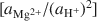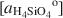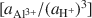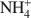Volume 50 - Issue 6 - December 2002
Research Article
Weathering of Chlorite: I. Reactions and Products in Microsystems Controlled by the Primary Mineral
- Mehrooz F. Aspandiar, Richard A. Eggleton
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 685-698
-
- Article
- Export citation
-
The weathering of chlorite in hydrothermally-altered basalt was studied with XRD, TEM and electron microprobe to determine the type and orientation of secondary minerals. Optical examination indicated chlorite assemblages to have altered in two distinct microsites: one microsite near micro-fissures traversing the regolith units, and the other away from the continuous passages. In this paper, weathering mechanisms and products of chlorite present in microsites distant from the micro-fissures are reported. In all the regolith units the original chlorite grain remained intact and was pseudomorphed by secondary products. In the saprock, chlorite altered to corrensite with possible random interstratifications of chlorite and corrensite and corrensite and vermiculite. In the saprolite, corrensite altered to vermiculite. Parallelism of two axes of the products with the host indicated topotactic alteration. In the fine saprolite, vermiculite was found to alter to kaolinite via a randomly interstratified kaolinite-vermiculite stage with a high proportion of kaolinite. Goethite crystallized in between packets of kaolinite, vermiculite and kaolinite-vermiculite. Though the disruption of the crystal structure of vermiculite is necessary in its alteration to kaolinite, the reaction was such as to maintain parallelism of the c axis. The alteration of chlorite to vermiculite was characterized by the loss of Mg and Fe and minor Al, all ions considered to be lost from the brucite-like sheet of chlorite. The Fe released during the alteration of vermiculite to kaolinite is likely to have migrated to micropores to form goethite. The presence of interstratifications of the end-members of layer silicates involved in the reaction sequence suggests that interstratifications are common during layer silicate weathering in environments where space is limited and consequently solution and ionic transport passages are restrictive.
Weathering of Chlorite: II. Reactions and Products in Microsystems Controlled by Solution Avenues
- Mehrooz F. Aspandiar, Richard A. Eggleton
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 699-709
-
- Article
- Export citation
-
Weathering of chlorite adjacent to macro- and micro-passages (fissures) in regolith units has been studied to determine the alteration mechanisms and products and compare them to those that prevail at a distance from the open passages. The micro-fissures in the saprolith units evolve as a function of weathering with an increase in micro-fissure density and their infilling with ferriargillans corresponding to an increase in weathering intensity. In the saprock and saprolite units, goethite-rich bands with surrounding reddish haloes invade the chlorite adjoining the micro-fissures. In the reddish haloes surrounding the goethite bands and fissures, the phyllosilicates alter to an intimate mixture of goethite, kaolinite, feroxyhyte and halloysite with the possible nanoscale presence of other fine-grained iron aluminosilicates such as hisingerite and smectite. In the fine saprolite, the weathering products after original chlorite adjoining the micro-fissures bearing ferriargillans, alter directly to ferriargillan products — kaolinite and goethite. The lack of orientation of the products with the parent phyllosilicates indicates the operation of a dissolution-precipitat ion mechanism which is in contrast to the topotactic alteration mechanism functioning at a distance within the phyllosilicate grain assemblage. The differences in alteration mechanisms and products of chlorite weathering in different microsites suggest the rate of weathering of chlorite can differ in microsites within individual regolith units. The presence of fine-grained metastable products in the form of feroxyhyte and halloysite adjoining the fissures suggests an Ostwald Step Rule sequence during alteration of phyllosilicates with rapid oxidation of Fe and the presence of Si in the microsite considered the main factor favoring fine-grained metastable products.
Zeolitization of a Phonolitic Ash Flow by Groundwater in the Laach Volcanic Area, Eifel, Germany
- Franz Bernhard, Ulrike Barth-Wirsching
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 710-725
-
- Article
- Export citation
-
Field and experimental studies were performed to understand the formation conditions of the Nettetal zeolite deposit, Laach volcanic area, Germany. This deposit shows pronounced small- (cm) and large-scale (tens of meters) variations of zeolitization, despite the same phonolitic precursor glass throughout the occurrence. Zeolitization of the pyroclastic ash flow is restricted to three distinct layers that are 0.15 to 10 m thick and separated by fresh ash. The glassy matrix is altered to chabazite, phillipsite, analcime and K-feldspar in various combinations, whereas the pumice clasts are altered predominantly to chabazite. Mass changes during zeolite formation appear to be small, and Ca enrichment in chabazite and phillipsite may have occurred after their formation by cation exchange.
The zeolites and zeolite assemblages observed in the Nettetal deposit were experimentally reproduced by reacting the phonolitic glass at 100–200°C with distilled water and 0.01 M alkaline solutions as well as with varying solid/liquid ratios and grain-sizes. Chabazite and phillipsite represented metastable transition phases with respect to analcime and K-feldspar. A high solid/liquid ratio accelerated the conversion of glass to zeolites.
None of the classic models of zeolite formation is fully applicable to the Nettetal deposit. The most probable environment for zeolitization in this deposit is the stagnant fringe water zone immediately above the groundwater table. In this zone, representing a relatively closed system, favorable solution compositions for zeolite formation could have been developed rather quickly by glass-water interaction, which is not possible within the more thoroughly flushed deeper parts of the groundwater system. The three distinct zeolite layers are probably the result of temporarily changing groundwater levels.
Clay Mineralogy of Spodosols with High Clay Contents in the Subalpine Forests of Taiwan
- Ching-Wei Lin, Zeng-Yei Hseu, Zueng-Sang Chen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 726-735
-
- Article
- Export citation
-
Three Ultic Haplorthods with significant illuviation of clay and spodic materials in the subalpine forests located in the Alishan area of central Taiwan were selected to identify the clay mineral compositions by X-ray diffraction (XRD) and to explain the transformation of the clay minerals. The three pedons are dominated by vermiculite and vermiculite-illite interstratified minerals, and they have minor kaolinite, quartz and gibbsite. No hydroxy interlayered vermiculite (HIV) was found in the E horizon of the three pedons because the forest soils are very acidic and have very low Al contents. The presence of HIV in the spodic (Bhs) and argillic horizons (Bt) of the three pedons was associated with greater free Fe and Al contents (Fed and Ald), more favorable pH ranges, and coatings of organo-metallic complexes which prevented continuous weathering. The specific pedogenic process, clay illuviation and podzolization occurred sequentially in the Alishan area, and induced the unusual clay mineral distribution and transformation. The largest amounts of illite are in the C horizon and the amounts of vermiculite increased with decreasing soil depths. A reverse distribution between illite and vermiculite through the soil profile was observed. Illite was transformed to vermiculite due to the strong weathering environment associated with extremely low exchangeable K contents. The weathering sequence of clay minerals of Spodosols with fine textures in the study area is proposed as: illite → vermiculite (or interstratified vermiculite-illite minerals) → HIV and vermiculite.
Formation of Sepiolite-Palygorskite and Related Minerals from Solution
- Rezan Birsoy
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 736-745
-
- Article
- Export citation
-
Most of the world's sepiolite-palyg orskite precipitates in lacustrine and perimarine environments. Although these minerals can transform from precursor minerals, the most common formation mechanism involves crystallization from solution. In this study, equilibrium activity diagrams are calculated for sepiolite-palygorskite in the seven component system MgO-CaO-Al2O3-SiO2-H2O-CO2-HCl, employing available thermodynamic data for related minerals, aqueous species and water. Stability fields are illustrated graphically on plots of log
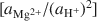 vs. log
vs. log 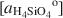 , using the activities for log
, using the activities for log 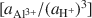 defined by an arbitrarily chosen value and the approximate saturation limits of pyrophyllite + amorphous silica, kaolinite + amorphous silica, kaolinite + pyrophyllite, pyrophyllite + quartz and gibbsite. The formation of sepiolite-palygorskite from solution is more favored in the presence of amorphous silica than quartz. Lower aqueous aluminum activities favor the non-aluminum phases sepiolite and kerolite relative to the aluminum-containing phases palygorskite and saponite. The stability ranges of worldwide associations of magnesite and dolomite with sepiolite and palygorskite are also illustrated as a function of aluminum activity.
defined by an arbitrarily chosen value and the approximate saturation limits of pyrophyllite + amorphous silica, kaolinite + amorphous silica, kaolinite + pyrophyllite, pyrophyllite + quartz and gibbsite. The formation of sepiolite-palygorskite from solution is more favored in the presence of amorphous silica than quartz. Lower aqueous aluminum activities favor the non-aluminum phases sepiolite and kerolite relative to the aluminum-containing phases palygorskite and saponite. The stability ranges of worldwide associations of magnesite and dolomite with sepiolite and palygorskite are also illustrated as a function of aluminum activity.
Transmission Electron Microscopy Evidence for Experimental Illitization of Smectite in K-Enriched Seawater Solution at 50°C and Basic pH
- A. Drief, F. Martinez-Ruiz, F. Nieto, N. Velilla Sanchez
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 746-756
-
- Article
- Export citation
-
Experimental illitization of smectite was studied by transmission electron microscopy (TEM) and X-ray diffraction (XRD). Experiments were performed on the <2 µm fraction consisting entirely of smectite separated from a soil formed on subvolcanic rock located in the External Zone of the Betic Cordilleras (southern Spain). Amounts of 0.25 g were added to different solutions: seawater, and three K-enriched seawater solutions prepared by adding KOH to seawater whose final [K] values were 0.1, 0.5 and 1 M, respectively. The experiments were performed at 50°C over a period of 30 days. The XRD patterns showed no mineralogical changes in residues from seawater or from the 0.1 M [K] solution. With increasing pH and K molarity, the smectite peak, initially at 1.4 nm, became broader. This change in the smectite peak was more significant in the residue from the 1 M [K] solution. The appearance of a small shoulder at 1.0 nm in the residue from a 0.5 M [K] solution showed the beginning of illite formation. However, its appearance was clearer in XRD patterns of the residue corresponding to the 1 M [K] solution. The XRD data from air-dried, glycolated, and heated samples from the 1 M [K] solution indicated the presence of smectite, disordered interstratified illite-smectite (I-S) and illite.
The TEM/AEM studies were performed on the residue corresponding to the 1 M [K] experiment. The HRTEM images revealed that smectite and illite occurred as separated packets with a ferroan lizardite, as a by-product of the smectite-to-illite reaction, interstratified and intergrown with illite. Smectite occurs both as ‘rims’ on the illite packet and in its core. The presence of smectite in the core of illite packet indicates that the lateral transition from smectite to illite was incomplete, taking place by direct replacement of smectite layers as a whole through a dissolution-precipitation mechanism. The experimental study shows that smectite may transform in a wide range of geological and artificial environments involving high-pH K-rich solutions.
Transmission and Analytical Electron Microscopy Evidence for High Mg Contents of 1M Illite: Absence of 1M Polytypism in Normal Prograde Diagenetic Sequences of Pelitic Rocks
- Donald R. Peacor, Blanca Bauluz, Hailiang Dong, David Tillick, Yonghong Yan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 757-765
-
- Article
- Export citation
-
The normal prograde diagenetic and low-grade metamorphic sequence of dioctahedral clay minerals including illite-rich I-S and illite, as observed by TEM, proceeds from a partially disordered 1Md stacking sequence to 2M1; i.e. 1M does not normally occur as an intermediate polytype. Examples of 1M illite stacking sequences have been studied, however, from the Golden Cross gold deposit, New Zealand, the Broadlands-Ohaaki geothermal system, New Zealand, the Potsdam Sandstone, New York, and the Silverton Caldera, Colorado. Specific clay-mineral packets identified by TEM techniques as 1M illite were found to have anomalously high Mg contents. The Broadlands illite provides the most definitive data, as separate packets of 1M and 2M1 illite coexist. Average compositions for 1M and 2M1 illite are (K1.66Ca0.04)Σ1.70(Al3.32Fe0.31Mg0.57Mn0.06)Σ4.26(Si6.43Al1.57)Σ8O20(OH)4 and (K1.57Na0.31Ca0.03)Σ1.91(Al3.58Fe0.05Mg0.29Mn0.01)Σ3.93(Si6.70Al1.30)Σ8O20(OH)4, respectively. In addition, 1Mdillite, which is the polytype occurring in the common 1Mdto 2M1 prograde sequence, is relatively Mg poor, but coexists with Mg-rich illite in the Silverton Caldera sample.
These data confirm that 1M stacking is caused by compositional anomalies, and thus explain the lack of the 1M stacking sequence in normal diagenetic sequences in pelitic rocks, as most illite in such environments has a relatively small phengitic component. The parameter Δz, a measure of the corrugation of the oxygen sheets, may be the key parameter reflecting the polytypic state of dioctahedral and trioctahedral micaceous minerals. Such composition-determined relations may be related to the occurrence of 1M polytypism in glauconite and celadonite, both dioctahedral 2:1 clay minerals having large Mg or Fe octahedral-cation components, and in trioctahedral micas. Insofar as the 1M stacking sequence does not have the same composition as 2M1 material, these data confirm that the different varieties of illite are not polytypes, sensu stricto.
Chemical Disaggregation of Kaolinitic Claystones (Tonsteins and Flint Clays)
- Don M. Triplehorn, Bruce F. Bohor, William J. Betterton
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 766-770
-
- Article
- Export citation
-
The coarse, non-clay fraction of many flint-like kaolinitic claystones often contains mineral grains diagnostic of the claystone's origin and, in the case of tonsteins (altered volcanic ashes), may also provide minerals suitable for radiometric dating. Separation of the non-clay mineral fraction is often difficult because flint clays and flint-like clays resist slaking in water and thus are difficult to disaggregate. Chemical disaggregation of resistant kaolinitic claystones may be achieved by immersion in either hydrazine monohydrate or DMSO for periods ranging from one day to several weeks. Generally, hydrazine monohydrate works more quickly and efficiently than DMSO to disaggregate most kaolinitic claystones and flint clays.
Effect of Acid and Alkali Treatments on Surface Areas and Adsorption Energies of Selected Minerals
- Grzegorz Jozefaciuk, Grzegorz Bowanko
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 771-783
-
- Article
- Export citation
-
Bentonite, biotite, illite, kaolin, vermiculite and zeolite were acidified or alkalized with hydrochloric acid or sodium hydroxide at concentrations of 0.1, 1.0 and 5.0 mole dm−3 at room temperature for two weeks. In acid treatments, dissolution of Al prevailed over Si and the opposite was observed in alkali treatments. The XRD patterns showed severe alteration of the crystal structure after acid treatments, whereas sharpening of the XRD peaks after alkali treatments was observed. Illite and kaolin were most resistant to acid attack. with a few exceptions, the surface areas of the minerals computed from both water and nitrogen adsorption isotherms increased with acid and alkali treatments. with increasing reagent concentration, the nitrogen surface area increased faster than the water surface area. well-defined trends were not noted in either changes of average water or nitrogen adsorption energies or in relative amounts of adsorption sites, indicating that the effects of acid and alkali attack are controlled by the individual character of the minerals.
Influence of Gibbsite Surface Area and Citrate on Ni Sorption Mechanisms at pH 7.5
- Noriko U. Yamaguchi, Andreas C. Scheinost, Donald L. Sparks
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 784-790
-
- Article
- Export citation
-
We investigated the sorption of Ni to gibbsite of two different surface areas at pH 7.5, in the presence and absence of citrate, over a time period of 180 days. Extended X-ray absorption fine-structure spectroscopy was employed to elucidate the sorption mechanisms at the molecular level. In agreement with former results, Ni-Al layered double hydroxide (LDH) formed in the presence of gibbsite of low surface area. However, gibbsite of high surface area suppressed the formation of the surface precipitate. Instead, two Al atoms neighboring Ni at distances of 2.95–2.98 Å indicated formation of an inner-sphere sorption complex, where each NiO6-octahedron shares edges with two AlO6-octahedra. Focused multiple scattering arising from Al atoms at a distance of 6 Å suggest that sorbed Ni(OH)2(OH2)4 monomers epitaxially extend the hexagonal arrangement of Al atoms in gibbsite. Only after 30 days or more was a small amount of LDH formed. The presence of citrate prevented the formation of LDH, while maintaining the formation of inner-sphere sorption complexes.
Emanation Thermal Analysis of Natural and Chemically-Modified Vermiculite
- J. Poyato, L. A. Pérez-Maqueda, A. Justo, V. Balek
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 791-798
-
- Article
- Export citation
-
Emanation thermal analysis (ETA), based on radon release measurements from previously labeled samples, was used for the first time in the characterization of the thermal behavior of natural Mg2+-vermiculite (Santa Olalla, Huelva, Spain) and of Na+- and
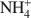 -exchanged vermiculite samples. In addition, vermiculite samples subjected to a chemical treatment with an aqueous solution of (NH4)2SiF6 and partially or totally re-saturated with Na+ ions were also investigated by ETA. The ETA results of natural Mg2+-vermiculite, Na+-vermiculite and
-exchanged vermiculite samples. In addition, vermiculite samples subjected to a chemical treatment with an aqueous solution of (NH4)2SiF6 and partially or totally re-saturated with Na+ ions were also investigated by ETA. The ETA results of natural Mg2+-vermiculite, Na+-vermiculite and 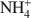 -vermiculite gave supplementary information about microstructure changes of the samples observed under dynamic heating conditions. The method has proved to be very useful for characterization of microstructure changes due to modification in the interlayer space of samples during the heat treatment. The crystallization of vermiculite into new phases, such as enstatite (for
-vermiculite gave supplementary information about microstructure changes of the samples observed under dynamic heating conditions. The method has proved to be very useful for characterization of microstructure changes due to modification in the interlayer space of samples during the heat treatment. The crystallization of vermiculite into new phases, such as enstatite (for 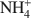 -vermiculite and Mg2+-vermiculite) and forsterite (for Na+-vermiculite) was also observed by ETA.
-vermiculite and Mg2+-vermiculite) and forsterite (for Na+-vermiculite) was also observed by ETA.
Preparation and Characterization of Eu-Magadiite Intercalation Compounds
- Naoko Mizukami, Masashi Tsujimura, Kazuyuki Kuroda, Makoto Ogawa
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 799-806
-
- Article
- Export citation
-
The intercalation of europium ions (Eu3+) into the interlayer space of a layered silicate, magadiite, was conducted by ion-exchange reactions between magadiite and europium(III) chloride. X-ray diffraction and elemental analysis results indicated that Eu3+ cations were intercalated into the interlayer space of magadiite. The ion exchange between Eu3+ and Na+ occurred preferentially so that the adsorbed Eu3+ amounts were controlled quantitatively. Thermal transformation of the original layered structure was suppressed by the intercalation of Eu3+. The resulting intercalation compounds exhibited photoluminescence arising from the intercalated Eu3+. The luminescence intensity varied in accordance with the amount of Eu3+ absorbed, suggesting that the self-quenching occurred at higher loading levels. The luminescence intensity was also changed by the heat treatment, corresponding to the change in the surroundings of the Eu3+ adsorbed, induced by the removal of the adsorbed water molecules and the hydroxyl groups of the silicate.
Diagenesis and Rheology of a Recent-Pleistocene Volcanogenic Sedimentary Sequence, Mexican Basin
- Liberto de Pablo-Galán, Juan J. de Pablo, M. de Lourdes Chávez-García
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 807-823
-
- Article
- Export citation
-
Diagenesis of the Holocene-Pleistocene volcanogenic sediments of the Mexican Basin produced, in strata of gravel and sand, 1H2O- and 2H2O-smectite, kaolinite, R3-2H2O-smectite (0.75)-kaolinite, R1-2H2O-smectite (0.75)-kaolinite, R3-kaolinite (0.75)-2H2O-smectite and R1-1H2O-smectite (0.75)-kaolinite. Smectite platelets were formed from volcanic glass by loss of Si4+. Partially-formed platelets have Si4+ between 4.55−4.10 a.p.f.u., Mg+Mn and the interlayer charge are relatively uniform while VIAl+Fe3++Ti varies between 0.98 and 1.63 a.p.f.u. Almost fully transformed platelets have Si4+ of 4.08−4.04 a.p.f.u.; Mg+Mn and the interlayer charge decrease proportionally to increasing VIAl+Fe3++Ti. Smectite-kaolinite mixed layers have octahedral occupancies of 2.01–2.15 a.p.f.u., IVAl3+ 0.09–0.55 a.p.f.u. and interlayer charges about half that of smectite; structural formulae corresponding to smectite (0.75–0.80)-kaolinite indicate octahedral occupancy of 2.50 a.p.f.u., tetrahedral replacement 0–0.31 a.p.f.u., and interlayer charge 0.24–0.51 equivalents, some indicating interstratifications of beidellite. Kaolinite is presumed to have formed from K-feldspar; smectite-kaolinite interstratifications sustain the transformation of kaolinite to smectite in an increasingly siliceous high-cation environment. In the mudstones of low-hydraulic conductivity and practically stagnant alkaline fluids, glass was transformed to 2H2O-smectite lamellae of IVAl between 0 and 0.47 a.p.f.u., octahedral occupancy 1.70–2.00 a.p.f.u. and interlayer charge of 0.23–1.21 equivalents, some corresponding to beidellite. The interlayer charge increases with IVAl and decreasing occupancy of the octahedral sheet; the abundance of Mg+Mn is inverse to that of VIAl+Fe3++Ti.
Clay suspensions containing 1H2O- and 2H2O-smectite, kaolinite and R3-2H2O-smectite (0.75)-kaolinite lead to low-energy edge-to-edge particle associations, non-Newtonian pseudoplastic behavior, and maximum apparent viscosity of 180 Pa s at 0.008 s−1 followed by rapid descent. Clay fractions with slightly larger 2H2O-smectite contents and smaller kaolinite contents reach maximum viscosity of 3611 Pa s at a shear rate of 0.0018 s−1 and of 3300 Pa s at 0.0024 s−1. They denote two high-energy face-to-face particle associations, followed by slow descent of the apparent viscosity under viscous flow. Suspensions change from elastic to viscous behavior at shear stresses of 1.03 and 5.91 Pa, respectively. Clay suspension vibrated at a frequency of 1 Hz develops a shear storage dynamic modulus greater than the shear dynamic loss modulus or the energy is preferentially stored, whereas at 5 Hz more energy is dissipated than stored.