10 results
Characterization of resistance to newer antimicrobials among carbapenem-resistant Klebsiella pneumoniae in the post–acute-care setting
- Helen L. Zhang, Kyle J. Gontjes, Jennifer H. Han, Kevin Alby, Zena Lapp, Evan Snitkin, Ellie J.C. Goldstein, Sean Muldoon, Pam Tolomeo, Ebbing Lautenbach
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 7 / July 2023
- Published online by Cambridge University Press:
- 28 July 2022, pp. 1159-1162
- Print publication:
- July 2023
-
- Article
- Export citation
-
We assessed susceptibility patterns to newer antimicrobial agents among clinical carbapenem-resistant Klebsiella pneumoniae (CRKP) isolates from patients in long-term acute-care hospitals (LTACHs) from 2014 to 2015. Meropenem-vaborbactam and imipenem-relebactam nonsusceptibility were observed among 9.9% and 9.1% of isolates, respectively. Nonsusceptibility to ceftazidime-avibactam (1.1%) and plazomicin (0.8%) were uncommon.
Genomic investigation to identify the source of SARS-CoV-2 infection among healthcare personnel
- Sarah Sansom, Hannah Barbian, Evan Snitkin, Christine Fukuda, Nicholas Moore, Lahari Thotapalli, Elias Baied, DO Young Kim, Mary Hayden, Michael Lin
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s74-s75
-
- Article
-
- You have access Access
- Open access
- Export citation
-
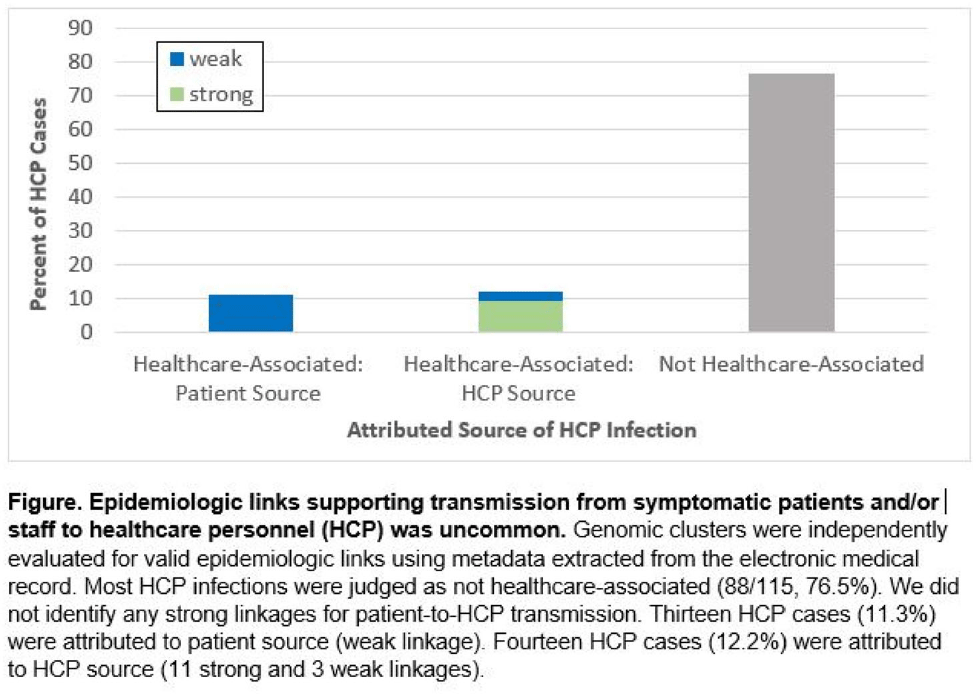
Background: Contact tracing alone is often inadequate to determine the source of healthcare personnel (HCP) COVID-19 when SARS-CoV-2 is widespread in the community. We combined whole-genome sequencing (WGS) with traditional epidemiologic analysis to investigate the frequency with which patients or other HCP with symptomatic COVID-19 acted as the source of HCP infection at a large tertiary-care center early in the pandemic. Methods: Cohort samples were selected from patients and HCP with PCR-positive SARS-CoV-2 infection from a period with complete retention of samples (March 14, 2021–April 10, 2020) at Rush University Medical Center, a 664-bed hospital in Chicago, Illinois. During this period, testing was limited to symptomatic patients and HCP. Recommended respiratory equipment for HCP evolved under guidance, including a 19-day period when medical face masks were recommended for COVID-19 care except for aerosol-generating procedures. Viral RNA was extracted and sequenced (NovaSeq, Illumina) from remnant nasopharyngeal swab samples in M4RT viral transport medium. Genomes with >90% coverage underwent cluster detection using a 2 single-nucleotide variant genetic distance cutoff. Genomic clusters were independently evaluated for valid epidemiologic links by 2 infectious diseases physicians (with a third adjudicator) using metadata extracted from the electronic medical record and according to predetermined criteria (Table 1). Results: In total, 1,031 SARS-CoV-2 sequences were analyzed, identifying 49 genomic clusters with HCP (median, 8; range, 2–43 members per cluster; total, 268 patients and 115 HCP) (Fig. 1). Also, 20,190 flowsheet activities were documented for cohort HCP and patient interactions, including 686 instances in which a cohort HCP contributed to a cohort patient’s chart. Most HCP infections were considered not healthcare associated (88 of 115, 76.5%). We did not identify any strong linkages for patient-to-HCP transmission. Moreover, 13 HCP cases (11.3%) were attributed to patient source (weak linkage). Also, 14 HCP cases (12.2%) were attributed to HCP source (11 strong and 3 weak linkages). Weak linkages were due to lack of epidemiologic data for HCP location, particularly nonclinical staff (eg, an environmental service worker who lacked location documentation to rule out patient-specific contact). Agreement for epidemiologic linkage between the 2 evaluators was high (κ, 0.91). Conclusions: Using genomic and epidemiologic data, we found that most HCP COVID-19 infections were not healthcare associated. We found weak evidence to support symptomatic patient-to-HCP transmission of SARS-CoV-2 and stronger evidence for HCP-to-HCP transmission. Large genomic clusters without plausible epidemiologic links were identified, reflecting the limited utility of genomic surveillance alone to characterize chains of transmission of SARS-CoV-2 during extensive community spread.
Funding: None
Disclosures: None

Nursing-Home Patient Functional and Microbiota Status Drive Environmental Contamination with Vancomycin-Resistant Enterococci
- Joyce Wang, Betsy Foxman, A. Krishna Rao, Lona Mody, Evan Snitkin
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, p. s68
-
- Article
-
- You have access Access
- Open access
- Export citation
-
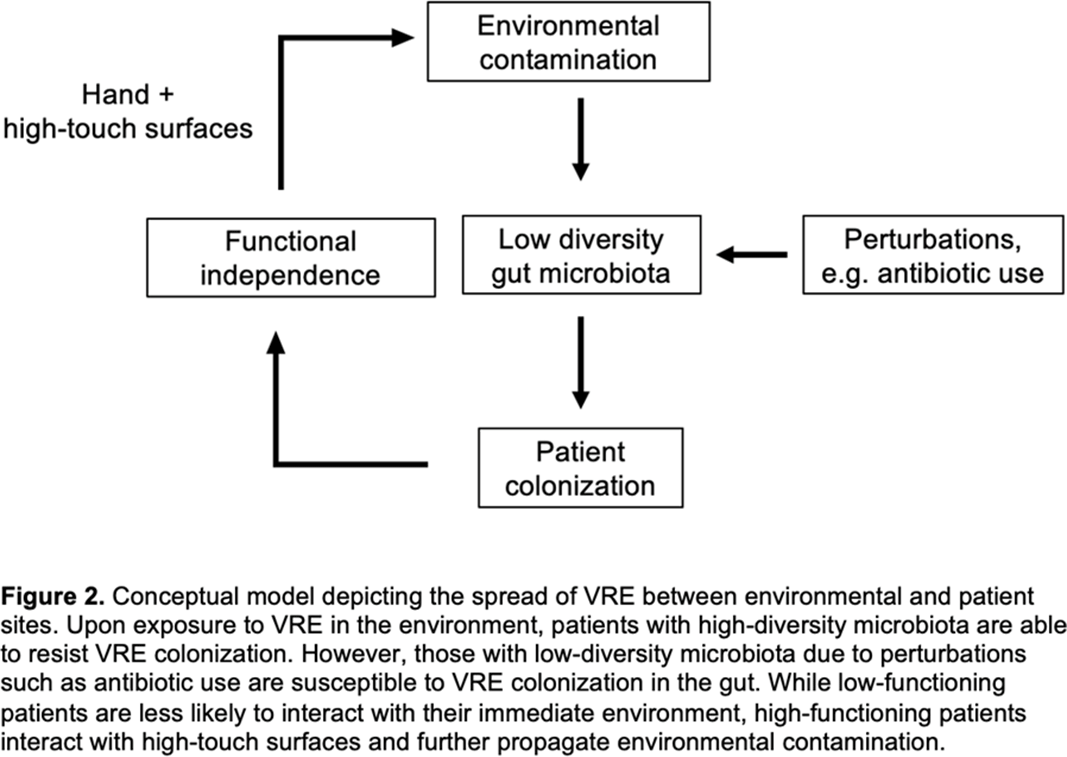
Background: Patient colonization and shedding of vancomycin-resistant enterococci (VRE) is a major source of environmental contamination leading to VRE transmission in nursing homes. We hypothesize that we can inform mitigation strategies by identifying patient clinical and microbiota features associated with environmental contamination with VRE. Methods: During a 6-month period of active surveillance in 6 Michigan nursing homes, 245 patients (with 806 follow-up visits) were enrolled. Patient clinical data and swabs for VRE were collected from multiple body sites and high-touch environmental surfaces. In total, 316 perirectal swabs were collected from 137 patients for gut microbiota analysis and community status type (CST) assignment based on taxonomic composition. The associations between VRE colonization pattern, gut microbial CST, and patient factors were examined using multivariable generalized estimating equations, adjusting for patient-and facility-level clustering. We used VRE colonization patterns to group study visits: “uncolonized” (patient−/environment−); “environment-only” (patient−/environment+); “patient-only” (patient+/environment−); “both” (patient+/environment+). Results: Across all study visits, VRE colonization on patient hand and groin/perirectal area was positively correlated with VRE contamination of high-touch environmental surfaces, suggesting direct transfer of VRE between patient and environment via patient hands (Figure 1A). We next set out to identify patient factors associated with patient colonization and environmental contamination. At baseline, while patients in the “both” group had anticipated risk factors such as longer prior hospitalization and more frequent broad-spectrum antibiotic use, they were unexpectedly younger than “uncolonized” patients and had similar functional status. This last feature contrasted with the “patient-only” group, characterized by higher urinary catheter use and higher functional dependence, suggestive of lower functional dependence facilitating patient contamination of their environment. No clinical features distinguished “uncolonized” and “environment-only” patients (Table 1). Lastly, in multivariable analyses, we determined the contribution of patient functional status and gut microbiota features to environmental contamination. Low-diversity CST, characterized by reduced anaerobic taxa, was weakly associated with “patient-only” and significantly associated with “both.” Notably, high functional dependence was significantly associated with “environment-only” and “patient-only” but not “both,” indicating high-functioning patients with disrupted gut microbiota as drivers of environmental contamination (Figure 1B). Conclusions: Our findings suggest that antimicrobial exposure disrupts patient gut microbiota, a significant mediator of colonization dynamics between patients and their environment, and that high-functioning patients may be more likely to spread VRE between their body sites and high-touch environmental surfaces (Figure 2). These findings highlight both antibiotic stewardship and patient hand hygiene as important targets for interrupting transmission mediated by environmental contamination.
Funding: No
Disclosures: None
Figure 1.
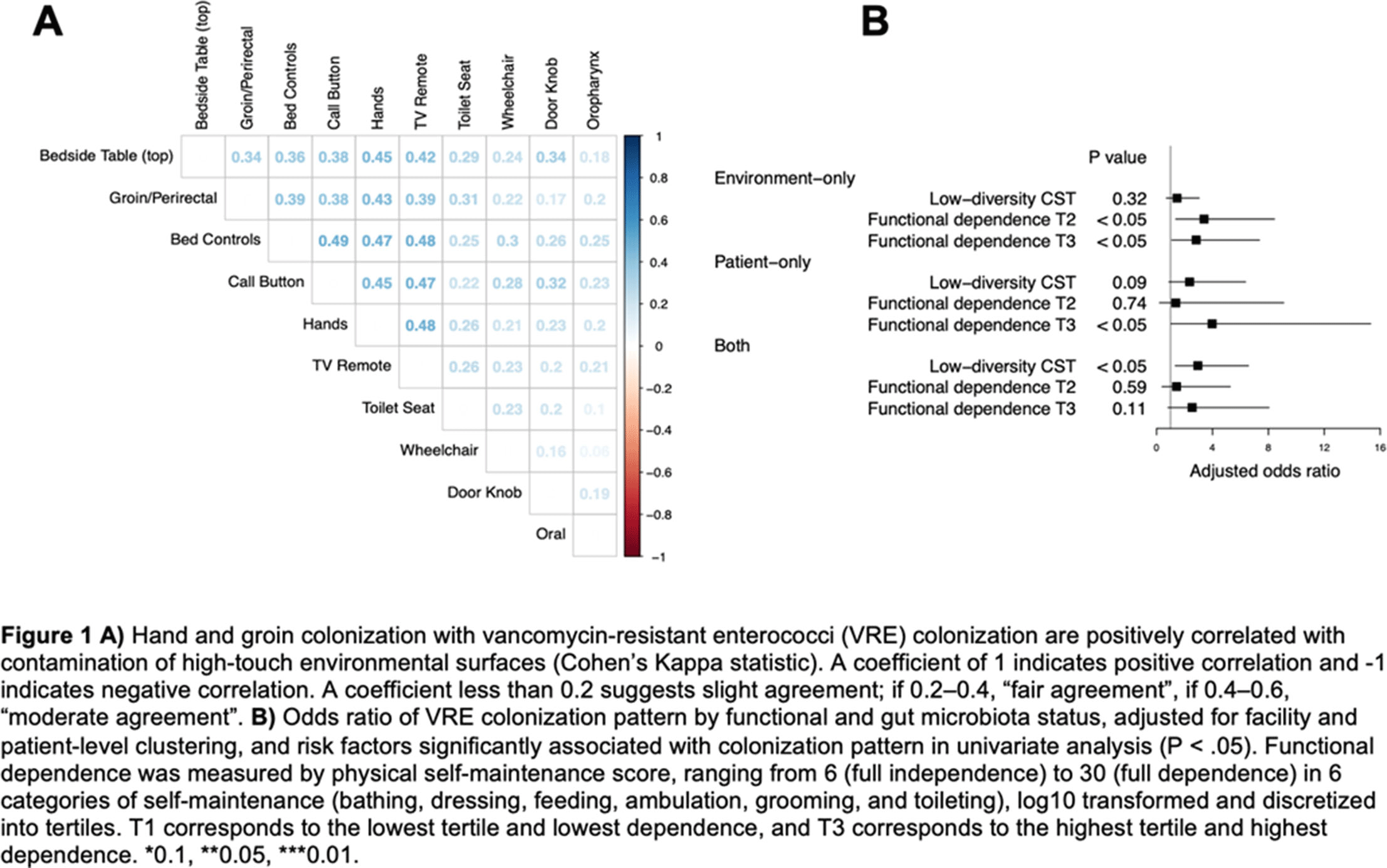
Table 1. 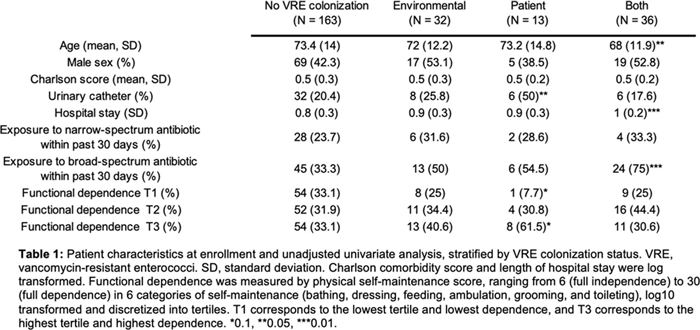
Figure 2.
Bad Bugs Move Alike: Regional Transmission of Antibiotic-Resistant Organisms
- Joyce Wang, Betsy Foxman, Ali Pirani, Zena Lapp, Lona Mody, Evan Snitkin
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s137-s138
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Upon admission, 56.8% of patients entering 6 Michigan nursing facilities from regional acute-care hospitals (ACHs) were colonized with 1 or more antibiotic-resistant organisms (AROs) (Mody 2018, CID). This observation raises 2 questions critical to regional infection control strategies: (1) Is the high ARO burden entering nursing facilities driven by dominant epidemic lineages or diverse circulating strains? and (2) What are the relative roles of patient characteristics (eg, high-risk patients) and exposure to specific ACHs (eg, high-risk facilities) in determining whether patients are colonized with AROs upon nursing facility admission? Here, we integrated whole-genome sequencing, patient transfer, and clinical data to answer these questions for the 4 most prevalent ARO species in the region: methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus faecalis (VREfc), Enterococcus faecium (VREfm), and ciprofloxacin-resistant Escherichia coli (CipREc). Methods: We studied colonizing isolates collected via active surveillance of 584 patients in 6 Michigan nursing facilities between 2013 and 2016. The whole genome of the first isolate of each ARO species collected from each patient was sequenced and analyzed to identify sequence types (STs) and to infer the transmission network by species. We determined the connectedness between nursing facilities based on the number of patients received from the same ACHs and assigned each ARO to the most recent ACH using curated transfer information. The associations between patient characteristics and recent ACH exposures with colonization by ARO were examined using multivariable models. Results: Most of the sequenced ARO isolates belonged to major healthcare-associated lineages: MRSA (ST5, N = 78 of 117); VREfc (ST6, N = 68 of 75); CipREc (ST131, N = 50 of 64); and closely related VREfm isolates (N = 129). Phylogenetically closely related isolates were found across study facilities, indicating that endemic ARO lineages have permeated local healthcare networks (Fig. 1). Patient characteristics played a dominant role in determining patient risk of ARO colonization on admission to a nursing facility. Only in the case of VREfm was a hospital significantly associated with colonization after adjustment for covariates (Table 1). Conclusions: ARO lineages were widely disseminated and colonization of specific ARO lineages at nursing facility entry could not be attributed to recent exposure to a specific ACH. Thus, for the ARO lineages studied here, a broader transmission system crosses ACHs, nursing facilities and probably the community. Therefore, the best indicators of ARO colonization were patient clinical characteristics, particularly poor functional status and antibiotic exposure. These findings suggest that intervention efforts targeting patients with characteristics associated with ARO colonization may help limit further spread among regional facilities.
Funding: None
Disclosures: None
Genomic Epidemiology of Clostridioides difficile Sequence Types 1 and 2 Across Three US Medical Centers
- Arianna Miles-Jay, Vincent Young, Eric Pamer, Tor Savidge, Mini Kamboj, Kevin Garey, Evan Snitkin
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s238
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background:Clostridioides difficile is a toxin-producing bacterium that is the foremost cause of healthcare-associated diarrhea in the United States. Recent epidemiologic and genomic evidence indicates that divergent C. difficile strains have varying propensities for transmission within healthcare settings. We investigated whether and how these differences are reflected in the genomic epidemiology of 2 common C. difficile strains—sequence type (ST) 1 (analogous to Ribotype 027) and ST2 (associated with Ribotypes 014/020)—across 3 geographically distinct US medical centers. Methods: Between 2011 and 2017, a convenience sample of ST1 and ST2 C. difficile clinical isolates were collected from 3 US sites: The University of Michigan Medical Center, Texas Medical Center Hospitals, and Memorial Sloan Kettering Cancer Center. Isolates underwent whole-genome sequencing and in silico multilocus sequence typing to verify strain types. Sequences were mapped to ST1 and ST2 reference genomes and single nucleotide variants (SNVs) were identified, filtered, and used to construct pairwise SNV distance matrices. A range of pairwise SNV distance thresholds were applied to assess genetic linkages consistent with recent transmission within ST1 compared to within ST2. Proportions of genetically linked isolates were compared using 2 tests. Results: We identified 200 ST1 and 188 ST2 isolates across the 3 collection sites. Overall, ST2 was more genetically diverse than ST1 (pairwise SNV distance range, 0–156 SNVs and 0–78 SNVs, respectively). ST2 isolates displayed significantly less evidence of recent transmission: 10 ST2 isolates (5.3%) were within 2 SNVs of another isolate compared to 88 (44%) ST1 isolates (P .001) (Fig. 1). As the SNV threshold increased to 5 and 10 SNVs, this trend was maintained (all P < .001). ST2 isolates were also more likely to be genetically linked to an isolate from a different collection site than ST1 isolates. Among isolates with genetic links to at least 1 other isolate at the 5 SNV and 10 SNV thresholds, 21 of 37 and 74 of 89 ST2 isolates (57%, 83%) were linked to an isolate from a different collection site, compared to 2 of 88 and 48 of 157 ST1 isolates (2% and 31%, respectively; both P < .001). Conclusions: Compared to C. difficile ST1 isolates, ST2 isolates displayed less evidence of recent healthcare transmission and were more likely to be genetically linked to isolates from divergent collection sites. Interpreting genetic linkages among C. difficile isolates requires an understanding of regional and strain-specific genetic diversity to avoid misattribution of genetic linkages to recent transmission.
Funding: None
Disclosures: None
Molecular Epidemiology of Community-Onset (CO), Community-Onset Healthcare-Associated (CO-HA) and Hospital-Onset (HO) Methicillin-Resistant Staphylococcus aureus (MRSA)
- Stephanie Thiede, Darjai Payne, Alla Aroutcheva, Michael Schoeny, Robert Weinstein, Evan Snitkin, Kyle Popovich
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s70-s71
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Previous work suggests an intermingling of community and hospital transmission networks driving the MRSA epidemic, but how those with CO-HA infections fit into the network remains unclear. We integrated epidemiologic data and whole-genome sequencing (WGS) from existing MRSA clinical isolates to determine whether there were distinguishable features of CO-HA MRSA infections that could guide interventions. Methods: We examined 955 existing clinical MRSA isolates from 2011 to 2013 from patients at Cook County Health, the major public healthcare network in Chicago, Illinois. We performed electronic and manual chart review to ascertain community (eg, illicit drug use, incarceration history) and healthcare exposures and comorbidities. WGS was performed on all sequences, and sequences were typed with multilocus sequence typing (MLST). We assessed the distribution of epidemiological factors and sequence type (ST) across onset type. Results: Infections were more frequent in males (70%); 61% of individuals with infection were African American and 21% were Hispanic. Overall, wound infections were the most common (81%) followed by blood (7%) and respiratory (6%). 82% of infections were ST8 (most USA300), 8% were ST5 (USA100) and 10% were other STs (Fig. 1a). Using standard epidemiologic definitions, we identified 523 CO, 295 CO-HA, and 137 HO infections. USA300 infections were common across CO, CO-HA, and HO categories, whereas USA100 was more frequently observed among CO-HA and HO. Current illicit drug use and history of incarceration—factors typically associated with CO-MRSA—were observed among both CO-HA and HO infections. 38% of CO-HA and 36% of HO had a history of MRSA infection or nasal colonization in the prior 6 months. As expected, 73% of CO-HA had a history of recent hospitalization, but this was also true for 44% of HO cases; points for intervention for both groups, especially CO-HA patients, include outpatient, inpatient, and ER care. Diabetes was common across categories, and HIV was more commonly observed among CO-HA cases (Fig. 1b). Conclusions: We characterized the genomic and epidemiologic features of CO-HA MRSA infections relative to CO and HO. By MLST and epidemiological analysis, CO-HA infections share similarities to both CO and HO. Although USA300 infections were the most common strain type, our findings highlight the need for WGS to discern relationships between individuals to understand the intermixing of healthcare and community networks for CO-HA infections. Higher resolution genomic analysis may help guide whether interventions need to be at hospital discharge or in the community to have the most impact on decreasing CO-HA MRSA infections.
Funding: Funding: from CDC Broad Agency Announcement: Genomic Epidemiology of Community-Onset Invasive USA300 MRSA Infections; Contract ID: 75D30118C02923
Disclosures: None
Cohorting KPC+ Klebsiella pneumoniae (KPC-Kp)–Positive Patients—A Genomic Exposé of Cross-Colonization Hazards
- Shawn Hawken, Mary Hayden, Karen Lolans, Rachel Yelin, Robert Weinstein, Michael Lin, Evan Snitkin
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s172-s173
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Long-term acute-care hospitals (LTACHs) are disproportionately burdened by multidrug-resistant organisms (MDROs) like KPC-Kp. Although cohorting KPC-Kp+ patients into rooms with other carriers can be an outbreak-control strategy and may protect negative patients from colonization, it is unclear whether cohorted patients are at unintended increased risk of cross colonization with additional KPC-Kp strains. Methods: Cohorting KPC-Kp+ patients at admission into rooms with other positive patients was part of a bundled intervention that reduced transmission in a high-prevalence LTACH. Rectal surveillance culturing for KPC-Kp was performed at the start of the study, upon admission, and biweekly thereafter, capturing 94% of patients. We evaluated whole-genome sequencing (WGS) evidence of acquisition of distinct KPC-Kp strains in a convenience sample of patients positive for KPC-Kp at study start or admission to identify plausible secondary KPC-Kp acquisitions. Results: WGS multilocus sequence type (MLST) strain variability was observed among the 452 isolates from the 254 patients colonized by KPC-Kp (Fig. 1). Among the 32 patients who were positive at the beginning of the study or admission and had a secondary isolate collected at a later date (median, 89 days apart, range, 2–310 days), 17 (53%) had secondary isolates differing by MLST from their admission isolate. Although 60% of the KPC-Kp in the study was ST258, there was substantial genomic variation within ST258 isolates from the same patient (range, 0–102 genetic variants), suggesting multiple acquisitions of distinct ST258 isolates. Among the 17 patients who imported ST258 and had ST258 isolated again later, 11 (65%) carried secondary isolates genetically closer to isolates from other importing patients than to their own ST258 (Fig. 2). Examination of spatiotemporal exposures among patients with evidence of multiple acquisitions revealed that 11 (65%) patients with multiple MLSTs shared a room with a patient who was colonized with an isolate matching the secondary MLST, and 6 (35%) patients who carried multiple distinct ST258 isolates shared a room with a patient who imported these closely related isolates prior to secondary acquisition. Conclusions: Half of patients who imported KPC-Kp and had multiple isolates available had genomically supported secondary acquisitions linked to roommates who carried the acquired strains. Although cohorting is intended to protect negative patients from acquiring MDROs, this practice may promote multiple strain acquisitions by colonized patients in the cohort, potentially prolonging the period of MDRO carriage and increasing time at risk of infection. Our findings add to the debate about single-patient rooms, which may be preferred to cohorts to minimize potential harms by reducing MDRO transmission.
Funding: None
Disclosures: None
4425 Anibiotic-Resistant Organism Acquisition in Nursing Facility Patients
- Joyce Wang, Marco Cassone, Kristen Gibson, Bonnie Lansing, Lona Mody, Evan Snitkin, Krishna Rao
-
- Journal:
- Journal of Clinical and Translational Science / Volume 4 / Issue s1 / June 2020
- Published online by Cambridge University Press:
- 29 July 2020, pp. 2-3
-
- Article
-
- You have access Access
- Open access
- Export citation
-
OBJECTIVES/GOALS: We investigated the association between gut microbiota features in newly admitted nursing facility (NF) patients and the acquisition of vancomycin-resistant Enterococcus (VRE) and/or resistant Gram-negative bacteria (rGNB) within 14 days. METHODS/STUDY POPULATION: Patients were recruited at 6 Michigan NFs from 09/16-08/18. VRE or rGNB colonization status was determined by culture swabs collected from multiple body sites at enrolment, day 7, and day 14. Our analysis focused on patients with no colonization at baseline, a perirectal swab collected at baseline, and at least one follow-up visit. The V4 hypervariable region of the 16S rRNA gene from bacterial DNA in each sample was PCR-amplified and sequenced on the MiSeq platform. Sequencing results were then processed with the mothur bioinformatics pipeline to classify bacterial taxa present in each sample. Taxa typically associated with the skin microbiota were removed. The primary outcome was acquisition of VRE and/or rGNB within 14 days. Exposures of interest included patient and microbiota characteristics. RESULTS/ANTICIPATED RESULTS: Among 61 patients, 18 (30%) acquired AROs within 14 days of enrolment (3 VRE, 13 rGNB, 2 both) (Table 1). The baseline microbiota features differed significantly in those who acquired a new ARO. Of the major 8 phyla found across samples, patients who acquired an ARO were depleted in the number of phyla present (5.74 ± 1.20 vs 5.06 ± 1.43; p = 0.037) (Fig. 1). The log10-transformed relative abundance of Enterococcus was enriched in patients who acquired an ARO (−0.32 ± 1.47) compared to those who did not (−1.68 ± 1.76; p = 0.021) (Fig. 2). Patients who did not acquire an ARO tended to harbour more butyrate-producing bacterial taxa and strict anaerobes, although the differences were not statistically significant (relative abundance of butyrate producer: 29.49 ± 22.09 vs 22.05 ± 17.76; anaerobes: 64.78 ± 23.54 vs 53.68 ± 27.61). DISCUSSION/SIGNIFICANCE OF IMPACT: Microbiota metrics calculated from perirectal samples are predictive of ARO acquisition. The clinical utility of perirectal samples thus warrants further assessment.
Cohorting KPC+ Klebsiella pneumoniae (KPC-Kp)–positive patients: A genomic exposé of cross-colonization hazards in a long-term acute-care hospital (LTACH)
- Shawn E. Hawken, Mary K. Hayden, Karen Lolans, Rachel D. Yelin, Robert A. Weinstein, Michael Y. Lin, Evan S. Snitkin
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue 10 / October 2020
- Published online by Cambridge University Press:
- 23 June 2020, pp. 1162-1168
- Print publication:
- October 2020
-
- Article
- Export citation
-
Objective:
Cohorting patients who are colonized or infected with multidrug-resistant organisms (MDROs) protects uncolonized patients from acquiring MDROs in healthcare settings. The potential for cross transmission within the cohort and the possibility of colonized patients acquiring secondary isolates with additional antibiotic resistance traits is often neglected. We searched for evidence of cross transmission of KPC+ Klebsiella pneumoniae (KPC-Kp) colonization among cohorted patients in a long-term acute-care hospital (LTACH), and we evaluated the impact of secondary acquisitions on resistance potential.
Design:Genomic epidemiological investigation.
Setting:A high-prevalence LTACH during a bundled intervention that included cohorting KPC-Kp–positive patients.
Methods:Whole-genome sequencing (WGS) and location data were analyzed to identify potential cases of cross transmission between cohorted patients.
Results:Secondary KPC-Kp isolates from 19 of 28 admission-positive patients were more closely related to another patient’s isolate than to their own admission isolate. Of these 19 cases, 14 showed strong genomic evidence for cross transmission (<10 single nucleotide variants or SNVs), and most of these patients occupied shared cohort floors (12 patients) or rooms (4 patients) at the same time. Of the 14 patients with strong genomic evidence of acquisition, 12 acquired antibiotic resistance genes not found in their primary isolates.
Conclusions:Acquisition of secondary KPC-Kp isolates carrying distinct antibiotic resistance genes was detected in nearly half of cohorted patients. These results highlight the importance of healthcare provider adherence to infection prevention protocols within cohort locations, and they indicate the need for future studies to assess whether multiple-strain acquisition increases risk of adverse patient outcomes.
A retrospective cohort study of antibiotic exposure and vancomycin-resistant Enterococcus recolonization
- Heather Y. Hughes, Robin T. Odom, Angela V. Michelin, Evan S. Snitkin, Ninet Sinaii, Aaron M. Milstone, David K. Henderson, Tara N. Palmore
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 4 / April 2019
- Published online by Cambridge University Press:
- 07 February 2019, pp. 414-419
- Print publication:
- April 2019
-
- Article
- Export citation
-
Objective:
In the National Institutes of Health (NIH) Clinical Center, patients colonized or infected with vancomycin-resistant Enterococcus (VRE) are placed in contact isolation until they are deemed “decolonized,” defined as having 3 consecutive perirectal swabs negative for VRE. Some decolonized patients later develop recurrent growth of VRE from surveillance or clinical cultures (ie, “recolonized”), although that finding may represent recrudescence or new acquisition of VRE. We describe the dynamics of VRE colonization and infection and their relationship to receipt of antibiotics.
Methods:In this retrospective cohort study of patients at the National Institutes of Health Clinical Center, baseline characteristics were collected via chart review. Antibiotic exposure and hospital days were calculated as proportions of VRE decolonized days. Using survival analysis, we assessed the relationship between antibiotic exposure and time to VRE recolonization in a subcohort analysis of 72 decolonized patients.
Results:In total, 350 patients were either colonized or infected with VRE. Among polymerase chain reaction (PCR)-positive, culture (Cx)-negative (PCR+/Cx−) patients, PCR had a 39% positive predictive value for colonization. Colonization with VRE was significantly associated with VRE infection. Among 72 patients who met decolonization criteria, 21 (29%) subsequently became recolonized. VRE recolonization was 4.3 (P = .001) and 2.0 (P = .22) times higher in patients with proportions of antibiotic days and antianaerobic antibiotic days above the median, respectively.
Conclusion:Colonization is associated with clinical VRE infection and increased mortality. Despite negative perirectal cultures, re-exposure to antibiotics increases the risk of VRE recolonization.








