7 results
DEVELOPMENT OF A NOVEL DIABETIC FOOT RISK ASSESSMENT FOR LOW-RESOURCE HEALTHCARE SETTINGS: A CASE STUDY OF LEAN DESIGN
- Madison Reddie, Daniel Frey
-
- Journal:
- Proceedings of the Design Society / Volume 3 / July 2023
- Published online by Cambridge University Press:
- 19 June 2023, pp. 1465-1474
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Interest in applying a “lean” philosophy to design has been slowly growing in recent years, but there are still few broadly applicable methodologies and illustrative cases published in the literature to guide lean design processes. Lean approaches promise cost reduction and increased product value, which could be particularly beneficial in product development for low- and middle-income country markets, where value demands are high. We use the clinical need of efficient diabetic foot risk assessment in lowresource healthcare settings to present an example of lean early-stage design of a medical device. The background of existing medical literature and commercial products is intentionally leveraged throughout the design process to streamline development and minimize the need for independent validation of product strategies and design features. Our approach resulted in an efficient design process that generated a novel, purely mechanical plantar pressure evaluation device that can indicate high risk of diabetic foot ulcer in resource-constrained settings. This case provides a practical example of how design processes can be adapted to be leaner where there are large gains from minimizing design cycle time and cost.
Susceptibility reporting and antibiotic prescribing for UTIs in the inpatient setting: A nudge toward improved stewardship
- Madison Ponder, Alan Kinlaw, Lindsay Daniels, Ashlyn Norris, Kevin Alby
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s17
-
- Article
-
- You have access Access
- Open access
- Export citation
-
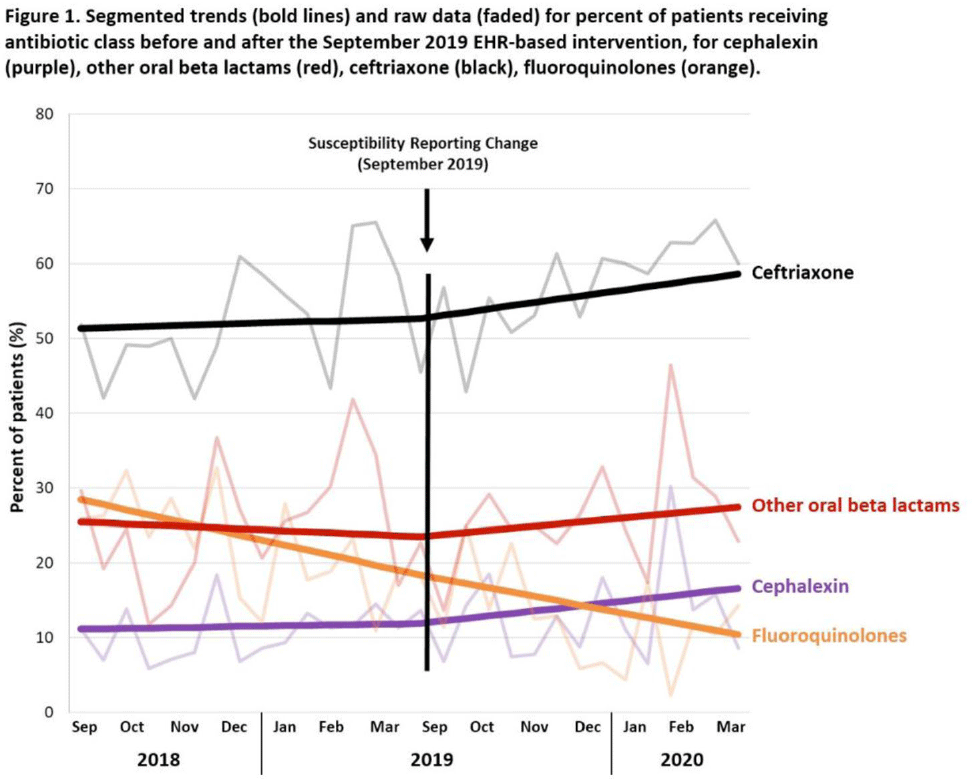
Background: Urinary tract infections (UTIs) are common in the inpatient, observation, and emergency department settings. Although many UTI-causing pathogens are susceptible to oral β-lactams, these agents are not tested directly, and susceptibility is extrapolated from other agents. To improve the use of these agents, the University of North Carolina Medical Center (UNCMC) added cephalexin to the susceptibility profile generated with urine culture results in the electronic health record (EHR). We evaluated prescribing trends of cephalexin, other oral β-lactams, fluoroquinolones, and other antibiotics for UTIs in the inpatient setting, before and after the susceptibility reporting change. Methods: An interrupted time-series analysis was conducted. Among 1,491 patients who had positive urine cultures with susceptibilities and received at least 1 antibiotic with a listed UTI indication during their inpatient stay at UNCMC, we measured the weekly prevalence (%) of patients who received each antibiotic group: cephalexin, other oral β-lactams (amoxicillin-clavulanate, cefdinir, cefuroxime), fluoroquinolones (levofloxacin, ciprofloxacin), and ceftriaxone. The study comprised a preintervention period (September 2018–March 2019) and a postintervention period (September 2019–March 2020). The prevalence of each antibiotic or group was plotted over time, and segmented linear regression was used to estimate the impact of the intervention on each antibiotic groups’ time trend. Results: At study baseline in September 2018, the weekly prevalence of antibiotic use was 11% for cephalexin, 26% for other oral β-lactams, 51% for ceftriaxone, and 29% for fluoroquinolones. Fluoroquinolone use decreased steadily throughout the study period, by 11% during the 7-month preintervention period (95% CI, −17% to −5%) and by 8% (95% CI, −13% to −3%) after the intervention (P for trend deflection, .70). In contrast, during the preintervention period, trends were flat for cephalexin, ceftriaxone, and other oral β-lactams (all P for nonzero preintervention slope were >.40). During the postintervention period, use increased for ceftriaxone (6%; 95% CI, 3%–9%). Post-intervention use also increased for cephalexin (5%; 95% CI, −3% to 12%) and other oral β-lactams (4%; 95% CI, −8%, 15%), but these trends were imprecise and not statistically significant at α = .05. Fig. 1 displays trends and raw data for each antibiotic group. Conclusions: The urine culture susceptibility reporting change was associated with small increases in cephalexin and ceftriaxone use, coincident with continued decreasing use of fluoroquinolones, for hospitalized patients with positive urine cultures and a listed UTI indication. Low-resource EHR-based interventions may confer considerable benefit for antimicrobial stewardship efforts in this clinical setting, and larger real-world studies are needed to replicate and contextualize these findings.
Funding: None
Disclosures: None
Evolution of severe acute respiratory coronavirus virus 2 (SARS-CoV-2) seroprevalence among employees of a US academic children’s hospital during coronavirus disease 2019 (COVID-19) pandemic
- Part of
- Brian T. Fisher, Anna Sharova, Craig L. K. Boge, Sigrid Gouma, Audrey Kamrin, Jesse Blumenstock, Sydney Shuster, Lauren Gianchetti, Danielle Collins, Elikplim Akaho, Madison E. Weirick, Christopher M. McAllister, Marcus J. Bolton, Claudia P. Arevalo, Eileen C. Goodwin, Elizabeth M. Anderson, Shannon R. Christensen, Fran Balamuth, Audrey R. Odom John, Yun Li, Susan Coffin, Jeffrey S. Gerber, Scott E. Hensley
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 43 / Issue 11 / November 2022
- Published online by Cambridge University Press:
- 02 December 2021, pp. 1647-1655
- Print publication:
- November 2022
-
- Article
- Export citation
-
Objective:
To describe the cumulative seroprevalence of severe acute respiratory coronavirus virus 2 (SARS-CoV-2) antibodies during the coronavirus disease 2019 (COVID-19) pandemic among employees of a large pediatric healthcare system.
Design, setting, and participants:Prospective observational cohort study open to adult employees at the Children’s Hospital of Philadelphia, conducted April 20–December 17, 2020.
Methods:Employees were recruited starting with high-risk exposure groups, utilizing e-mails, flyers, and announcements at virtual town hall meetings. At baseline, 1 month, 2 months, and 6 months, participants reported occupational and community exposures and gave a blood sample for SARS-CoV-2 antibody measurement by enzyme-linked immunosorbent assays (ELISAs). A post hoc Cox proportional hazards regression model was performed to identify factors associated with increased risk for seropositivity.
Results:In total, 1,740 employees were enrolled. At 6 months, the cumulative seroprevalence was 5.3%, which was below estimated community point seroprevalence. Seroprevalence was 5.8% among employees who provided direct care and was 3.4% among employees who did not perform direct patient care. Most participants who were seropositive at baseline remained positive at follow-up assessments. In a post hoc analysis, direct patient care (hazard ratio [HR], 1.95; 95% confidence interval [CI], 1.03–3.68), Black race (HR, 2.70; 95% CI, 1.24–5.87), and exposure to a confirmed case in a nonhealthcare setting (HR, 4.32; 95% CI, 2.71–6.88) were associated with statistically significant increased risk for seropositivity.
Conclusions:Employee SARS-CoV-2 seroprevalence rates remained below the point-prevalence rates of the surrounding community. Provision of direct patient care, Black race, and exposure to a confirmed case in a nonhealthcare setting conferred increased risk. These data can inform occupational protection measures to maximize protection of employees within the workplace during future COVID-19 waves or other epidemics.
Electron microscopy explorations of the human brain: using immunofluorescence to address challenges
- Kristina Micheva, Marc Perez, Anish Simhal, Richard Weinberg, Daniel Madison
-
- Journal:
- Microscopy and Microanalysis / Volume 27 / Issue S1 / August 2021
- Published online by Cambridge University Press:
- 30 July 2021, p. 1142
- Print publication:
- August 2021
-
- Article
-
- You have access Access
- Export citation
The religious/spiritual beliefs and needs of cancer survivors who underwent cancer-directed surgery
- Elizabeth Palmer Kelly, Anghela Z. Paredes, Stephanie DiFilippo, Madison Hyer, Diamantis I. Tsilimigras, Daniel Rice, Junu Bae, Timothy M. Pawlik
-
- Journal:
- Palliative & Supportive Care / Volume 19 / Issue 2 / April 2021
- Published online by Cambridge University Press:
- 28 August 2020, pp. 175-181
-
- Article
- Export citation
-
Objective
We sought to characterize patients’ preferences for the role of religious and spiritual (R&S) beliefs and practices during cancer treatment and describe the R&S resources desired by patients during the perioperative period.
MethodA cross-sectional survey was administered to individuals who underwent cancer-directed surgery. Data on demographics and R&S beliefs/preferences were collected and analyzed.
ResultsAmong 236 participants, average age was 58.8 (SD = 12.10) years; the majority were female (76.2%), white (94.1%), had a significant other or spouse (60.2%), and were breast cancer survivors (43.6%). Overall, more than one-half (55.9%) of individuals identified themselves as being religious, while others identified as only spiritual (27.9%) or neither (16.2%). Patients who identified as religious wanted R&S integrated into their care more often than patients who were only spiritual or neither (p < 0.001). Nearly half of participants (49.6%) wanted R&S resources when admitted to the hospital including the opportunity to speak with an R&S leader (e.g., rabbi; 72.1%), R&S texts (64.0%), and journaling materials (54.1%). Irrespective of R&S identification, 68.0% of patients did not want their physician to engage with them about R&S topics.
Significance of resultsAccess to R&S resources is important during cancer treatment, and incorporating R&S into cancer care may be especially important to patients that identify as religious. R&S needs should be addressed as part of the cancer care plan.
The Potential Effects on United States Agriculture of an Avian Influenza Outbreak
- Scott Brown, Daniel Madison, H.L. Goodwin, F. Dustan Clark
-
- Journal:
- Journal of Agricultural and Applied Economics / Volume 39 / Issue 2 / August 2007
- Published online by Cambridge University Press:
- 28 April 2015, pp. 335-343
-
- Article
- Export citation
-
The U.S. poultry industry has spent considerable resources to date preparing for an outbreak of avian influenza in this country. This research quantifies the potential effects of two alternative avian influenza scenarios on the poultry industry. In addition, this research looks at effects on other agriculture sectors including the loss of feed demand from an outbreak and the impacts on aggregate measures like farm income and consumer food expenditures. The economic sector model maintained by the Food and Agricultural Policy Research Institute (FAPRI) is employed for these scenarios.
Contributors
-
- By Mitchell Aboulafia, Frederick Adams, Marilyn McCord Adams, Robert M. Adams, Laird Addis, James W. Allard, David Allison, William P. Alston, Karl Ameriks, C. Anthony Anderson, David Leech Anderson, Lanier Anderson, Roger Ariew, David Armstrong, Denis G. Arnold, E. J. Ashworth, Margaret Atherton, Robin Attfield, Bruce Aune, Edward Wilson Averill, Jody Azzouni, Kent Bach, Andrew Bailey, Lynne Rudder Baker, Thomas R. Baldwin, Jon Barwise, George Bealer, William Bechtel, Lawrence C. Becker, Mark A. Bedau, Ernst Behler, José A. Benardete, Ermanno Bencivenga, Jan Berg, Michael Bergmann, Robert L. Bernasconi, Sven Bernecker, Bernard Berofsky, Rod Bertolet, Charles J. Beyer, Christian Beyer, Joseph Bien, Joseph Bien, Peg Birmingham, Ivan Boh, James Bohman, Daniel Bonevac, Laurence BonJour, William J. Bouwsma, Raymond D. Bradley, Myles Brand, Richard B. Brandt, Michael E. Bratman, Stephen E. Braude, Daniel Breazeale, Angela Breitenbach, Jason Bridges, David O. Brink, Gordon G. Brittan, Justin Broackes, Dan W. Brock, Aaron Bronfman, Jeffrey E. Brower, Bartosz Brozek, Anthony Brueckner, Jeffrey Bub, Lara Buchak, Otavio Bueno, Ann E. Bumpus, Robert W. Burch, John Burgess, Arthur W. Burks, Panayot Butchvarov, Robert E. Butts, Marina Bykova, Patrick Byrne, David Carr, Noël Carroll, Edward S. Casey, Victor Caston, Victor Caston, Albert Casullo, Robert L. Causey, Alan K. L. Chan, Ruth Chang, Deen K. Chatterjee, Andrew Chignell, Roderick M. Chisholm, Kelly J. Clark, E. J. Coffman, Robin Collins, Brian P. Copenhaver, John Corcoran, John Cottingham, Roger Crisp, Frederick J. Crosson, Antonio S. Cua, Phillip D. Cummins, Martin Curd, Adam Cureton, Andrew Cutrofello, Stephen Darwall, Paul Sheldon Davies, Wayne A. Davis, Timothy Joseph Day, Claudio de Almeida, Mario De Caro, Mario De Caro, John Deigh, C. F. Delaney, Daniel C. Dennett, Michael R. DePaul, Michael Detlefsen, Daniel Trent Devereux, Philip E. Devine, John M. Dillon, Martin C. Dillon, Robert DiSalle, Mary Domski, Alan Donagan, Paul Draper, Fred Dretske, Mircea Dumitru, Wilhelm Dupré, Gerald Dworkin, John Earman, Ellery Eells, Catherine Z. Elgin, Berent Enç, Ronald P. Endicott, Edward Erwin, John Etchemendy, C. Stephen Evans, Susan L. Feagin, Solomon Feferman, Richard Feldman, Arthur Fine, Maurice A. Finocchiaro, William FitzPatrick, Richard E. Flathman, Gvozden Flego, Richard Foley, Graeme Forbes, Rainer Forst, Malcolm R. Forster, Daniel Fouke, Patrick Francken, Samuel Freeman, Elizabeth Fricker, Miranda Fricker, Michael Friedman, Michael Fuerstein, Richard A. Fumerton, Alan Gabbey, Pieranna Garavaso, Daniel Garber, Jorge L. A. Garcia, Robert K. Garcia, Don Garrett, Philip Gasper, Gerald Gaus, Berys Gaut, Bernard Gert, Roger F. Gibson, Cody Gilmore, Carl Ginet, Alan H. Goldman, Alvin I. Goldman, Alfonso Gömez-Lobo, Lenn E. Goodman, Robert M. Gordon, Stefan Gosepath, Jorge J. E. Gracia, Daniel W. Graham, George A. Graham, Peter J. Graham, Richard E. Grandy, I. Grattan-Guinness, John Greco, Philip T. Grier, Nicholas Griffin, Nicholas Griffin, David A. Griffiths, Paul J. Griffiths, Stephen R. Grimm, Charles L. Griswold, Charles B. Guignon, Pete A. Y. Gunter, Dimitri Gutas, Gary Gutting, Paul Guyer, Kwame Gyekye, Oscar A. Haac, Raul Hakli, Raul Hakli, Michael Hallett, Edward C. Halper, Jean Hampton, R. James Hankinson, K. R. Hanley, Russell Hardin, Robert M. Harnish, William Harper, David Harrah, Kevin Hart, Ali Hasan, William Hasker, John Haugeland, Roger Hausheer, William Heald, Peter Heath, Richard Heck, John F. Heil, Vincent F. Hendricks, Stephen Hetherington, Francis Heylighen, Kathleen Marie Higgins, Risto Hilpinen, Harold T. Hodes, Joshua Hoffman, Alan Holland, Robert L. Holmes, Richard Holton, Brad W. Hooker, Terence E. Horgan, Tamara Horowitz, Paul Horwich, Vittorio Hösle, Paul Hoβfeld, Daniel Howard-Snyder, Frances Howard-Snyder, Anne Hudson, Deal W. Hudson, Carl A. Huffman, David L. Hull, Patricia Huntington, Thomas Hurka, Paul Hurley, Rosalind Hursthouse, Guillermo Hurtado, Ronald E. Hustwit, Sarah Hutton, Jonathan Jenkins Ichikawa, Harry A. Ide, David Ingram, Philip J. Ivanhoe, Alfred L. Ivry, Frank Jackson, Dale Jacquette, Joseph Jedwab, Richard Jeffrey, David Alan Johnson, Edward Johnson, Mark D. Jordan, Richard Joyce, Hwa Yol Jung, Robert Hillary Kane, Tomis Kapitan, Jacquelyn Ann K. Kegley, James A. Keller, Ralph Kennedy, Sergei Khoruzhii, Jaegwon Kim, Yersu Kim, Nathan L. King, Patricia Kitcher, Peter D. Klein, E. D. Klemke, Virginia Klenk, George L. Kline, Christian Klotz, Simo Knuuttila, Joseph J. Kockelmans, Konstantin Kolenda, Sebastian Tomasz Kołodziejczyk, Isaac Kramnick, Richard Kraut, Fred Kroon, Manfred Kuehn, Steven T. Kuhn, Henry E. Kyburg, John Lachs, Jennifer Lackey, Stephen E. Lahey, Andrea Lavazza, Thomas H. Leahey, Joo Heung Lee, Keith Lehrer, Dorothy Leland, Noah M. Lemos, Ernest LePore, Sarah-Jane Leslie, Isaac Levi, Andrew Levine, Alan E. Lewis, Daniel E. Little, Shu-hsien Liu, Shu-hsien Liu, Alan K. L. Chan, Brian Loar, Lawrence B. Lombard, John Longeway, Dominic McIver Lopes, Michael J. Loux, E. J. Lowe, Steven Luper, Eugene C. Luschei, William G. Lycan, David Lyons, David Macarthur, Danielle Macbeth, Scott MacDonald, Jacob L. Mackey, Louis H. Mackey, Penelope Mackie, Edward H. Madden, Penelope Maddy, G. B. Madison, Bernd Magnus, Pekka Mäkelä, Rudolf A. Makkreel, David Manley, William E. Mann (W.E.M.), Vladimir Marchenkov, Peter Markie, Jean-Pierre Marquis, Ausonio Marras, Mike W. Martin, A. P. Martinich, William L. McBride, David McCabe, Storrs McCall, Hugh J. McCann, Robert N. McCauley, John J. McDermott, Sarah McGrath, Ralph McInerny, Daniel J. McKaughan, Thomas McKay, Michael McKinsey, Brian P. McLaughlin, Ernan McMullin, Anthonie Meijers, Jack W. Meiland, William Jason Melanson, Alfred R. Mele, Joseph R. Mendola, Christopher Menzel, Michael J. Meyer, Christian B. Miller, David W. Miller, Peter Millican, Robert N. Minor, Phillip Mitsis, James A. Montmarquet, Michael S. Moore, Tim Moore, Benjamin Morison, Donald R. Morrison, Stephen J. Morse, Paul K. Moser, Alexander P. D. Mourelatos, Ian Mueller, James Bernard Murphy, Mark C. Murphy, Steven Nadler, Jan Narveson, Alan Nelson, Jerome Neu, Samuel Newlands, Kai Nielsen, Ilkka Niiniluoto, Carlos G. Noreña, Calvin G. Normore, David Fate Norton, Nikolaj Nottelmann, Donald Nute, David S. Oderberg, Steve Odin, Michael O’Rourke, Willard G. Oxtoby, Heinz Paetzold, George S. Pappas, Anthony J. Parel, Lydia Patton, R. P. Peerenboom, Francis Jeffry Pelletier, Adriaan T. Peperzak, Derk Pereboom, Jaroslav Peregrin, Glen Pettigrove, Philip Pettit, Edmund L. Pincoffs, Andrew Pinsent, Robert B. Pippin, Alvin Plantinga, Louis P. Pojman, Richard H. Popkin, John F. Post, Carl J. Posy, William J. Prior, Richard Purtill, Michael Quante, Philip L. Quinn, Philip L. Quinn, Elizabeth S. Radcliffe, Diana Raffman, Gerard Raulet, Stephen L. Read, Andrews Reath, Andrew Reisner, Nicholas Rescher, Henry S. Richardson, Robert C. Richardson, Thomas Ricketts, Wayne D. Riggs, Mark Roberts, Robert C. Roberts, Luke Robinson, Alexander Rosenberg, Gary Rosenkranz, Bernice Glatzer Rosenthal, Adina L. Roskies, William L. Rowe, T. M. Rudavsky, Michael Ruse, Bruce Russell, Lilly-Marlene Russow, Dan Ryder, R. M. Sainsbury, Joseph Salerno, Nathan Salmon, Wesley C. Salmon, Constantine Sandis, David H. Sanford, Marco Santambrogio, David Sapire, Ruth A. Saunders, Geoffrey Sayre-McCord, Charles Sayward, James P. Scanlan, Richard Schacht, Tamar Schapiro, Frederick F. Schmitt, Jerome B. Schneewind, Calvin O. Schrag, Alan D. Schrift, George F. Schumm, Jean-Loup Seban, David N. Sedley, Kenneth Seeskin, Krister Segerberg, Charlene Haddock Seigfried, Dennis M. Senchuk, James F. Sennett, William Lad Sessions, Stewart Shapiro, Tommie Shelby, Donald W. Sherburne, Christopher Shields, Roger A. Shiner, Sydney Shoemaker, Robert K. Shope, Kwong-loi Shun, Wilfried Sieg, A. John Simmons, Robert L. Simon, Marcus G. Singer, Georgette Sinkler, Walter Sinnott-Armstrong, Matti T. Sintonen, Lawrence Sklar, Brian Skyrms, Robert C. Sleigh, Michael Anthony Slote, Hans Sluga, Barry Smith, Michael Smith, Robin Smith, Robert Sokolowski, Robert C. Solomon, Marta Soniewicka, Philip Soper, Ernest Sosa, Nicholas Southwood, Paul Vincent Spade, T. L. S. Sprigge, Eric O. Springsted, George J. Stack, Rebecca Stangl, Jason Stanley, Florian Steinberger, Sören Stenlund, Christopher Stephens, James P. Sterba, Josef Stern, Matthias Steup, M. A. Stewart, Leopold Stubenberg, Edith Dudley Sulla, Frederick Suppe, Jere Paul Surber, David George Sussman, Sigrún Svavarsdóttir, Zeno G. Swijtink, Richard Swinburne, Charles C. Taliaferro, Robert B. Talisse, John Tasioulas, Paul Teller, Larry S. Temkin, Mark Textor, H. S. Thayer, Peter Thielke, Alan Thomas, Amie L. Thomasson, Katherine Thomson-Jones, Joshua C. Thurow, Vzalerie Tiberius, Terrence N. Tice, Paul Tidman, Mark C. Timmons, William Tolhurst, James E. Tomberlin, Rosemarie Tong, Lawrence Torcello, Kelly Trogdon, J. D. Trout, Robert E. Tully, Raimo Tuomela, John Turri, Martin M. Tweedale, Thomas Uebel, Jennifer Uleman, James Van Cleve, Harry van der Linden, Peter van Inwagen, Bryan W. Van Norden, René van Woudenberg, Donald Phillip Verene, Samantha Vice, Thomas Vinci, Donald Wayne Viney, Barbara Von Eckardt, Peter B. M. Vranas, Steven J. Wagner, William J. Wainwright, Paul E. Walker, Robert E. Wall, Craig Walton, Douglas Walton, Eric Watkins, Richard A. Watson, Michael V. Wedin, Rudolph H. Weingartner, Paul Weirich, Paul J. Weithman, Carl Wellman, Howard Wettstein, Samuel C. Wheeler, Stephen A. White, Jennifer Whiting, Edward R. Wierenga, Michael Williams, Fred Wilson, W. Kent Wilson, Kenneth P. Winkler, John F. Wippel, Jan Woleński, Allan B. Wolter, Nicholas P. Wolterstorff, Rega Wood, W. Jay Wood, Paul Woodruff, Alison Wylie, Gideon Yaffe, Takashi Yagisawa, Yutaka Yamamoto, Keith E. Yandell, Xiaomei Yang, Dean Zimmerman, Günter Zoller, Catherine Zuckert, Michael Zuckert, Jack A. Zupko (J.A.Z.)
- Edited by Robert Audi, University of Notre Dame, Indiana
-
- Book:
- The Cambridge Dictionary of Philosophy
- Published online:
- 05 August 2015
- Print publication:
- 27 April 2015, pp ix-xxx
-
- Chapter
- Export citation




