3 results
Preventing measles transmission in ambulatory pediatric settings during peak respiratory viral season
- Katia Halabi, Tammie Hull, Randi Pfeffer, Payal Patel, Sarah King, Randal De Souza, Matthew Washam
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s119
-
- Article
-
- You have access Access
- Open access
- Export citation
-
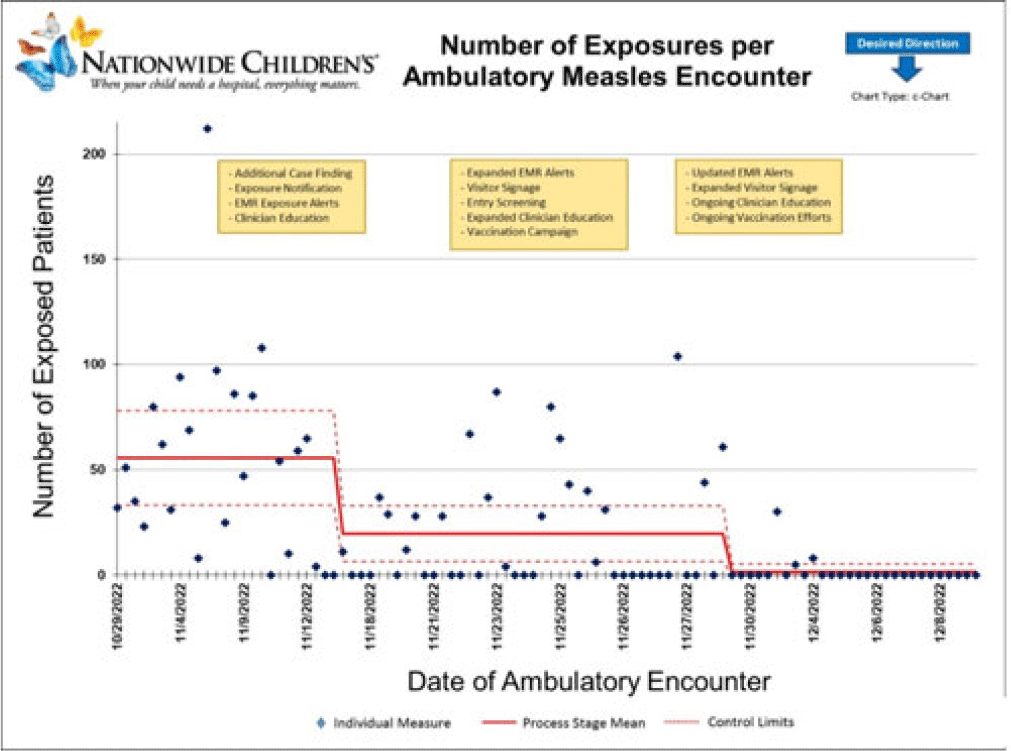
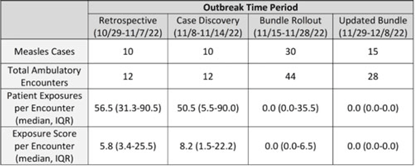
Background: Measles is a highly transmissible respiratory virus that presents with nonspecific prodromal symptoms followed by a characteristic cephalocaudal rash. In the prodromal phase, children with measles can be challenging to differentiate from children with other circulating respiratory viral infections. A measles outbreak in Central Ohio starting in October 2022 coincided with a national surge in children with viral respiratory infections which presented unique challenges in preventing healthcare transmission in the pediatric ambulatory setting. Methods: Following initial identification of presumed community transmission of measles in Central Ohio in November 2022, a multidisciplinary measles response team was convened at Nationwide Children’s Hospital (NCH) to prevent secondary healthcare transmission via rapid-cycle quality improvement. Prevention efforts were focused broadly across NCH ambulatory locations in Central Ohio, including the main campus and offsite emergency departments, regional urgent cares, and primary care network. Preliminary risk factors were identified via chart review of initial cases, which included vaccine status, ZIP code of residence, and known daycare or household exposure. These risk factors were used to guide an intervention bundle comprising enhanced screening at registration and triage, creation of electronic medical record alerts to identify at-risk patients, increased clinician education, and expanded community messaging. As the outbreak evolved, risk factors were updated, and interventions were adjusted to adapt response. Outcome metrics included total patient exposures as well as the relative exposure score. The exposure score was an internal metric derived using the vaccine status of exposed patients and ventilation at the site of exposure to assess likelihood of secondary cases occurring from an exposure. Results: In total, 65 patients with measles were seen at NCH facilities between October 29 and December 8, 2022. The outbreak response was divided into 4 periods: (1) cases identified retrospectively prior to first diagnosis (October 29–November 7, 2022), (2) initial case discovery (November 8–14, 2022), (3) implementation of prevention bundle (November 15–28, 2022), and (4) updates to the response (November 29–December 8, 2022). Ambulatory healthcare exposures and incidence of secondary cases decreased over the outbreak periods in response to implementation of the prevention bundle (Fig. and Table). Conclusions: An outbreak of measles occurring simultaneously with peak respiratory viral season presented challenges in early identification of suspected cases and mitigation of healthcare exposure. Transmission was effectively prevented following rapid deployment of a prevention bundle adjusted in real-time through rapid-cycle quality improvement. Ongoing longitudinal vaccination efforts are needed to sufficiently mitigate transmission risk in communities with under-vaccinated populations.
Disclosures: None
Successful diagnostic stewardship for Clostridioides difficile testing in pediatrics
- Katia C. Halabi, Barbara Ross, Karen P. Acker, Jean-Marie Cannon, Maria Messina, Diane Mangino, Krystal Balzer, Alexandra Hill-Ricciuti, Daniel A. Green, Lars F. Westblade, Christine M. Salvatore, Lisa Saiman
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 2 / February 2023
- Published online by Cambridge University Press:
- 15 June 2022, pp. 186-190
- Print publication:
- February 2023
-
- Article
- Export citation
-
Objective:
To reduce both inappropriate testing for and diagnosis of healthcare-onset (HO) Clostridioides difficile infections (CDIs).
Design:We performed a retrospective analysis of C. difficile testing from hospitalized children before (October 2017–October 2018) and after (November 2018–October 2020) implementing restrictive computerized provider order entry (CPOE).
Setting:Study sites included hospital A (a ∼250-bed freestanding children’s hospital) and hospital B (a ∼100-bed children’s hospital within a larger hospital) that are part of the same multicampus institution.
Methods:In October 2018, we implemented CPOE. No testing was allowed for infants aged ≤12 months, approval of the infectious disease team was required to test children aged 13–23 months, and pathology residents’ approval was required to test all patients aged ≥24 months with recent laxative, stool softener, or enema use. Interrupted time series analysis and Mann-Whitney U test were used for analysis.
Results:An interrupted time series analysis revealed that from October 2017 to October 2020, the numbers of tests ordered and samples sent significantly decreased in all age groups (P < .05). The monthly median number of HO-CDI cases significantly decreased after implementation of the restrictive CPOE in children aged 13–23 months (P < .001) and all ages combined (P = .003).
Conclusion:Restrictive CPOE for CDI in pediatrics was successfully implemented and sustained. Diagnostic stewardship for CDI is likely cost-saving and could decrease misdiagnosis, unnecessary antibiotic therapy, and overestimation of HO-CDI rates.
Successful Diagnostic Stewardship for Clostridioides difficile Testing in Pediatrics
- Katia Halabi, Lisa Saiman, Philip Zachariah, Karen Acker, Jean-Marie Cannon, Maria Messina, Diane Mangino, Krystal Balzer, Daniel Green, Christine Salvatore
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s394
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: As many as 40% of infants aged ≤12 months and 10%–28% of children aged 13–24 months are colonized by Clostridioides difficile. The IDSA and the SHEA recommend that testing should never be routinely recommended for infants ≤12 months of age and should not be routinely performed for children 1–2 years of age unless other causes are excluded. We report implementation of C. difficile diagnostic stewardship at 2 children’s hospitals. Methods: We implemented age-based restrictions for C. difficile testing at hospital A (∼200-bed, free-standing, children’s hospital) and hospital B (∼100-bed children’s hospital within a larger hospital). Both sites are part of the same multicampus institution, and both used nucleic acid amplification testing to detect C. difficile throughout the study. In May 2018, we implemented an electronic order set for C. difficile that provided alerts to avoid testing young infants and patients with recent use of laxatives, stool softeners, or enemas, but providers could order C. difficile testing at their discretion. In October 2018, we implemented a more restrictive diagnostic stewardship algorithm for C. difficile. No testing was allowed for infants aged ≤12 months. Approval pediatric infectious diseases staff was required to test children aged 13–24 months. Pathology resident approval was required to test children aged ≥24 months who had received laxatives, stool softeners, or enemas within ≤24 hours. Clinical microbiology laboratory supervisors reinforced rejection of nondiarrheal stool specimens for testing. Providers at both campuses were informed about the new testing guidelines by e-mail. We compared the number of tests sent and positive cases of healthcare facility-onset C. difficile (HO-CDI) by age strata before and after the implementation of the restrictive testing algorithm. Results: After the intervention, the number of tests in infants significantly declined; 2 infants aged ≤12 months and 4 infants aged 13–24 months were tested for C. difficile (Table). After the intervention, the number of tests per month declined at hospital A, as did the number of HO-CDI cases at both hospitals. Rejections of nondiarrheal stools significantly increased after the intervention (P < .001). Conclusions:C. difficile diagnostic stewardship for children was successfully implemented using a rule-based alert system in the electronic health record. This intervention was associated with a reduced number of tests sent and cases of HO-CDI. This strategy was cost-saving and prevented misdiagnosis, unnecessary antibiotic therapy, and overestimation of HO-CDI rates.
Funding: None
Disclosures: None





