon Attell, Sarah Kabbani, Melinda Neuhauser, Sarah Yi, Kelly Hatfield, Sujan Reddy, Lauri Hicks
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s26
-
- Article
-
- You have access Access
- Open access
- Export citation
-
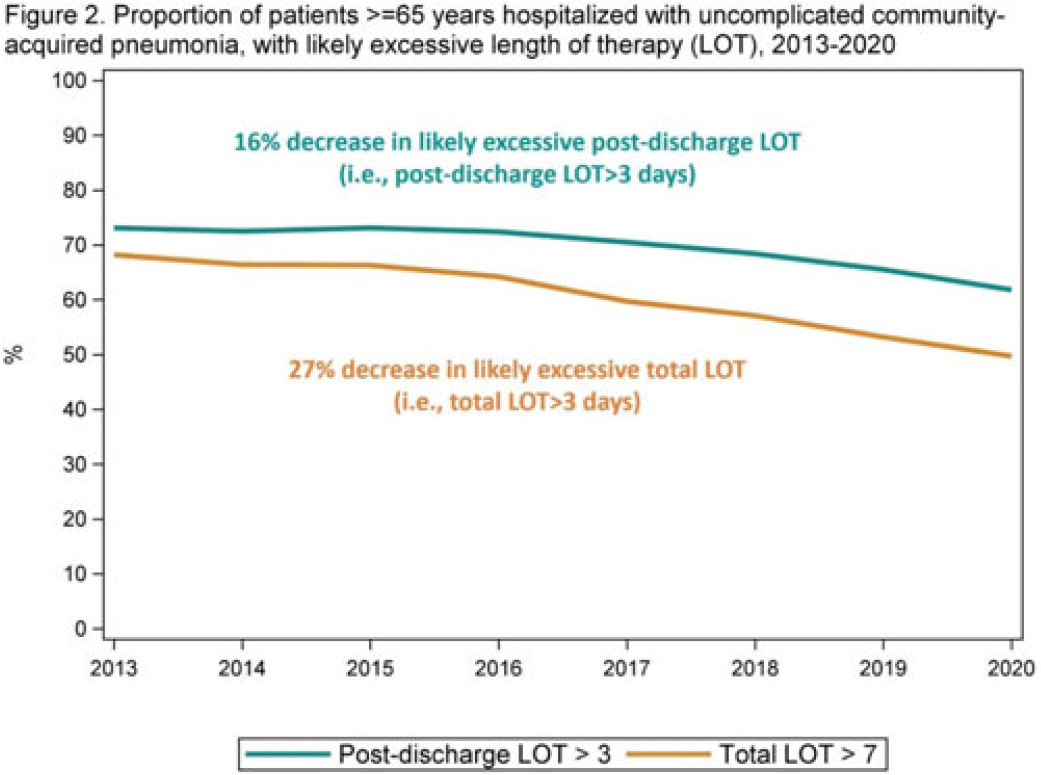
Background: The 2014 US National Strategy for Combating Antibiotic-Resistant Bacteria aimed to reduce inappropriate inpatient antibiotic use by 20% for monitored conditions, such as community-acquired pneumonia (CAP), by 2020. Clinical guidelines recommend treating uncomplicated CAP with a minimum of 5 days of antibiotic therapy. Total length of therapy (LOT) >7 days or >3 days after clinical improvement is rarely necessary. In a previous study estimating LOT in uncomplicated CAP patients, 71% of patients ≥65 years exceeded recommended duration of antibiotics in 2012–2013 (Yi et al, 2018). We evaluated annual trends in LOT in adults ≥65 years hospitalized with uncomplicated CAP from 2013 to 2020. Methods: We conducted a retrospective cohort study among patients in the CMS database with a primary diagnosis of bacterial or unspecified pneumonia using International Classification of Diseases 9th and 10th Revision codes, length of stay (LOS) of 2–10 days, discharged home with self-care, and not rehospitalized in the 3 days following discharge. Discharge home was used as a surrogate for clinical improvement. Because inpatient LOT is not available in CMS data, we used linear regression to model inpatient LOT as a function of LOS using data on CAP patients ≥65 years from the PINC AI healthcare database. Postdischarge LOT was based on prescriptions filled following discharge. Total LOT was calculated by summing estimated inpatient LOT and actual postdischarge LOT (Fig. 1). Total LOT >7 days and postdischarge LOT >3 days were considered indicators of likely excessive LOT. We reported trends in the proportion of patients with likely excessive LOT during the study period. Results: From 2013 through 2020, there were 400,928 uncomplicated CAP hospitalizations among patients aged ≥65 years. Patients were more likely to be female (55%), and they had a median age of 76 years and a median LOS of 3 days. The median total LOT decreased from 9.5 days in 2013 to 7.7 days in 2020. The proportion of patients with total LOT >7 days decreased from 68% in 2013 to 50% in 2020 (% change, −27%); the proportion with postdischarge LOT >3 days decreased from 73% in 2013 to 62% in 2020 (% change, −16%) (Fig. 2). Conclusions: Likely excessive total LOT for adults ≥65 years hospitalized with uncomplicated CAP decreased by 27% in 2020, a considerable improvement from 2013. However, the high proportion of patients with likely excessive postdischarge LOT in 2020 (62%) demonstrates the need for antibiotic stewardship to optimize prescribing at hospital discharge.
Disclosures: None
Empiric antibiotic selection for community-acquired pneumonia in US hospitals, 2013–2020
- Hannah Wolford, Brandon Attell, James Baggs, Sujan Reddy, Sarah Kabbani, Melinda Neuhauser, Lauri Hicks
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s26-s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
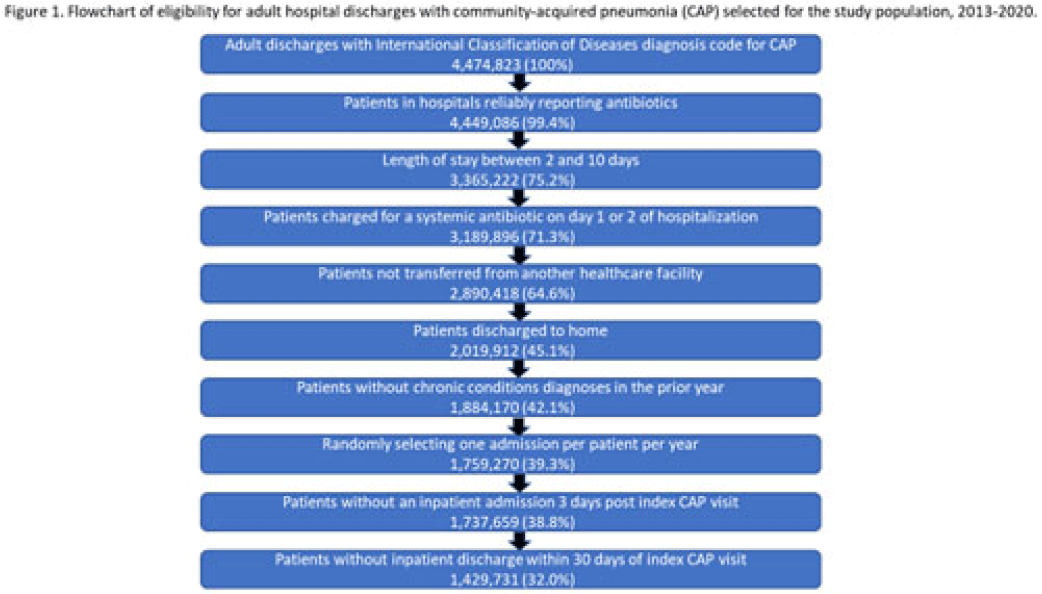
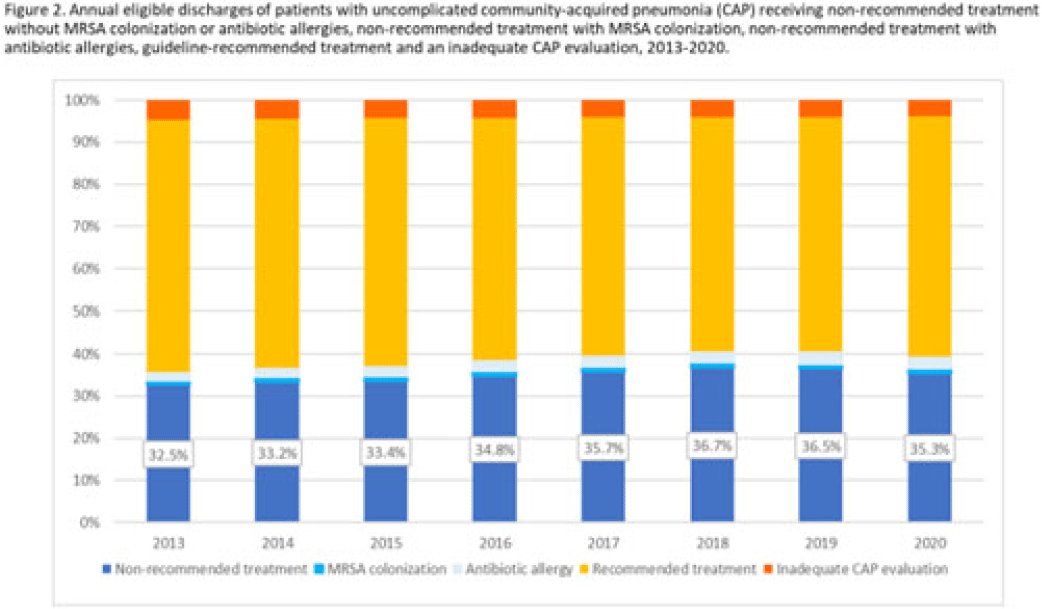
Background: Community-acquired pneumonia (CAP) is a common indication for antibiotic prescribing in hospitalized patients. Professional societies’ clinical guidelines recommend specific antibiotics for empiric treatment of CAP based on clinical factors. Manual assessments of appropriateness are time-consuming and are often conducted on a smaller scale. We evaluated empiric antibiotic selection among a large cohort of adults hospitalized with CAP using electronic health records. Methods: In this study, we used the PINC-AI healthcare database to define a cohort of adults hospitalized with CAP from 2013 to 2020. CAP was identified by International Classification of Diseases (ICD) diagnosis codes. Exclusions were applied to identify uncomplicated CAP (Fig. 1). Treatment was only evaluated if a chest radiograph or computerized tomography (CT) scan was charged during the first 2 days of hospitalization, otherwise it was considered an inadequate CAP evaluation. Administrative billing data were used to identify antibiotics charged within the first 2 days of hospitalization. Empiric guideline-recommended treatment was determined based on 2019 CAP guidelines and more recent studies. Patients who received nonrecommended treatment were evaluated for antibiotic allergies in the current hospitalization or methicillin-resistant Staphylococcus aureus (MRSA) colonization or infection in the year prior or on admission using International Classification of Disease, Tenth Revision (ICD-10) diagnosis codes. Results: We identified 4.47 million adult hospitalizations with CAP from 2013 to 2020; 32% (1.43 million) were included in this analysis (Fig. 1). Among discharges with adequate CAP evaluation (1.37 million), 59.7% received recommended antibiotics in the first 2 days of hospitalization, ranging from 62.6% in 2013 to 57.5% in 2019. Overall, 34.8% of our study population received a nonrecommended antibiotic without documentation of an antibiotic allergy or MRSA colonization (2013: 32.5%; 2018: 36.7%) (Fig. 2). Most patients in our study population received >1 antibiotic (92.3%) in the first 2 days of hospitalization. The most common antibiotics among patients receiving recommended treatment were ceftriaxone (74.2% of patients receiving recommended treatment), azithromycin (67.2%), and levofloxacin (31.8%) (Fig. 3a). The most common nonrecommended antibiotics were vancomycin (57.2% of patients receiving nonrecommended treatment), piperacillin-tazobactam (48.1%), and cefepime (25.7%) (Fig. 3b). From 2013 to 2020, cefepime charges consistently increased among CAP patients treated with nonrecommended antibiotics, whereas levofloxacin charges consistently decreased among CAP patients treated with only recommended antibiotics. Conclusions: Approximately one-third of patients with uncomplicated CAP received nonrecommended empiric antibiotics, and from 2013 to 2020 that proportion increased by 9%. Additional strategies are needed to help identify opportunities to optimize antibiotic selection among patients with CAP.
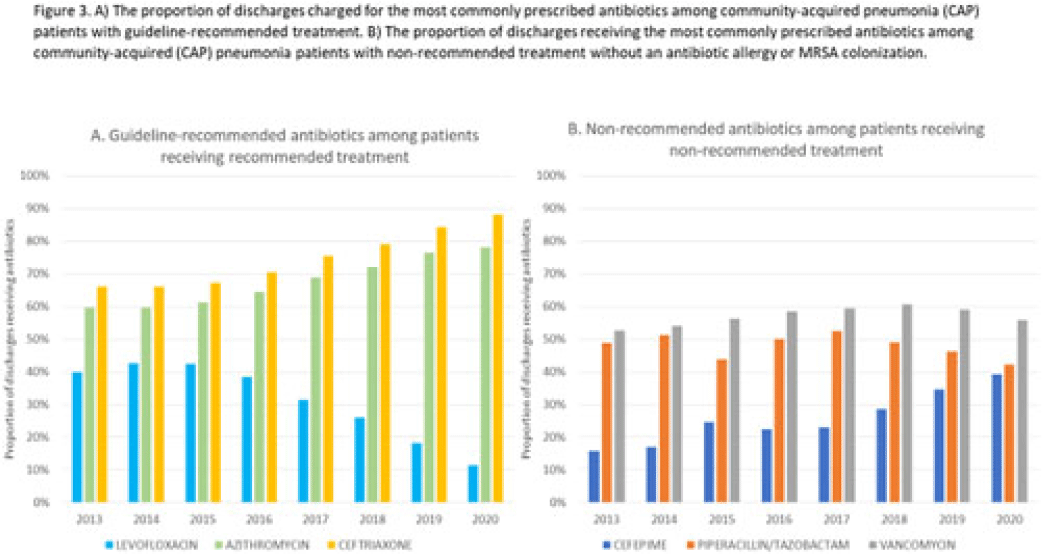
Disclosures: None
Implementing a health-system–wide antibiotic stewardship program in ambulatory surgery centers
- Kasey Hickman, Nicolas Forcade, Mandelin Cooper, Shivanne Bhagwandeen, Brandy Russell
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: In 2016, the CDC released the Core Elements of Outpatient Antibiotic Stewardship, which extended the requirements previously released for hospital facilities and nursing homes to the outpatient setting. Several regulatory agencies focused on outpatient antimicrobial use. However, The Joint Commission and the Ambulatory Surgery Center (ASC) Leapfrog Group excluded ambulatory surgery centers from their medication management standards and questions. Due to the public health and patient safety benefits of implementing an antimicrobial stewardship program (ASP) and increasing regulatory interest in the matter, the Hospital Corporation of America (HCA) Ambulatory Surgery Division formally launched a nationwide ASP for its ambulatory surgery centers in March 2021. Methods: HCA is a large healthcare system with 146 ASCs in 16 states in 2021. The structure of the ASCs are local surgery centers with a medical director, a nurse responsible for infection prevention, and a pharmacist at a regional level. The types of surgeries vary based on location and ASC site. In 2019, a multidisciplinary team formed the corporate planning committee. The program was modeled after the CDC Core Elements and The Joint Commission’s requirements for an ASP. Each ASC was asked to build a local ASP team, led by a local physician and a regionally based pharmacist. In addition, a stewardship goal was established to update all preoperative antibiotic surgical-site infection prophylaxis order sets. The corporate committee provided educational resources, including evidence-based guidelines for appropriate antibiotic selection for surgical-site infections. They collected antibiotic cost per case as a baseline metric to track and analyze. Pediatric, ophthalmic, and gastrointestinal endoscopic procedures were excluded from the program. Results: From January 1, 2020, through December 31, 2021, including only centers that were operational during this period and excluding single specialty endoscopy centers, antibiotic cost per case decreased annually from $2.38 to $1.84 (t = 4.157; P < .005), and the postoperative infection rate also declined from 0.370 to 0.304 (t = 2.079; P = .040). Conclusions: Our findings suggest that implementing a health-system–wide outpatient antibiotic stewardship program in the ambulatory surgery center setting is feasible and may contribute to decreased antibiotic cost per case and improved postoperative surgical site infection rates.
Disclosures: None
Prevalence of and risk factors for bacteremic UTIs in hospitalized adults without definitive signs or symptoms of UTI
- Sonali Advani, David Ratz, Jennifer Horowitz, Lindsay Petty, Kenneth Schmader, Tawny Czilok, Anurag Malani, Tejal Gandhi, Scott Flanders, Valerie Vaughn
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s27-s28
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: IDSA guidelines recommend withholding treatment in patients with asymptomatic bacteriuria in the absence of systemic signs of infection. However, some patients with bacteriuria may not be able to express symptoms either due to presence of indwelling catheter, underlying complicated urologic anatomy, dementia, or altered mental status (AMS). Clinicians frequently treat bacteriuria in this population with antimicrobial therapy due to concern for sepsis. To determine treatment need, we aimed to review prevalence and risk factors for bacteremic urinary tract infection (UTI) in a cohort of hospitalized inpatients without definitive signs and symptoms of a UTI. Methods: This retrospective cohort study of inpatients with a positive urine culture who presented without definitive signs or symptoms of a UTI was conducted between July 1, 2017, and June 30, 2022, in 68 academic and community hospitals (Michigan Hospital Medicine Safety Consortium). Signs and symptoms were obtained from medical record review 3 days before and after urine-culture collection. Bacteremic UTI was defined as any positive blood culture growing at least 1 organism matching the urine culture. Risk factors for bacteremic UTI were assessed using multivariable logistic regression models with results expressed as odds ratios (ORs) for dichotomous variables and relative risks (RRs) for continuous variables. Results: Of 11,793 patients meeting study criteria, 73.6% were female with a median age of 78.2 years. Overall, 41.8% had AMS, 33.8% had dementia, 15.6% had an indwelling urinary catheter, and 54.6% had complicated urologic history (eg, urologic surgery). Of these, 166 patients (1.4%) developed bacteremic UTI. On adjusted analysis, male sex, hypotension, heart rate >90, urinary retention, fatigue, log of serum leukocytosis [1 log increase in serum WBC = 2.718 × serum white blood cell count (WBC)], and pyuria with >25 WBC per high-powered field (WBC/hpf) on urinalysis were associated with bacteremic UTI (Table). Older age, presence of an indwelling catheter, complicated urologic history, functional decline, AMS, dementia, and change in urine were not associated with higher odds for bacteremic UTI (Table). Of patients with AMS and no definitive signs or symptoms of a UTI, only 89 (1.8%) of 4,932 developed a bacteremic UTI. Conclusions: Bacteremic UTI is relatively rare in hospitalized inpatients presenting with bacteriuria without symptoms of UTI. Predictors of bacteremic UTI included male sex, hypotension, tachycardia, urinary retention, fatigue, serum leukocytosis, and higher levels of pyuria (>25 WBC/hpf) on urinalysis. Our findings provide stewards a framework to risk stratify inpatients of older age who present with positive urine cultures but without (or are unable to express) signs or symptoms of UTI.
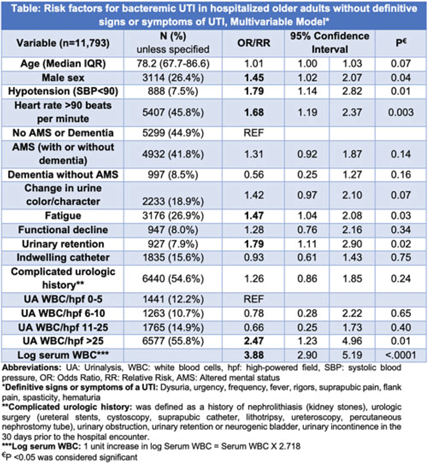
Disclosures: None
Antibiotic use among SARI patients according to the AWaRe classification before and during the COVID-19 pandemic in Bangladesh
- Md Ariful Islam, Md. Zakiul Hassan, Mohammad Abdul Aleem, Zubair Akhtar, Tanzir Ahmed Shuvo, Md Kaousar Ahmmed, Syeda Mah-E-Muneer, Md Abdullah Al Jubayer Biswas, Ayesha Afrin, Probir Kumar Ghosh, Fahmida Chowdhury
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s28
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Irrational antibiotic use among hospitalized patients can lead to antibiotic resistance. For rational use, the WHO introduced the Access, Watch, and Reserve (AWaRe) classification of antibiotics. We explored antibiotic use according to the AWaRe classification among patients hospitalized with severe acute respiratory infection (SARI) between the prepandemic and COVID-19 pandemic periods in Bangladesh. Methods: From June 2017 to November 2022, we analyzed SARI inpatient data from the hospital-based influenza surveillance platform at 9 tertiary-level hospitals in Bangladesh. We defined June 2017–February 2020 as the prepandemic period and March 2020–November 2022 as the pandemic period. Physicians identified inpatients meeting the WHO SARI case definition and recorded patient demographics, clinical characteristics, and antibiotics received during hospitalization. We used descriptive statistics to summarize the data. Results: We enrolled 20,640 SARI patients (median age, 20 years; IQR, 1.6–50; 63% male); and among them, 18,197 (88%) received antibiotics (26% of those received >1 different course of antibiotics). Compared to the prepandemic period, the proportion of antibiotic use among SARI patients was higher during the pandemic: 93% (9,887 of 10,655) versus 83% (8,310 of 9,985) (P < .001). According to AWaRe classification, Access, Watch, and Reserve groups accounted for 32% (n = 2,623), 86% (n = 7,158), and 0.05% (n = 4), respectively, before the pandemic and 32% (n = 3,194), 90% (n = 8,850), and 0.08% (n = 8), respectively, during the pandemic (Fig.). The most common antibiotic prescribed for children aged <5 years during the prepandemic was ceftriaxone (n = 1,940, 74%), followed by amikacin (n = 325, 13%) and flucloxacillin (n = 300, 12%); similarly, during the pandemic, most common antibiotic prescribed was ceftriaxone (n = 3,097, 79%), followed by amikacin (n = 723, 18%) and flucloxacillin (n = 348, 9%). The most common antibiotic prescribed for patients aged ≥5 years during the prepandemic period was ceftriaxone (n = 3,174, 54%), followed by amoxicillin-clavulanic acid (n = 1,304, 22%) and azithromycin (n = 1,038, 18%). During the pandemic, the most common antibiotic prescribed for patients aged ≥5 years was ceftriaxone (n = 3,793, 64%), followed by amoxicillin-clavulanic acid (n = 1,327, 22%) and clarithromycin (n = 797, 13%). Among children aged <5 years, use of the Watch group of antibiotics during the prepandemic and pandemic periods was similar: 94% (n = 3,688) versus 95% (n = 2,347) (P = .099). However, among patients aged ≥5 years, the use of Watch antibiotics was higher during the pandemic compared to the prepandemic period: 87% (n = 5,163) versus 82% (n = 4,811) (P < .001). Conclusions: Use of antibiotics in the Watch group was predominant among SARI patients both before and during the COVID-19 pandemic, and it increased among SARI patients aged ≥5 years during the pandemic period in Bangladesh. Promoting antibiotic stewardship programs for physicians, including in-service training on antibiotic use, could reduce irrational antibiotic use, which might contribute to mitigating antibiotic resistance in the country.
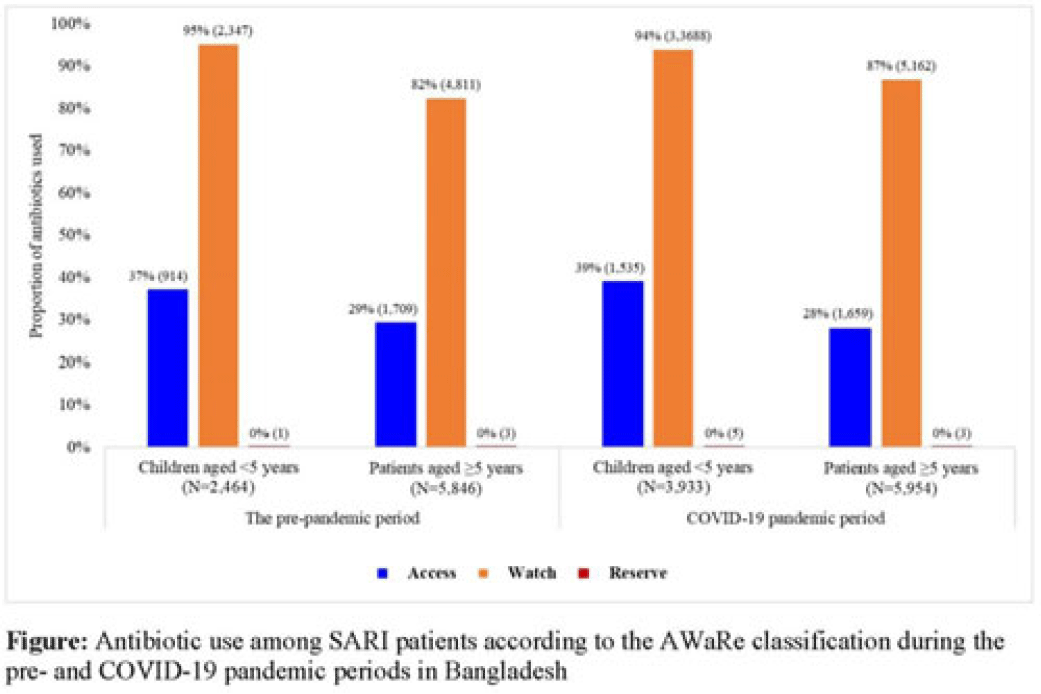
Financial support: This study was funded by the CDC.
Disclosures: None
Electronic health record–based identification of inpatients receiving antibiotic treatment for community-acquired pneumonia
- David Yang, Leigh Cressman, Keith Hamilton, Lauren Dutcher
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s28-s29
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Inappropriate antibiotic use for community-acquired pneumonia (CAP) is common. Although antibiotic stewardship activities require real-time, accurate identification of patients being treated for CAP, there are few reliable methods to identify such patients using the electronic health record (EHR). We conducted a retrospective study to assess the performance of provider-selected antibiotic indication in identifying patients being treated for CAP among a cohort of hospitalized adults. Methods: We randomly selected 440 patients from a cohort of patients who received at least 1 systemic antibiotic within 48 hours of admission between January 1, 2019, and December 31, 2021, at 3 acute-care hospitals. The reference standard for treatment of CAP was defined as intention to treat for pneumonia by inpatient provider(s) within 48 hours of admission, as assessed by chart review of provider notes. Treatment for pneumonia using any terminology except with “hospital-acquired pneumonia” (HAP) or “ventilator-associated pneumonia” (VAP) were counted. Provider-selected indication of CAP (in an antibiotic order) was compared against this reference standard; sensitivity, specificity, and positive and negative predictive values were calculated. Performance characteristics of International Classification of Disease, Tenth Revision (ICD-10) codes for pneumonia in identifying CAP patients were assessed against the same reference standard. A secondary analysis including terms HAP and VAP in the reference standard was performed. Results: Provider-selected antibiotic indication for CAP had a sensitivity of 64.4%, specificity of 96.3%, positive predictive value (PPV) of 73.1%, and negative predictive value (NPV) of 96.1%, giving comparable performance to ICD-10 codes (Table 1). Of those with 21 false-negative results, 13 (61.9%) had a healthcare-associated lower respiratory tract infection and 14 (66.7%) had sepsis indicated in at least 1 antibiotic order. Conclusions: Provider-selected antibiotic indication showed moderate sensitivity and high specificity for identifying CAP-treated cases. Importantly, use of this method can be deployed for real-time antibiotic stewardship interventions for CAP.
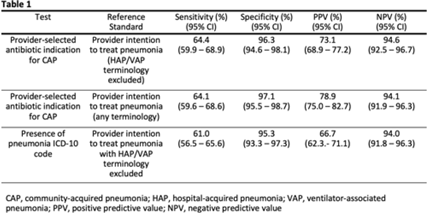
Disclosures: None
Sources of antibiotics for acute respiratory infection in children aged <5 years children in South Asia: A multicountry study
- Md Abdullah Al Jubayer Biswas, Mohammad Riashad Monjur, South Eastern Sydney Local Health District, Md. Zakiul Hassan, Nusrat Homaira
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s29
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: In South Asia, a region of almost 2 billion people across 8 countries, acute respiratory infections (ARIs) are associated with significant morbidity and mortality in children aged <5 years. Although ~80% of ARIs are due to viral etiology and are often self-limiting, they remain the single largest reason for antibiotic use in children aged <5 years in South Asia. We investigated the sources and dispensing pattern of antibiotics for ARIs in children aged <5 years in South Asia. Methods: We analyzed nationally representative, population-based, publicly available household survey data from 6 South Asian countries’ Demographic and Health Surveys (DHS): Afghanistan, Bangladesh, India, Maldives, Nepal, and Pakistan. The outcome of interest was the source of antibiotics for children aged <5 years who reportedly had symptoms compatible with ARI (cough, fever, and runny nose) and had received antibiotics for the ARI episode in the 2 weeks preceding the survey. We used a generalized estimating equation with an exchangeable correlation structure to account for country-specific cluster-level correlation to estimate the odds of sources of antibiotics usage. Models were adjusted for age, sex, type of place of residence, wealth index, and parents’ education. To analyze the data, we used the sample weight supplied by the DHS to ensure that our results appropriately reflect the target population in each of the countries studied. Results: In total, across the 6 South Asian countries, 24,104 children aged <5 years had symptoms of ARI, 7,587 (31%; 95% CI, 30–33) from received antibiotics. A higher proportion of antibiotic usage for ARIs episodes occurred in Afghanistan (66%), followed by Maldives (53%), Pakistan (45%), and Nepal (43%). Regarding the source of antibiotics, a higher proportion of antibiotics was obtained from the private medical sector in India, followed by unqualified sources in Bangladesh, and the public sector in Afghanistan. Our adjusted multivariable analysis revealed that, in comparison to the public sector, participants were 2.6 times (aOR, 2.6; 95% CI, 1.6–4.3) more likely to receive antibiotics from private medical sector drug sources in Nepal and 1.3 times more likely (aOR, 1.3; 95% CI, 1.1–1.5) in Afghanistan. Conclusions: In South Asian countries, the private medical sector was the most common primary source of antibiotics for children with ARIs. Targeted efforts to create awareness around antibiotic dispensing and guidelines to improve practices may curtail the use of antibiotics for ARIs in children aged <5 years in South Asia.
Disclosures: None
Fluconazole resistance in non-albicans Candida species in the United States, 2012-2021
- Emily Jenkins, Meghan Lyman, Brendan Jackson, Shawn Lockhart, Hannah Wolford, Sujan Reddy, James Baggs
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s29-s30
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Candida spp can cause a variety of infections known as candidiasis, ranging from severe invasive infections to superficial mucosal infections of the mouth and vagina. Fluconazole, a triazole antifungal, is commonly prescribed to treat candidiasis but increasing fluconazole resistance is a growing concern for several Candida spp. Although C. albicans has historically been the most common cause of candidiasis, other species are increasingly common and antifungal resistance is more prevalent in these non-albicans species, including C. glabrata, C. parapsilosis, and C. tropicalis, which were the focus of this analysis. Methods: We used the PINC AI healthcare data (PHD) database to examine fluconazole resistance for inpatient isolates between 2012 and 2021 from 187 US acute-care hospitals with at least 1 Candida spp culture with a fluconazole susceptibility result over the entire period. We calculated annual percentage fluconazole resistance for C. glabrata, C. tropicalis, and C. parapsilosis isolates using the clinical laboratory interpretation for resistance. Results: We identified 4,264 C. glabrata, 2,482 C. parapsilosis, and 2,283 C. tropicalis isolates between 2012 and 2021 with susceptibility results. The percentage of C. glabrata isolates resistant to fluconazole doubled between 2020 and 2021 (14.6% vs 29.3%) (Fig. 1a). The percentage of C. parapsilosis isolates resistant to fluconazole steadily increased since 2017 (Fig. 1b), with an 82% increase in 2021 compared with 2020 (3.8% in 2020 vs 6.9% in 2021). Fluconazole resistance among C. tropicalis isolates varied over the years, with a 0.3% decrease in 2021 from 2020 (Fig. 1c). Of hospitals reporting at least 1 result each year 2020–2021, 44% observed an increase in the proportion of C. glabrata isolates resistant to fluconazole in 2021 compared to 2020. Conclusions: Our analysis highlights a concerning increase in fluconazole resistance among C. glabrata and C. parapsilosis isolates in 2021 compared with previous years. Further investigation of the observed increases in fluconazole resistance among these Candida spp could provide further insight on potential drivers of resistance or limitations in reported results from large databases. More analyses are needed to understand rates, sites of Candida infections, and risk factors (eg, antifungal exposure) associated with resistance.
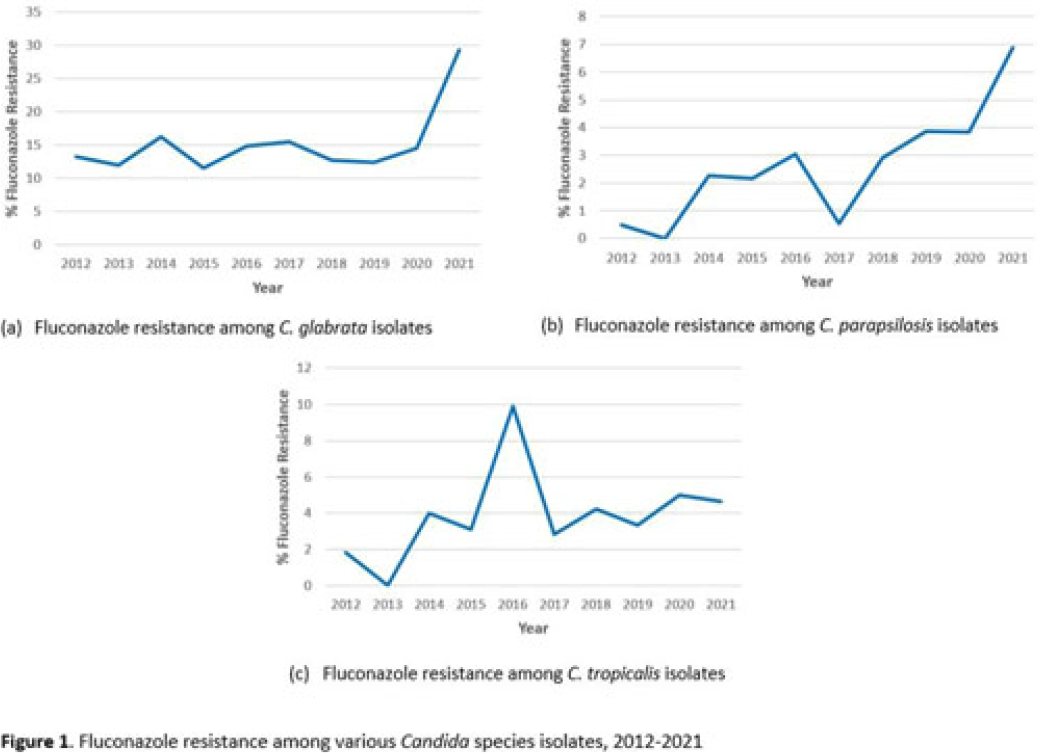
Disclosures: None
Development of a multiyear pediatric antibiogram in Georgia identifies antibiotic resistance trends
- Matt Linam, Madeleine Goldstein, Robert Jerris, Mark Gonzalez
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s30
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antibiograms are used to monitor antibiotic resistance trends and help guide empiric antibiotic treatment. Community pediatricians may not have access to or be comfortable using children’s hospital antibiograms. Creating and disseminating a statewide pediatric antibiogram can help inform antibiotic stewardship efforts.
Objective: To develop a pediatric-specific antibiogram for the state of Georgia. Methods: Annual pediatric antibiograms for the 5 children’s hospitals in Georgia from 2014 through 2021 were collected. All sites complied with the Clinical and Laboratory Standards Institute guidelines for antibiomicrobic breakpoints and antibiogram development. Antibiogram data were combined, and the most common bacteria were selected to incorporate into the statewide antibiogram: Staphylococcus aureus, Streptococcus pneumoniae, Enterococcus faecalis, Escherichia coli, Klebsiella pneumoniae, Enterobacter cloacae complex, and Pseudomonas aeruginosa. Antibiogram data were reported as percentage susceptible and total number of isolates. Interhospital susceptibility differences were compared for methicillin-susceptible S. aureus (MSSA), methicillin-resistant S. aureus (MRSA), E. coli, and K. pneumoniae from 2018 through 2021. P < .05 was considered significant. The combined antibiogram data from 2014 through 2021 were used to show antibiotic susceptibility trends over time. Results: The 2021 antibiogram is shown in the Table. For MSSA and MRSA, clindamycin susceptibility was 80% and 85%, respectively. K. pneumoniae susceptibility to amoxicillin-clavulanate was 91%. For E. coli, using urine-specific breakpoints, susceptibility to cefazolin was 89%. A few statistically significant differences in antibiotic susceptibility were detected between hospitals, but most were unlikely to be clinically relevant (all susceptibilities ≥90% or < 80%). A notable exception was trimethoprim-sulfamethoxazole susceptibility for K. pneumoniae, which ranged from 74% to 98% in 2020 and from 74% to 86% in 2021. From 2014 to 2021, the percentage of MRSA decreased from 49% to 34%. Over the 8 years, susceptibility to ceftriaxone for E. coli ranged from 93% to 95% and from 90% to 95% for K. pneumoniae. Susceptibility to meropenem for E. coli and K. pneumoniae ranged from 99% to 100%. Conclusions: Antibiotic susceptibility for pediatric bacterial isolates in Georgia remained stable over time and supported the narrow-spectrum empiric antibiotic treatment recommended in national evidence-based guidelines for skin and soft-tissue infections, community-acquired pneumonia, and uncomplicated urinary tract infections. MRSA rates decreased over time and multidrug-resistant gram-negative bacilli were uncommon and remained stable.
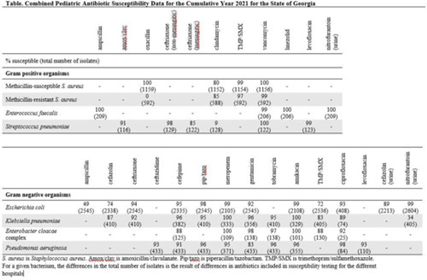
Disclosures: None
Initial blood culture collection practices and the associated factors upon continued empiric piperacillin-tazobactam usage
- Satoshi Kitaura, Koh Okamoto, Ryo Yamaguchi, Takehito Yamamoto, Toshiyuki Kishida, Daisuke Inoue, Hirotaka Miyashita, Masayuki Ueda, Hideki Hashimoto, Sohei Harada, Shu Okugawa, Takeya Tsutsumi, Kyoji Moriya
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s30-s31
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Approaches to the prescription behavior of broad-spectrum antibiotics, including preauthorization and prospective audit and feedback (PAF), are a focus of antimicrobial stewardship (ASP). However, preprescription behavior, such as blood-culture collection before empiric prescription, is understudied and merits more attention given its influence on the usage of broad-spectrum antibiotics. At the University of Tokyo Hospital, carbapenems are subject to PAF, which has resulted in a compensatory increase in piperacillin-tazobactam use. To evaluate the inherent preprescription behavior associated with a broad-spectrum antibiotic, we investigated the initial blood-culture collection practices upon hospitalization in patients who were continued on empiric piperacillin-tazobactam. Methods: A retrospective observational study was conducted at the University of Tokyo Hospital, a tertiary-care hospital in Tokyo, Japan. Patients who were administered piperacillin-tazobactam on the day of hospitalization between April 2016 and December 2017 were included. Patients aged <=18 years and/or patients who discontinued piperacillin-tazobactam within two days were excluded. Only 1 admission per patient was kept for analysis. The medical records of 250 randomly selected patients were reviewed to obtain data on demographics, blood-culture collection, severity, specialties, and risk factors for multidrug-resistant organisms. A multivariable logistic regression analysis was used to identify factors associated with blood-culture collection. Results: In total, 960 discrete patients fulfilled the study criteria. Of the randomly selected 250 patients, blood cultures were collected from 162 patients (64.8%), and microbial growth was observed in 30 cases (18.5%). Enterobacterales and anaerobes accounted for 73.3% of the microbial population. Gastroenterologists (94, 37.6%) and general surgeons (52, 20.8%) were the most common prescribers. Hepatobiliary (83, 33.2%), respiratory (58, 23.2%), and intra-abdominal infections (IAI; 34, 13.6%) were the major suspected diagnoses. Blood-culture collection was associated with the use of immunosuppressive agents (OR, 3.48; 95% CI, 1.49–8.99), intrabdominal infection (OR, 0.28; 95% CI, 0.12–0.67), systemic inflammatory response syndrome criteria ≥ 2 (OR, 4.50; 95% CI, 2.25–9.42), and surgical specialty (OR, 0.33; 95% CI, 0.18–0.60). Conclusions: More than one-third of patients requiring hospitalization and empiric piperacillin-tazobactam did not undergo blood-culture collection. The finding that blood cultures were less likely to be obtained in patients with suspected IAI requiring hospitalization and by surgical specialties raises a concern regarding suboptimal evaluation. Further assessment of the appropriateness of blood-culture collection in the setting of broad-spectrum antibiotic prescription and tailored promotion of blood-culture collection to surgical specialties may be warranted.
Disclosures: S.K.: The author (during graduate school (PhD) was involved in antiviral research relevant to a neglected tropical disease and favipiravir. During this graduate school research, favipiravir was provided by FUJIFILM Toyama Chemical Co. Ltd
Risk Factors and outcomes associated with inappropriate empiric broad-spectrum antibiotic use in hospitalized patients with community-acquired pneumonia
- Tejal Gandhi, Lindsay Petty, Valerie Vaughn, Anurag Malani, David Ratz, Tawny Czilok, Jennifer Horowitz, Elizabeth McLaughlin, Lisa Dumkow, Stephanie Burdick, Danielle Osterholzer, Mariam Younas, Steven Bernstein, Scott Flanders
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s31-s32
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Inappropriate broad-spectrum antibiotic use targeting methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa can result in increased adverse events, antibiotic resistance, and Clostridioides difficile infection. In 2019, revised ATS/IDSA community-acquired pneumonia (CAP) guidelines removed healthcare-associated pneumonia (HCAP) as a clinical entity and modified patient factors warranting empiric broad-spectrum antibiotic (BSA) use. As a result, most patients hospitalized with CAP should receive empiric antibiotics targeting standard CAP pathogens. Based on revised guidelines, we evaluated predictors and outcomes associated with inappropriate BSA use among hospitalized patients with CAP. Methods: Between November 2019 and July 2022, trained abstractors collected data on non-ICU adult medical patients admitted with CAP at 67 Michigan hospitals who received either an inappropriate empiric BSA on hospital day 1 or 2 or a standard CAP regimen. Inappropriate empiric BSA use was defined as use of an anti-MRSA or anti-pseudomonal antibiotic in a patient eligible for standard CAP coverage per IDSA guidelines. Patients with immune compromise, moderate or severe chronic obstructive pulmonary disease (COPD), pulmonary complication, or guideline-concordant treatment with BSA were excluded. Data collected included comorbidities, antibiotic use and hospitalizations in the preceding 90 days, cultures in the preceding year, signs or symptoms of pneumonia, hospital characteristics, and 30-day postdischarge patient outcomes. Data were collected through chart review and patient phone calls. Predictors of inappropriate empiric BSA were evaluated using logistic general estimating equation (GEE) models, accounting for hospital-level clustering. We assessed the effect of inappropriate empiric BSA (vs standard CAP therapy) on 30-day patient outcomes using logistic GEE models controlling for predictors associated with the outcome and probability of treatment. Results: Of 8,286 included patients with CAP, 2,215 (26.7%) were empirically treated with inappropriate BSA. The median BSA treatment was 3 days (IQR, 2.5). After adjustments, factors associated with inappropriate empiric BSA treatment included hospitalization or treatment with high-risk antibiotics in preceding 90 days, transfer from a postacute care facility, hemodialysis, support with ≥3 L supplemental oxygen, severe sepsis, leukocytosis, and higher pneumonia severity index (Fig. 1). After adjustments, patients with inappropriate empiric BSA treatment had higher readmissions 30 days after discharge, more transfers to the intensive care unit, more antibiotic-associated adverse events, and longer hospitalizations (Fig. 2). Conclusions: Patients hospitalized with CAP often received inappropriate BSA as empiric coverage, and this inappropriate antibiotic selection was associated with worse patient outcomes. To improve patient outcomes, stewardship efforts should focus on reducing inappropriate BSA use in patients hospitalized for CAP with historic HCAP risk factors or severe CAP without other guideline-directed indications for BSA.
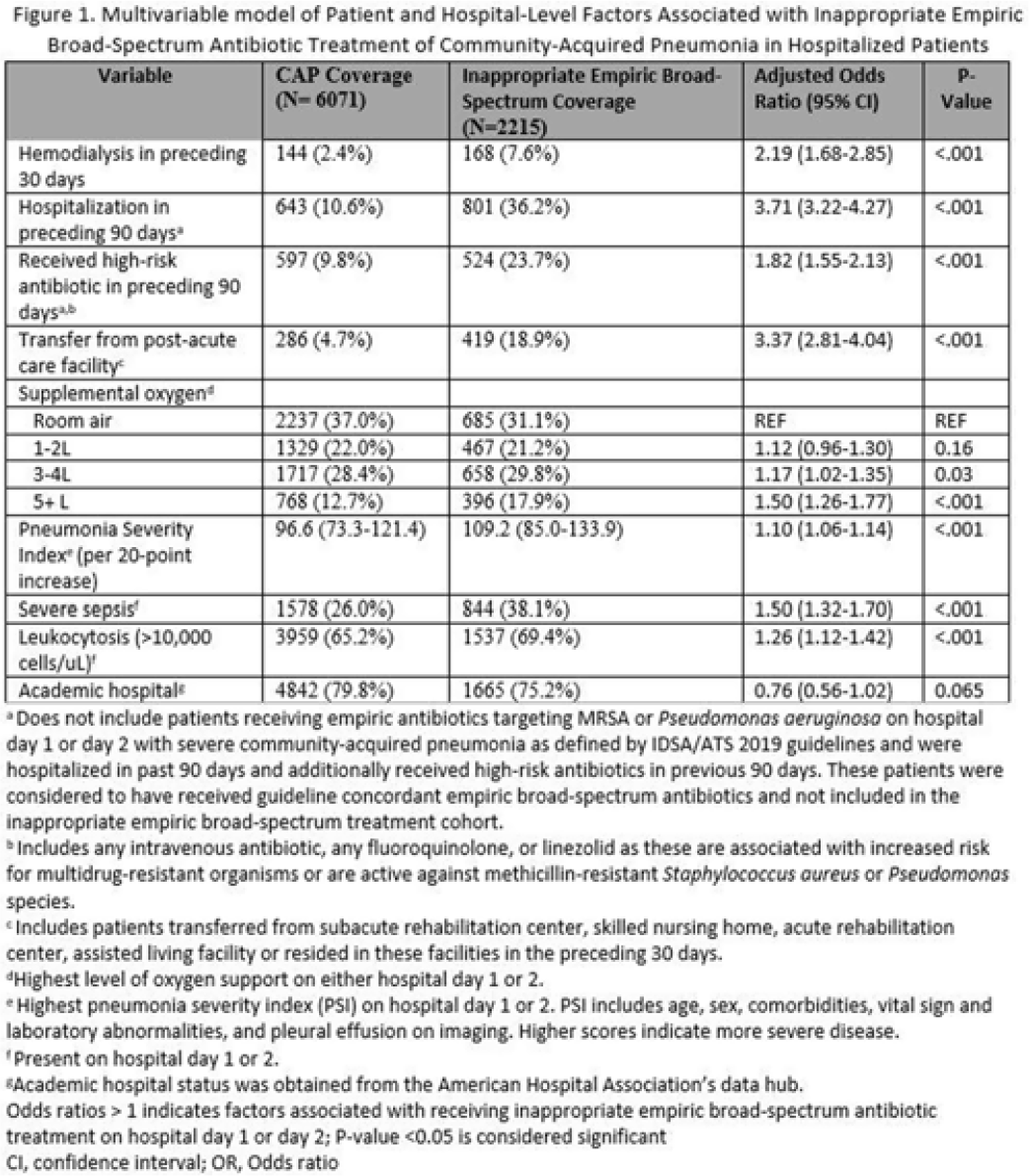
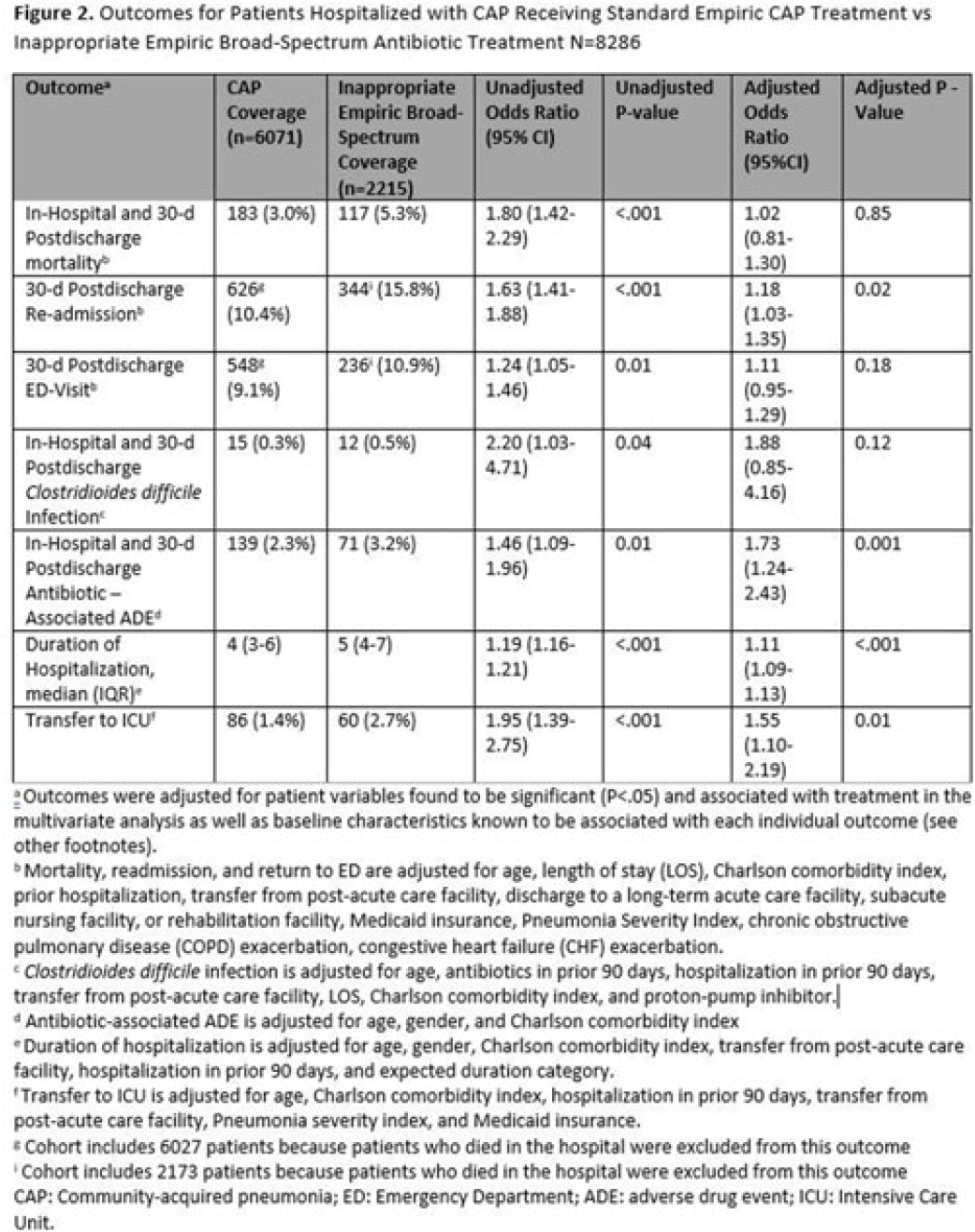
Financial support. H.M.S. initiative is underwritten by Blue Cross and Blue Shield of Michigan.
Disclosures: None
Effect of antifungal stewardship on micafungin prescribing practices in intensive care units at a tertiary-care hospital
- Radhika Arya, Sarah Norman, Farah Daas, Sheena Ramdeen
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s32
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Fungal diseases are associated with substantial global mortality and economic burden, especially in critically ill or immunocompromised patients. Antifungal resistance has emerged as a barrier to treating invasive fungal infections, but antifungal stewardship is still a developing effort due to limited data. Here, we describe the antifungal prescribing practices and the impact of antifungal stewardship on micafungin days of therapy (DOTs) in critical care units. Methods: This retrospective study included patients who were admitted to the intensive care unit (ICU) at a tertiary-care hospital in Washington, DC. The preintervention group included baseline micafungin use data between January 1, 2021, and May 31, 2021. The postintervention group included prospective audits, feedback on micafungin orders by a clinical pharmacist, and education on the appropriateness of the antifungal agents. The postintervention group included patients admitted between June 1, 2021, and December 31, 2021. Approval was obtained from the institutional review board. Results: The overall average of micafungin days of therapy (DOT) per 1,000 patient days present in the preintervention group versus the postintervention group was 33 versus 24 days, respectively. Moreover, 121 patients were randomly selected for a more detailed retrospective review to define micafungin prescribing practices further. Of these, 73 patients (60.3%) were male; the median age was 63 years. The most common cause for prescribing micafungin in both groups was empiric antifungal coverage (62.8%), followed by fungemia (12.4%). The most common organism isolated was Candida albicans. For other sources of infection and organisms isolated, refer to Table 1. In-hospital mortality occurred in 63 (52.06%) patients in both groups. Conclusions: Antifungal stewardship through prospective audit and feedback and education by clinical pharmacists decreased micafungin DOTs in critical care units. Empiric prescribing of micafungin is highly prevalent in the ICU despite the low incidence of invasive fungal infections. Although periodic drug utilization reviews and pharmaceutical surveillance can help reduce the prolonged duration of micafungin therapy in the ICU, more robust and routine antifungal stewardship is key to the appropriate use of micafungin to avoid the emergence of antifungal resistance.
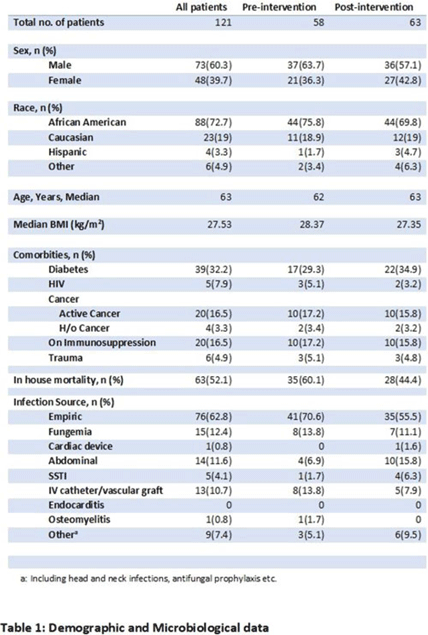
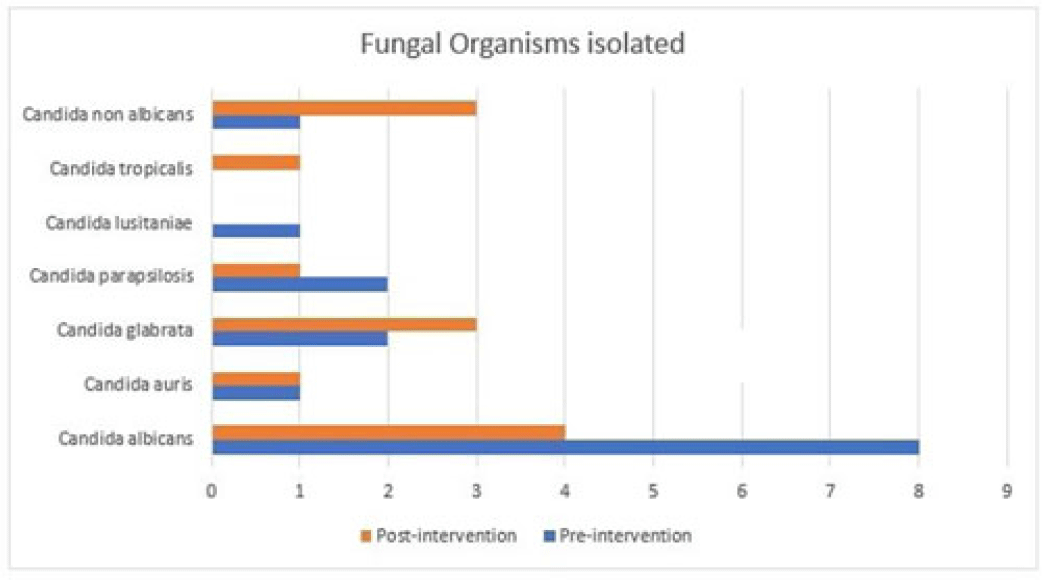
Disclosures: None
Serotonergic agents and linezolid: Impact of exposure to more than one agent concomitantly on risk of adverse effects
- Xuping Yan, Christopher McCoy, Ryan Chapin, Matthew Lee, Howard Gold, Kendall Donohoe
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s32-s33
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The off-target effects linezolid have the potential to cause serotonin syndrome when given in conjunction with serotonergic agents. Despite package insert labeling as a contraindication, several postmarketing studies have demonstrated a low incidence of serotonin syndrome with the concomitant use of linezolid and other serotonergic agents. Linezolid provides a convenient oral option for gram-positive infections. However, due to concerns for serotonin syndrome, the use of linezolid is sometimes avoided. Methods: We performed a single-center, retrospective, medical record review of all adult inpatients from September 2021 to September 2022. Patients included had 1 administration of linezolid and 1 inpatient administration of a selective serotonin reuptake inhibitor (SSRI) or serotonin and norepinephrine reuptake inhibitor (SNRI) within 14 days. The primary outcome was the incidence of serotonin syndrome as defined by the Hunter serotonin toxicity criteria, which were retrospectively applied to each patient based on medical-record documentation. We compared patients receiving 1 versus multiple serotonergic agents. Secondary outcomes included duration of hospitalization and adverse outcomes based on concerns for serotonin syndrome such as need for rescue, ICU admission, or change in medication. Results: Of the 50 included patients from a convenience sample, 27 (54%) were on linezolid and >1 serotonergic agent. Patients had similar baseline characteristics (Table 1). The most common concomitant agent used was an SSRI. Other agents that predispose patients to serotonin syndrome included opioid analgesics and other classes of antidepressants (Fig. 1). Serotonin syndrome occurred within 48 hours in 1 patient on an SNRI and a continuous fentanyl drip. There was no need for rescue or ICU admission due to serotonin syndrome. No patients were readmitted due to serotonin syndrome, and no differences were observed in hospital lengths of stay. Conclusions: Exposure to a single serotonergic agent combined with receipt of linezolid was not associated with any cases of serotonin syndrome. Exposure to multiple serotonergic agents was not associated with a high incidence of serotonin syndrome. This small series supports previous reports demonstrating relative safety of linezolid given with serotonergic agents and encourages review of interruptive drug–drug interaction alerts for linezolid within the electronic ordering system.
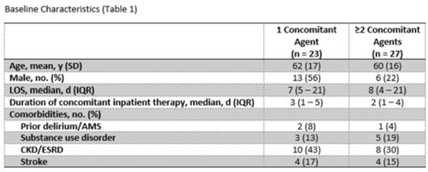

Disclosures: None
Assessing inpatient antibiotic use during COVID-19 surges with or without infectious diseases consultation
- Nicole Tommasi, Shira Doron, Gabriela Andujar-Vazquez, Maureen Campion
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s33-s34
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Throughout the COVID-19 pandemic, increased inappropriate antibiotic use (AU) drove concern for antimicrobial resistance. Antimicrobial stewardship efforts are critical for combatting antimicrobial resistance. Our objective was to compare AU between SARS-CoV-2 delta and omicron variant surge periods in COVID-19 patients hospitalized at Tufts Medical Center (TMC) in Boston. Infectious diseases consultation (IDC) was mandatory for patients diagnosed with COVID-19 throughout the SARS-CoV-2 delta variant surge. During the SARS-CoV-2 omicron variant surge, IDC was optional for certain patient populations. Instead, the antibiotic stewardship program (ASP) reviewed these patients for appropriate medical management. We hypothesized that AU would increase during the SARS-CoV-2 omicron variant surge compared to the delta variant surge due to optional IDC because IDC would reduce inappropriate AU for suspected viral pneumonia. Methods: Retrospective medical record review of patients hospitalized with COVID-19 during the SARS-CoV-2 delta and omicron variant surges was conducted. We collected data regarding vital signs, white blood cell count (WBC), length of stay (LOS), steroid use, IDC, and AU (defined as percentage of patients receiving at least 1 antibiotic dose), with a separate category for antibiotics commonly used for bacterial pneumonia (ampicillin-sulbactam, azithromycin, cefepime, cefpodoxime, ceftazidime, ceftriaxone, doxycycline, piperacillin-tazobactam, vancomycin). We determined that 71 patients from each group were needed to detect an absolute difference of 20% in AU between surges with 75% power, based on the CDC estimate that 80% of patients hospitalized with COVID-19 receive an antibiotic. Unpaired t tests and χ2 analyses were conducted on demographic data. Inferential statistics assessed for differences between the 2 SARS-CoV-2 variant surges in AU and days of therapy (DOT), supplemental oxygen (SaO2), steroid use, and IDC utilizing a Wilcoxon rank-sum test and logistic regression analyses. Results: Results showed no significant differences in AU between surges (38.0% during the SARS-CoV-2 delta variant surge vs 42.3% during the SARS-CoV-2 omicron variant surge; P = .131). Disease severity was not different between surges as measured by steroid use, initial WBC, and SaO2. WBC was a predictor for AU in both surges (delta surge, P = 0.007; omicron surge, P = .002). Average LOS was higher throughout the SARS-CoV-2 delta variant surge for all patients (11.58 days during the delta surge, vs 5.97 days during the omicron variant surge; P = .047) and those who received antibiotics (18.44 days during the delta variant surge vs 6.70 days dring the omicron variant surge; P = .210). Total DOT was significantly longer during the SARS-CoV-2 delta variant surge for all antibiotics (463 DOT during the delta variant surge vs 277 DOT during the omicron variant surge; P = .047) and antibiotics commonly used for bacterial pneumonia (315 DOT during the delta variant surge vs 202 DOT during the omicron variant surge; P = .021). Conclusions: Making IDC optional for certain patient populations diagnosed with COVID-19 did not affect AU in a large, urban academic medical center with a comprehensive ASP.
Disclosures: None
Creating an electronic antibiogram using visualization software: Easily updatable and removes the need for yearly manual review
- Ashley Dauphin, Christopher McCoy, Robert Bowden, Matthew Lee, Howard Gold, Ryan Chapin
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s34
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Previously, our hospital manually built a static antibiogram from a surveillance system (VigiLanz) culture report. In 2019, a collaboration between the antimicrobial stewardship team (AST) and the infection control (IC) team set out to leverage data automation to create a dynamic antibiogram. The goal for the antibiogram was the ability to easily distribute and update for hospital staff, with the added ability to perform advanced tracking and surveillance of organism and drug susceptibilities for AST and IC. By having a readily available, accurate, and Clinical and Laboratory Standards Institute (CLSI)–compliant antibiogram, clinicians have the best available data on which to base their empiric antibiotic decisions. Methods: First, assessment of required access to hospital databases and selection of a visualization software (MS Power BI) was performed. Connecting SQL database feeds to Power BI enabled creation of a data model using DAX and M code to comply with the CLSI, generating the first isolate per patient per year. Once a visual antibiogram was created, it was validated against compiled antibiograms using data from the microbiology laboratory middleware (bioMerieux, Observa Integrated Data Management Software). This validation process uncovered some discrepancies between the 2 reference reports due to cascade reporting of susceptibilities. The Observa-derived data were used as the source of truth. The antibiogram prototype was presented to AST/IC members, microbiology laboratory leadership, and other stakeholders to assess functionality. Results: Following feedback and revisions by stakeholders, the new antibiogram was published on a hospital-wide digital platform (Fig. 1). Clinicians may view the antibiogram at any time on desktops from a firewall (or password)–protected intranet. The antibiogram view defaults to the current calendar year and users may interact with the antibiogram rows and columns without disrupting the integrity of the background databases or codes. Each year, simple refreshing of the Power BI antibiogram and changing of the calendar year allows us to easily and accurately update the antibiogram on the hospital-wide digital platform. Conclusions: This interdisciplinary collaboration resulted in a new dynamic, CLSI-compliant antibiogram with improved usability, increased visibility, and straightforward updating. In the future, a mobile version of the antibiogram may further enhance accessibility, bring more useful information to providers, and optimize AST/IC guidelines and education.
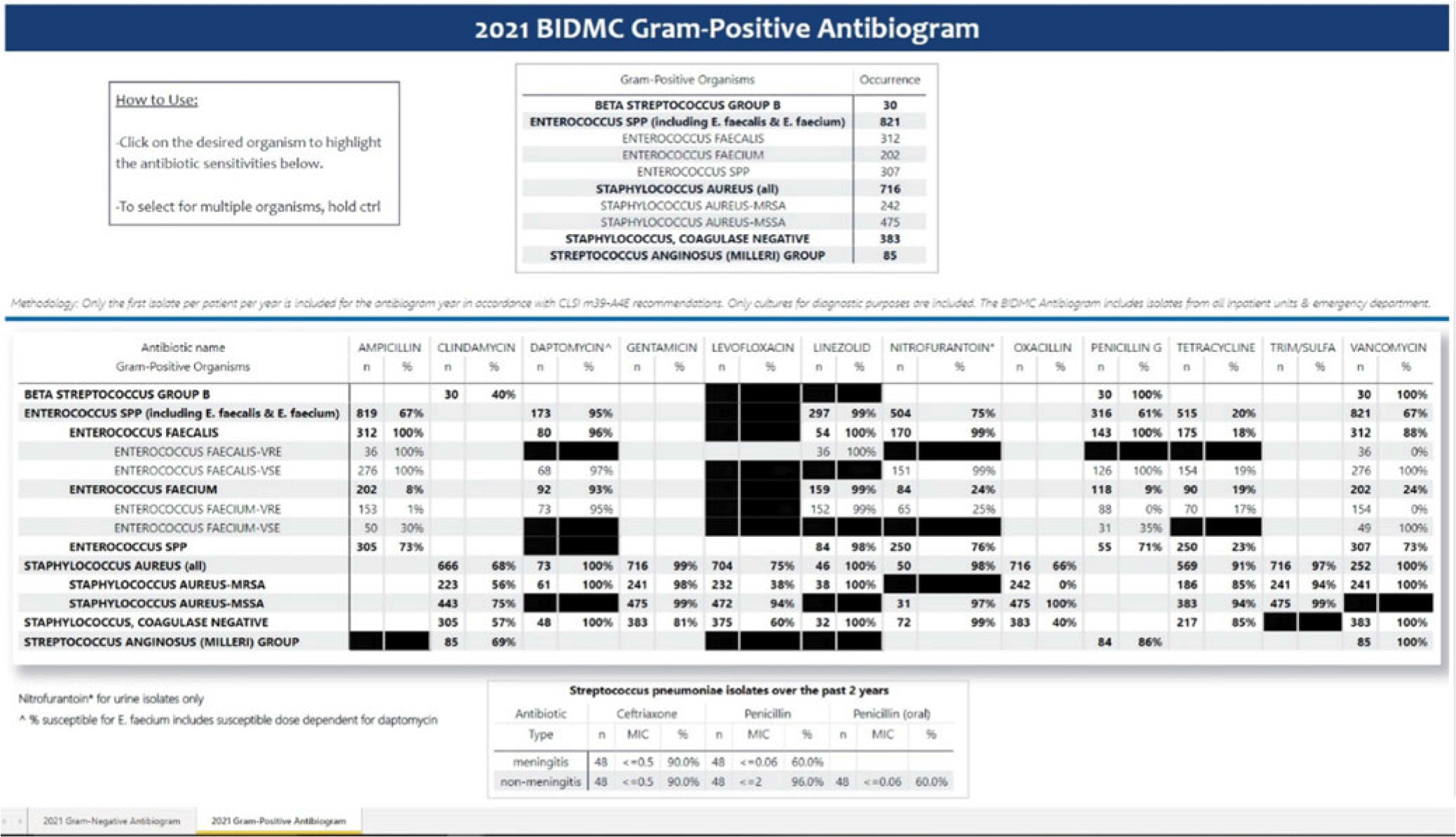
Disclosures: None
Identifying the relationship between hospital rurality and antibiotic overuse
- Hannah Hardin, Valerie Vaughn, Andrea White, Jennifer Horowitz, Elizabeth McLaughlin, Julia Szymczak, Lindsay Petty, Anurag Malani, Scott Flanders, Tejal Gandhi
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s34-s35
-
- Article
-
- You have access Access
- Open access
- Export citation
-
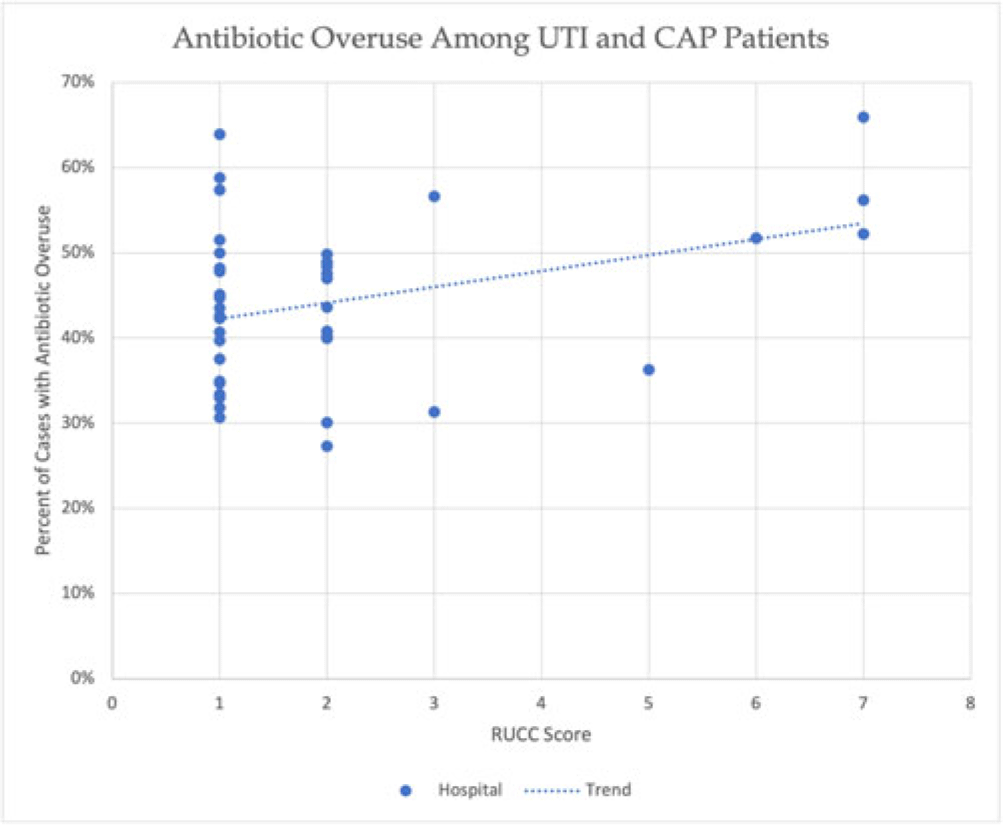
Background: Antibiotic overuse and the resulting patient outcomes span all hospitals. However, although antibiotic stewardship can improve antibiotic use, effective stewardship programs require expertise and an infrastructure that are not present in all hospitals. Rural hospitals have less access to resources, infectious disease expertise, and participation in academic research. Thus, we compared antibiotic overuse at discharge between rural and nonrural hospitals for patients diagnosed with community-associated pneumonia (CAP) or urinary tract infection (UTI)—the 2 most common hospital infections. Methods: To determine whether antibiotic overuse at discharge was higher among rural versus nonrural hospitals, we analyzed data from a 41-hospital prospective cohort of patients treated for CAP or UTI between July 1, 2017, and July 30, 2019, in Michigan. Antibiotic overuse was defined as treatment that was unnecessary (ie, patient did not have an infection), excessive (ie, duration >4 days for CAP), or included suboptimal fluoroquinolone use (ie, safer alternative available). Overuse was determined based on patient risk factors, symptoms, allergies, diagnostic results, and time to stability. Hospital rurality was defined using the Rural–Urban Continuum Codes (RUCC) score. We defined rural as a score ≥4 and very rural as a score of 7–9. We used t tests to compare the mean percentage of patients with antibiotic overuse at discharge between nonrural and rural (and very rural) hospitals. Results: Across 41 hospitals, we included 23,449 patients with CAP or UTI. There were 5 rural (and 3 very rural) hospitals with 2,039 (and 1,082) patients. Antibiotic overuse at discharge was present in 43.1% of patient cases in nonrural hospitals, 52.5% in rural hospitals (P = .04 vs nonrural) and 58.1% in very rural hospitals (P = .007 vs nonrural). Compared to nonrural hospitals, the mean percentage of cases with antibiotic overuse at discharge in rural hospitals was 9.4% higher (15.1% higher in very rural hospitals). Results were similar in a subgroup analysis of only patients with UTI (47.0% in rural vs 37.5% in nonrural, mean difference, 9.5%; P = .03) but were not statistically significant in patients with CAP (53.8% vs 48.0%, respectively; mean difference, 5.8%; P = 0.23). Conclusions: In this retrospective study, rural hospitals—especially very rural hospitals, had higher rates of antibiotic overuse at discharge than nonrural hospitals. Our findings suggest that antibiotic stewardship interventions tailored toward the unique differences in infrastructure, resources, and needs of rural hospitals are essential to community health.
Disclosures: None
Validation of antibiotic stewardship metrics for genitourinary infection management in Veterans Affairs outpatient settings
- Jordan Braunfeld, Matthew Samore, Jacob Crook, McKenna Nevers, Kelly Echevarria, Ben Brintz, Matthew Goetz, Karl Madaras-Kelly
-
- Published online by Cambridge University Press:
- 29 September 2023, p. s35
-
- Article
-
- You have access Access
- Open access
- Export citation
-
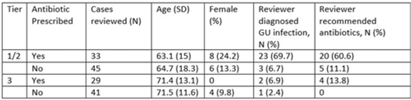
Background: Diagnosis and management of suspected urinary tract infection (UTI) in outpatient settings has been shown to be suboptimal. We previously developed a set of stewardship metrics for UTIs based on electronic health record (EHR) data (Antimicrobial Stewardship & Healthcare Epidemiology 2022;2 suppl 1:S5–S6. doi:10.1017/ash.2022). A tier-based approach was used to more fully capture antibiotic use associated with genitourinary (GU) symptoms and diagnoses. Herein we report a preliminary analysis of validity and reliability of these metrics based on chart abstraction. Methods: The study cohort consisted of patients who visited Veterans Affairs emergency departments or primary care clinics between 2015 and 2022 and who had a GU diagnosis based on International Classification of Disease, Tenth Revision (ICD-10) codes, divided into 3 categories: tier 1 (antibiotics always indicated), tier 2 (antibiotics sometimes indicated), and tier 3 (antibiotics not indicated). Visits related to urological procedures, nontarget settings, or concomitant non-GU infections were excluded. Cases were randomly sampled for manual review from within 8 strata based on tier, use of antibiotics, and visit type. An infectious disease physician and pharmacist abstracted charts using a standardized data-collection instrument. Clinical judgments regarding diagnosis and treatment were recorded on a Likert scale without knowledge of how the patient was managed. The intraclass correlation coefficient (ICC) was used to estimate interrater reliability. Results: To date, 148 cases have been reviewed (50 by both reviewers). Mean (SD) age was 67.5 (15.3) years and 12.2% were female. In a majority of tier 1 and 2 visits in which antibiotics were given, the reviewers found evidence for GU infection (69.7%) and favored prescribing of antibiotics (60.6%) (Table). In contrast, most patients in the tier 3 category who received antibiotics were judged to have noninfectious conditions (eg, benign prostatic hypertrophy) and to not require antibiotics. In the subset of records examined by both reviewers, the interrater reliability of judgments of whether antibiotics were warranted was good (ICC = .704). Conclusions: This preliminary validation provides support for a tier-based approach for stewardship metrics for GU conditions that relies upon electronic data to identify patients for whom antibiotics are generally not indicated.
Disclosures: None
Understanding clinician perspectives on antibiotic associated adverse events to inform feedback
- Jerald Cherian, George Jones, Taylor Helsel, Zunaira Virk, Alejandra Salinas, Suzanne Grieb, Sara Keller, Pranita Tamma, Sara Cosgrove
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s35-s36
-
- Article
-
- You have access Access
- Open access
- Export citation
-
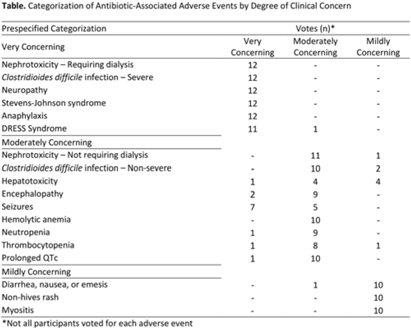
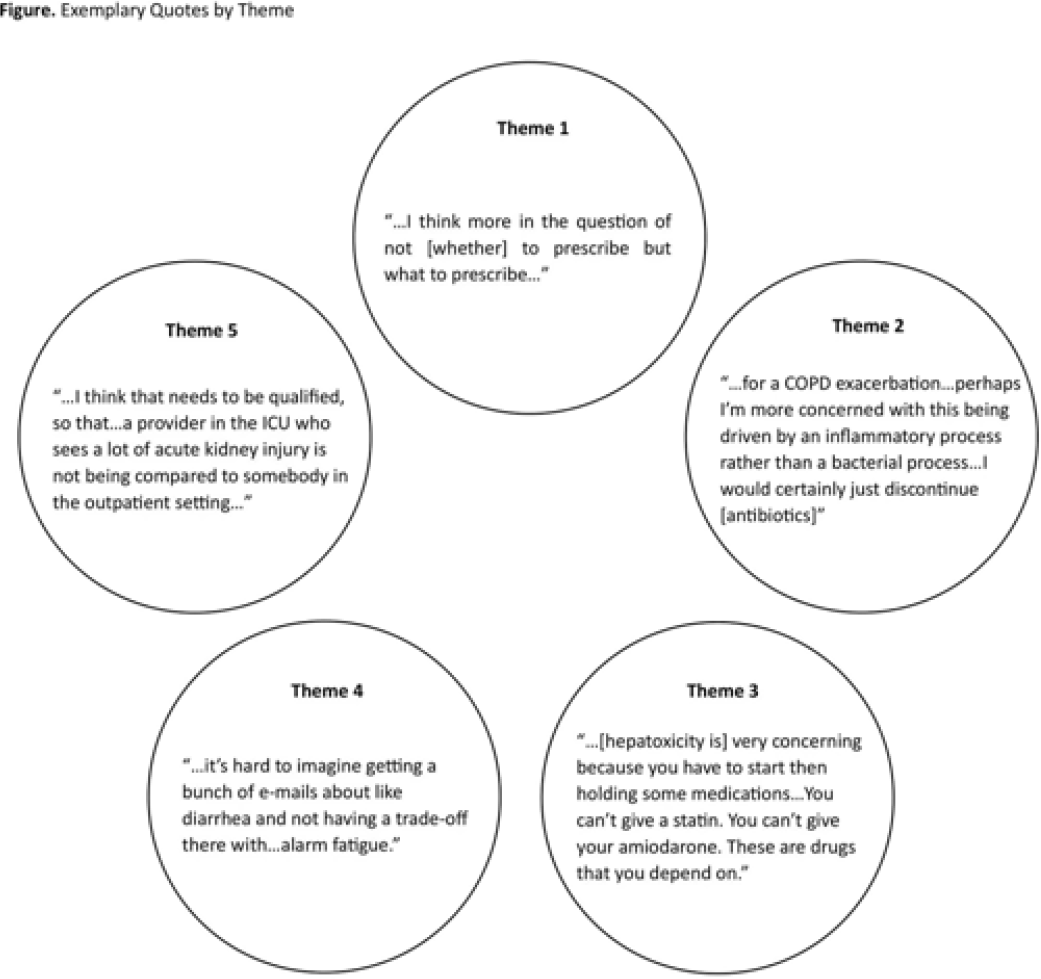
Background: Feedback regarding antibiotic-associated adverse events (ABX-AEs) may assist clinicians with antibiotic decision making. We sought to understand how clinicians account for ABX-AEs when prescribing and their preferences for ABX-AE feedback. Methods: We conducted 1-hour virtual focus groups with 3–5 physicians or advance practice practitioners (APPs) per session at Johns Hopkins Hospital. Participants discussed the role of ABX-AEs in antibiotic decision making and feedback preferences. Participants evaluated prespecified categorization (mildly, moderately, or very concerning) of several ABX-AEs. Focus groups were recorded and transcribed. Transcripts were coded inductively by 2 independent reviewers; discrepancies were resolved by consensus. Codes were used to conduct thematic analysis. Results: Overall, 3 focus groups were conducted with 12 participants: 41.6% were house staff, 16.7% were attending physicians, and 41.6% were APPs. Most were female (91.6%) and were white (41.7%) or Asian (41.7%). Clinicians generally agreed with the prespecified categorizations of ABX-AEs based on degree of clinical concern (Table). We identified 5 themes: (1) The risk of ABX-AE is considered during initial prescribing but influences agent selection more than the decision to prescribe antibiotics. (2) The occurrence of an ABX-AE leads to assessment of need for continued antibiotic therapy. (3) The impact of an ABX-AE on other management decisions is as important as the direct harm of the ABX-AE when assessing severity. (4) Feedback must be curated to prevent clinicians from being overwhelmed with data. (5) Clinicians will be more receptive to feedback regarding ABX-AEs if feedback is contextualized (Fig.). Conclusions: The themes identified and assessment of ABX-AEs of greatest clinical concern may help inform the development of effective ABX-AE feedback methods to improve antibiotic safety.
Disclosures: None
Teams in transition: Increasing role of advanced practice providers in inpatient antimicrobial use
- Reinaldo Perez, Michael Yarrington, Connor Deri, Michael Smith, Jillian Hayes, Rebekah Wrenn, Rebekah Moehring
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s36-s37
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antimicrobial stewardship strategies must be tailored to effectively engage prescribers with distinct training, experiences, and career paths. Advanced practice providers (APPs) have taken on increasing roles as primary team members in acute-care hospitals, but the impact of this practice shift on antimicrobial prescribing is unknown. We describe longitudinal trends in antimicrobial days of therapy (DOT) by attributed provider type in 3 hospitals. Methods: We performed a retrospective time-series analysis of antimicrobial use for the 7-year period of July 2015–June 2022 to investigate the changes by provider type at 3 hospitals: a major university hospital and 2 community hospitals. DOT, antibacterial, and antifungal agent groups were defined using National Healthcare Safety Network methods. We included anti-influenza and antiherpesvirus agents in the antiviral group. We defined protected agents as those targeted by hospital antimicrobial stewardship program policy (eg, requiring preauthorization). Provider type was defined by electronic health record user profiles in 3 categories: physician, trainees (residents, fellows and medical students), and APPs (nurse practitioners, physician assistants, and nurse anesthetists). We evaluated DOT per 1,000 days present over time by agent group to assess quarterly rate trends. Then, we calculated the percentage of total DOT by provider group. We used multinomial logistic regression to measure changes in percentage DOT across the clinician groups over time using physicians as the referent. Results: Across hospitals and provider groups, we observed an overall decrease in use rates for antibacterial and protected agents (17% each) and increased use rates for antiviral agents (38%) and antifungal agents (4%) (Table 1). Baseline distribution of DOT by provider group and change in distribution over time varied by hospital and agent group (Fig. and Table 2). The largest increases in percentage DOT attributed to APPs compared with physicians occurred in the university hospital with the following average increases per quarter: 1.5% for antibacterials, 3.9% for antivirals, 3.3% for antifungals, and 3.8% for protected agents (Table 3). Community hospitals had higher initial percentage DOT attributed to physicians, but both hospitals experienced increased percentage DOT attributed to APPs. Percentage DOT attributed to trainees varied significantly across agent groups and hospitals. Conclusions: Hospitals had differing baseline patterns of DOT attributed to provider groups, but all experienced increases in DOT attributed to APPs. APPs have increasing involvement in antimicrobial use decisions and should be engaged in future antimicrobial stewardship initiatives.
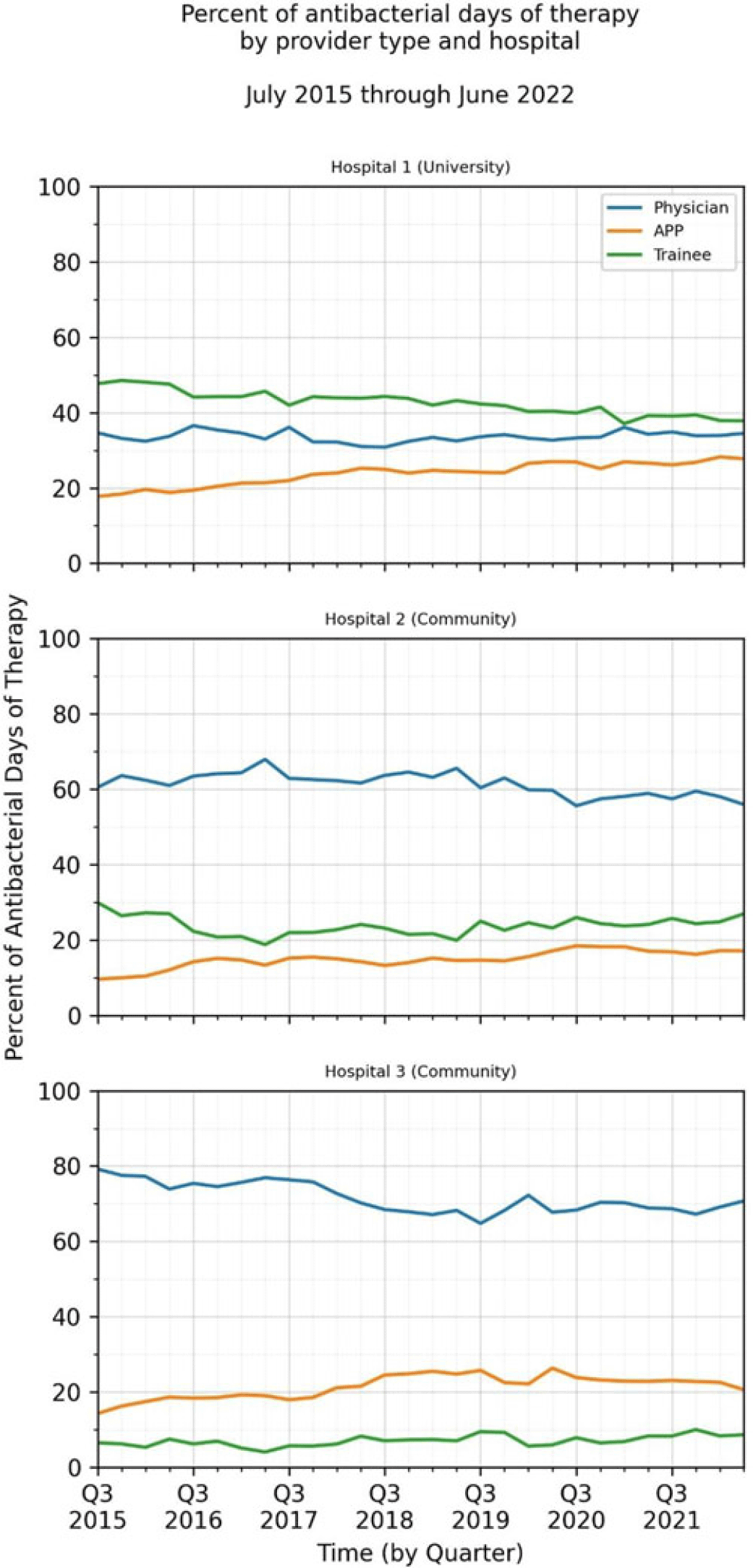
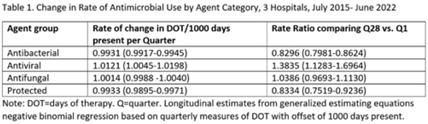
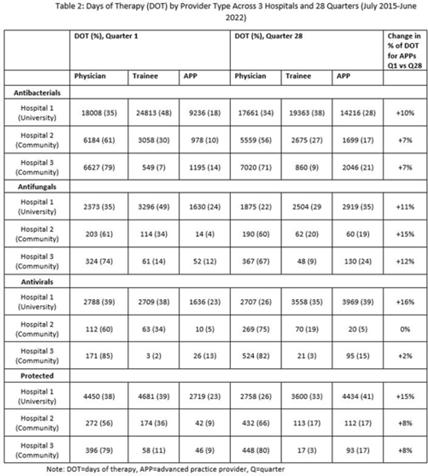
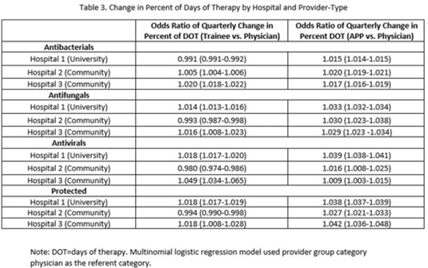
Disclosures: None
Handshake stewardship on adult acute-care surgical services
- Elizabeth Neuner, Kevin Hsueh, Michael Durkin, Sena Sayood
-
- Published online by Cambridge University Press:
- 29 September 2023, pp. s37-s38
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Handshake stewardship is a variation of prospective audit and feedback that entails the individual review of patient charts by a physician–pharmacist collaborative team followed by in-person feedback to primary teams to communicate recommendations regarding optimal antibiotic use. Handshake stewardship has been shown to have durable effects in reducing antimicrobial use in children’s hospitals, but data regarding this intervention in adult hospitals are scarce. In particular, no data are available regarding the impact of this type of stewardship intervention on adult surgical units. We examined the effect of a handshake stewardship intervention at a large academic medical center on adult trauma and acute- and critical-care surgery (ACCS) units. Methods: The antimicrobial stewardship program (ASP) at Barnes-Jewish Hospital launched a handshake stewardship intervention targeting surgical floor teams in January 2022. These teams included the ACCS teams and a number of other surgical services. The intervention consisted of once weekly reviews and in-person rounds with the surgical floor teams along with the establishment of a 7 day per week “hotline” in which the surgical teams could contact an ID physician or pharmacist with questions regarding antibiotic use. Patients with formal ID consultations were not reviewed. Recommendations were tracked including the type, the antibiotic targeted, and recommendation acceptance or rejection. Descriptive statistics were performed to analyze these results. At the end of 12 months, antibiotic use in the floors covered by the ACCS teams were pulled from the NHSN AU module to perform an interrupted time-series analysis 12 months before and after the intervention. Results: Overall, 3,127 charts were reviewed during the intervention period and 637 recommendations were made to all the surgical teams. Opportunities for antibiotic use optimization were identified in ~20% of antibiotic orders. The overall recommendation acceptance rate was 71%. In the ACCS units, 272 interventions were recommended, with an acceptance rate of 67%. The most frequent recommendations were for antibiotic discontinuation (37%), antibiotic de-escalation (17%), shortening duration (12%), and broadening coverage (12%). Antibiotic usage trends (Fig. 1) on the ACCS floors, which were showing a nonsignificant increasing trend (P = .70) before and after the intervention, now show a nonsignificant decreasing trend (P = .20). Conclusions: There are numerous opportunities for antibiotic optimization on adult surgical floors. Although handshake stewardship is a labor-intensive intervention, preliminary findings after 1 year show that, on ACCS units, there may be a trend toward a sustained impact.
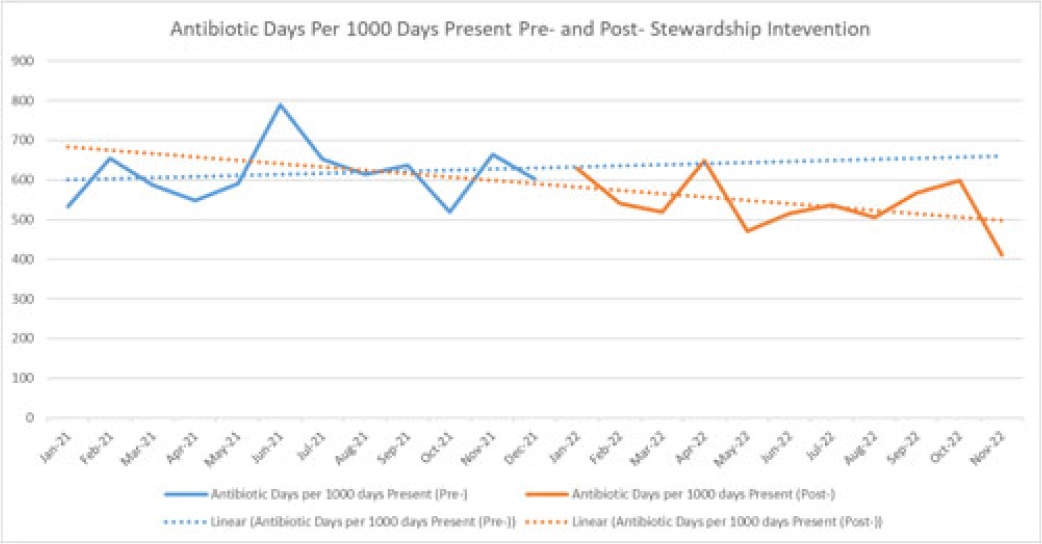

Disclosures: None





























