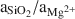3 results
Clay Mineral Diagenesis in Core KM-3 of Searles Lake, California
- R. L. Hay, S. G. Guldman, J. C. Matthews, R. H. Lander, M. E. Duffin, T. K. Kyser
-
- Journal:
- Clays and Clay Minerals / Volume 39 / Issue 1 / February 1991
- Published online by Cambridge University Press:
- 02 April 2024, pp. 84-96
-
- Article
- Export citation
-
Core KM-3 at Searles Lake, California, comprises 693.4 m of lacustrine sediments deposited over the past 3.2 m.y. The lake water evolved from moderately saline, slightly alkaline, and dominated by Na, Ca, Cl, SO4, and HCO3 + CO3 to a highly alkaline brine dominated by Na and CO, ions. Sediments are chiefly muds and evaporites. Montmorillonite and illite are the principal detrital clay minerals supplied to Searles Lake at present and probably during the late Pliocene and Pleistocene.
The drill core is divided into three diagenetic zones on the basis of clay-mineral reactions. The upper zone (0-291.1 m) contains authigenic Fe-illite, Mg-smectite, K-feldspar, and analcime, which average 60–70% of the <2-μm silicate fraction of mud samples. The principal silicate reactants are detrital montmorillonite and kaolinite, which have been largely consumed. The middle zone (291.1–541.6 m) also contains authigenic Fe-illite, Mg-smectite, K-feldspar, and analcime, but they form only 20 to 30% of the <2-μm silicate fraction, and considerable detrital montmorillonite and kaolinite remain unaltered. In muds of the lower zone (541.6–693.4 m), a small amount of clinoptilolite is the only authigenic silicate mineral identified, although authigenic montmorillonite probably coexists here as well. Vitric ash is the silicate reactant, and detrital clay minerals apparently remain unaltered in the lower zone.
Diagenetic zoning reflects the pore-water chemistry, in which pH may have been the most important parameter. Sediments of the upper zone were deposited in highly alkaline lakes of variable salinity, and sinking brines with a pH of 9.0–10.0 have saturated all sediments. Sediments of the middle zone were deposited in lakes of moderate to high salinity. The pH was overall lower than in the upper zone, although it probably exceeded 9.0, at least locally, during silicate diagenesis. Moderate salinity and a slightly alkaline pH (~ 7.5–8.0) are inferred for both the lake and pore water of the lower zone.
Oxygen-isotope values of authigenic Fe-illite, Mg-smectite, and K-feldspar, and phillipsite in the upper diagenetic zone reflect a high degree of evaporative concentration and presumably of salinity. Equilibrium water values calculated for 22°C from the oxygen-isotopic composition of authigenic phyllosilicates range from −3.5 to +1.9%, averaging −1.2% (SMOW). Higher salinities are suggested for K-feldspar, for which water values range from +1.8 to +4.8%, averaging +3.4‰ The water value for phillipsite is +0.3‰ By comparison, rainfall at Searles Lake has an average δ18O value of about −9.8%, and a brine sample has a value of +4.0‰
Mud samples of the middle and upper diagenetic zones tend to be rich in Fe-illite or Mg-smectite, but not in both, indicating that the two minerals are favored to some extent by different environments. Fe-illite seems to be generally favored by oxidizing conditions and probably by a playa environment, and Mg-smectite seems to be favored by reducing conditions and an open-water environment. One strati-graphic unit of the upper zone does not fit this pattern and contains abundant Fe-illite in sediments of a deep perennial lake.
Magnesium-Rich Clays of the Meerschaum Mines in the Amboseli Basin, Tanzania and Kenya
- R. L. Hay, R. E. Hughes, T. K. Kyser, H. D. Glass, J. Liu
-
- Journal:
- Clays and Clay Minerals / Volume 43 / Issue 4 / August 1995
- Published online by Cambridge University Press:
- 28 February 2024, pp. 455-466
-
- Article
- Export citation
-
The Sinya Beds of the Amboseli Basin in Tanzania and Kenya consist largely of carbonate rocks and Mg-rich clays that are intensely deformed where exposed in and near former meerschaum mines. The carbonate rocks consist of limestone and dolomite in Tanzania, but only dolomite has been identified in Kenya. Sepiolite and mixed-layered kerolite/stevensite (Ke/St) are subordinate constituents of the carbonate rocks. The carbonate rocks and overlying bedded sepiolite were deposited in a semiarid lake basin at the foot of the large volcano Kilimanjaro. Calcite and dolomite of the carbonate rocks have δ18O values 4–6‰ lower than calcite and dolomite of the late Pleistocene Amboseli Clays, suggesting that the Sinya Beds were deposited in the middle or early Pleistocene under a different climatic regime when meteoric water had lower δ18O values than at present.
Mg-rich clay minerals form veins and fill cavities in the Sinya Beds. The principal clay minerals are sepiolite and Ke/St, some of which contains substantial Al and Fe (Al-Ke/St). NEWMOD® modeling and other X-ray diffraction (XRD) data suggest that most of the Ke/St contains 25–50 percent kerolite layers, but minor amounts of kerolite-rich Ke/St are present in some samples. Illite with an inferred high content of Fe or Mg is a minor constituent of the samples with Al-Ke/St. The cavity-filling clays were chemically precipitated, as shown by field relationships and SEM study. The early-deposited clays of veins and cavities are principally Ke/St with minor sepiolite, and the latest clay is sepiolite (meerschaum), generally with minor Ke/St.
The δ18O values of cavity-filling Ke/St range from 22.5–25.6‰ and correlate with mineral composition, with the highest values associated with the highest content of stevensite and the lowest values with the highest content of kerolite. This relation suggests that high salinities favored stevensite and low salinities favored kerolite. δ18O values of sepiolite (meerschaum) fall in the middle of the range for Ke/St, suggesting that salinity was not the main control on sepiolite precipitation. High values of
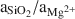 may have been a major factor in sepiolite precipitation.
may have been a major factor in sepiolite precipitation.Different mixtures of dilute ground water and saline, alkaline lake water in pore fluids may largely account for the differences in clay mineralogy of cavity-filling clays. Sepiolite is the dominant clay mineral in lacustrine sediments of the Amboseli Basin, and the cavity-filling sepiolite may reflect a high proportion of lake water. The low-Al Ke/St may have formed from fluids with a higher proportion of ground water. Detrital clay was very likely a factor in forming the Al-Ke/St, for which δ18O values suggest a saline environment.
Uraninite-Water Interactions in an Oxidizing Environment
- M. Fayek, T. K. Kyser, R. C. Ewing, M. L. Miller
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 465 / 1996
- Published online by Cambridge University Press:
- 03 September 2012, 1201
- Print publication:
- 1996
-
- Article
- Export citation
-
Exceptionally low δ 18O values of primary uraninite and pitchblende (i.e. -32 per mil to -19.5 per mil) from Proterozoic unconformity-type uranium deposits in Saskatchewan, Canada, in conjunction with theoretical uraninite-water oxygen isotope fractionation factors suggest that primary uranium mineralization is not in oxygen isotopie equilibrium with clays and silicates. The low δ 18O values have been interpreted to have resulted from the recrystallization of primary uranium mineralization in the presence of modern meteoric fluids having low δ 18O values of ca. -18 per mil. The absence of apparent alteration in many of the uraninite and pitchblende samples requires that the uranium minerals exchange oxygen isotopes with fluids, with only minor disturbances to their original chemical compositions and textures. However, experiments on the interaction between water and natural uraninites, from these deposits, and detailed electron micro-probe analyses of natural uraninite and pitchblende indicate that, in the presence of water, old uraninite rapidly alters to curite (Pb2U5O174H2O). The hydration of uraninite to curite releases uranium and calcium into solution and becquerelite (Ca(UO2)6O4(OH)6H2O) is precipitated. In the presence of Si-saturated waters, uranium silicate minerals, soddyite ((UO2)2(SiO4)2H2O) and kasolite (Pb(UO2)SiO4H2O are precipitated in addition to, curite and schoepite ((UO2)8O2(OH)12(H2O)12). The mineral paragenesis observed in these experiments is similar to sequences observed in oxidized zones in uranium deposits and UO2-water experiments. Therefore, it is unlikely that natural uraninite and pitchblende can simply exchange oxygen with an oxidizing fluid without a concomitant change in phase chemistry or structure, nor will oxidation of uraninite lead to the formation of U3O7. as predicted by theoretical calculations used in natural analogue studies for the disposal of high level nuclear waste.