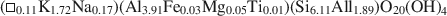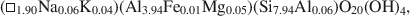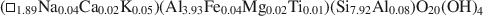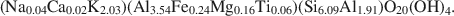Volume 38 - February 1990
Research Article
Transmission and Analytical Electron Microscopic Study of Mixed-Layer Illite/Smectite Formed as an Apparent Replacement Product of Diagenetic Illite
- Wei-Teh Jiang, Donald R. Peacor, R. J. Merriman, B. Roberts
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 449-468
-
- Article
- Export citation
-
Ordered illite/smectite (I/S) and illite in a pelitic rock from a prograde metamorphic sequence in North Wales were observed by transmission electron microscopy. The dominant phyllosilicate noted was diagenetic-metamorphic illite, occurring as subparallel packets of layers, each about a few hundred Ångstroms thick. It exhibited two-layer polytypism (presumably 2M1) and numerous strain features and had a composition of (K1.21Na0.12)(Al3.36Fe0.31Mg0.33)(Si6.28Al1.72)O20(OH)4.
I/S occurred as thick packets of wavy layers having 10-Å subperiodicity and sharp differences in contrast in successive lattice fringes. All stages in a replacement series were noted, from one or two layers of smectite within illite, through thin packets of I/S, to thick packets that contained inherited deformation textures of diagenetic-metamorphic illite. Deformed illite was replaced by I/S more commonly than was undeformed illite. The I/S replacing undeformed original illite had significantly greater order, primarily of R1 type (ISISIS…), than that replacing deformed illite. R> 1 I/S occurred as small crystallites and contained relatively less smectite than the ordered I/S, Single smectite layers were spaced within several illite layers, forming curved packets of layers. IISIIS… (R2) and IIISIIIS… (R3) ordering were present locally, as was discrete smectite. Analytical electron microscopic analyses indicated that the I/S, (K0.46Na0.43)(Al3.75Fe0.06Mg0.19)(Si6.26Al1.74)O20(OH)4, was rectorite-like in composition and had smaller (Mg + Fe) contents and greater Al/Si ratios than the coexisting illite, which was also anomalous in terms of general crystal-chemical relationships between coexisting illite and I/S in burial diagenesis environments. The I/S appears to have formed by replacement of diagenetic-metamorphic illite, presumably at very low temperatures under hydrous conditions via dissolution and crystallization.
Adsorption of Benzene, Toluene, and Xylene by Two Tetramethylammonium-Smectites Having Different Charge Densities
- Jiunn-Fwu Lee, Max M. Mortland, Cary T. Chiou, Daniel E. Kile, Stephen A. Boyd
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 113-120
-
- Article
- Export citation
-
A high-charge smectite from Arizona [cation-exchange capacity (CEC) = 120 meq/100 g] and a low-charge smectite from Wyoming (CEC = 90 meq/100 g) were used to prepare homoionic tetra-methylammonium (TMA)-clay complexes. The adsorption of benzene, toluene, and o-xylene as vapors by the dry TMA-clays and as solutes from water by the wet TMA-clays was studied. The adsorption of the organic vapors by the dry TMA-smectite samples was strong and apparently consisted of interactions with both the aluminosilicate mineral surfaces and the TMA exchange ions in the interlayers. In the adsorption of organic vapors, the closer packing of TMA ions in the dry high-charge TMA-smectite, compared with the dry low-charge TMA-smectite, resulted in a somewhat higher degree of shape-selective adsorption of benzene, toluene, and xylene. In the presence of water, the adsorption capacities of both samples for the aromatic compounds were significantly reduced, although the uptake of benzene from water by the low-charge TMA-smectite was still substantial. This lower sorption capacity was accompanied by increased shape-selectivity for the aromatic compounds. The reduction in uptake and increased selectivity was much more pronounced for the water-saturated, high-charge TMA-smectite than for the low-charge TMA-smectite. Hydration of the TMA exchange ions and/or the mineral surfaces apparently reduced the accessibility of the aromatic molecules to interlamellar regions. The resulting water-induced sieving effect was greater for the high-charge TMA-smectite due to the higher density of exchanged TMA-ions. The low-charge Wyoming TMA-smectite was a highly effective adsorbent for removing benzene from water and may be useful for purifying benzene-contaminated water.
High-Resolution Transmission Electron Microscopy and Electron Diffraction of Mixed-Layer Illite/Smectite: Experimental Results
- David R. Veblen, George D. Guthrie, Jr., Kenneth J. T. Livi, Robert C. Reynolds, Jr.
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 1-13
-
- Article
- Export citation
-
High-resolution transmission electron microscopy (HRTEM) and electron diffraction experiments have been performed on R1 and R> 1 illite/smectite (I/S) samples that from X-ray powder diffraction (XRD) experiments appear to contain well-ordered layer sequences. The HRTEM images confirmed earlier computer image simulations, which suggested that periodicities due to I/S ordering can be imaged in TEM instruments of moderate resolution. The experiments also confirmed that in instruments of this sort, the strongest contrast arising from the compositional difference between I and S layers occurs under rather unusual imaging conditions of strong overfocus. Some selected-area electron diffraction (SAD) patterns showed additional diffraction spots consistent with R1 and R3 ordering. SAD patterns and cross-fringes arising in HRTEM images from non-00l reciprocal lattice rows indicated that the stacking vectors of most adjacent 2:1 layers were not randomly oriented with respect to each other. Thus, the I/S was not fully turbostratic, but instead consisted of very thin, coherently stacked crystallites that extended across the fundamental particles postulated by Nadeau and coworkers.
S/(I + S) ratios were determined for about seventy HRTEM images obtained and interpreted by three different TEM operators. These ratios were consistent with those obtained from standard XRD procedures, suggesting that results obtained by XRD can be used to infer the initial structural state of mixed-layer I/S prior to treatment of samples for XRD experiments. The HRTEM experiments thus demonstrated that the two specimens examined consisted of ordered I/S existing as small crystals, most of which contained more layers than the fundamental particles of Nadeau and coworkers. The non-turbostratic stacking suggests an energetic interaction between the individual fundamental particles, leading to at least two alternative thermodynamic descriptions of these materials. Although the I/S crystals in the present experiments probably were disaggregated into fundamental particles during sample preparation for XRD, the I/S crystals appear to have separated only along the smectite interlayers. If the term “fundamental particle” is to be used for primary, untreated I/S, its original definition should be modified to include not only free particles, but also those that occur as layers within small crystals. It further should be recognized that these particles can interact thermodynamically and crystallographically with their neighbors.
Molecular-Scale Imaging of Clay Mineral Surfaces with the Atomic Force Microscope
- H. Hartman, Garrison Sposito, Andrew Yang, S. Manne, S. A. C. Gould, P. K. Hansma
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 337-342
-
- Article
- Export citation
-
Specimen samples of Crook County montmorillonite and Silver Hill illite, purified and prepared in the Na-form, were imaged under 80% relative humidity using an atomic force microscope. The direct images showed clearly the hexagonal array of hexagonal rings of oxygen ions expected for the basal planes of 2:1 phyllosilicates. Fourier transformation of the digital information obtained by the microscope scanning tip led to an estimate of 5.1 ± 0.3 Å for the nearest-neighbor separation, in agreement with the ideal nearest-neighbor spacing of 5.4 Å for hexagonal rings as derived from X-ray powder diffraction data. The atomic force microscope should prove to be a useful tool for the molecular-scale resolution of clay mineral surfaces that contain adsorbed macromolecules.
Transmission Electron Microscopic Study of Coexisting Pyrophyllite and Muscovite: Direct Evidence for the Metastability of Illite
- Wei-Teh Jiang, Eric J. Essene, Donald R. Peacor
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 225-240
-
- Article
- Export citation
-
Transmission electron microscopy has been used to characterize coexisting pyrophyllite and muscovite in low-grade metamorphosed pelites from Witwatersrand and northeastern Pennsylvania. The Witwatersrand sample consisted largely of porphyroblasts of interlayered muscovite and pyrophyllite in a fine-grained matrix of the same phases. In both textures, muscovite and pyrophyllite occurred as interlayered packets (with apparently coherent interfaces) from about 300 Å to a few micrometers in thickness, with no mixed layering. Their compositions were determined with a scanning transmission electron microscope to be
$$(\Box{_{{\rm{0}}{\rm{.11}}}}{\rm{ }}{{\rm{K}}_{{\rm{1}}{\rm{.72}}}}{\rm{Na}_{{\rm{0}}{\rm{.17}}})(A}{{\rm{l}}_{{\rm{3}}{\rm{.91}}}}{\rm{ F}}{{\rm{e}}_{{\rm{0}}{\rm{.03}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.05}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.01}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{6}}{\rm{.11}}}}{\rm{ Al}}{{\rm{l}}_{{\rm{1}}{\rm{.89}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}$$and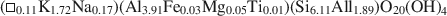 $$(\Box_{{\rm{1}}{\rm{.90}}}{\rm{N}}{{\rm{a}}_{{\rm{0.06}}}}{{\rm{K}}_{{\rm{0}}{\rm{.04}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.94}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.01}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.05}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{7}}{\rm{.94}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.06}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}},$$respectively.
$$(\Box_{{\rm{1}}{\rm{.90}}}{\rm{N}}{{\rm{a}}_{{\rm{0.06}}}}{{\rm{K}}_{{\rm{0}}{\rm{.04}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.94}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.01}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.05}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{7}}{\rm{.94}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.06}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}},$$respectively.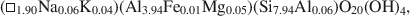
The pyrophyllite and muscovite in the Pennsylvania shale likewise occurred only as coexisting coherent to sub-parallel packets as thin as 200 Å, with compositions of
$$(\Box_{{\rm{1}}{\rm{.89}}}{\rm{N}}{{\rm{a}}_{{\rm{0.04}}}}{\rm{C}}{{\rm{a}}_{{\rm{0}}{\rm{.02}}}}{{\rm{K}}_{{\rm{0}}{\rm{.05}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.93}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.04}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.02}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.01}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{7}}{\rm{.92}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.08}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}$$and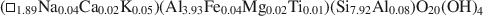 $$({\rm{N}}{{\rm{a}}_{{\rm{0}}{\rm{.04}}}}{\rm{C}}{{\rm{a}}_{{\rm{0}}{\rm{.02}}}}{{\rm{K}}_{{\rm{2}}{\rm{.03}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.54}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.24}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.16}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.06}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{6}}{\rm{.09}}}}{\rm{A}}{{\rm{l}}_{{\rm{1}}{\rm{.91}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}.$$The textures of both samples were consistent with an equilibrium relationship between pyrophyllite and muscovite. The Pennsylvania sample also contained NH4-rich illite, kaolinite, and an illite-like phase having intermediate Na/K, which collectively imply non-equilibrated low-grade conditions.
$$({\rm{N}}{{\rm{a}}_{{\rm{0}}{\rm{.04}}}}{\rm{C}}{{\rm{a}}_{{\rm{0}}{\rm{.02}}}}{{\rm{K}}_{{\rm{2}}{\rm{.03}}}}{\rm{)(A}}{{\rm{l}}_{{\rm{3}}{\rm{.54}}}}{\rm{F}}{{\rm{e}}_{{\rm{0}}{\rm{.24}}}}{\rm{M}}{{\rm{g}}_{{\rm{0}}{\rm{.16}}}}{\rm{T}}{{\rm{i}}_{{\rm{0}}{\rm{.06}}}}{\rm{)(S}}{{\rm{i}}_{{\rm{6}}{\rm{.09}}}}{\rm{A}}{{\rm{l}}_{{\rm{1}}{\rm{.91}}}}{\rm{)}}{{\rm{O}}_{{\rm{20}}}}{{\rm{(OH)}}_{\rm{4}}}.$$The textures of both samples were consistent with an equilibrium relationship between pyrophyllite and muscovite. The Pennsylvania sample also contained NH4-rich illite, kaolinite, and an illite-like phase having intermediate Na/K, which collectively imply non-equilibrated low-grade conditions.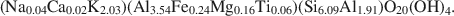
The compositions of these coexisting pyrophyllite and muscovite define a solvus with steep limbs and extremely limited solid solution. Illite is a white mica, intermediate in composition between pyrophyllite and muscovite, formed at much lower temperatures than muscovite. These relations show that illite is metastable relative to pyrophyllite + muscovite in all of its diagenetic and low-grade metamorphic occurrences. This further implies that illite precursor phases, such as smectite, are also metastable. The prograde reactions involving smectite, illite, and muscovite are therefore inferred to represent Ostwald-step-rule-like advances through a series of metastable phases toward the equilibrium states attained in the greenschist facies. “Illite crystallinity” can therefore be a measure of reaction progress, for which temperature is only one of several determining factors.
Mineralogy, Chemistry, and Diagenesis of Tuffs in the Sucker Creek Formation (Miocene), Eastern Oregon
- Stephen P. Altaner, Ralph E. Grim
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 561-572
-
- Article
- Export citation
-
The lacustrine Sucker Creek Formation (Miocene) of eastern Oregon includes unaltered vitric tuffs as well as tuffs altered to the following diagenetic fades: (1) bentonite, (2) interbedded bentonite and opal-CT, (3) K-clinoptilolite, and (4) Ca-clinoptilolite. Bentonite beds contain Fe-rich smectite (8–10 wt. % Fe2O3), quartz, plagioclase, and Ca-clinoptilolite. Opal-CT-rich layers contain inorganic silica (opal-CT), Fe-rich smectite, and minor diatoms. K-clinoptilolite beds typically contain clinoptilolite that can be extremely K-rich (≤7.6 wt. % K2O), opal-CT, smectite, plagioclase, and K-feldspar. This diagenetic facies also includes smectitic tuff and unaltered tuff. Ca-clinoptilolite beds contain Ca-clinoptilolite, quartz, K-feldspar, smectite, and illite.
Based on its chemistry and mineralogy, the bentonite appears to have been derived from dacitic volcanic ash. Chemical considerations and the close spatial relationship between beds of bentonite and opal-CT suggest that the diagenetic alteration of glass to smectite provided silica to the adjacent opal-CT beds. Based on the presence of late-stage Ca-clinoptilolite, alteration appears to have proceeded in a relatively closed chemical system.
Based on the composition of preserved vitric tuff, the zeolitic tuffs appear to be derived from rhyolitic ash, which diagenetically altered in an open hydrologic system and produced vertical zonations in mineralogy. In this model, bentonite horizons at the top of the K-clinoptilolite diagenetic fades formed by reaction of volcanic glass with dilute fluids that had a relatively low (Na+ + K+ + Ca2+)/H+ activity ratio and
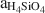 , whereas the underlying K-clinoptilolite beds formed from reactions between glass and dilute fluids having a higher (Na+ + K+ + Ca2+)/H+ activity ratio and
, whereas the underlying K-clinoptilolite beds formed from reactions between glass and dilute fluids having a higher (Na+ + K+ + Ca2+)/H+ activity ratio and 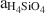 . Unaltered vitric ash between these beds may represent zones of higher permeability that inhibited secondary mineral alteration. Ca-clinoptilo-lite-rich beds appear to have undergone alteration similar to K-clinoptilolite-rich beds as well as to have been subjected to later, low-temperature (perhaps 75°–150°C) hydrothermal alteration which enhanced cation exchange in the zeolite and formed quartz from opal-CT.
. Unaltered vitric ash between these beds may represent zones of higher permeability that inhibited secondary mineral alteration. Ca-clinoptilo-lite-rich beds appear to have undergone alteration similar to K-clinoptilolite-rich beds as well as to have been subjected to later, low-temperature (perhaps 75°–150°C) hydrothermal alteration which enhanced cation exchange in the zeolite and formed quartz from opal-CT.
Transformation of Akaganéite Into Goethite and Hematite in Alkaline Media
- R. M. Cornell, R. Giovanoli
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 469-476
-
- Article
- Export citation
-
The conversion of akaganéite to goethite and/or hematite in alkaline media has been followed by X-ray powder diffraction and transmission electron microscopy (TEM). The rate of transformation fell and the amount of hematite in the product increased as the [OH−] decreased to < 1 M. Kinetic studies and TEM indicated that the transformation involved dissolution of akaganéite followed by reprecipitation of goethite and/or hematite. The rate-determining step was the dissolution of akaganéite.
Silicate species retarded the formation of goethite + hematite principally by inhibiting dissolution of akaganéite; to a lesser extent, they interfered with the nucleation of goethite. Silicate modified the morphology of goethite, but not hematite.
Comparison of the transformation behavior of akaganéite with that previously observed for ferrihydrite indicated that the composition of the reaction product depended strongly on the transformation conditions, i.e., pH and the presence of foreign species. The nature of the solid precursor was important insofar as its degree of crystallinity governed the dissolution kinetics and its surface properties influenced interaction with any foreign species in the system.
Influence of Hydrazine on the Vibrational Modes of Kaolinite
- C. T. Johnston, D. A. Stone
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 121-128
-
- Article
- Export citation
-
Raman and Fourier-transform-infrared (FTIR) spectroscopic methods and X-ray powder diffraction (XRD) techniques have been used to study the influence of hydrazine on the vibrational modes of kaolinite. Strong vibrational perturbations of the OH-stretching and -deformation bands were observed in the Raman and FTIR spectra on intercalation. The intensities of the Raman- and IR-active OH-stretching bands decreased significantly upon intercalation; the intensities of the Raman bands were reduced to a greater extent than the IR bands. The deformation bands were also strongly perturbed by the presence of hydrazine in the interlamellar region. Upon evacuation of the intercalate, two new bands at 3628 and 912 cm−1 were noted, indicating the presence of a different structural conformation of the complex under vacuum. Similar results were obtained using XRD, on evacuation of the kaolinite-hy-drazine (KH) complex the d(001) value decreased from 10.4 to 9.6 Å. Partial collapse of the intercalate from 10.4 to 9.6 Å was probably due to keying of the -NH2 moiety of hydrazine into the siloxane ditrigonal cavity, as indicated by a blue-shift of the inner-OH band from 3620 to 3628 cm-1. Structural OH vibrational modes may therefore be useful probes of amine interactions with clay mineral surfaces.
Hydrophobicity of Clay Surfaces: Sorption of 1,2-Dibromoethane and Trichloroethene
- B. L. Sawhney, M. P. N. Gent
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 14-20
-
- Article
- Export citation
-
Sorption and desorption of two neutral, nonpolar organic compounds, 1,2-dibromoethane (EDB, a soil fumigant) and trichloroethene (TCE, an industrial solvent and common ground-water contaminant), by pyrophyllite, kaolinite, illite, and smectite were investigated. For sorption, vapors of the compounds in a stream of dry N2 gas were passed through columns of the powdered clay minerals for different periods of time. The compounds retained by the clays were extracted with methanol and analyzed by gas chromatography (GC). For desorption, N2 was passed through the treated samples, and the desorbed compounds were collected in hexane traps and analyzed by GC.
Initially sorption was rapid for several hours but then proceeded at a slower rate for many hours. Surprisingly, the clays sorbed large quantities of these neutral compounds; for example, the pyrophyllite, kaolinite, illite, and smectite sorbed about 3, 5, 6, and 9% EDB, respectively, by weight. The amounts of TCE sorbed, which is more volatile than EDB, were somewhat less. Only a portion of the compounds sorbed over a period of time were desorbed in the same period of time. Desorption was rapid initially but then proceeded at a slow rate; slow desorption continued for over 100 hr from samples which had undergone sorption for only 1–2 hr. A two-compartment efflux model was used to describe the sorption behavior. A rapidly desorbing component was considered to be present on the outer surfaces of the clay aggregates and a slowly desorbing component, in the interior pores of the aggregates.
Characterization of Illitization of Smectite in Bentonite Beds at Kinnekulle, Sweden
- Atsuyuki Inoue, Takashi Watanabe, Norihiko Kohyama, Ann Marie Brusewitz
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 241-249
-
- Article
- Export citation
-
Structure, morphology, and chemical composition of illite/smectite (I/S) containing 30–50% smectite layers (% S) from Kinnekulle bentonites, Sweden, of diagenetic origin were examined using X-ray powder diffraction (XRD) and transmission electron microscopy (TEM). Interlayer arrangements of I/S changed from random interstratification to short-range ordered at about 40% S. The transition from random to ordered structure proceeded continuously as reflected by the gradual decrease in probability of two smectite neighbors (Pss) towards zero. TEM observations of water-dispersed samples that had not been cation-exchanged showed that the I/S consisted dominantly of flakes coexisting with laths having a length/width ratio of about 4, regardless of % S. The thickness of the I/S particles ranged from 30 to 100 Å, and no systematic variation in thickness was detected with decreasing % S. The chemical composition of the I/S also changed continuously with decreasing % S. These observations suggest no dissolution of smectite layers and no recrystallization of illite layers during the formation of the I/S in these bentonites; rather, cationic substitutions occurred within a smectite precursor (termed a solid-state transformation mechanism). A comparison of interlayer order, particle texture, and chemistry of the I/S from various types of rocks suggests that the mechanism of smectite-to-illite conversion in the range 100% S-30% S was related to the porosity and permeability of original rocks. The solid-state transformation mechanism appears to have predominated in rocks of low porosity and permeability.
Polarized Single-Crystal Fourier-Transform Infrared Microscopy of Ouray Dickite and Keokuk Kaolinite
- C. T. Johnston, S. F. Agnew, D. L. Bish
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 573-583
-
- Article
- Export citation
-
Single-crystal Fourier-transform infrared (FTIR) spectra of Keokuk kaolinite and Ouray dickite were obtained with an FTIR microscope. Although numerous IR, FTIR, and Raman spectra of polycrystalline kaolinite and dickite can be found in the literature, the present data represent the first reported single-crystal vibrational spectra for these clay minerals. The orientation of the crystallographic axes of dickite was determined using a cross-polarizing optical microscope fitted with an 550-nm optical retardation plate. Assignment of the inner hydroxyl group OH1 to the 3623-cm-1 band was confirmed, and the angle of this OH group to the b-axis was determined to be 47° based upon the measured dichroic ratio. The 3702-3710-cm−1 absorption feature appeared to consist of two closely spaced bands having slightly different polarization behavior. The inner-surface hydroxyl group OH3 was assigned to the absorption bands at 3710 cm−1. The calculated angle of the OH3 groups to the b-axis was found to be 22°, which agrees well with the angles determined by X-ray powder diffraction and neutron diffraction. The remaining hydroxyl groups, OH2 and OH4, were assigned to the 3656 cm-1 band; the angle of the OH2 and OH4 groups to the b-axis was measured at 45°. The polarization behavior of the OH-deformation bands of dickite at 911, 937, and 952 cm−1 was found to be similar to that observed in the OH-stretching region. Single-crystal FTIR spectra of Keokuk kaolinite showed that rotation of the electric vector around the c/z axis in the ab plane of kaolinite resulted in a behavior distinct from that of dickite. The OH-stretching bands of kaolinite were found to be considerably more polarized than the corresponding bands of dickite. This is related directly to the fact that dickite possesses a glide plane (space group Cc) compared with kaolinite, which does not (space group C1).
Role of Water in the Smectite-to-Illite Reaction
- Gene Whitney
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 343-350
-
- Article
- Export citation
-
A series of hydrothermal experiments was performed to determine the effect of fluid abundance on the reaction of smectite to illite. Experiments were conducted on K-saturated montmorillonite (<0.1-µm fraction) in a closed system at 250° to 400°C using run times of 1, 7, 14, 30, and 60 days at 100 MPa (1 kbar) pressure. In fluid-deficient systems (pore spaces not saturated), the rate and extent of illitization was significantly inhibited. A rock: water ratio of 20:1 (mass: mass) produced an R0 illite/smectite (I/S) having 82% smectite layers after 60 days at 250°C, whereas a rock: water ratio of 1:1 produced an I/S having 57% smectite layers under the same conditions. The effect became less pronounced at higher temperatures, with the 20:1 and the 1:1 experimental products differing by only 11% expandability at 400°C after 60 days. In addition, the low-fluid experiments produced fewer crystalline byproducts (quartz, cristobalite, chlorite) than did the fluid-rich runs, and the I/S was more difficult to disperse and orient in the fluid-deficient samples, suggesting enhanced cementation at grain contacts or the production of particle morphologies that did not lend themselves to orientation. The difference in reactivity of the smectite and I/S as a function of water content appears to be attributable to the reduced capacity for low volumes of water to mediate the dissolution, solute transport, and precipitation reactions that make up the series of reactions collectively termed illitization. Of these variables, solute transport is likely to be affected most by reduction of fluid.
Far-Infrared Study of Potassium in Micas
- R. Prost, V. Laperche
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 351-355
-
- Article
- Export citation
-
The characterization of cations associated with clay materials has generally been approached by the sequential use of specific chemical reagents. To avoid the disturbing effect of the chemical reagents on the state and location of compensating cations of clays and to get information in situ, far-infrared spectroscopy was used. The far-infrared vibrational spectra of the potassium cation in muscovite, phlogopite, and biotite were recorded before and after heating at the dehydroxylation temperature. The vibrational frequency of K in micas before dehydroxylation was found to be a function of the di- or trioctahedral character and of the Fe content. After dehydroxylation or deprotonation, shifts of the K absorption band to lower frequencies were observed for the heated muscovite, in which K exchangeability increased, and to higher frequencies for the heated biotite, in which K exchangeability decreased. These results suggest that the vibrational frequency of potassium is characteristic of the state of K in these minerals and of its ability to be exchanged.
Mica Alteration Reactions in Jurassic Reservoir Sandstones from the Haltenbanken Area, Offshore Norway
- Sadoon Morad
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 584-590
-
- Article
- Export citation
-
Petrographic observations indicate that dolomite, siderite, pyrite, Ti oxides, quartz, and K-feldspar formed as by-products of mica alteration during diagenesis of Jurassic reservoir sandstones of the Haltenbanken area (offshore central Norway). These minerals precipitated on a mica-grain scale or a thin-section scale. Modal analyses and mass-balance calculations of muscovite alteration to kaolinite and of biotite to kaolinite, illite, and chlorite suggest limited elemental mobility during sandstone diagenesis. The alteration of mica to kaolinite occurred in sandstones buried <2200 m deep (present temperature < 70°C), whereas illitization and chloritization of biotite occurred during burial to depths > 3000 m (present temperature >95°C).
Thermal Stability of Halloysite by High-Pressure Differential Thermal Analysis
- Sheryl L. Johnson, Stephen Guggenheim, A. F. Koster van Groos
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 477-484
-
- Article
- Export citation
-
Platy (Te Puke, New Zealand), cylindrical (Spruce Pine, North Carolina), and spherical (North Gardiner mine, Huron, Lawrence County, Indiana) halloysite samples were analyzed by high-pressure differential thermal analysis (HP-DTA) to determine the effect of morphological and chemical differences on their respective thermal stability. In halloysite, these morphological differences imply structural features. The metastable phase relations of each are analogous to those of kaolinite. At 1 bar, the platy, cylindrical, and spherical samples showed peak temperatures (maximum deflection in the dehydroxylation endotherm) of 560°, 578°, and 575°C, respectively, whereas at about 600 bars the peak temperatures were 622°, 655°, and 647°C. At low pressures the observed reaction is related to dehydroxylation: halloysite (H) metahalloysite (MH) + vapor (V), whereas higher pressures produce melting reactions, either H + V = metaliquid (ML) for conditions of P(H2O) = P(total), or H + MH = ML for P(H2O) < P(total). The PT conditions of the invariant point, H + MH + ML + V, for each system are: Te Puke, 612° ± 4°C, 25 ± 7 bars; Spruce Pine, 657 ± 2°C, 30 ± 7 bars; North Gardiner, 652° ± 2°C, 34 ± 7 bars. The lower thermal stability of the Te Puke sample may be related to its higher iron content, although additional data are necessary to confirm that it is not related also to the platy structure. Furthermore, morphological differences between the cylindrical and spherical varieties appear to have had little effect on the energy required to dehydroxylate these halloysite structures. Exceptionally high values obtained for the dehydroxylation enthalpies using the van't Hoff equation, compared with values derived using other methods, may be explained by a 10–15-bar excess in the intracrystalline H2O fugacity during dehydroxylation. Intracrystalline fugacity is defined here as the H2O fugacity within crystallites and is not related to the partial pressure of H2O around individual particles.
Reactivity of Anisoles on Clay and Pillared Clay Surfaces
- Kathleen A. Carrado, Ryoichi Hayatsu, Robert E. Botto, Randall E. Winans
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 250-256
-
- Article
- Export citation
-
Pillared bentonites were found to be efficient catalysts for the O-methyl bond cleavage of anisoles (e.g., m-methylanisole, guaiacol, and creosol) under very mild, static conditions (150°C, a few hours, inert atmosphere). The O-methyl bond cleavage led to phenolic products. Gas chromatographymass spectrometry and solid-state 13C nuclear magnetic resonance (NMR) techniques used to probe 13C-labeled anisoles revealed that dealkylation and transalkylation reactions occurred to a large extent, and that conversion was efficient at >95% after two days. Ortho- and para-isomers were observed exclusively, without any evidence of meta-substitution. Volatile products were determined by mass spectrometry to be 13CH3OH and (13CH3)2O. Magic-angle spinning 13C NMR experiments showed that the molecules were fairly mobile in the clay micropores prior to catalysis. After catalysis, cross-polarization NMR showed that molecular motion had decreased markedly. Ultraviolet-visible spectroscopy of the colored complexes suggested some quinone formation. The trend of clay reactivity was found to be: pillared bentonite ≫ acid-washed montmorillonite > untreated bentonite > pillared fluorhectorite ≃ untreated fluorhectorite.
Effect of Cysteine and Manganese on the Crystallization of Noncrystalline Iron(III) Hydroxide at pH 8
- R. M. Cornell, R. Giovanoli, W. Schneider
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 21-28
-
- Article
- Export citation
-
To provide a greater understanding of the crystallization of iron oxides under natural aqueous conditions, the combined effect of an inorganic ion (Mn2+) and a reducing organic ligand (L-cysteine) on the conversion of noncrystalline ferric hydroxide to goethite and/or hematite was investigated at pH 8.
At cysteine: Fe ratios ≥ 0.2, L-cysteine caused noncrystalline iron(III) hydroxide to transform rapidly into goethite at pH 8; in the absence of the organic ligand, hematite was the predominant reaction product. The presence of Mn (≥9 mole %) in the cysteine-ferric hydroxide system retarded crystallization and reduced the goethite-promoting effect of cysteine.
Polarographic measurements showed that the adsorption of cysteine on noncrystalline iron(III) hydroxide was immediately followed by the oxidation of cysteine to the disulfide with simultaneous reduction of a proportion of the interracial ferric ions. The partly reduced noncrystalline iron(III) hydroxide dissolved at pH 8 more rapidly than the original material, thus facilitating the formation of goethite. In Mn(II)-noncrystalline iron(III) hydroxide coprecipitates, the interfacial oxidation/reduction reaction with cysteine (and hence the partial reduction of the noncrystalline phase) was reduced, due to replacement of some interfacial Fe(III) by non-reducible Mn.
At pH 8, uptake of Mn by crystalline iron oxides was low (< 5 mole %). Mn precipitated preferentially as pure Mn phases, either rhodochrosite (in NaHCO3 buffer) or hausmannite (in NH4Cl/NH3 buffer).
Identification of Noncrystalline (Fe,Cr)(Oh)3 by Infrared Spectroscopy
- James E. Amonette, Dhanpat Rai
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 129-136
-
- Article
- Export citation
-
Iron-chromium hydroxides are important solid phases governing the aqueous concentrations of Cr(III) in soils and fly ashes. Although direct identification of noncrystalline (Fe,Cr)(OH)3 is difficult, the infrared spectra of noncrystalline Fe(OH)3 and Cr(OH)3, coprecipitated (Fe,Cr)(OH)3, and physical mixtures of Fe(OH)3 and Cr(OH)3 can be distinguished on the basis of the asymmetric stretching doublet (v3) of structural carbonate anions. As the Cr mole fraction of the coprecipitated (Fe,Cr)(OH)3 increases, the position of the low-frequency v3 peak (v3″) changes progressively to higher frequencies, and the carbonate v3 splitting decreases. No change in carbonate v3 splitting or v3″ location was observed for physical mixtures of Fe(OH)3 and Cr(OH)3. The changes in v3 splitting are believed to be caused by different degrees of polarization of the carbonate ligand by the Fe and Cr cations.
Pure Cr(OH)3 exhibits a strong affinity for carbonate and H2O and tends to remain noncrystalline even at very high pHs. In contrast, pure Fe(OH)3 gradually converts to crystalline goethite at high pH, to the exclusion of much of the H2O and carbonate. The presence of Cr in (Fe,Cr)(OH)3 solid solutions seems to inhibit the transformation to crystalline goethite. The strong association of carbonate with Cr and the kinetic inertness of Cr(III) inner-sphere complexes in general may account for the maintenance of noncrystalline solid-solution materials in lieu of transformation to a crystalline end product.
Preparation of a Kaolinite-Polyacrylamide Intercalation Compound
- Yoshiyuki Sugahara, Shigeo Satokawa, Kazuyuki Kuroda, Chuzo Kato
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 137-143
-
- Article
- Export citation
-
Acrylamide has been polymerized between the layers of kaolinite by heat treatment. Acrylamide monomer was first intercalated by the displacement reaction between a kaolinite-N-methylformamide (NMF) intercalation compound and a 10% acrylamide aqueous solution. The resulting intercalation compound showed a basal spacing of 11.3 Å. Infrared (IR) spectroscopy and 13C nuclear magnetic resonance spectroscopy with cross polarization and magic angle spinning (13C CP/MAS-NMR) indicated the replacement of NMF by acrylamide. IR spectroscopy also showed the formation of hydrogen bonds with the hydroxyls of kaolinite. When the kaolinite-acrylamide intercalation compound was heated at 300°C for 1 hr, the basal spacing increased to 11.4 Å, and IR and 13C CP/MAS-NMR showed the disappearance of C=C bonds, indicating the polymerization of acrylamide. The heat-treated kaolinite-acrylamide intercalation compound was resistant to 30 min-washing with water, whereas the untreated kaolinite-acrylamide intercalation compound collapsed after the same treatment, an observation consistent with acrylamide polymerization between the layers of kaolinite. IR spectroscopy revealed that polyacrylamide was hydrogen bonded to kaolinite, but in a manner different from the hydrogen bonding of acrylamide.
Study of Some Physicochemical Properties of Pillared Montmorillonites: Acid-Base Potentiometric Titrations and Electrophoretic Measurements
- Marcelo J. Avena, Raúl Cabrol, Carlos P. De Pauli
-
- Published online by Cambridge University Press:
- 02 April 2024, pp. 356-362
-
- Article
- Export citation
-
The surface charges and the zeta potential of a Na-montmorillonite (Na-mont) and two pillared montmorillonite (MP1 and MP2) samples with different aluminum contents were determined by Potentiometric titrations and electrophoretic measurements. At pH >9 the two pillared montmorillonite samples showed zeta potentials similar to those of Na-mont, but at pH <8, the negative zeta potential shifted to lower negative values as the aluminum content increased. Sample MP1, which had a greater Al content, showed an isoelectric point (IEP) of 5.0–5.5. Titration curves obtained by acid-base Potentiometrie titration for sample MP1 showed a well-defined cross-over point at pH = 5.0, whereas this point was not observed for sample MP2 in the pH range studied. The results indicate, in principle, that both techniques can be used to characterize surface charges in this type of material. An attempt was also made to relate the data obtained from electrophoretic mobility and Potentiometrie titrations.