Poster Presentation
Antibiotic Stewardship
Analysis of Recurrent Urinary Tract Infection Management in Outpatient Settings Reveals Opportunities for Antibiotic Stewards
- Marissa Valentine-King, Barbara Trautner, Roger Zoorob, George Germanos, Jason Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s34
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Studies of antibiotic prescribing choice and duration have typically excluded women with recurrent UTI (rUTI), yet the Infectious Disease Society of America (IDSA) UTI treatment guidelines are applicable to recurrent and sporadic cystitis. We sought to better understand prescribing practices among uncomplicated rUTI patients in terms of choice of drug, duration of therapy, and the risk factors for receiving guideline-discordant therapy. Methods: We performed a retrospective database study by extracting electronic health record data from adults seen at academic primary care, internal medicine, or urology practices between November 2016 and December 2018. Inclusion criteria included having ≥2 or ≥3 International Classification of Diseases Tenth Edition (ICD-10) cystitis codes recorded within a 6- or 12-month period, respectively. We excluded patients with ICD-10 codes indicating any structural or functional genitourinary comorbidities, interstitial cystitis, vaginosis, compromised immune systems, or pregnancy in the prior year. Patients were also excluded if they had signs or symptoms of pyelonephritis at presentation. Results: Overall, 232 patients presented for 597 outpatient visits. Most were married (52.2%), non-Hispanic white (62.9%), and female (92.2%), with a median age of 58 years (IQR, 41–68). Only 21% of visits with an antibiotic prescribed for treatment consisted of a first-line therapy agent prescribed for the recommended duration. In terms of antibiotic choice, these agents were prescribed in 58.4% of scenarios, which primarily included nitrofurantoin (37.8%) and trimethoprim-sulfamethoxazole (TMP-SMX) (20.3%). Guideline-discordant choices of fluoroquinolones (28.8%), and β-lactams (11.2%) were the second and third most commonly prescribed drug categories, respectively. Multinomial logistic regression identified age (OR, 1.02; 95% CI, 1.002–1.04) or having a telephone visit (OR, 3.17; 95% CI, 1.54–6.52) as independent risk factors for receiving a β-lactam. The duration exceeded the 3-day guideline recommendation in 87.6% of fluoroquinolones and 73% of TMP-SMX (73%) prescriptions, and 61% of nitrofurantoin prescriptions exceeded the recommended 5-day duration. Multiple logistic regression analysis revealed that seeking care at a urology clinic (OR, 2.81; 95% CI, 1.59–5.17) served as an independent factor for therapy duration exceeding guideline recommendations. Conclusions: This retrospective study revealed shortcomings in prescribing practices in the type and duration of therapy for rUTI. rUTI as well as sporadic UTI are important targets for outpatient antibiotic stewardship interventions.
Funding: This investigator-initiated research study was funded by Rebiotix Inc, a Ferring Company.
Disclosures: None
Blood Culture Utilization at Six Southeastern US Hospitals
- Bobby Warren, Rebekah Moehring, Michael Yarrington, Deverick Anderson, Christopher Polage
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s34-s35
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Group Name: Duke Center for Antimicrobial Stewardship and Infection Prevention
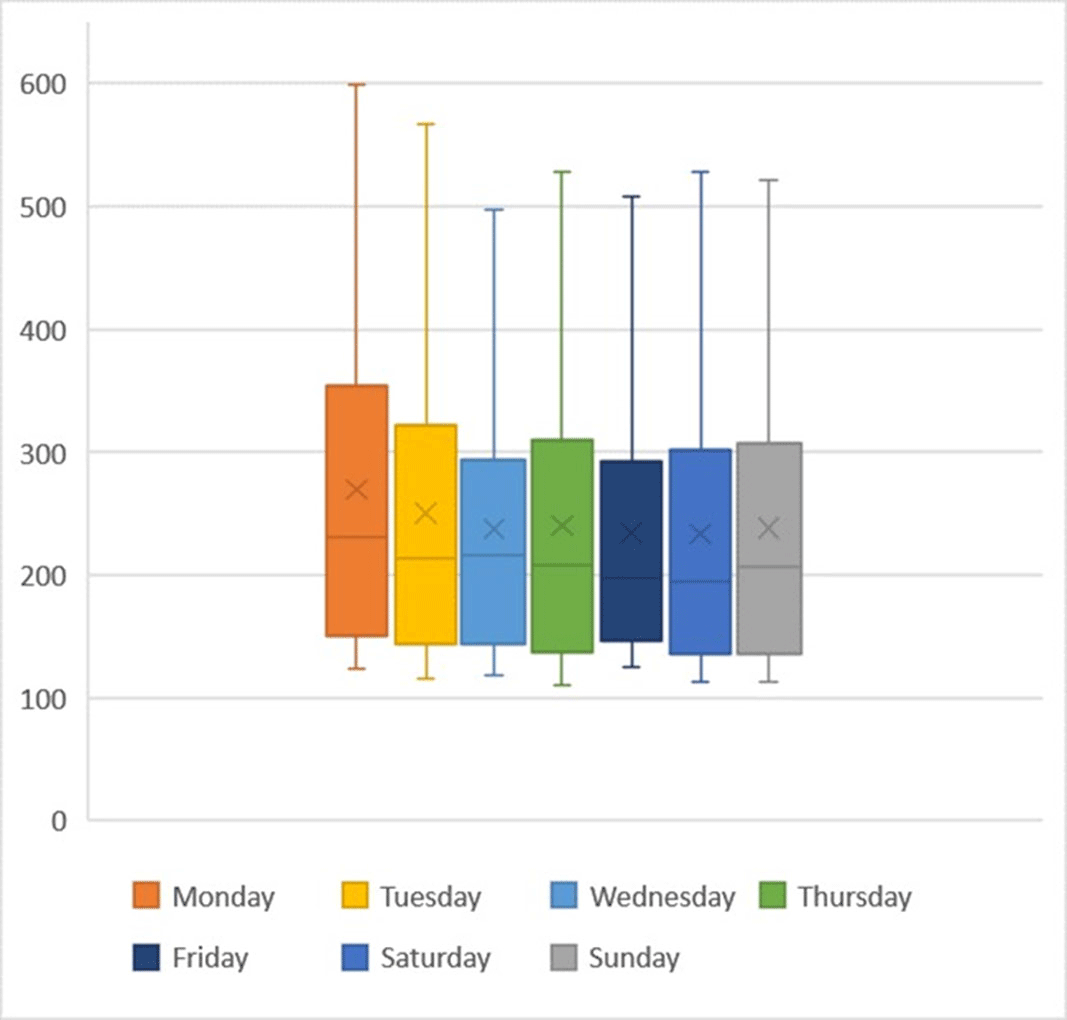
Background: Blood cultures are an essential diagnostic test, but over- and underutilization may cause harm. Methods: We analyzed blood culture utilization at 6 hospitals in the southeastern United States including 1 academic hospital (A) and 5 community hospitals (B–F) from May 2019 to April 2020. We measured blood culture utilization rate (BCUR) per 1,000 patient days and blood cultures per encounter. We counted blood cultures by laboratory accession number and measured utilization per 1,000 patient days and encounter. A likely contaminant was defined as 1 of 2 blood cultures collected in the same calendar day positive for a common skin commensal (CSC), as defined by the NHSN, and not identified from subsequent cultures. A likely pathogen was defined as a culture with a pathogen not on the CSC list or a CSC not meeting the contaminant definition. Hospital-level BCUR included samples for culture collected in the emergency department (ED) and inpatient areas divided by inpatient days. Results: The analysis included 117,897 blood cultures and 662,723 patient days with a median BCUR of 209.7 per hospital and median blood culture per encounter of 2 (Table 1). One community hospital (C) demonstrated a substantially higher BCUR than others. Cultures were frequently collected in the ED (54%; range, 36%–78%); most encounters with cultures in the ED were subsequently admitted to an inpatient unit (84%; range, 73%–89%). Higher BCURs were observed in intensive care and oncology units. The proportion of first blood cultures drawn after initiation of antibiotics was 6% (range, 3%–9%. Mondays had higher BCURs than other days of the week (Figure 1). The average BCUR by month was 176.1 (range, 164.3–181.4) with no seasonal patterns observed. Overall, 7.7% (range, 4.5%–9.1%) of blood cultures identified a likely pathogen and 2.1% (range, 1.3%–3.2%) identified a likely contaminant. The 3 hospitals with BCURs >200 also had contaminant rates >2% and >60% ED cultures. Conclusions: Blood culture utilization varied by hospital, unit, and day of the week. We observed higher rates of likely contaminants among hospitals with higher BCURs and ED culture rates. Comparisons may assist in identifying opportunities to optimize practice around blood-culture ordering and collection.
Funding: No
Disclosures: None
Table 1. 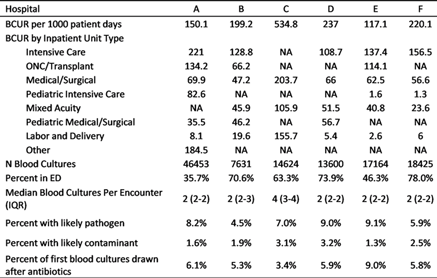
Figure 1.
Outpatient Antibiotic Use for Common Infectious Diagnoses: Patterns in Telehealth During the Emergence of COVID-19
- Brigid Wilson, Taissa Bej, Sunah Song, Janet M Briggs, Richard Banks, Robin Jump, Federico Perez, Ukwen Akpoji
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s35-s36
-
- Article
-
- You have access Access
- Open access
- Export citation
-
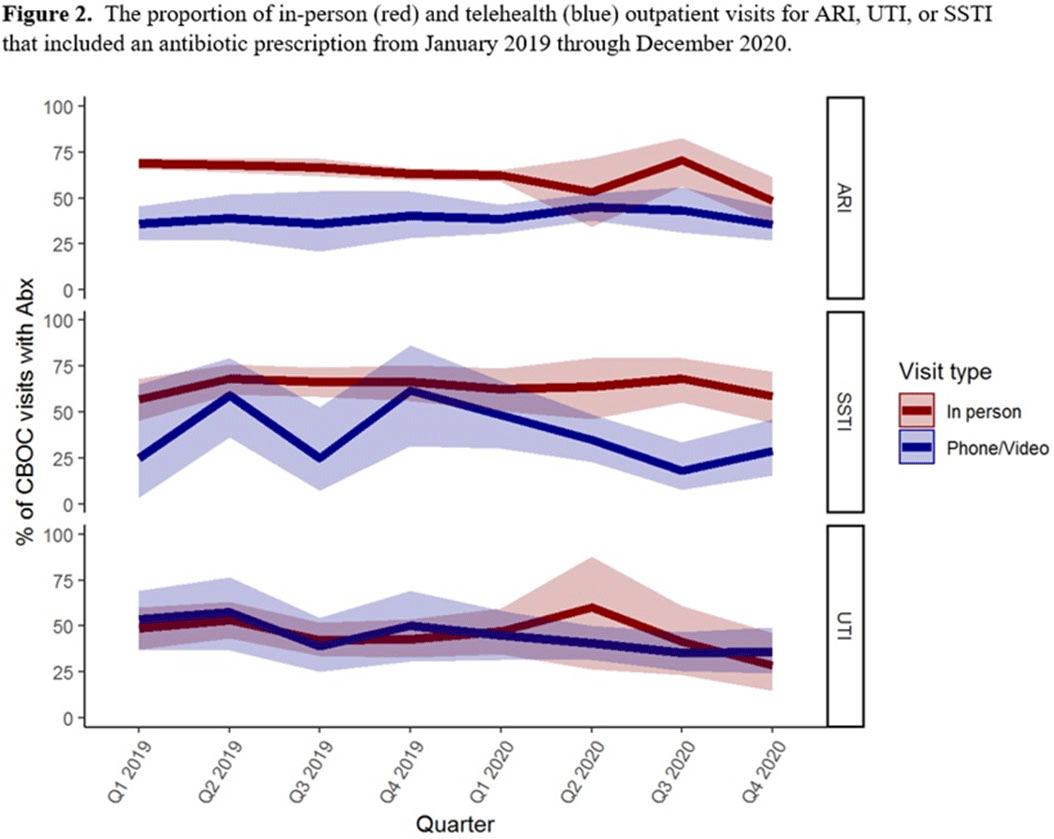
Background: The influence of increased use of telehealth during the emergence of COVID-19 on antibiotic prescriptions in outpatient settings is unknown. The VA Northeast Ohio Healthcare System has 13 community-based outpatient clinics (CBOCs) that provide primary and preventive care. We assessed changes in antibiotic prescriptions that occurred as care shifted from in-person to telehealth visits. Methods: Using VHA administrative databases, we identified all primary care CBOC visits between January 1, 2019, and December 31, 2020, that included a diagnosis for an acute respiratory infection (ARI), a urinary tract infection (UTI), or a skin or soft-tissue infection (SSTI), excluding visits with >1 of these diagnoses or with additional infectious diagnoses (eg, pneumonia, influenza). We summarized the proportion of telehealth visits and the proportion of patients prescribed antibiotics at quarterly intervals. We specifically assessed outpatient visits from April to December 2019 compared to the same months in 2020 to account for seasonality while analyzing diagnosis and antibiotic trends in the emergence of the COVID-19 pandemic. Results: The patients receiving care in April–December 2019 compared to April–December 2020 were similar (Table 1). From April through December 2019, 90% of CBOC primary care visits with a diagnosis for ARI, UTI, or SSTI were in-person, and antibiotics were prescribed at 63%, 46%, and 65% of visits in either modality, respectively (Figure 1). From April through December 2020, only 33% of CBOC primary care visits for ARI, UTI, and SSTI were in person, and antibiotics were prescribed at 46%, 38%, and 47% of visits in either modality, respectively. Comparing April–December in 2019 and 2020, the number of CBOC visits for ARI fell by 76% (2,152 visits to 509 visits), with a more modest decline of 20% and 35% observed for UTI and SSTI visits. In-person visits for ARIs and SSTIs were more likely than telehealth visits to result in an antibiotic prescription (Figure 2). Conclusions: Among the CBOCs at our healthcare system, an increase in the proportion of telehealth visits and a reduction in ARI diagnoses occurred after the emergence of COVID-19. In this setting, we observed a reduction in the proportion of visits for ARIs, UTIs, and SSTIs that included an antibiotic prescription.
Funding: Merck
Disclosures: None
Table 1. 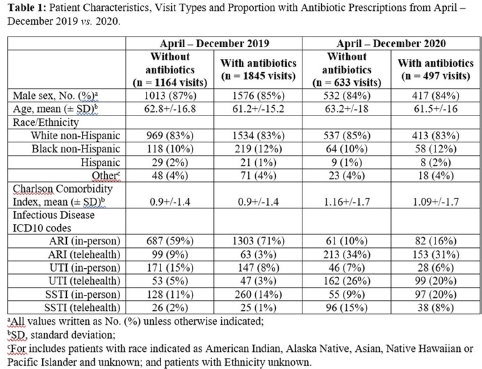
Figure 1.
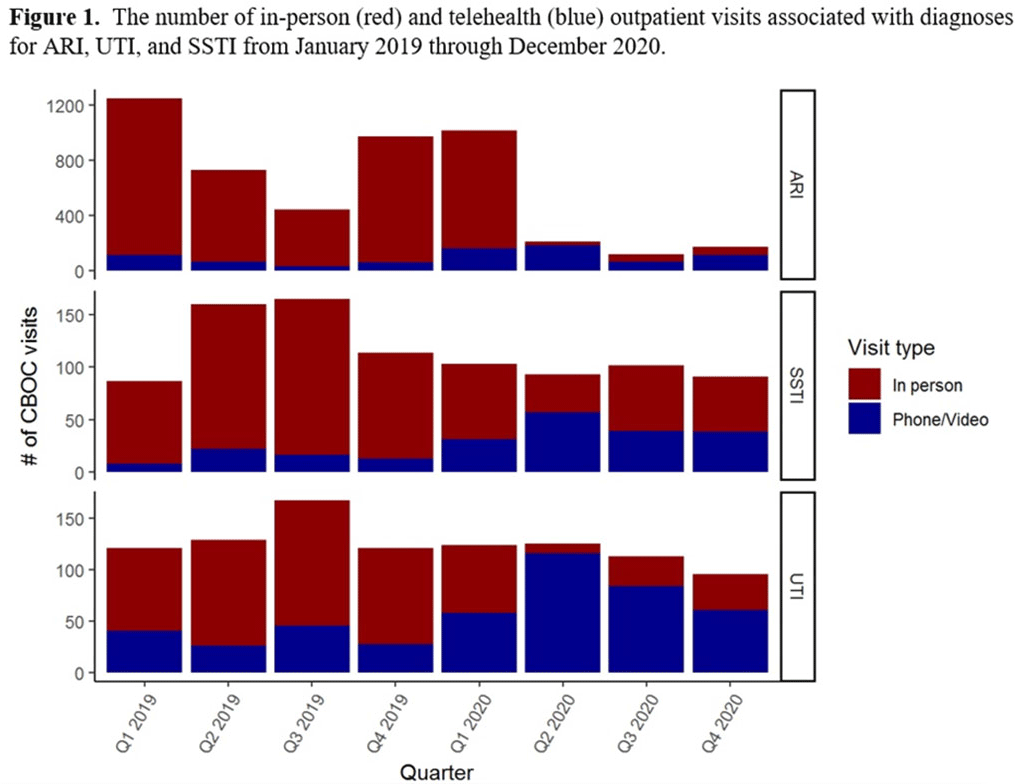
Figure 2.
Antimicrobial Stewardship Standards and Patient Safety: A Case Study in Blood Culture Contamination
- Connie Schaefer
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s36
-
- Article
-
- You have access Access
- Open access
- Export citation
-
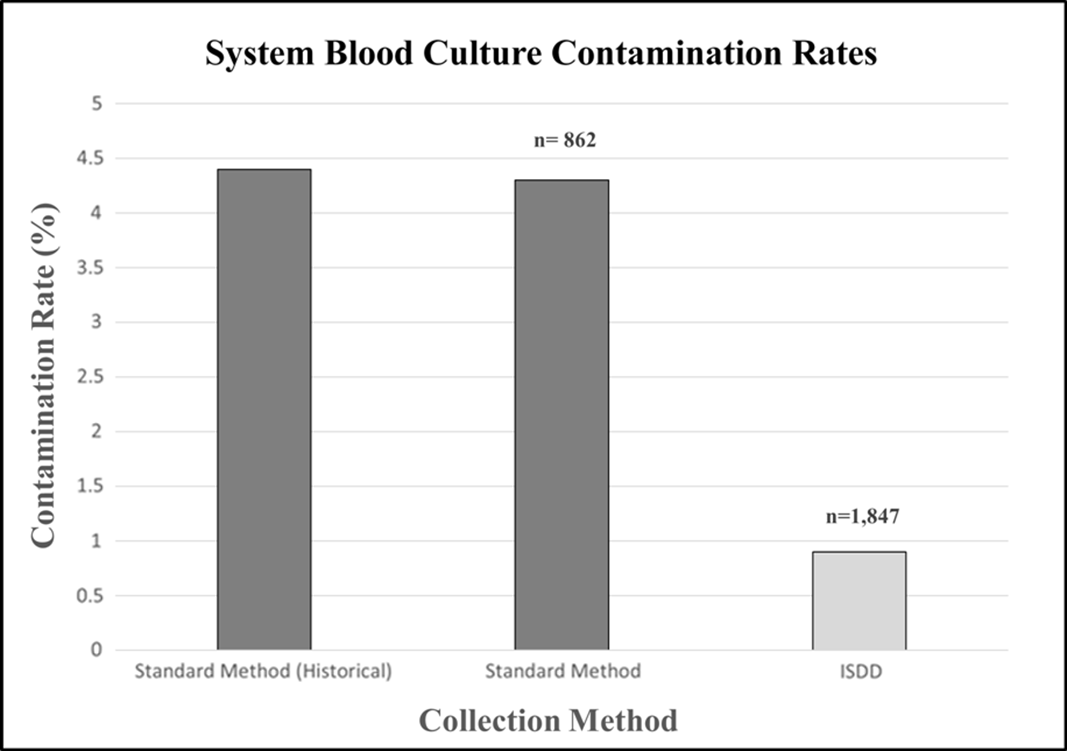
Background: Blood culture is a crucial diagnostic tool for healthcare systems, but false-positive results drain clinical resources, imperil patients with an increased length of stay (and associated hospital-acquired infection risk), and undermine global health initiatives when broad-spectrum antibiotics are administered unnecessarily. Considering emerging technologies that mitigate human error factors, we questioned historically acceptable rates of blood culture contamination, which prompted a need to promote and trial these technologies further. In a 3-month trial, 3 emergency departments in a midwestern healthcare system utilized an initial specimen diversion device (ISDD) to draw blood cultures to bring their blood culture contamination rate (4.4% prior to intervention) below the 3% benchmark recommended by the Clinical & Laboratory Standards Institute. Methods: All emergency department nursing staff received operational training on the ISDD for blood culture sample acquisition. From June through August 2019, 1,847 blood cultures were drawn via the ISDD, and 862 were drawn via the standard method. Results: In total, 16 contamination events occurred when utilizing the ISDD (0.9%) and 37 contamination events occurred when utilizing the standard method (4.3%). ISDD utilization resulted in an 80% reduction in blood culture contamination from the rate of 4.4% rate held prior to intervention. Conclusions: A midwestern healthcare system experienced a dramatic reduction in blood culture contamination across 3 emergency departments while pilot testing an ISDD, conserving laboratory and therapeutic resources while minimizing patient exposure to unnecessary risks and procedures. If the results obtained here were sustained and the ISDD utilized for all blood culture draws, nearly 400 contamination events could be avoided annually in this system. Reducing unnecessary antibiotic use in this manner will lower rates of associated adverse events such as acute kidney injury and allergic reaction, which are possible topics for further investigation. The COVID-19 pandemic has recently highlighted both the importance of keeping hospital beds available and the rampant carelessness with which broad-spectrum antibiotics are administered (escalating the threat posed by multidrug-resistant organisms). As more ambitious healthcare benchmarks become attainable, promoting and adhering to higher standards for patient care will be critical to furthering an antimicrobial stewardship agenda and to reducing treatment inequity in the field.
Funding: No
Disclosures: None
Figure 1.
Minimal Mortality Among Veterans with Urine Cultures Positive for Group B Streptococcus
- Nicole Mongilardi, Robin Jump, Federico Perez, Taissa Bej, Janet M Briggs, Richard Banks, Brigid Wilson, Sunah Song
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s36-s37
-
- Article
-
- You have access Access
- Open access
- Export citation
-
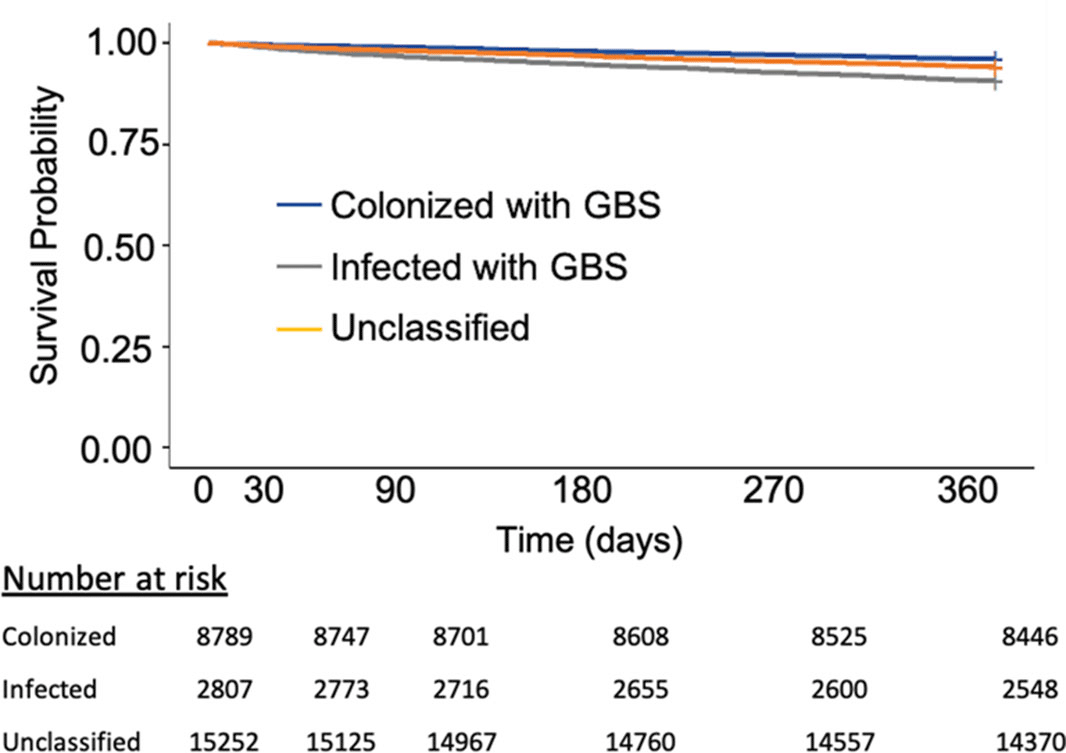
Background: Group B Streptococcus (GBS) can cause life-threating invasive infections, yet GBS is also a normal component of the intestinal and genitourinary tract. Although it is regarded as a potential urinary pathogen, the morbidity and mortality associated with recovery of GBS from urine cultures of nonpregnant adults is not well understood. We evaluated characteristics and mortality among nonpregnant adults with urine cultures that grew GBS. Methods: Using administrative data from the Veterans’ Healthcare Administration (VHA), we conducted a retrospective cohort study of VA healthcare system users from January 1, 2008, through December 31, 2017, with monomicrobial urine cultures growing ≥100,000 colony-forming units of GBS. Urinary tract infection (UTI) cases were defined as urinalysis positive for leukocyte esterase and pyuria (≥10 white blood cells), an International Classification of Diseases (ICD) code for UTI, and an antibiotic prescription. Cases with colonization were defined as negative for leukocyte esterase and pyuria, no ICD code for UTI, and no antibiotic prescription. Cases not meeting either definition were deemed unclassifiable. We compared demographics, comorbidities, and all-cause mortality among these 3 groups. Results: Over the 10-year study period, 26,848 veterans had 30,740 urine cultures positive for GBS. Applying the definitions above, there were 2,807 cases of infection, 8,789 cases of colonization, and 15,252 cases that were unclassifiable. Patients with a GBS UTI were slightly older compared to those who were colonized, with a higher Charlson comorbidity index and greater burden of chronic renal disease (Table 1). Individuals with infection versus colonization had 30-day mortality rates of 1% and 0%, respectively, and 1-year mortality rates of 9% and 4%, respectively (Figure 1). Conclusions: The association of a greater burden of illness among veterans who met our definition of UTI compared to colonization might be more reflective of providers’ responses to patients with chronic medical conditions rather than a difference in GBS as a cause of UTI. Overall, the prospect of a urine culture that grows GBS does not appear to be associated with adverse long-term outcomes.
Funding: No
Disclosures: None
Table 1. 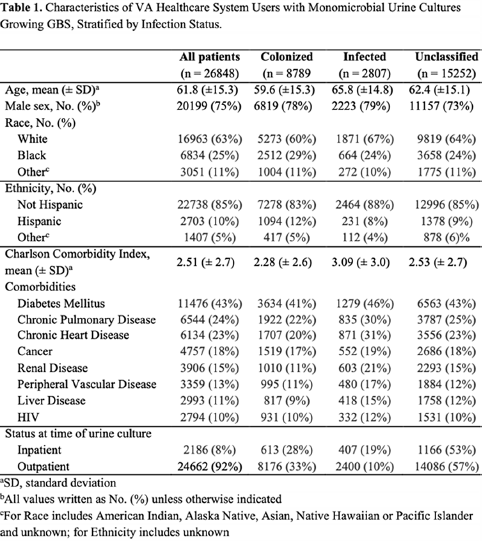
Figure 1.
Assessing Baccalaureate Nursing Students’ Antibiotic Stewardship Knowledge Using Virtual Standardized Patient Simulations
- Mary Lou Manning, Monika Pogorzelska-Maziarz, David Jack, Lori Wheeler
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s37
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: According to the Centers for Disease Control and Prevention, the single most important factor leading to the development of antibiotic resistance (AMR) is the use of antibiotics. Studies indicate that up to 50% of hospitalized patients receive at least 1 antibiotic, half of which are inappropriate. The outpatient setting accounts for >60% of antibiotic use and over half of these prescriptions are inappropriate. Antibiotic stewardship programs improve appropriate antibiotic use, reduce AMR, decrease complications of antibiotic use, and improve patient outcomes. Building a nursing workforce with necessary AMR and antibiotic stewardship knowledge and skill is critical. Nursing graduates can translate knowledge into practice, promoting the judicious use of antibiotics to keep patients safe from antibiotic harm. Methods: Third-year baccalaureate nursing students enrolled in a fall 2020 health promotion course at an urban university affiliated with an academic medical center participated. Students received a 3-hour lecture on antibiotics, AMR and antibiotic stewardship nursing practices and actively engaged in antibiotic stewardship simulations using standardized patient (SP) encounters. The SP participants were specifically trained for these activities. Simulations included a 30-minute brief before and a 60-minute briefing after the activities. All activities occurred via video conferencing. Case scenarios, developed by the authors, focused on penicillin-allergy delabeling of an adolescent prior to elective surgery and appropriate use of antibiotics in managing pediatric urinary tract infections and acute otitis media (AOM). Before-and-after tests were used to assess the impact on AMR and antibiotic stewardship knowledge. Results: Over a period of 4 days, all enrolled students (n = 165) participated in 1 three-hour virtual simulation session. Using Zoom video conferencing with multiple breakout rooms, the activities were easily managed. During the simulations, students often struggled with reading an antibiogram and applying the concept of “watchful waiting” in AOM management. Significant differences were found in before-and-after test results, with significant improvement in students’ general and specific knowledge and awareness of antibiotics (P < .01). During the debriefing sessions, students reported increased awareness related to their role in advancing the judicious use of antibiotics. Conclusions: Initially, we planned to conduct in-person SP simulations. Due to the COVID-19 pandemic, faculty and students demonstrated remarkable flexibility and resilience as we successfully converted to a virtual format. Virtual lecture and SP simulations, followed by debriefing, was an effective approach to educate baccalaureate nursing students about AMR and their role in antibiotic stewardship. Areas for course content improvement were identified.
Funding: No
Disclosures: None
How the COVID-19 Pandemic Affected Antimicrobial Prescribing Practices at a Tertiary-Care Healthcare System in Detroit, Michigan
- Angela Beatriz Cruz, Jennifer LeRose, Avnish Sandhu, Teena Chopra
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s37-s38
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Inappropriate antimicrobial use continues to threaten modern medicine. The ongoing pandemic likely exacerbated this problem because COVID-19 presents similarly to bacterial pneumonia, confusion exists regarding treatment guidelines, and testing turnaround times (TATs) are slow. Our primary object was to quantify antimicrobial use changes during the pandemic to rates before the crisis. A subanalysis within the COVID-19 cohort was completed based on SARS-CoV-2 status. Methods: The pre–COVID-19 period was January–May 2019 and the COVID-19 period was January–May 2020. Subanalyses were used to explore differences in antibiotics use between persons not under investigation (non-PUIs), SARS-CoV-2–negative PUIs, and SARS-CoV-2–positive PUIs. Non-PUI patients were those without respiratory symptoms and/or fever. The χ2 and Wilcoxon signed rank-sum tests were used for analysis. Results: During the 2019 and 2020 study periods, 7,909 and 7,283 patients received >1 antimicrobial, respectively (Figure 1). Overall, antibiotic therapy per 1,000 patient days increased from 633.1 before COVID-19 to 678.5 during COVID-19, a 7.2% increase (Table 1). Notably, broad-spectrum respiratory antibiotics demonstrated a significant increase between pre–COVID-19 and COVID-19 cohorts (p < 0.001). Of the 7,283 patients within the COVID-19 cohort, 34.7% (n = 2,532) were PUI and 13.8% (n = 1,002) of these patients tested SARS-CoV-2 positive. Again, broad-spectrum respiratory antibiotics use was significantly increased for COVID-19 patients (p < 0.001). Of note, the proportion of patients receiving respiratory antibiotics steadily decreased over time (R2 = 0.99). Conclusions: There was a significant increase in antibiotic use during the COVID-19 pandemic. Encouragingly, antimicrobial use decreased over time, likely due to (1) faster TATs, (2) real-time education to clinicians and subsequent de-escalation of unnecessary antimicrobials, and (3) development of treatment guidelines as new research emerged.
Funding: No
Disclosures: None
Figure 1.
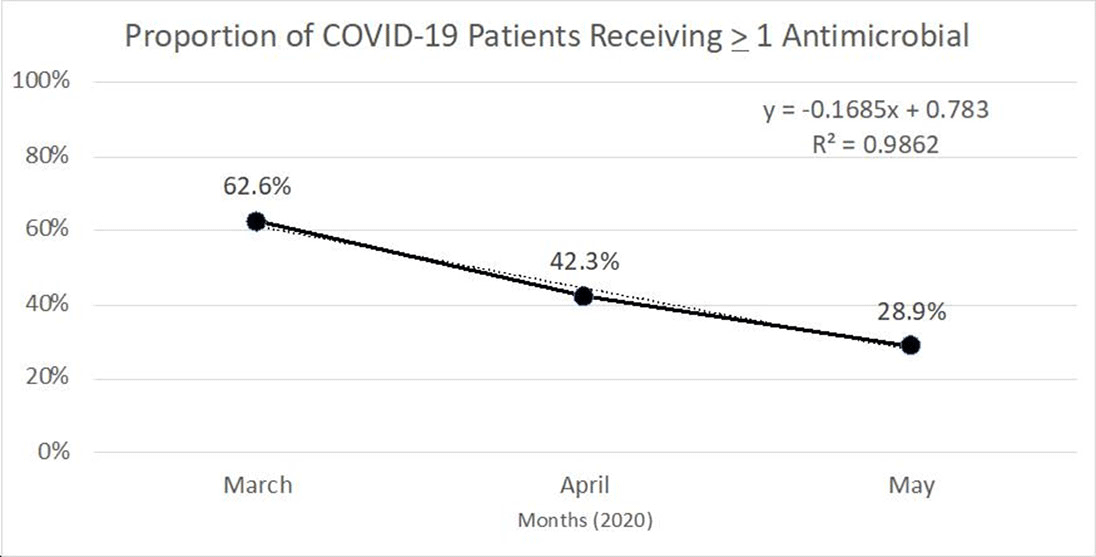
Table 1. 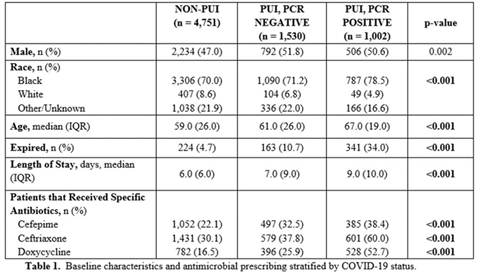
Evaluation of Penicillin Allergy Prevalence and Antibiotic Prescribing Patterns for Patients within the Emergency Department
- Ashlyn Norris, Lindsay Daniels, Nikolaos Mavrogiorgos, Kalynn Northam, Mildred Kwan, Gary Burke, Renae Boerneke
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s38
-
- Article
-
- You have access Access
- Open access
- Export citation
-
As the point of entry into healthcare for many patients, the emergency department (ED) is an ideal setting in which to assess penicillin (PCN) allergies. An estimated 10% of the United States population has a reported PCN allergy; however, few studies have evaluated the prevalence and impact of PCN allergies on antibiotic selection within the ED. Patients with a documented PCN allergy are more likely to be exposed to costly alternative broad-spectrum antibiotics that have higher rates of adverse events, including C. difficile infections. We sought to determine the prevalence of PCN allergies within the UNC Medical Center ED. Key secondary outcomes included the percentage of patients with a documented PCN allergy who (1) received alternative antibiotics (carbapenems, aztreonam, fluoroquinolones, clindamycin, vancomycin), (2) received β-lactam antibiotics and experienced an allergic reaction during their ED visit, and/or (3) had received a β-lactam antibiotic during a past hospitalization or ED visit without their chart being appropriately updated. A retrospective evaluation included patients aged >18 years with a documented PCN allergy who were discharged from the ED between January 1, 2017, and December 31, 2019. Over the study period, there were 14,635 patient encounters with a documented PCN allergy that comprised 8,573 unique patients. The prevalence of PCN allergies was 14.3% for all ED encounters. PCN allergy–labeled patients received alternative antibiotics in 59.4% of ED encounters in which antibiotics were prescribed. Of the 454 β-lactam antibiotics (62 penicillins, 380 cephalosporins, 12 carbapenems) administered to PCN allergy-labeled patients within the ED, there were zero allergic reactions. Also, 18.6% of PCN allergy-labeled patients had received and tolerated a β-lactam antibiotic during prior hospitalizations or ED visits (1.7% penicillins, 14.4% cephalosporins, 2.6% carbapenems) without appropriate updated documentation to reflect β-lactam antibiotic tolerance. These findings confirm the utilization of non–β-lactam antibiotics in PCN allergy-labeled patients, highlighting the importance of accurate and updated allergy documentation in the electronic medical record. These findings also demonstrate the need for improved allergy documentation and protocols to proactively assess penicillin allergy labels while in the ED.
Funding: No
Disclosures: None
Impact of an Inpatient Nurse-Initiated Penicillin Allergy Delabeling Questionnaire
- Lauren Dutcher, Hilary Bediako, Christina Harker, Aditi Rao, Kristen Sigafus, Keith Hamilton, Olajumoke Fadugba
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s38-s39
-
- Article
-
- You have access Access
- Open access
- Export citation
-
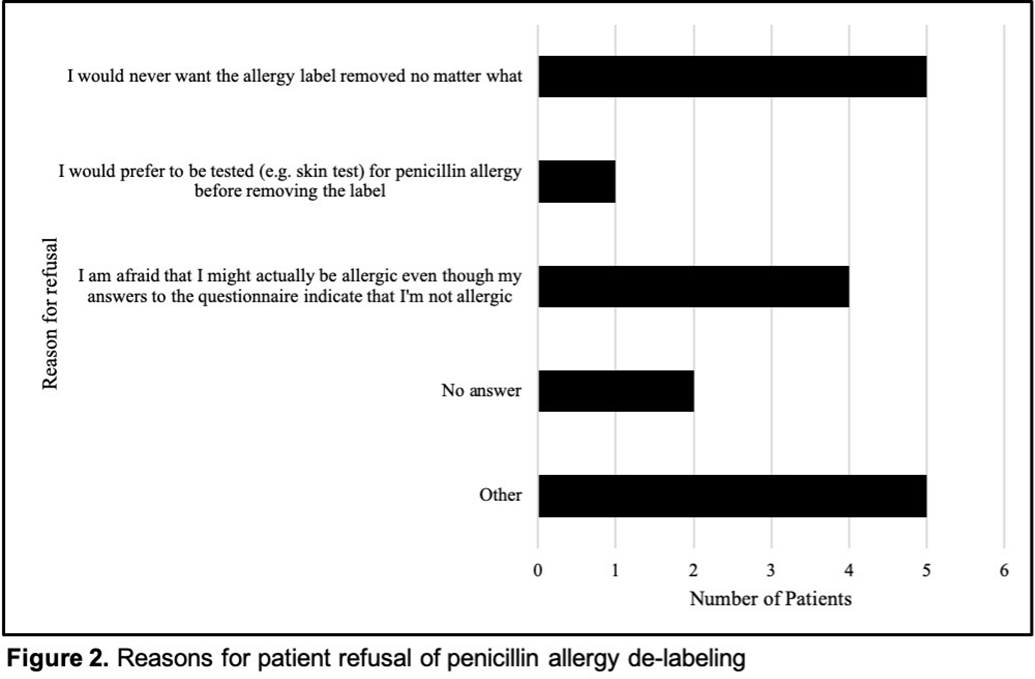
Background: Penicillin allergy is the most common drug allergy, with ~10% of all patients in the United States reporting a penicillin allergy. A penicillin allergy label is associated with the use of inappropriate or broad-spectrum antibiotics, worse patient outcomes, increased bacterial resistance, and increased healthcare costs, yet no studies have explored the unique role nurses may play in allergy delabeling through history taking as a part of broader antimicrobial stewardship efforts. Here, we describe the impact of using an inpatient nurse-initiated penicillin-allergy questionnaire. Methods: We implemented a nurse-driven intervention focused on penicillin allergy delabeling in inpatient noncritical care units (surgery, neurology, medicine, oncology, and cardiovascular medicine) at an academic hospital from July 9, 2019, to July 24, 2020. Patients with a penicillin allergy listed in the electronic health record (EHR) were identified and invited to participate. The intervention consisted of a questionnaire administered by nurses who elicited details of penicillin allergy history. If a patient was deemed eligible for penicillin allergy removal, nurses requested approval from both the patient as well as a physician member of the study team. Results: In total, 306 patients with a penicillin allergy label were identified in the EHR, of whom 242 patients were eligible for and agreed to participate in the delabeling interview (Figure 1). Of the 34 (14%) of 242 patients potentially eligible for delabeling by the questionnaire based on their history, the study physicians agreed with delabeling for 23 (68%) of 34 patients. Of these 34 patients, 18 (53%) agreed with delabeling (pending physician approval), and 16 (47%) of these 34 patients were ultimately delabeled. For those who declined delabeling, never wanting the label removed under any circumstance and uncertainty about accuracy of the survey results were common reasons for refusal (Figure 2). Additionally, for the 13 patients who refused delabeling, 9 patients did not want or were unsure about following up with an allergy specialist. Conclusions: The nurse-driven penicillin-allergy delabeling questionnaire is a no-cost intervention that can successfully identify patients to delabel. In this study, this measure resulted in the removal of 16 (7%) of 242 penicillin allergy labels. However, patients frequently opted to keep penicillin allergy labels, expressing uncertainty and fear of removal. Future work should explore optimal methods to engage nurses and patients in allergy delabeling, as well as the impact on antibiotic use and patient outcomes.
Funding: No
Disclosures: None
Figure 1.
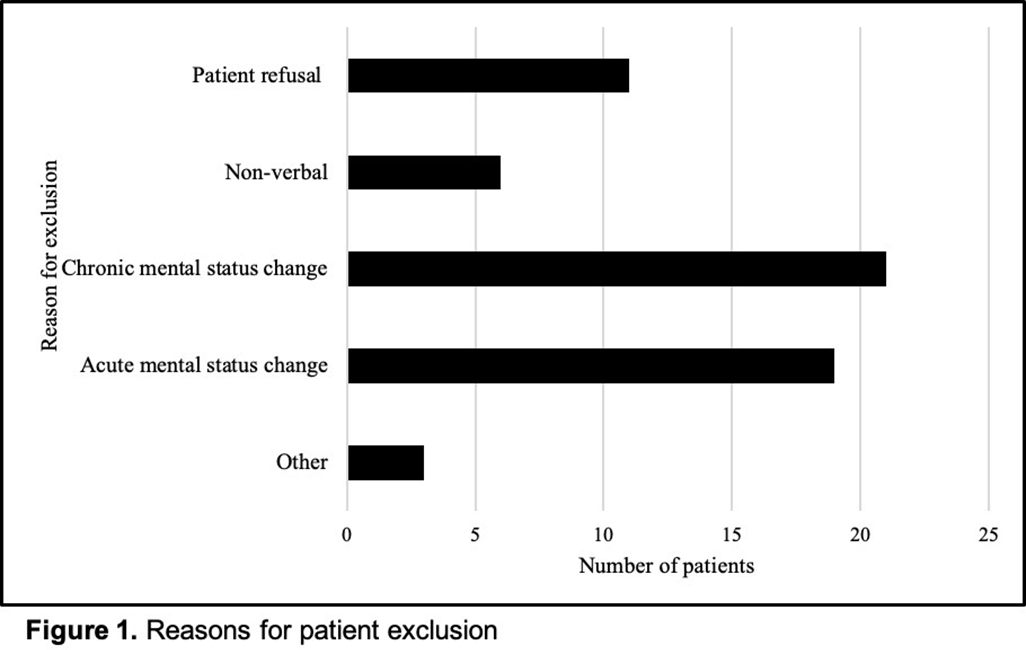
Figure 2.
Evaluation of Penicillin Allergies and an Allergy Assessment Pilot in the Emergency Department
- Ashlyn Norris, Kalynn Northam, Lindsay Daniels, Mildred Kwan, Gary Burke, Nikolaos Mavrogiorgos, Renae Boerneke
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s39
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Penicillin (PCN) allergy is one of the most frequently reported medication allergies, with ~10% of the US population reporting a PCN allergy. However, studies have shown that only 1% of the US population have a true IgE-mediated reaction to PCN. Delabeling and appropriately updating patient allergy profiles could decrease the use of alternative broad-spectrum antibiotics, rates of infectious complications [C. difficile, methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE)], antibiotic resistance, and overall healthcare cost. The emergency department (ED) is an important setting in which to assess PCN allergies and to delabel patients when appropriate because there are >130 million ED visits in the United States each year. We sought to determine the percentage of PCN allergy–labeled patients who could be delabeled through a PCN allergy assessment interview in an ED. Key secondary outcomes included the percentage of interviewed patients who could not be delabeled based on history alone but would be eligible for an amoxicillin oral challenge or a PCN skin test (PST). A prospective PCN allergy assessment pilot was performed for patients aged >18 years presenting to the UNC Medical Center ED between December 1 and December 17, 2020, with a documented PCN allergy. A pharmacist conducted penicillin allergy assessments on a convenience sample of patients presenting to the ED between 8 a.m. and 3 p.m. on weekdays. Based on patients’ reported and documented histories, charts were updated with the most accurate information and allergies were delabeled if appropriate. In total, 95 patients were assessed; 62 (65.3%) were interviewed and 15 (24.2%) were delabeled. In addition, 26 patients (41.9%) were deemed eligible for an oral amoxicillin challenge, 19 (30.6%) qualified for a PST, and 2 (3.2%) patients did not qualify for further assessment due to having a an IgE-mediated reaction in the past 5 years. Of the 15 patients who were delabeled, 6 (40.0%) received antibiotics during their admission: 4 (73.3%) of those patients received a penicillin and 2 (36.7%) received a cephalosporin, all without adverse reactions. Patient assessments took ~20 minutes to complete, including chart review, patient interview, and postinterview chart updating. The results from this pilot study demonstrate the impact of performing PCN allergy assessments in ED. Interdisciplinary opportunities should be explored to develop processes that will improve the efficiency and sustainability of PCN allergy assessments within the ED to allow this important stewardship intervention to continue.
Funding: No
Disclosures: None
Associations Between Patient Neighborhood Characteristics and Inappropriate Antimicrobial Use
- Joseph Engeda, Jane Kriengkauykiat, Erin Epson
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s39
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antimicrobials are among the most commonly prescribed medications in US hospitals; an estimated 50% of hospitalized patients receive an antimicrobial. Research has shown that antimicrobial prescriptions to vary by patient- and hospital-level factors; however, disparities by patient neighborhood characteristics have not been examined. We evaluated associations between hospital and neighborhood indicators of socioeconomic status (SES) and antimicrobial use (AU) for gram-positive bacterial infections (GPBs), and broad-spectrum use for community-acquired infections (BSCAs) and hospital-onset infections (BSHOs). Methods: This analysis was conducted among 86 acute-care hospitals in California that submitted AU data via the NHSN in 2019. Hospital-level AU was measured as standardized antimicrobial administration ratios (SAARs) calculated by dividing observed antimicrobial use by risk-adjusted predicted antimicrobial use for GPB, BSCA, and BSHO antimicrobial groupings and categorized as binary (>1 or <1); SAARs >1 indicate potential inappropriate prescribing. California Office of Statewide Health Planning and Development 2018 data were used to obtain hospital characteristics and patient age, race or ethnicity, insurance, and comorbidities (defined by Charlson comorbidity index) for hospitalizations where AU may have been indicated, based on International Classification of Diseases Tenth Revision (ICD-10) diagnosis codes. The California Healthy Places Index (HPI) was used to obtain composite neighborhood SES indicators for each patient at the ZIP code level, measured as tertiles. Covariates were aggregated to the hospital level. Poisson regressions were used to evaluate the association between hospital and neighborhood SES indicators and SAAR scores, controlling for potential hospital-level confounders. Results: Among 86 hospitals included in the analysis, the mean patient age for hospitalizations where AU may have been indicated was 66 years, the proportion of white patients was 55%, and the mean proportion of Medi-Cal users was 19%. After adjusting for confounders including age, race or ethnicity, insurance status, comorbidities, and number of hospital beds; higher median values of patient SES had a protective effect against hospitals having GP SAAR scores > 1 (relative risk [RR], 0.68; 95% CI, 0.50–0.93) but was not significantly associated with hospitals having BSCA SAAR scores >1 (RR, 0.79; 95% CI, 0.62–1.02) or BSHO SAAR scores >1 (RR, 0.80; 95% CI, 0.61–1.04). Conclusions: Considering SES in addition to summary antimicrobial use scores such as SAARs may help identify populations potentially at risk for inappropriate AU; however, patient-level information is still necessary to evaluate appropriateness of antimicrobial prescribing.
Funding: No
Disclosures: None
Prospective Audit for Antimicrobial Use and Stewardship Practices in Intensive Care Unit at a Tertiary-Care Center in India
- Parul Singh, Purva Mathur, Kamini Walia, Anjan Trikha
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s40
-
- Article
-
- You have access Access
- Open access
- Export citation
-
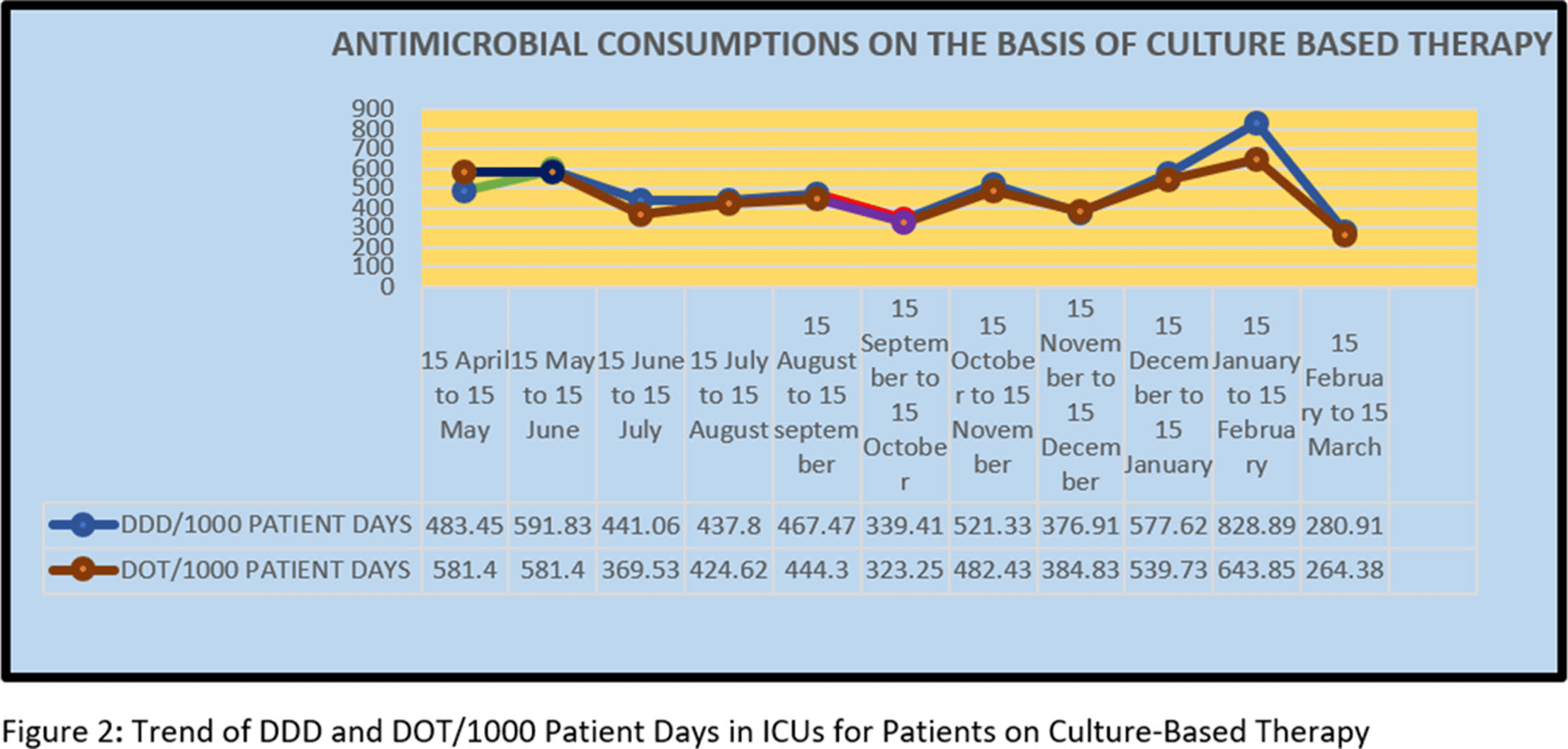
Background: Antimicrobial decision making in the ICU is challenging. Injudicious use of antimicrobials contributes to the development of resistant pathogens and drug-related adverse events. However, inadequate antimicrobial therapy is associated with mortality in critically ill patients. Antimicrobial stewardship programs are increasingly being implemented to improve prescribing. Methods: This prospective study was conducted over 11 months, during which the pharmacist used a standardized survey form to collect data on antibiotic use. Evaluation of antimicrobial use and stewardship practices in a 12-bed polytrauma ICU and a 20-bed neurosurgery ICU of the 248-bed AIIMS Trauma Center in Delhi, India. Antimicrobial consumption was measured using WHO-recommended defined daily dose (DDD) of given antimicrobials and days of therapy (DOT). Results: Antibiotics were ranked by frequency of use over the 11-month period based on empirical therapy and culture-based therapy. The 11-month DDD and DOT averages when empiric antibiotics were used were 532 of 1,000 patient days and 484 per 1,000 patient days, respectively (Figure 1). When cultures were available, DDD was 486 per 1,000 patient days and DOT was 442 per 1,000 patient days (Figure). Conclusions: The quantity and frequency of antibiotics used in the ICUs allowed the AMSP to identify areas to optimize antibiotic use such as educational initiatives, early specimen collection, and audit and feedback opportunities.
Funding: No
Disclosures: None
Figure 1.
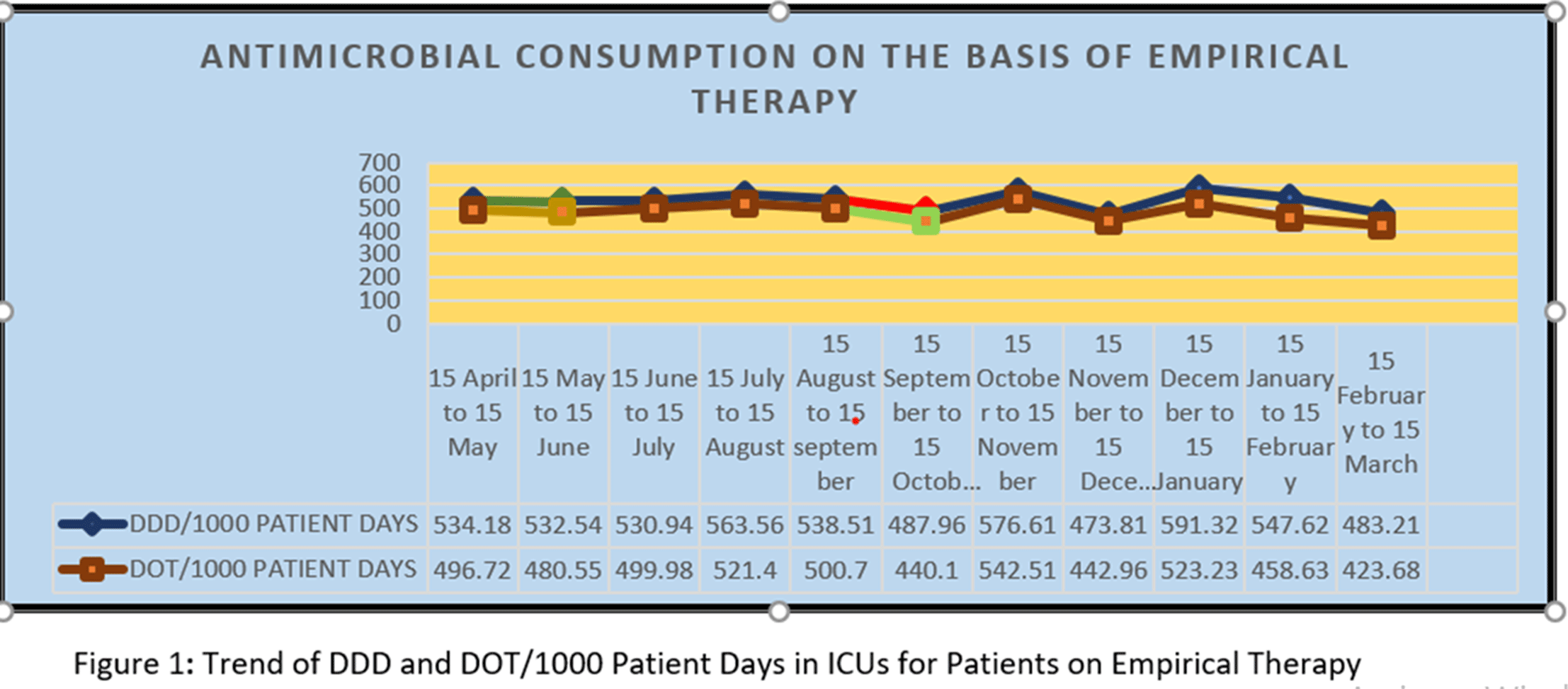
Figure 2.
Evaluating the Prevalence of Leading Practices in Antimicrobial Stewardship
- Barbara Braun, Salome Chitavi, Eddie Stenehjem, Mushira Khan, David Baker, David Hyun
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s41
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Most hospitals have a basic infrastructure in place for antimicrobial stewardship programs (ASPs). Although this is a critical first step, we need to ensure that ASPs are working to implement effective evidence-based approaches nationally. In 2018, a group of leading antibiotic stewardship organizations met and identified specific, effective, and recommended ASP activities based on current scientific evidence and their experience (Baker et al, Joint Comm J Qual Pat Saf 2019;45:517–523). To determine the extent to which hospitals are currently implementing the recommended practices, we conducted an electronic questionnaire–based assessment. Methods: A 50-item questionnaire-based assessment was sent via QualtricsTM to the hospital’s designated ASP leader. The sample comprised 992 Joint Commission accredited hospitals. The practices of interest related to (1) development of facility-specific treatment guidelines, (2) measuring appropriate use and concordance of care with these guidelines, (3) engaging clinicians while the patient is on the unit, (4) diagnostic stewardship, (5) measurement of antimicrobial utilization data, and (6) measuring hospital-acquired Clostridioides difficile infection (CDI) rates. Sampling weights were used to adjust the results for nonresponse using R software. Results: In total, 288 hospitals completed the questionnaire. Small and nonteaching hospitals were significantly less likely to respond (p < 0.005, p=0.01 respectively), however there were no differences by healthcare system membership or urban/rural location. 49% of respondents had the specialist term ASP or infectious disease (ID) in their title. Most hospitals (93.1%) had developed facility-specific treatment guidelines for specific inpatient conditions, often community-acquired pneumonia (85%), sepsis (81%), UTI (75%), and SSTI (69%). However, only 37% had formally assessed compliance with 1 or more of these guidelines. Also, 83% reported having a process for prospective audit and feedback, of which 43% do this 4–5 days per week. Similarly, 49% reported that they review all antimicrobials ordered. Recommendations are commonly given by the ASP pharmacist (69%) via some combination of telephone (78%), face-to-face (69%), text message (54%), and/or EHR alert (36%). Overall, 66% of hospitals had procedures in place to prevent inappropriate diagnostic testing for C. difficile, and 39% of hospitals had similar policies for urine specimens. Furthermore, >80% were routinely measuring days of therapy and CDI rates. Conclusions: Most hospitals have facility-specific treatment guidelines and measure CDI and days of therapy. Practices for active engagement with frontline staff in prospective audit and feedback vary widely. Greater understanding of barriers to assessing adherence to hospitals’ treatment guidelines is critical to improving this practice.
Funding: The Pew Charitable Trusts
Disclosures: None
C. difficile
Clostridioides difficile Is Not Difficult to Predict in Hospital Settings
- Kinta Alexander, Frances Petersen, Sean Brown
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s41
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background:Clostridioides difficile is a gram-positive bacteria that is the most common cause of hospital-associated infectious diarrhea among traditional and nontraditional high-risk populations. Excess healthcare costs associate with C. difficile infection (CDI) prevalence, morbidity, and mortality is shown to economically impact the US healthcare system with at least an additional $1 billion in annual cost. Exposure to antimicrobial agents resulted in increased risk for hospital-onset CDI (HO-CDI) at an inner-city hospital during 2010 and 2011. Methods: A retrospective case-control study of all persons with HO-CDI in the MICU was conducted at an inner-city hospital between January 1, 2010, and December 31, 2011. A patient was considered to have developed HO-CDI if diarrhea developed after 72 hours of admission into the MICU and a confirmed laboratory stool specimen for Clostridioides difficile infection (CDI) was obtained. A non–HO-CDI person was randomly selected using “risk set sampling.” After the application of inclusion and exclusion criteria, 88 cases were eligible for the study. Of these cases, 29 met the definition for HO-CDI, and 59 met the definition for non–HO-CDI. The relationship between antimicrobial use and the development of HO-CDI in patients in the MICU at an inner-city hospital was investigated using a logistic regression model in which the variable of total antibiotics was used as a possible predictor for predicting a positive HO-CDI. Results: Logistic regression was utilized to determine the relationships between selected study variables and presence or absence of HO-CDI. Total antibiotics was significantly related to HO-CDI. The results of this analysis showed that total antibiotics was a significant predictor for HO-CDI. The total value of the coefficient B for this predictor was 0.47, and the exponentiated value (exp[B]) of this coefficient was 1.60 (95% CI, 1.08–2.35). In this sample, patients who had 1 or more antibiotics were at a 60% greater risk of having a positive HO-CDI culture. There was a significant association between the use of metronidazole and HO-CDI (p < .001). Conclusions: Antimicrobial stewardship is an integral part of patient safety. The findings from this study were instrumental in the implementation of a fledging antimicrobial stewardship program and the use of evidence-based practices at this inner-city hospital.
Funding: No
Disclosures: None
Clinical Characteristics and Fecal Microbiome in Recurrent Versus Nonrecurrent Clostridioides difficile Infection
- Swapnil Lanjewar, Ashley Kates, Lauren Watson, Nasia Safdar
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s41-s42
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Up to 30% of patients with Clostridioides difficile infection (CDI) develop recurrent infection, which is associated with a 33% increased risk of mortality at 180 days. The gut microbiome plays a key role in initial and recurrent episodes of CDI. We examined the clinical characteristics and gut microbial diversity in patients with recurrent (rCDI) versus nonrecurrent CDI at a tertiary-care academic medical center. Methods: Stool samples were collected from 113 patients diagnosed with CDI between 2018 and 2019. Clinical and demographic data were extracted from the electronic medical record (Table 1), and 16S rRNA sequencing of the v4 region was carried out on the Illumina MiSeq using 2×250 paired-end reads. Sequences were binned into operational taxonomic units (OTUs) using mothur and were classified to the genus level whenever possible using the ribosomal database project data set version 16. Alpha diversity was calculated using the Shannon diversity index. Β diversity was calculated using the Bray-Curtis dissimilarity matrix. Differential abundance testing was done using DESeq to assess taxonomic differences between groups. A P value of .05 was used to assess significance. Results: In total, 55 patients had rCDI (prior positive C. difficile polymerase chain reaction in last 7–365 days) and 58 had nonrecurrent CDI (Table 1). Patients with rCDI had a higher frequency of organ transplant and comorbidity. No differences in α not β diversity were observed between groups. Also, 4 OTUs were more abundant in those with rCDI: Ruminococcus (n = 2), Odoribacter, and Lactobacillus. Patients with rCDI had microbiomes with greater proportions of Bacteroidetes (27% of OTUs) compared to the nonrecurrent group (18%) as well as fewer OTUs belonging to the Firmicutes phyla compared to the nonrecurrent patients (56% vs 59%). Among the rCDI patients, those experiencing 2 or more recurrences had greater abundances of Bacteroides and Ruminococcus, while those experiencing only 1 recurrence had significantly greater abundances of Akkermensia, Ruminococcus, Streptococcus, Roseburia, Clostridium IV, and Collinsella compared to those with only 1 recurrence (Table 2). Conclusions: Patients with rCDI had a more impaired microbiome than those with initial CDI. Ruminococcus OTUs have been previously indicated as a risk factor for recurrence and treatment failure, and they were significantly more abundant in those with rCDI and among those with multiple recurrences. The greatest differences in the microbiome were observed between those with 1 recurrence compared to those with multiple recurrences. Interventions for gut microbiome restoration should focus particularly on those with recurrent CDI.
Funding: No
Disclosures: None
Table 1. 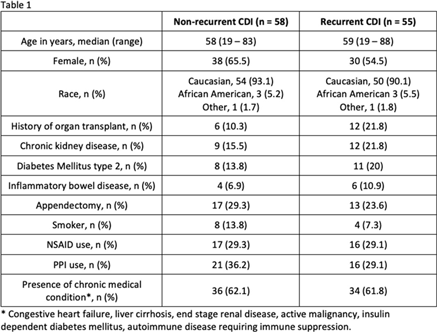
Table 2. 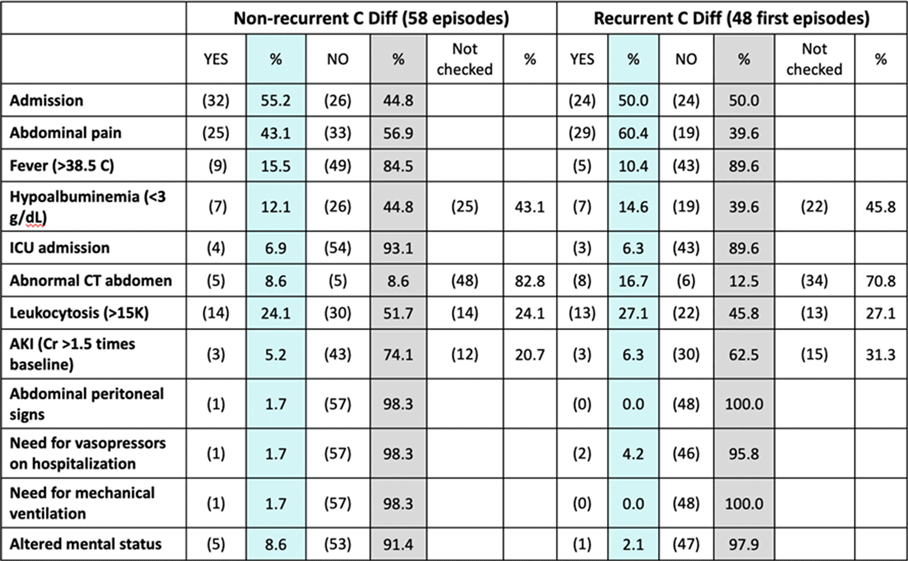
Impact of Two-Step Testing Algorithm on Reducing Hospital-Onset Clostridioides difficile Infections
- Bhagyashri Navalkele, Wendy Winn, Sheila Fletcher, Regina Galloway, Jason Parham, William Daley, Patrick Kyle, Vonda Clack, Kathy Sheilds
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s42-s43
-
- Article
-
- You have access Access
- Open access
- Export citation
-
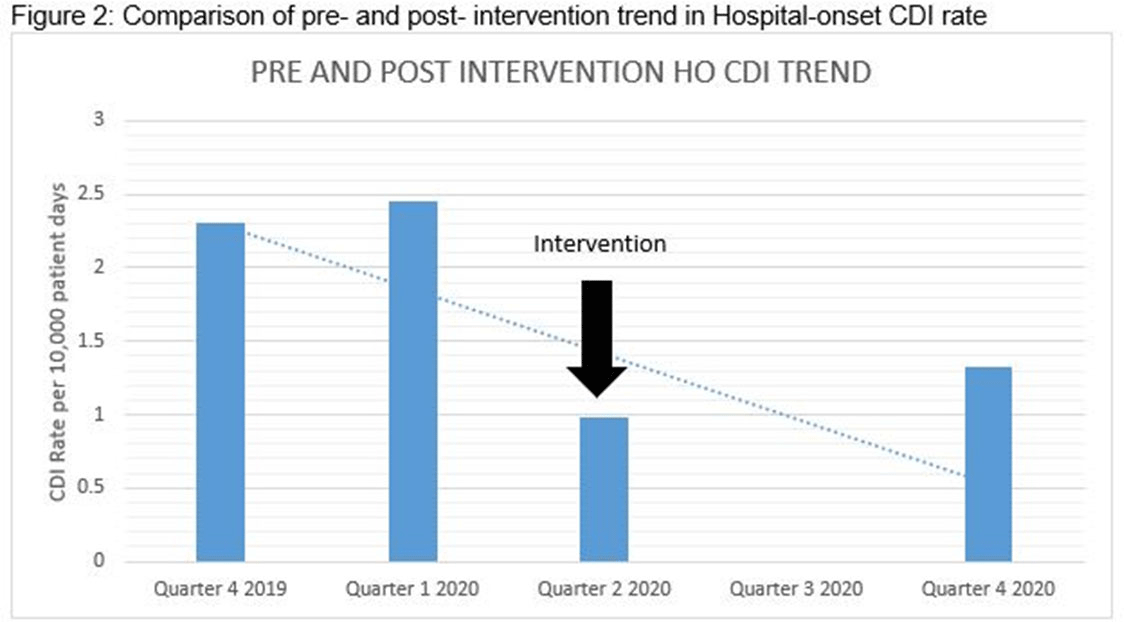
Clostridioides difficile infection (CDI) is one of the leading causes of hospital–onset infections. Clinically distinguishing true CDI versus colonization with C. difficile is challenging and often requires reliable and rapid molecular testing methods. At our academic center, we implemented a 2-step testing algorithm to help identify true CDI cases. The University of Mississippi Medical Center is a 700+ bed academic facility located in Jackson, Mississippi. Hospital-onset (HO) CDI was defined based on NHSN Laboratory Identified (LabID) event as the last positive C. difficile test result performed on a specimen using a multistep testing algorithm collected >3 calendar days after admission to the facility. HO-CDI data were collected from all inpatient units except the NICU and newborn nursery. HO-CDI outcomes were assessed based on standardized infection ratio (SIR) data. In May 2020, we implemented a 2-step testing algorithm (Figure 1). All patients with diarrhea underwent C. difficile PCR testing. Those with positive C. difficile PCR test were reflexed to undergo enzyme immunoassay (EIA) glutamate dehydrogenase antigen (Ag) testing and toxin A and B testing. The final results were reported as colonization (C. difficile PCR+/EIA Ag+/Toxin A/B−) or true CDI case (C. difficile PCR+/EIA +/Toxin A/B +) or negative (C. difficile PCR−). All patients with colonization or true infection were placed under contact isolation precautions until diarrhea resolution for 48 hours. During the preintervention period (October 2019–April 2020), 25 HO-CDI cases were reported compared to 8 cases in the postintervention period (June 2020–December 2020). A reduction in CDI SIR occurred in the postintervention period (Q3 2020–Q4 2020, SIR 0.265) compared to preintervention period (Q4 2019–Q1 2020, SIR 0.338) (Figure 2). We successfully reduced our NHSN HO-CDI SIR below the national average after implementing a 2-step testing algorithm for CDI. The 2-step testing algorithm was useful for antimicrobial stewardship to guide appropriate CDI treatment for true cases and for infection prevention to continue isolation of infected and colonized cases to reduce the spread of C. difficile spores.
Funding: No
Disclosures: None
Figure 1.
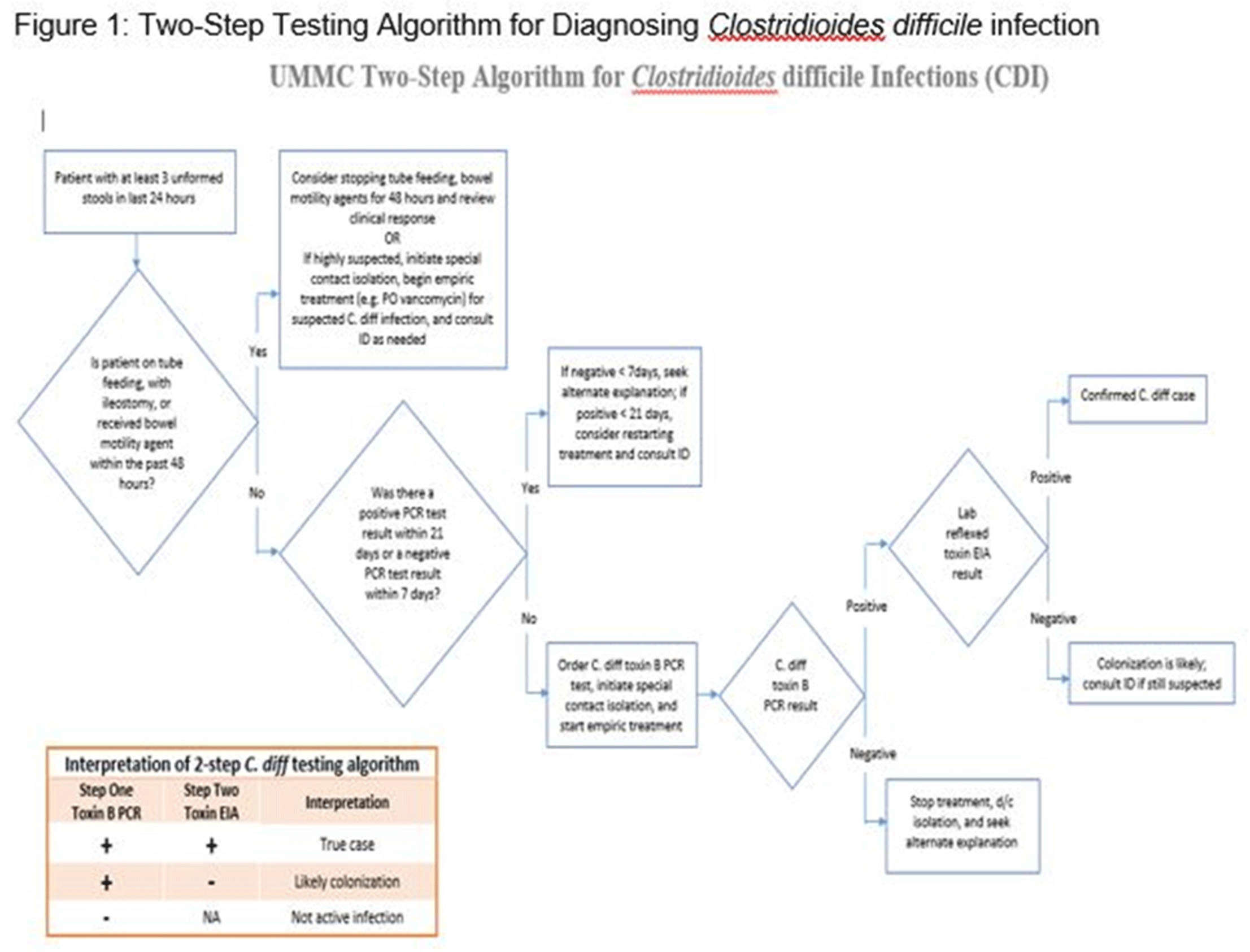
Figure 2.
Did Clostridioides difficile Testing and Infection Rates Change During the COVID-19 Pandemic?
- Armani Hawes, Payal Patel, Angel Desai
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s43
-
- Article
-
- You have access Access
- Open access
- Export citation
-
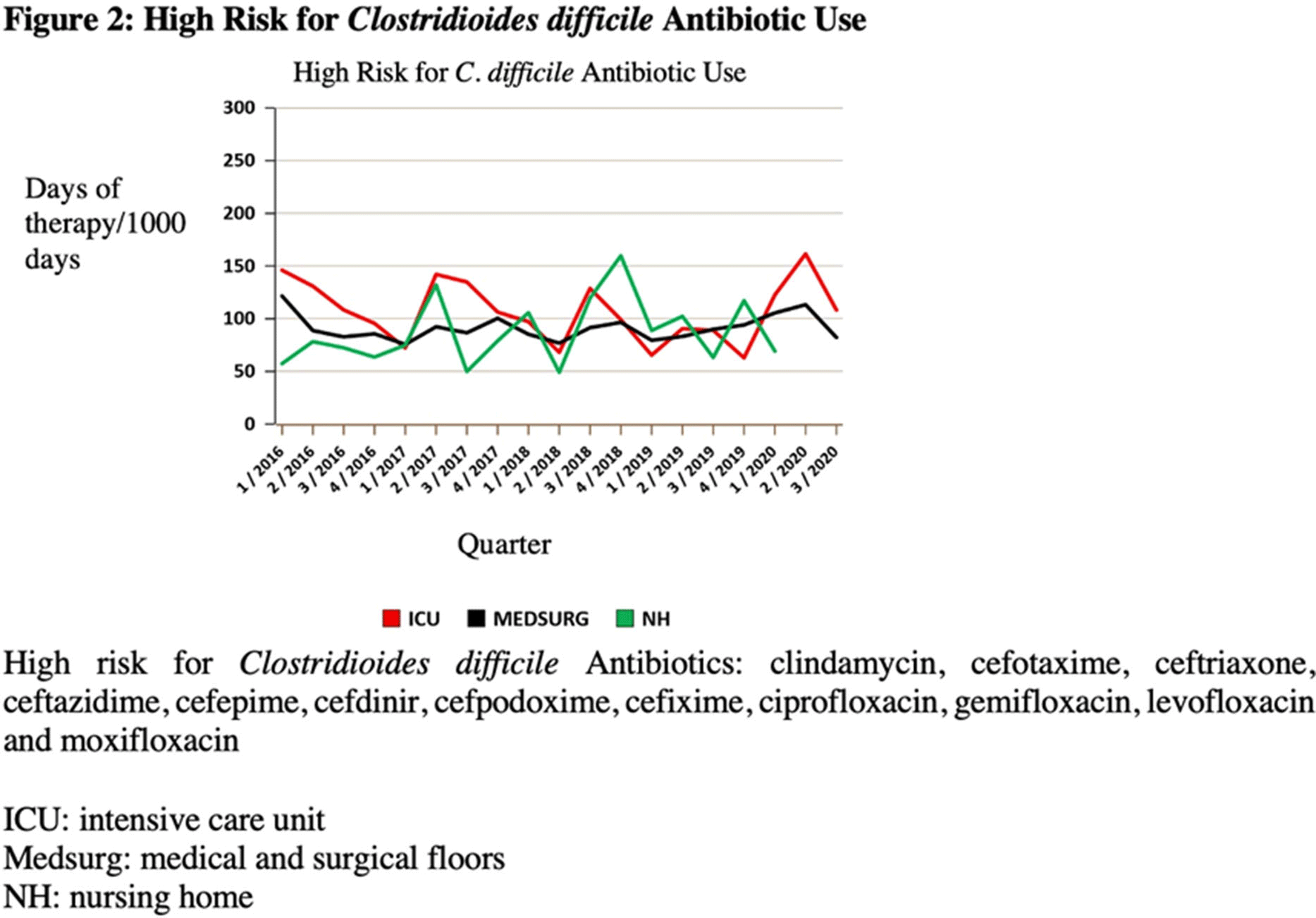
Background: The COVID-19 pandemic has underscored the importance of ongoing infection prevention efforts. Increased adherence to infection prevention recommendations, increased antibiotic use, improved hand hygiene, and correct donning and doffing of personal protective equipment may have influenced healthcare-associated infections (HAIs) in the United States during the pandemic. In this study, we investigated testing for Clostridioides difficile infection (CDI) and incidence during the initial surge of the pandemic. We hypothesized that strict adherence to contact precautions may have resulted in a decreased incidence of CDI in hospitalized patients during the first peak of the COVID-19 pandemic and that CDI testing may have increased even in the absence of directed diagnostic stewardship efforts. Methods: We conducted a single-center, retrospective, observational study at the Veterans’ Affairs (VA) Hospital in Ann Arbor, Michigan, between January 2019 and June 2020. We compared data on CDI tests from January 2019 through February 2020 to data from March 2020 (the admission of the first patient with COVID-19 at our institution) through June 2020. Pre-peak and peak periods were defined by confirmed cases in Washtenaw County. No novel diagnostic or CDI-focused stewardship interventions were introduced by the antimicrobial stewardship program during the study period. An interrupted time series analysis was performed using STATA version 16.1 software (StataCorp LLC, College Station, TX). Results: There were 6,525 admissions and 34,533 bed days between January 1, 2019, and June 30, 2020. Also, 900 enzyme immunoassay (EIA) tests were obtained and 104 positive cases of CDI were detected between January 2019 and June 2020. A statistically significant decrease in EIA tests occurred after March 1, 2020 (the COVID-19 peak in our region) compared to January 1, 2019–March 1, 2020 (Figure 1). After March 1, 2020, the number of EIA tests obtained decreased by 10.2 each month (95% CI, −18.7 to −1.7; P = .02). No statistically significant change in the incidence of CDI occurred. The use of antibiotics that were defined as high risk for CDI increased in the months of April–June 2020 (Figure 2). Conclusions: In this single-center study, we observed a stable incidence of CDI but decreased testing during the first peak of the COVID-19 pandemic. Understanding local HAI reporting is critical because changes in HAI reporting structures and exemptions during this period may have affected national reporting. Further research should be undertaken to investigate the effect of COVID-19 on other HAI reporting within the US healthcare system.
Funding: No
Disclosures: None
Figure 1.
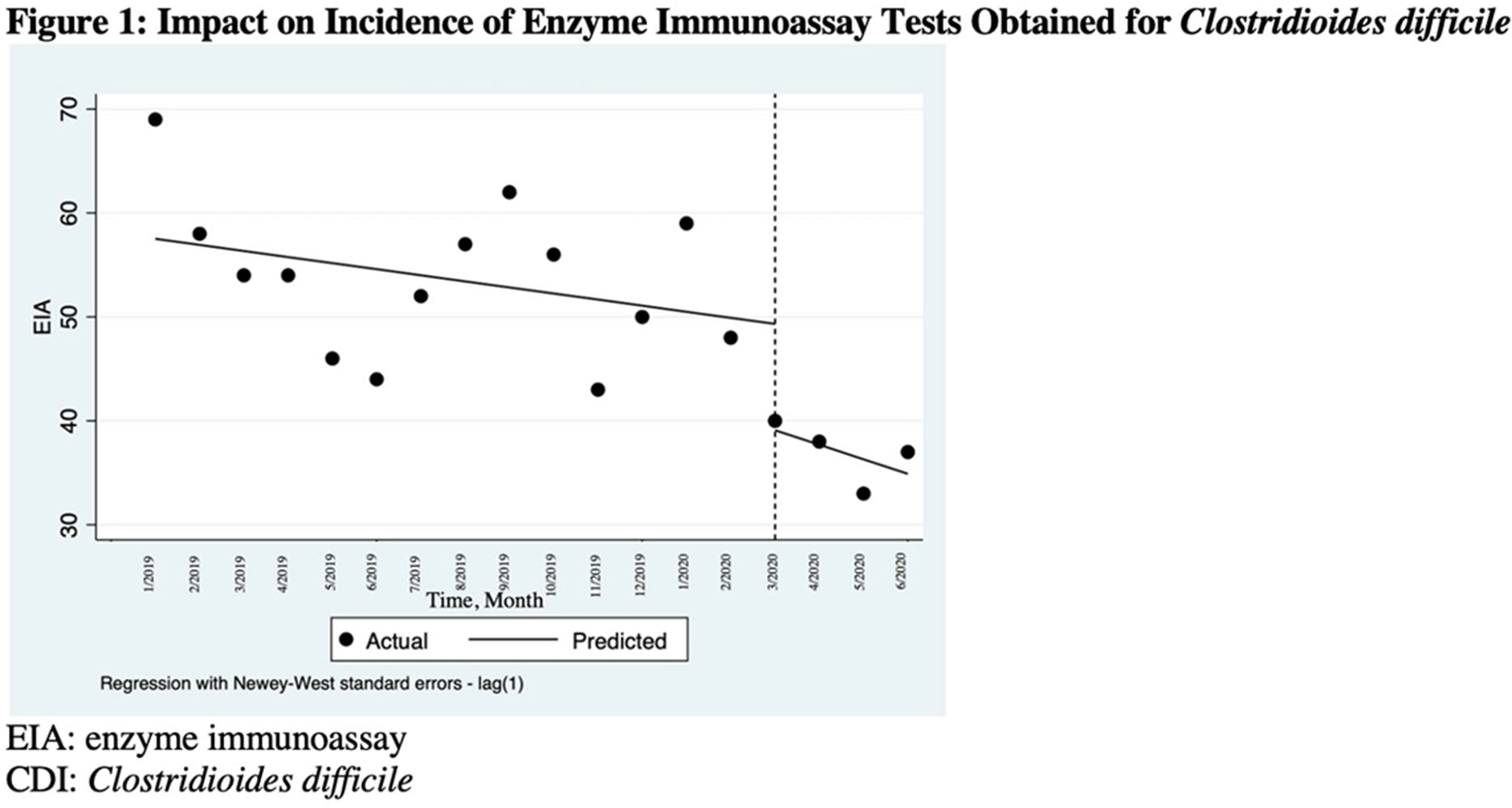
Figure 2.
CLABSI
Inequities in CLABSI Rates in a Children’s Hospital by Race, Ethnicity, and Language Preference
- Caitlin McGrath, Matthew Kronman, Danielle Zerr, Brendan Bettinger, Tumaini Coker, Shaquita Bell
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s43-s44
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Systemic racism results in health inequities based on patient race, ethnicity, and language preference. Whether these inequities exist in pediatric central-line–associated bloodstream infections (CLABSIs) is unknown. Methods: This retrospective cohort study included patients with central lines hospitalized from October 2012 to June 2019 at our tertiary-care children’s hospital. Self-reported race, ethnicity, language preference, demographic, and clinical factors were extracted from the electronic health record. The primary outcome was non–mucosal barrier injury (non-MBI) CLABSI episodes as defined by the NHSN. CLABSI rates between groups were compared using χ2 tests and Cox proportional hazard regression. We adjusted for care unit, age, immunosuppressed status, diapered status, central-line type, line insertion within 7 days, daily CLABSI maintenance bundle compliance, number of blood draws and IV medication doses, and need for total parental nutrition, extracorporeal membrane oxygenation, and renal replacement therapy. In mid-2019, we engaged stakeholders in each care unit to describe preliminary findings and to identify and address potential drivers of observed inequities. Results: We included 337 non-MBI CLABSI events over 230,699 central-line days (CLDs). The overall non-MBI CLABSI rate during the study period was 1.46 per 1,000 CLDs. Unadjusted CLABSI rates for black or African American (henceforth, “black”), Hispanic, non-Hispanic white, and Asian (the 4 largest race or ethnicity groups by CLDs) patients were 2.74, 1.53, 1.42, 1.24 per 1,000 CLDs, respectively (P < .001) (Table 1). Unadjusted CLABSI rates for patients with limited-English proficiency (LEP) and English-language preference were 1.98 and 1.38 per 1,000 CLDs, respectively (P = .014). After adjusting for covariates, the hazard ratio (HR) point estimate for CLABSI rate remained higher for black patients (HR, 1.50; 95% CI, 0.99–2.28) and patients with LEP (HR, 1.33; 95% CI, 0.87–2.05), compared to the reference group based on largest CLD. The differences in CLABSI rate by race or ethnicity and language were more pronounced in 2 of our 6 care units. Stakeholder engagement and analysis of hospital data revealed opportunities on those units for improved (1) interpreter utilization and (2) line maintenance observation practices by race/ethnicity and language preference (data not shown). These findings and CLABSI rates over time by race/ethnicity and language preference (Figures 1 and 2) were shared with frontline staff. Conclusions: In our children’s hospital, CLABSI rates differed based on patients’ self-reported race, ethnicity, and language preference, despite controlling for factors commonly associated with CLABSI. Identifying inequities in CLABSI rates and mitigating their determinants are both essential to the goal of achieving equitable care.
Funding: No
Disclosures: None
Table 1. 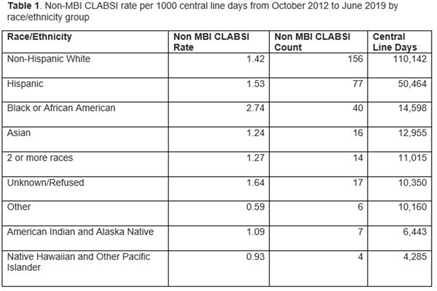
Figure 1.

Figure 2.

COVID-19
Use of COVID-19 Serologic Testing in Healthcare Workers with Acute Respiratory Tract Infection
- Amy Ray
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s44
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Diagnostic tests for COVID-19 are in high demand. Serologic assays are of interest as diagnostic adjuncts to SARS-CoV-2 quantitative polymerase chain reaction (PCR); however, many of the commercially available assays have limited validation data and clinical utility is unknown. We describe the utilization of SARS-CoV-2 IgG enzyme-linked immunosorbent assay (ELISA) for healthcare workers with acute respiratory tract infection (ARTI) who underwent SARS-CoV-2 PCR testing. Methods: The MetroHealth System is the largest public hospital system in Ohio, employing ~8,000 staff. COVID-19 detection began in early March 2020. EDI novel coronavirus COVID-19 IgG ELISA (KT-1032) targeting antibody response to viral nucleocapsid was obtained for diagnostic and seroprevalence analyses. Manufacturer reports of sensitivity and specificity of the assay are 100% and 99%, respectively. A 2-part test strategy for employees with symptoms of ARTI was implemented. Qualifying symptoms for SARS-CoV-2 PCR testing included fever and either cough or shortness of breath. Additional symptoms were included to reflect expanding knowledge of COVID-19. Employees who underwent SARS-CoV-2 PCR testing (Luminex ARIES) were offered serologic testing on day 14 following PCR result. Education accompanied the offer for serologic testing as well as the receipt of test result to aide interpretation. Results: From April 16, 2020, through July 6, 2020, 588 employees underwent PCR testing. Overall, 70 cases of COVID-19 were detected. Of the 197 employees who opted for serologic testing, IgG positivity was 12.6%. The mean time to IgG collection following PCR result was 30 days (range, 10–79). Using PCR results obtained in the clinical setting of ARTI as the diagnostic gold standard, IgG was 84.6% sensitive and 98.2% specific (Figure 1). Conclusions: In a population of symptomatic healthcare workers, SARS-CoV2 IgG testing was specific for COVID-19 diagnosis. Sensitivity was inadequate compared to the positive predictive agreement of 90% or greater required for US Food and Drug Administration emergency use authorization. In a low-prevalence environment for COVID-19 (<5%), a positive SARS-CoV-2 IgG has a low positive predictive value, which may falsely imply immunity and may negatively affect infection prevention practices.
Funding: No
Disclosures: None
Figure 1.

COVID-19 and Ventilator-Associated Event Discordance
- Kelly Cawcutt, Mark Rupp, Lauren Musil
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s45
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The COVID-19 pandemic has challenged healthcare facilities since its discovery in late 2019. Notably, the subsequent COVID-19 pandemic has led to an increase in healthcare-acquired infections such as ventilator associated events (VAEs). Many hospitals in the United States perform surveillance for the NHSN for VAEs by monitoring mechanically ventilated patients for metrics that are generally considered to be objective and preventable and that lead to poor patient outcomes. The VAE definition is met in a stepwise manner. Initially, a ventilator-associated condition (VAC) is met when there an increase in ventilator requirements after a period of stability or improvement. An IVAC is then met when there is evidence of an infectious process such as leukocytosis or fever and a new antimicrobial agent is started. Finally, possible ventilator-associated pneumonia (PVAP) is met when there is evidence of microbial growth or viral detection. Since the beginning of the COVID-19 pandemic, our hospital has seen an increase in VAEs, which is, perhaps, not unexpected during a respiratory illness pandemic. However, the NSHN definitions of VAE, and PVAP in particular, do not account for the novelty and nuances of COVID-19. Methods: We performed a chart review of 144 patients who had a VAE reported to the NHSN between March 1 and December 31, 2020. Results: Of the 144 patients with a VAE reported to NHSN, 39 were SARS-CoV-2 positive. Of the 39 patients, 4 patients (10.25%) met the NHSN PVAP definition due to a positive SARS-CoV-2 PCR that was collected in the prolonged viral shedding period of their illness (< 90 days). One of the four patients also had a bacterial infection in addition to their subsequent positive COVID-19 result. All these patients were admitted to the hospital with a COVID-19 diagnosis and their initial PCR swab was performed upon admission. Conclusions: We believe that the PVAP definition was inappropriately triggered by patients who were decompensating on the ventilator due to a novel respiratory virus that was present on admission. Early in the pandemic, frequent swabbing of these patients was performed to try and understand the duration of viral shedding and to determine when it would be safe to transfer patients from isolation after prolonged hospitalization. The NSHN definition should take into consideration the prolonged viral shedding period of COVID-19 and natural history of the illness, and subsequent COVID-19 testing within 90 days of an initial positive should not require classification as a hospital-acquired PVAP.
Funding: No
Disclosures: None


























