Oral Presentation
MDR GNR
How Does Antimicrobial Resistance Increase Medical Costs in Community-Acquired Acute Pyelonephritis?
- Bongyoung Kim, Taul Cheong, Jungmo Ahn
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s23
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The proportion of antimicrobial-resistant Enterobacterales that are causative pathogens for community-acquired acute pyelonephritis (CA-APN) has been increasing. We examined the effect of antimicrobial resistance on medical costs in CA-APN. Methods: A single-center retrospective cohort study was conducted at a tertiary-care hospital in Korea between January 2018 to December 2019. All hospitalized patients aged ≥19 years who were diagnosed with CA-APN were recruited, and those with Enterobacterales as a causative pathogen were included. Comparisons between CA-APN caused by extended-spectrum β-lactamase (ESBL)–producing pathogens (ESBL+ group) and those by non–ESBL-producing organisms (ESBL– group) as well as CA-APN caused by ciprofloxacin-resistant pathogens (CIP-R group) and those by ciprofloxacin-sensitive pathogens (CIP-S group) were performed. Log-linear regression was performed to determine the risk factors for medical costs. Results: In total, 241 patients were included in this study. Of these, 75 (31.1%) had an ESBL-producing pathogen and 87 (36.1%) had a ciprofloxacin-resistant pathogen. The overall medical costs were significantly higher in the ESBL+ group compared with the ESBL− group (US$3,730.18 vs US$3,119.32) P <0.001) as well as in CIP-R group compared with CIP-S group (3,730.18 USD vs. 3,119.32 USD, P =0.005). In addition, length of stay was longer in ESBL+ group compared with ESBL-group (11 vs. 8 days, P <0.001) as well as in CIP-R group compared with CIP-S group (11 vs. 8 days, P <0.001). There were no significant difference in the proportion of clinical failure between ESBL+ and ESBL- groups; CIP-R and CIP-S groups. Based on the log-linear regression model, the costs associated with ESBL-producing Enterobacterales as the causative pathogen would be, on average, 27% higher or US$1,211 higher than its counterpart (P = .026). By the same token, a patient who is a year older would incur US$23 higher cost (P = .040). Having any structural problem in urinary tract would incur US$1,231 higher cost (P = .015). A unit increase in Pitt score would incur US$767 USD higher cost (P < 0.001) higher cost, all other things constant. Conclusions: Medical costs for hospitalized patients with CA-APN are increased by the existence of ESBL-producing Enterobacterales but not by the existence of ciprofloxacin-resistant Enterobacterales.
Funding: No
Disclosures: None
Figure 1.
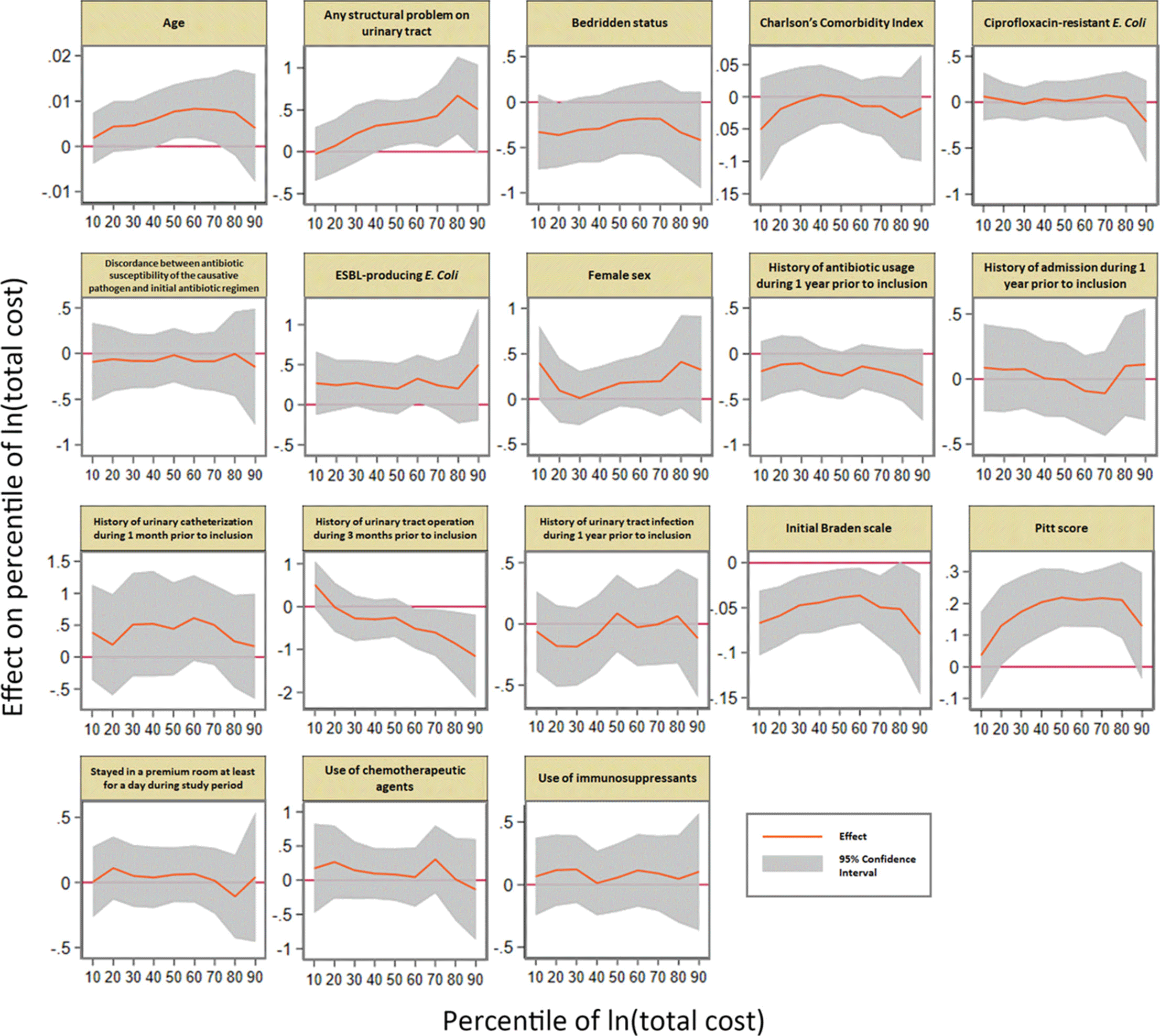
Table 1. 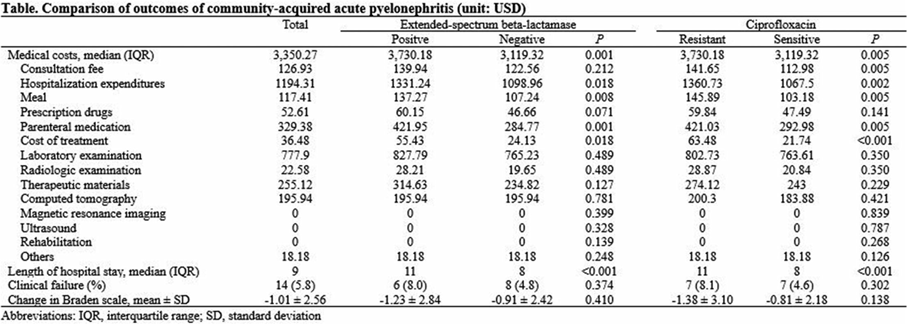
Table 2. 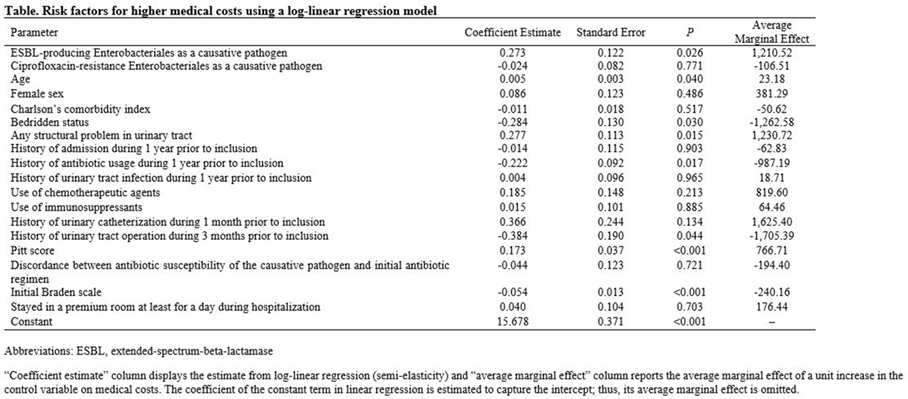
Original Article
Experiences in outpatient parenteral antimicrobial therapy (OPAT): Barriers and challenges from the front lines
- Nicole Ng, Pamela Bailey, Rachel Pryor, Lillian Fung, Christine Veals, Kenneth Sabouri, Julie Reznicek
-
- Published online by Cambridge University Press:
- 03 November 2021, e42
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background:
Outpatient parenteral antimicrobial therapy (OPAT) is now the standard of care for managing patients who no longer need inpatient care but require prolonged intravenous antimicrobial therapy. OPAT increases patient satisfaction, reduces the lengths of hospital stay, lowers emergency department readmission rates, and decreases total healthcare spending.
Objective:To investigate Virginia Commonwealth University Health System’s experience with OPAT and to highlight the obstacles patients and clinicians face when navigating and utilizing this program.
Design:We conducted this descriptive study at a large, academic, tertiary-care hospital in Central Virginia.
Methods:We performed manual reviews of electronic medical records of 602 patient, and we evaluated the records of those receiving OPAT between 2017 and 2020. Reviews included antimicrobial agents, diagnoses requiring OPAT, adverse effects related to antimicrobials, adverse effects related to peripherally inserted central catheters (PICC), readmission rate, discharge destination, and death. We evaluated our program with descriptive statistics.
Results:Among 602 patients who received OPAT, most were diagnosed with bacteremia or musculoskeletal infections. Patients were either discharged home or to another healthcare facility, with the former comprising most of the rehospitalizations. Ertapenem and vancomycin were associated with the most adverse drug events among our cohort. Elevated transaminase levels were noted in 23% of patients. The rate of PICC-line adverse events in this study population was 0.05%.
Conclusions:Our findings highlight the barriers and challenges that patients and providers face when receiving OPAT, and they can inform efforts to improve patient clinical outcomes.
Oral Presentation
Medical Informatics
Automated Nationwide Benchmarking Dashboard for Antimicrobial Stewardship Programs within the Veterans’ Health Administration
- Michihiko Goto, Eli Perencevich, Alexandre Marra, Bruce Alexander, Brice Beck, Daniel Livorsi, Julia Friberg, Christopher Richards, DeShauna Jones, Michael Sauder
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s23-s24
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Group Name: VHA Center for Antimicrobial Stewardship and Prevention of Antimicrobial Resistance (CASPAR) Background: Antimicrobial stewardship programs (ASPs) are advised to measure antimicrobial consumption as a metric for audit and feedback. However, most ASPs lack the tools necessary for appropriate risk adjustment and standardized data collection, which are critical for peer-program benchmarking. We created a system that automatically extracts antimicrobial use data and patient-level factors for risk-adjustment and a dashboard to present risk-adjusted benchmarking metrics for ASP within the Veterans’ Health Administration (VHA). Methods: We built a system to extract patient-level data for antimicrobial use, procedures, demographics, and comorbidities for acute inpatient and long-term care units at all VHA hospitals utilizing the VHA’s Corporate Data Warehouse (CDW). We built baseline negative binomial regression models to perform risk-adjustments based on patient- and unit-level factors using records dated between October 2016 and September 2018. These models were then leveraged both retrospectively and prospectively to calculate observed-to-expected ratios of antimicrobial use for each hospital and for specific units within each hospital. Data transformation and applications of risk-adjustment models were automatically performed within the CDW database server, followed by monthly scheduled data transfer from the CDW to the Microsoft Power BI server for interactive data visualization. Frontline antimicrobial stewards at 10 VHA hospitals participated in the project as pilot users. Results: Separate baseline risk-adjustment models to predict days of therapy (DOT) for all antibacterial agents were created for acute-care and long-term care units based on 15,941,972 patient days and 3,011,788 DOT between October 2016 and September 2018 at 134 VHA hospitals. Risk adjustment models include month, unit types (eg, intensive care unit [ICU] vs non-ICU for acute care), specialty, age, gender, comorbidities (50 and 30 factors for acute care and long-term care, respectively), and preceding procedures (45 and 24 procedures for acute care and long-term care, respectively). We created additional models for each antimicrobial category based on National Healthcare Safety Network definitions. For each hospital, risk-adjusted benchmarking metrics and a monthly ranking within the VHA system were visualized and presented to end users through the dashboard (an example screenshot in Figure 1). Conclusions: Developing an automated surveillance system for antimicrobial consumption and risk-adjustment benchmarking using an electronic medical record data warehouse is feasible and can potentially provide valuable tools for ASPs, especially at hospitals with no or limited local informatics expertise. Future efforts will evaluate the effectiveness of dashboards in these settings.
Funding: No
Disclosures: None
Figure 1.
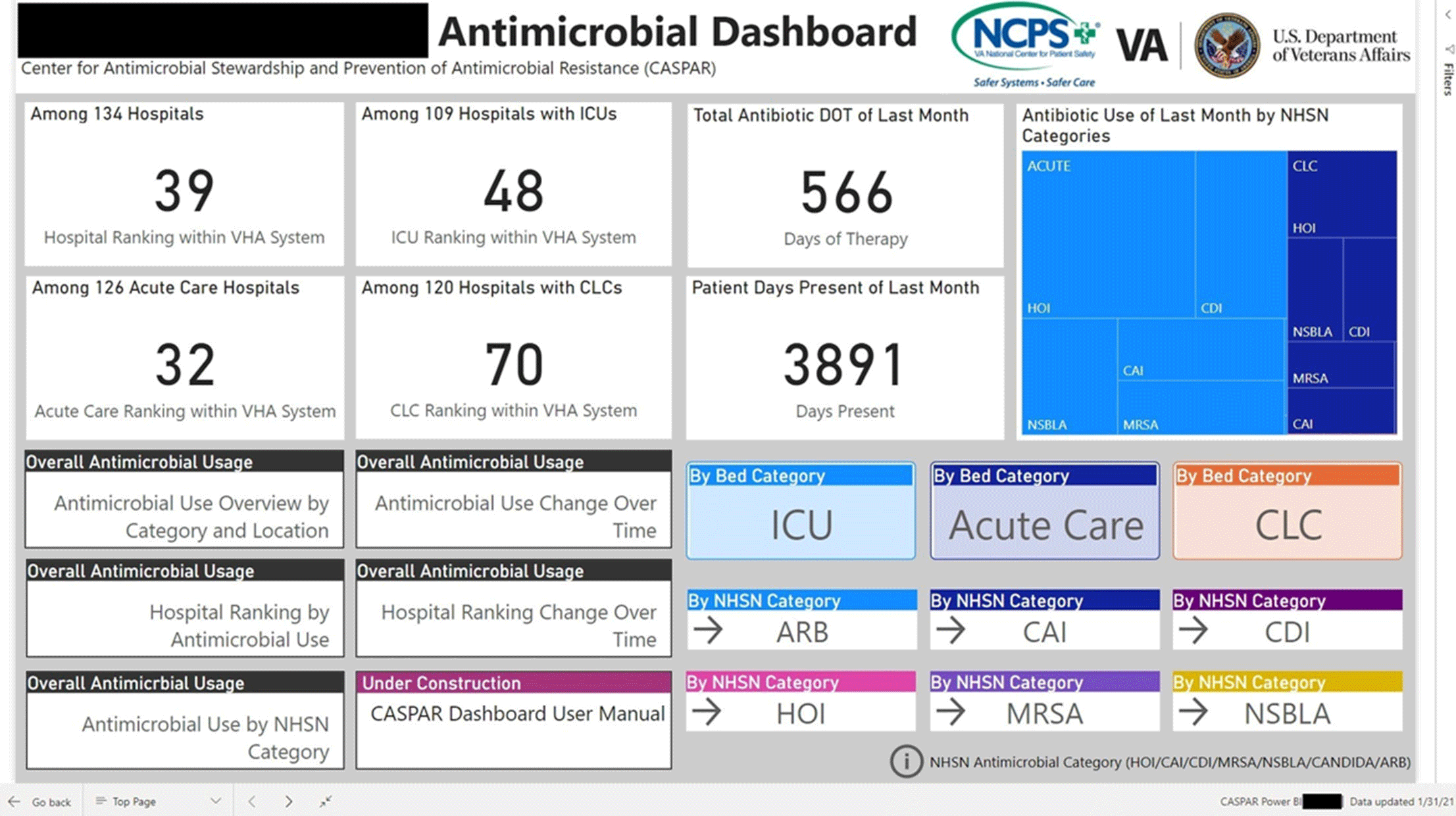
Figure 2.
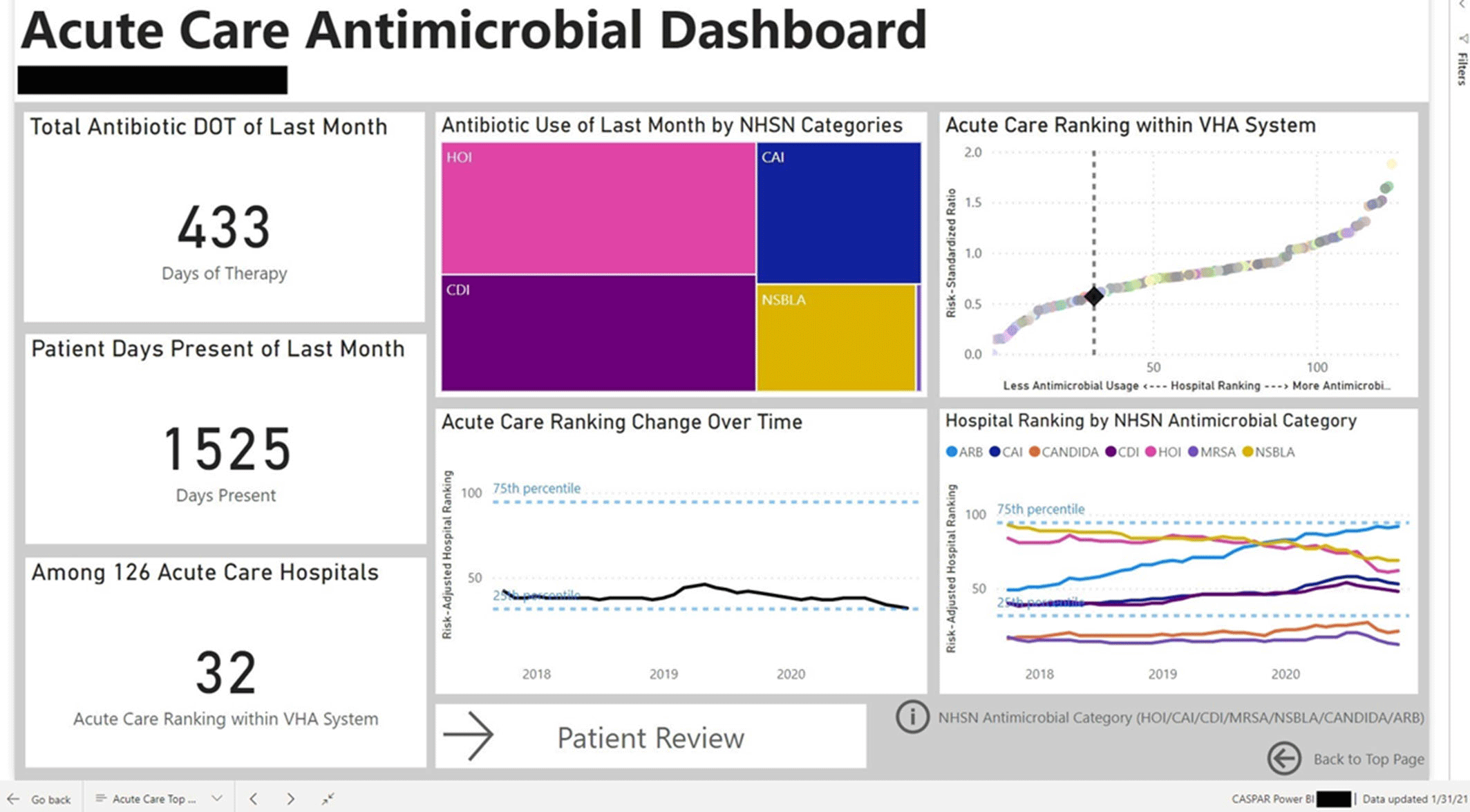
Figure 3.
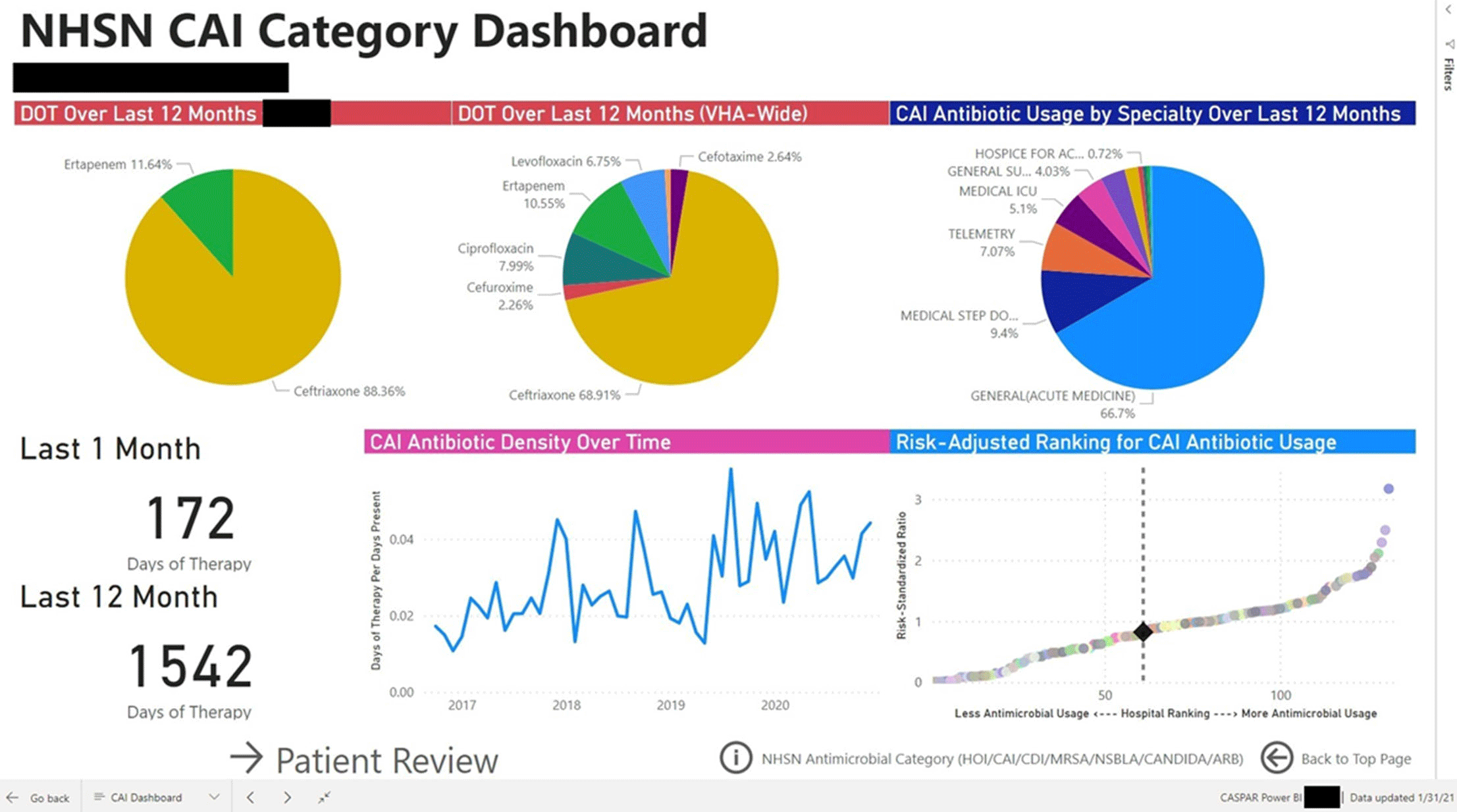
Original Article
Improving antibiotic prescribing for pediatric acute respiratory tract infections: A cluster randomized trial to evaluate individual versus clinic feedback
- Herbert W. Clegg, Stephen J. Ezzo, Kelly B. Flett, William E. Anderson
-
- Published online by Cambridge University Press:
- 03 November 2021, e43
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective:
To assess the effect of individual compared to clinic-level feedback on guideline-concordant care for 3 acute respiratory tract infections (ARTIs) among family medicine clinicians caring for pediatric patients.
Design:Cluster randomized controlled trial with a 22-month baseline, 26-month intervention period, and 12-month postintervention period.
Setting and participants:In total, 26 family medicine practices (39 clinics) caring for pediatric patients in Virginia, North Carolina, and South Carolina were selected based upon performance on guideline-concordance for 3 ARTIs, stratified by practice size. These were randomly allocated to a control group (17 clinics in 13 practices) or to an intervention group (22 clinics in 13 practices).
Interventions:All clinicians received an education session and baseline then monthly clinic-level rates for guideline-concordant antibiotic prescribing for ARTIs: upper respiratory tract infection (URI), acute bacterial sinusitis (ABS), and acute otitis media (AOM). For the intervention group only, individual clinician performance was provided.
Results:Both intervention and control groups demonstrated improvement from baseline, but the intervention group had significantly greater improvement compared with the control group: URI (odds ratio [OR], 1.62; 95% confidence interval [CI], 1.37–1.92; P < 0.01); ABS (OR, 1.45; 95% CI, 1.11–1.88; P < 0.01); and AOM (OR, 1.59; 95% CI, 1.24–2.03; P < 0.01). The intervention group also showed significantly greater reduction in broad-spectrum antibiotic prescribing percentage (BSAP%): odds ratio 0.80, 95% CI 0.74-0.87, P < 0.01. During the postintervention year, gains were maintained in the intervention group for each ARTI and for URI and AOM in the control group.
Conclusions:Monthly individual peer feedback is superior to clinic-level only feedback in family medicine clinics for 3 pediatric ARTIs and for BSAP% reduction.
Trial registration:ClinicalTrials.gov identifier: NCT04588376, Improving Antibiotic Prescribing for Pediatric Respiratory Infection by Family Physicians with Peer Comparison.
A modified Delphi approach to develop a trial protocol for antibiotic de-escalation in patients with suspected sepsis
- Michael E. Yarrington, Rebekah W. Moehring, Michael Z. David, Keith W. Hamilton, Michael Klompas, Chanu Rhee, Kevin Hsueh, Elizabeth Dodds Ashley, Ronda L. Sinkowitz-Cochran, Matthew Ryan, Deverick J. Anderson, for the DETOURS Expert Panel of the CDC Prevention Epicenters Program
-
- Published online by Cambridge University Press:
- 08 November 2021, e44
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background:
Early administration of antibiotics in sepsis is associated with improved patient outcomes, but safe and generalizable approaches to de-escalate or discontinue antibiotics after suspected sepsis events are unknown.
Methods:We used a modified Delphi approach to identify safety criteria for an opt-out protocol to guide de-escalation or discontinuation of antibiotic therapy after 72 hours in non-ICU patients with suspected sepsis. An expert panel with expertise in antimicrobial stewardship and hospital epidemiology rated 48 unique criteria across 3 electronic survey rating tools. Criteria were rated primarily based on their impact on patient safety and feasibility for extraction from electronic health record review. The 48 unique criteria were rated by anonymous electronic survey tools, and the results were fed back to the expert panel participants. Consensus was achieved to either retain or remove each criterion.
Results:After 3 rounds, 22 unique criteria remained as part of the opt-out safety checklist. These criteria included high-risk comorbidities, signs of severe illness, lack of cultures during sepsis work-up or antibiotic use prior to blood cultures, or ongoing signs and symptoms of infection.
Conclusions:The modified Delphi approach is a useful method to achieve expert-level consensus in the absence of evidence suifficient to provide validated guidance. The Delphi approach allowed for flexibility in development of an opt-out trial protocol for sepsis antibiotic de-escalation. The utility of this protocol should be evaluated in a randomized controlled trial.
Presentation Type: Oral Presentation
MRSA/VRE
Discontinuation of Contact Precautions in Patients with Nosocomial MRSA and VRE Infections During the COVID-19 Pandemic
- Marisa Hudson, Mayar Al Mohajer
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s24
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Gaps exist in the evidence supporting the benefits of contact precautions for the prevention of methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci (VRE). The Centers for Disease Control and Prevention allow suspending contact precautions for MRSA and VRE in cases of gown shortages, as we have seen during the COVID-19 pandemic. We evaluated the impact of discontinuing isolation precautions in hospitalized patients with MRSA and VRE infection, due to gown shortage, on the rate of hospital-acquired (HA) MRSA and VRE infections. Methods: A retrospective chart review was performed on adult patients (n = 2,200) with established MRSA or VRE infection at 5 hospitals in CommonSpirit Health, Texas Division, from March 2019 to October 2020. Data including demographics, infection site, documented symptoms, and antibiotic use were stratified based on patient location (floor vs ICU). Rates of hospital-acquired MRSA and VRE infection before and after the discontinuation of isolation (implemented in March 2020) were compared. Incidence density rate was used to assess differences in the rate of MRSA and VRE infections between pre- and postintervention groups. Results: The rate of hospital-acquired (HA) MRSA infection per 10,000 patient days before the intervention (March 19–February 20) was 12.19, compared to 10.64 after the intervention (March 20–July 20) (P = .038). The rates of HA MRSA bacteremia were 1.13 and 0.93 for the pre- and postintervention groups, respectively (P = .074). The rates of HA VRE per 10,000 patient days were 3.53 and 4.44 for the pre- and postintervention groups, respectively (P = .274). The hand hygiene rates were 0.93 before the intervention and 0.97 after the intervention (P = .028). Conclusions: Discontinuing isolation from MRSA and VRE in the hospital setting did not lead to a statistically significant increase in hospital-acquired MRSA or VRE infections. In fact, rates of hospital-acquired MRSA decreased, likely secondary to improvements in hand hygiene during this period. These results support the implementation of policies for discontinuing contact isolation for hospitalized patients with documented MRSA or VRE infection, particularly during shortages of gowns.
Funding: No
Disclosures: None
Figure 1.
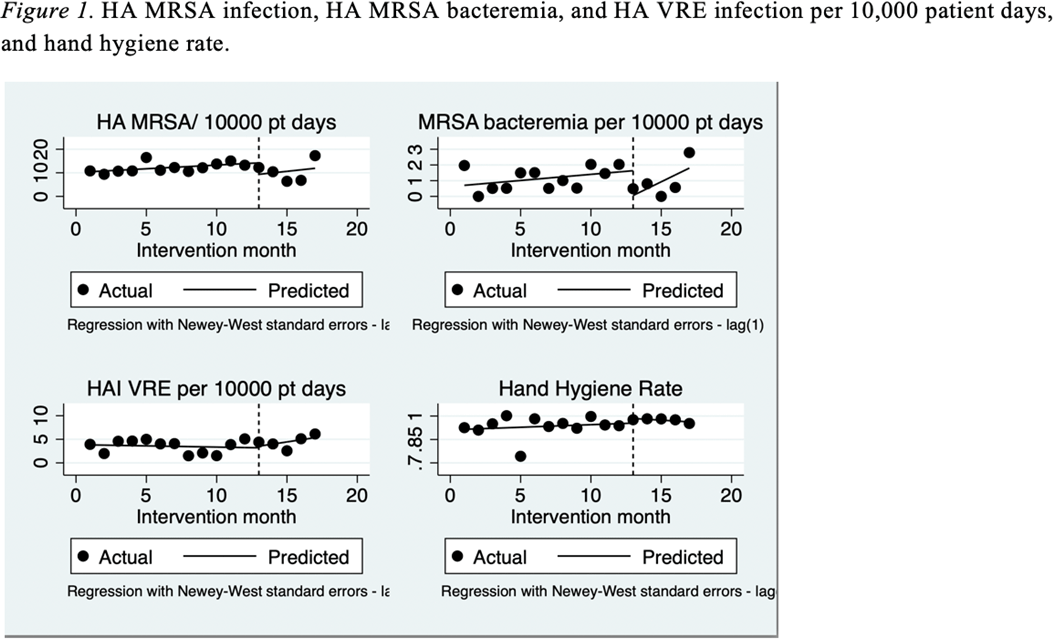
Table 1. 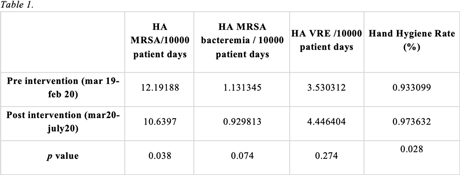
Oral Presentation
Outbreaks
Mycobacterium abscessus Surgical Site Infections Due to Modular Cooler-Heater Units in Cardiac Surgery
- Ahmed Abdul Azim, Sharon Wright, Bryan Connors, Patrick Gordon, Preeti Mehrotra
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s25
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: In the spring of 2020, we identified 3 patients with organ-space surgical site infections (SSIs) secondary to Mycobacterium abscessus (Table 1). All 3 patients underwent cardiac surgery in the same operating room (OR) during which the CardioQuip Modular Cooler-Heaters (MCHs) were used. We describe key aspects of our cluster investigation, which ultimately led to release of a national safety alert by the Food and Drug Administration (FDA). Methods: For environmental cultures, we obtained samples from 9 MCHs in circulation; 2 scrub sink samples; ice from the OR ice machine; water samples from sinks in the cardiovascular critical care unit, and water samples from floors above the cardiac ORs. All samples were sent for molecular genotyping. For pathway studies, an external environmental engineering team was consulted who conducted smoke pathway tests in 3 different ORs. The team also conducted a particle generator experiment, simulating the set-up of a cardiac bypass surgery case. To assess disinfection practices, we reviewed the manufacturer instructions for use (IFU) protocol of the MCHs and audited our own policies and procedures to ensure compliance. Results: For environmental cultures, molecular typing from 5 of 9 MCHs and all 3 patient SSI isolates returned positive for the identical hybrid species M. abscessus bolleti. All other samples with mycobacterial growth returned with different species. For pathway studies, the particle-generator experiment demonstrated particle movement from the MCH to the sterile field with facilities-guidelines–compliant OR ventilation and despite MCH manufacturing design. For disinfection practices, despite compliance with the stated IFU, and in consultation with experts, we implemented disinfection of associated Quick-connect devices (otherwise not stated in the IFU), and we also initiated a precleaning step prior to disinfection. Conclusions: Our investigation concluded that 3 patients developed SSIs with Mycobacterium abscessus that was aerosolized from the CardioQuip MCH. This finding led to the national FDA safety report alerting providers to risks associated with the device and the need for continued vigilance around disinfection. In addition, we implemented other control measures including placement of MCHs outside all ORs; creation of a separate MCH fleet for non-OR use; and use of modified disinfection protocols. To date, no additional cases have been identified.
Funding: No
Disclosures: None
Table 1. 
Original Article
Bacterial coinfections with coronavirus disease 2019 (COVID-19)
- Glen Huang, Daisuke Furukawa, Bryant D. Yang, Brian J. Kim, Arthur C. Jeng
-
- Published online by Cambridge University Press:
- 10 November 2021, e45
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background:
The pandemic caused by severe acute respiratory coronavirus virus 2 (SARS-CoV-2) has dramatically increased cheshospitalizations, and it is often difficult to determine whether there is a bacterial or fungal coinfection at time of presentation. In this study, we sought to determine the rates of coinfection and utilization of antibiotics in SARS-CoV-2 disease.
Methods:Retrospective chart review of patients hospitalized with COVID-19 pneumonia from April 13, 2020, to July 14, 2020.
Results:In total, 277 patients were hospitalized for COVID-19 pneumonia during this period. Patients that received antibiotics within 48 hours of presentation were more likely to be febrile (59.3% vs 41.2%; P = .01) and to have leukocytosis (23.9% vs 5.9%; P < .01) and were less likely to have a procalcitonin level <0.25 ng/mL (58.8% vs 74.5%; P = .04). In total, 45 patients had positive blood cultures collected during hospitalization, 16 of which were clinically significant. Of the clinically significant blood cultures, 5 were collected <48 hours of admission. Moreover, 18 sputum cultures were clinically significant, 2 of which were collected within 48 hours of admission.
Conclusion:Bacterial and fungal coinfections in COVID-19 appear to be rare on presentation; thus, this factor may be a good target for enhanced antibiotic stewardship.
Oral Presentation
Outbreaks
Outbreak of Pseudomonas aeruginosa Bacteremia Infections among Stem-Cell Transplant Patients Related to Change in Prophylaxis
- Kerrie VerLee, Chau Nguyen, Russell Lampen, Jim Codman, Tunisia Peters
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s25-s26
-
- Article
-
- You have access Access
- Open access
- Export citation
-
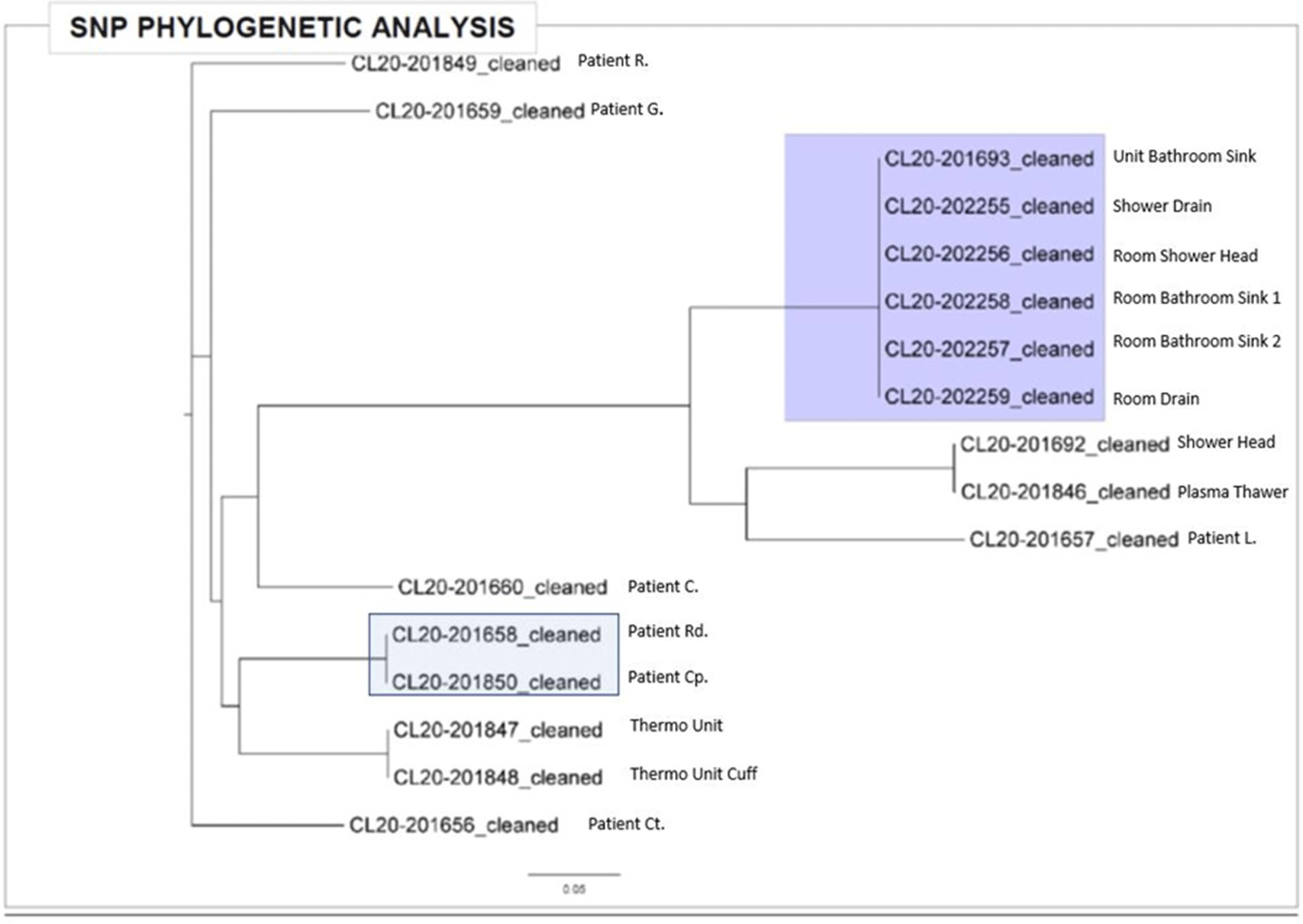
Background:Pseudomonas aeruginosa outbreaks can originate from various sources and can cause severe complications in posttransplant patients. Antibiotic prophylaxis can decrease posttransplant infections; however, consideration must be given to P. aeruginosa coverage as we outline an outbreak among the stem-cell transplant (SCT) population. Methods: A multidisciplinary outbreak investigation was conducted to evaluate sources of contamination and changes in clinical processes. Positive blood cultures from SCT patients and environmental isolates were analyzed using whole-genome sequencing (WGS). Incidence density rates for P. aeruginosa blood cultures from January 2019 through October 2020 were calculated per 10,000 patient days and stratified by unit, specimen, and transplant type. Statistical analysis was calculated with significance at p < 0.05. Results: A cluster of 8 SCT patients was identified between May and September 2020. Moreover, 10 environmental samples were positive for P. aeruginosa including drains, water sources prior to the point-of-use (POU) filter and blood-bank thaw machines. Phylogenetic analysis revealed 1 cluster of 2 patients who shared the same room, 5 patients with unique P. aeruginosa isolates, and 2 separate clusters of environmental isolates with relatedness only to each other. Review of clinical processes showed a change from fluoroquinolone prophylaxis to cephalosporin in the spring of 2020. Also, 5 P. aeruginosa bacteremia infections occurred prior to June (11.78 cases per 10,000 patient days). During the period of cephalosporin use, 8 infections were identified (58.27 cases per 10,000 patient days) (P = .006). Following the restart of fluoroquinolone, zero infections have occurred to date, as of January 28, 2021. Conclusions: Discontinuation of fluoroquinolone prophylaxis was associated with P. aeruginosa bacteremia infections in SCT patients. Use of fluoroquinolone prophylaxis in SCT patients is protective from P. aeruginosa bacteremia infections. There have been no further infections in the following 3 months after the change back to the use of fluoroquinolone. Additionally, WGS showed that most patient isolates did not have a common source, suggesting that P. aeruginosa gastrointestinal colonization may play a role in seeding these bacteremia infections.
Funding: No
Disclosures: None
Figure 1.
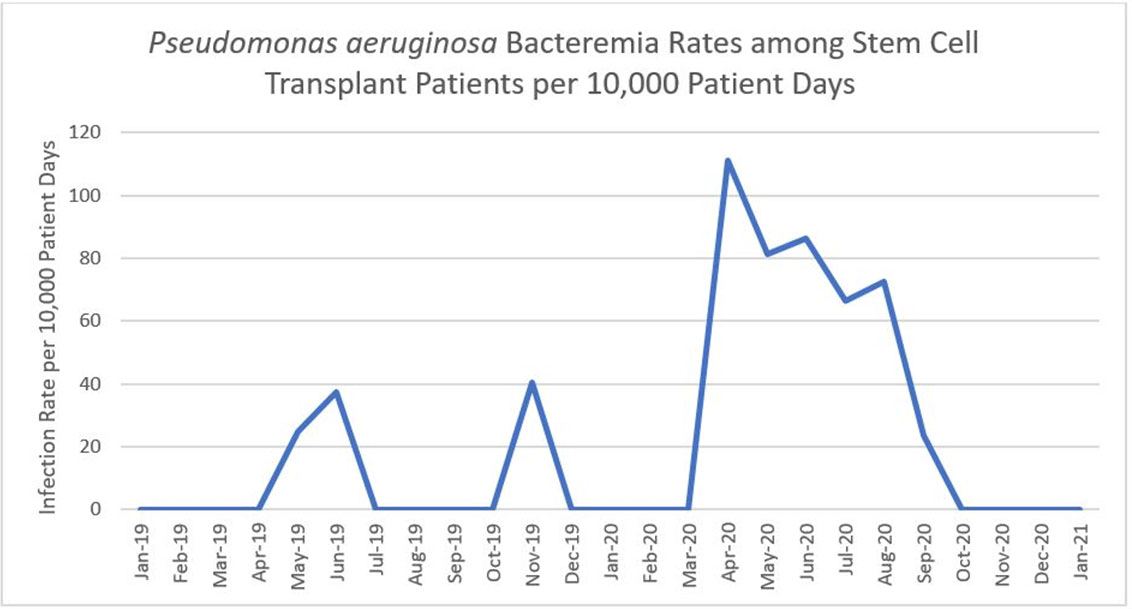
Figure 2.
Review
Interventions to optimize antimicrobial stewardship
- Nick J. Tinker, Rachel A. Foster, Brandon J. Webb, Souha Haydoura, Whitney R. Buckel, Edward A. Stenehjem
-
- Published online by Cambridge University Press:
- 10 November 2021, e46
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Developing and improving an antimicrobial stewardship program successfully requires evaluation of numerous factors. As technology progresses and our understanding of antimicrobial resistance grows, careful consideration should be taken to ensure that a program meets the needs of the institution and is achievable given the available resources. In this review, we explore fundamental initiatives and strategies for both new and established antimicrobial stewardship programs, including the specific areas to target and key elements required for sustainable implementation.
Oral Presentation
Outbreaks
Verona Integron-Encoded Metallo-Beta-Lactamase (VIM)–Producing Pseudomonas aeruginosa Outbreak Associated with Acute Care
- Allison Chan, Alicia Shugart, Albert Burks, Christina Moore, Paige Gable, Heather Moulton-Meissner, Gillian McAllister, Alison Halpin, Maroya Walters, Amelia Keaton, Kelley Tobey, Katie Thure, Sarah Schmedes, Paige Gable, Henrietta Hardin, Adrian Lawsin
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s26
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Contaminated healthcare facility plumbing is increasingly recognized as a source of carbapenemase-producing organisms (CPOs). In August 2019, the Tennessee State Public Health Laboratory identified Tennessee’s twelfth VIM-producing carbapenem-resistant Pseudomonas aeruginosa (VIM-CRPA), from a patient in a long-term acute-care hospital. To determine a potential reservoir, the Tennessee Department of Health (TDH) reviewed healthcare exposures for all cases. Four cases (33%), including the most recent case and earliest from March 2018, had a history of admission to intensive care unit (ICU) room X at acute-care hospital A (ACH A), but the specimens were collected at other facilities. The Public Health Laboratory collaborated with ACH A to assess exposures, perform environmental sampling, and implement control measures. Methods: TDH conducted in-person infection prevention assessments with ACH A, including a review of the water management program. Initial recommendations included placing all patients admitted to room X on contact precautions, screening for CPO on room discharge, daily sink basin and counter cleaning, and other sink hygiene measures. TDH collected environmental and water samples from 5 ICU sinks (ie, the handwashing and bathroom sinks in room X and neighboring room Y [control] and 1 hallway sink) and assessed the presence of VIM-CRPA. Moreover, 5 patients and 4 environmental VIM-CRPA underwent whole-genome sequencing (WGS). Results: From February to June 2020, of 21 patients admitted to room X, 9 (43%) underwent discharge screening and 4 (44%) were colonized with VIM-CRPA. Average room X length of stay was longer for colonized patients (11.3 vs 4.8 days). Drain swabs from room X’s bathroom and handwashing sinks grew VIM-CRPA; VIM-CRPA was not detected in tap water or other swab samples. VIM-CRPA from the environment and patients were sequence type 253 and varied by 0–13 single-nucleotide variants. ACH A replaced room X’s sinks and external plumbing in July. Discharge screening and contact precautions for all patients were discontinued in November, 5 months following the last case and 12 consecutive negative patient discharge screens. Improved sink hygiene and mechanism testing for CRPA from clinical cultures continued, with no new cases identified. Conclusions: An ICU room with a persistently contaminated sink drain was a persistent reservoir of VIM-CRPA. The room X attack rate was high, with VIM-CRPA acquisition occurring in >40% of patients screened. The use of contaminated plumbing fixtures in ACH have the potential to facilitate transmission to patients but may be challenging to identify and remediate. All healthcare facilities should follow sink hygiene best practices.
Funding: No
Disclosures: None
Original Article
Effects of antibiotic suppression on three healthcare systems’ National Healthcare Safety Network Antibiotic Resistance Option data
- Christopher D. Evans, Matthew D. Estes, Youssoufou Ouedraogo, Daniel Muleta, Marion A. Kainer, Pamela P. Talley
-
- Published online by Cambridge University Press:
- 10 November 2021, e47
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective:
The National Healthcare Safety Network (NHSN) Antibiotic Resistance (AR) Option is a valuable tool that can be used by acute-care hospitals to track and report antibiotic resistance rate data. Selective and cascading reporting results in suppressed antibiotic susceptibility results and has the potential to adversely affect what data are submitted into the NHSN AR Option. We describe the effects of antibiotic suppression on NHSN AR Option data.
Methods:NHSN AR Option data were collected from 14 hospitals reporting into an existing NHSN user group from January 1, 2017, to December 31, 2018, and linked to commercial automated antimicrobial susceptibility testing instruments (cASTI) that were submitted as part of unrelated Tennessee Emerging Infections Program surveillance projects. A susceptibility result was defined as suppressed if the result was not found in the NHSN AR Option data but was reported in the cASTI data. Susceptibility results found in both data sets were described as released. Proportions of suppressed and released results were compared using the Pearson χ2 and Fisher exact tests.
Results:In total, 852 matched isolates with 3,859 unique susceptibilities were available for analysis. At least 1 suppressed antibiotic susceptibility result was available for 726 (85.2%) of the isolates. Of the 3,859 susceptibility results, 1,936 (50.2%) suppressed antibiotic susceptibility results were not reported into the NHSN AR option when compared to the cASTI data.
Conclusion:The effect of antibiotic suppression described in this article has significant implications for the ability of the NHSN AR Option to accurately reflect antibiotic resistance rates.
Oral Presentation
Outbreaks
Successful Control of Human Parainfluenza Type 3 Outbreak in a Level IV Neonatal Intensive Care Unit
- Bhagyashri Navalkele, Sheila Fletcher, Sanjosa Martin, Regina Galloway, April Palmer
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s26-s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
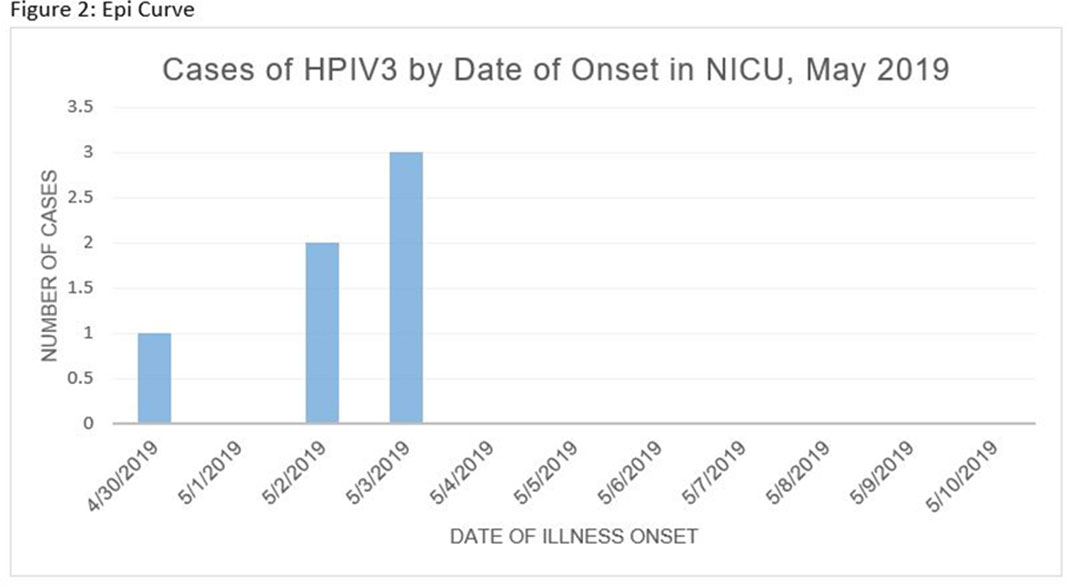
Human parainfluenza (HPIV) is a common cause for upper respiratory tract illnesses (URTI) and lower respiratory tract illnesses (LRTI) in infants and young children. Here, we describe successful control of an HPIV type 3 (HPIV3) outbreak in a neonatal intensive care unit (NICU). NICU babies with new-onset clinical signs or symptoms of RTI and positive HPIV-3 nasopharyngeal specimen by respiratory pathogen panel (RPP) test on hospital day 14 or later were diagnosed with hospital-onset (HO) HPIV-3 infection. After 3 NICU babies were diagnosed with HO HPIV-3, an outbreak investigation was initiated on May 3, 2019, and continued for 2 incubation periods since the last identified case. Enhanced infection prevention measures were immediately implemented. All positive cases were placed in a cohort in a single pod of the NICU and were placed on contact precautions with droplet isolation precautions. Dedicated staffing and equipment were assigned. Environmental cleaning and disinfection with hospital-approved disinfectant wipes was performed daily. Visitors were restricted in the NICU. All employees entering the NICU underwent daily symptom screening for respiratory tract illness. All NICU babies were screened daily for respiratory tract illness with prompt isolation and RPP testing on positive screen. To determine the source of the HPIV3 outbreak, all HPIV3-positive specimens from the NICU and available temporally associated community-onset (CO) controls collected from non-NICU units were sent to the Centers for Disease Control and Prevention (CDC) for whole-genome sequencing (WGS) analysis. The first and last cases of HPIV-3 were diagnosed on May 1 and May 5, 2019, respectively. In total, 7 HO HPIV3 cases were reported: 1 in newborn nursery (NBN) and 6 in NICU. The case from the NBN was determined to be unrelated to the outbreak and the source was linked to a sick visitor. Of the 6 NICU babies, 5 had an LRTI and 1 had a URTI. Average time from admission to diagnosis was 71 days (range, 24–112). None had severe illnesses requiring intubation, and all had full recovery. No CO HPIV3 cases were reported from the NICU during the investigation. A maximum likelihood phylogenetic tree of HPIV3 WGS (Figure 1) showed that sequences from the 6 HO cases clustered together separately from the 3 CO controls, suggesting a single source of transmission, and 3 CO cases were not related to the HO cases or source of the outbreak. Early diagnosis and isolation of respiratory tract viral infections is important to prevent an outbreak. Successful control of outbreak in NICU requires prompt implementation of infection prevention measures with focus on symptom screening, cohorting, and disinfection practices.
Funding: No
Disclosures: None
Figure 1.
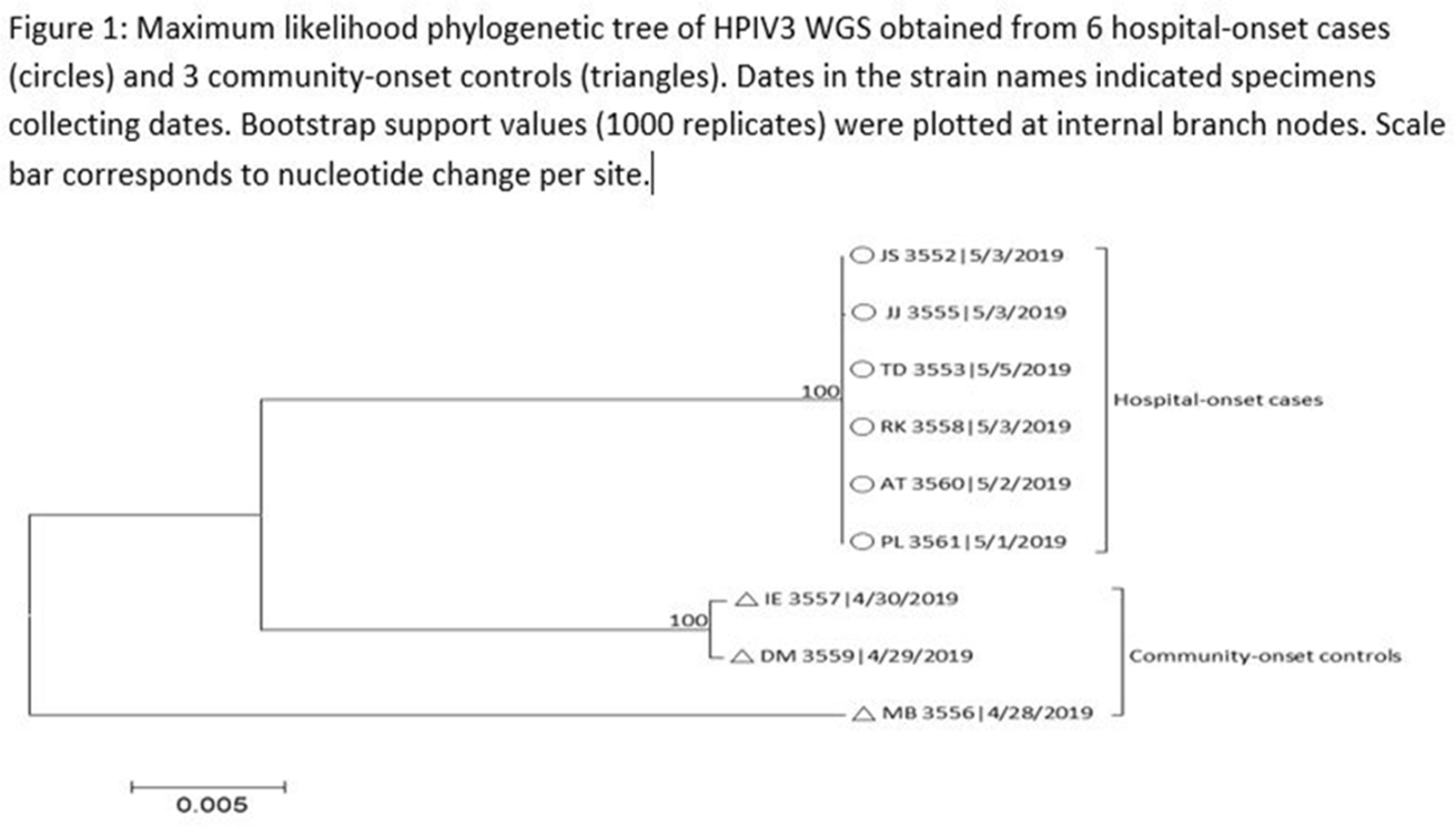
Figure 2.
Original Article
Survey of physician and pharmacist steward perceptions of their antibiotic stewardship programs
- Shana A. B. Burrowes, Mari-Lynn Drainoni, Maria Tjilos, Jorie M. Butler, Laura J. Damschroder, Matthew Bidwell Goetz, Karl Madaras-Kelly, Caitlin M. Reardon, Matthew H. Samore, Jincheng Shen, Edward Stenehjem, Yue Zhang, Tamar F. Barlam
-
- Published online by Cambridge University Press:
- 12 November 2021, e48
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective:
To examine how individual steward characteristics (eg, steward role, sex, and specialized training) are associated with their views of antimicrobial stewardship program (ASP) implementation at their institution.
Design:Descriptive survey from a mixed-methods study.
Setting:Two large national healthcare systems; the Veterans’ Health Administration (VA) (n = 134 hospitals) and Intermountain Healthcare (IHC; n = 20 hospitals).
Participants:We sent the survey to 329 antibiotic stewards serving in 154 hospitals; 152 were physicians and 177 were pharmacists. In total, 118 pharmacists and 64 physicians from 126 hospitals responded.
Methods:The survey was grounded in constructs of the Consolidated Framework for Implementation Research, and it assessed stewards’ views on the development and implementation of antibiotic stewardship programs (ASPs) at their institutions We then examined differences in stewards’ views by demographic factors.
Results:Regardless of individual factors, stewards agreed that the ASP added value to their institution and was advantageous to patient care. Stewards also reported high levels of collegiality and self-efficacy. Stewards who had specialized training or those volunteered for the role were less likely to think that the ASP was implemented due to a mandate. Similarly volunteers and those with specialized training felt that they had authority in the antibiotic decisions made in their facility.
Conclusions:Given the importance of ASPs, it may be beneficial for healthcare institutions to recruit and train individuals with a true interest in stewardship.
Concise Communication
The coronavirus disease 2019 (COVID-19) delta wave: Refocusing effort and remaining resilient in the face of evolving infection prevention and antimicrobial stewardship challenges
- Michael P. Stevens, Jacob W. Pierce, Rachel Pryor, Michelle E. Doll, Gonzalo M. Bearman
-
- Published online by Cambridge University Press:
- 18 November 2021, e49
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Challenges for infection prevention and antimicrobial stewardship programs have arisen with the fourth wave of the coronavirus disease 2019 (COVID-19) pandemic, fueled by the delta variant. These challenges include breakthrough infections in vaccinated individuals, decisions to re-escalate infection prevention measures, critical medication shortages, and provider burnout. Various strategies are needed to meet these challenges.
Oral Presentation
Pediatrics
Results of a Multicenter Diagnostic Stewardship Collaborative to Optimize Blood Culture Use in Critically Ill Children
- Danielle Koontz, Charlotte Woods-Hill, Annie Voskertchian, Anping Xie, Marlene Miller, James Fackler, Elizabeth Colantuoni, Aaron Milstone, Ximin Li
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Group Name: Bright STAR Authorship Group
Background: Blood cultures are fundamental in the diagnosis and treatment of sepsis. Culture practices vary widely, and overuse can lead to false-positive results and unnecessary antibiotics. Our objective was to describe the implementation of a multisite quality improvement collaborative to reduce unnecessary blood cultures in pediatric intensive care unit (PICU) patients, and its 18-month impact on blood culture rates and safety metrics. Methods: In 2018, 14 PICUs joined the Blood Culture Improvement Guidelines and Diagnostic Stewardship for Antibiotic Reduction in Critically Ill Children (Bright STAR) Collaborative, designed to understand and improve blood culture practices in critically ill children. Guided by a centralized multidisciplinary study team, sites first reviewed existing evidence for safe reduction of unnecessary blood cultures and assessed local practices and barriers to change. Subsequently, local champions developed and implemented clinical decision-support tools informed by local patient needs to guide new blood-culture practices. The coordinating study team facilitated regular evaluations and discussions of project progress through monthly phone calls, site visits if requested by sites or the study team, and collaborative-wide teleconferences. The study team collected monthly blood culture rates and monitored for possible delays in obtaining blood cultures using a standardized review process as a safety balancing metric. We compared 24 months of baseline data to 18 months of postimplementation using a Poisson regression model accounting for the site-specific patient days and correlation of culture use within a site over time. Results: Across the 14 sites, before implementation, 41,768 blood cultures were collected over 259,701 PICU patient days. The mean preimplementation site-specific blood culture rate was 15.7 cultures per 100 patient days (rate range, 9.6–48.2 cultures per 100 patient days). After implementation, 22,397 blood cultures were collected over 208,171 PICU patient days. The mean postimplementation rate was 10.4 cultures per 100 patient days (rate range, 4.7–28.3 cultures per 100 patient days), which was 33.6% lower than the preimplementation (relative rate 0.66; 95% CI, 0.65–0.68 p <0.01). In 18 months post-implementation, sites reviewed 793 positive blood cultures, and identified only one suspected delay in culture collection possibly attributable to the site’s blood culture reduction program. Conclusions: Multidisciplinary quality improvement teams safely facilitated a 33.6% average reduction in blood culture use in critically ill children at 14 hospitals. Future collaborative work will determine the impact of blood culture diagnostic stewardship on antibiotic use and other important patient safety outcomes.
Funding: No
Disclosures: None
Figure 1.
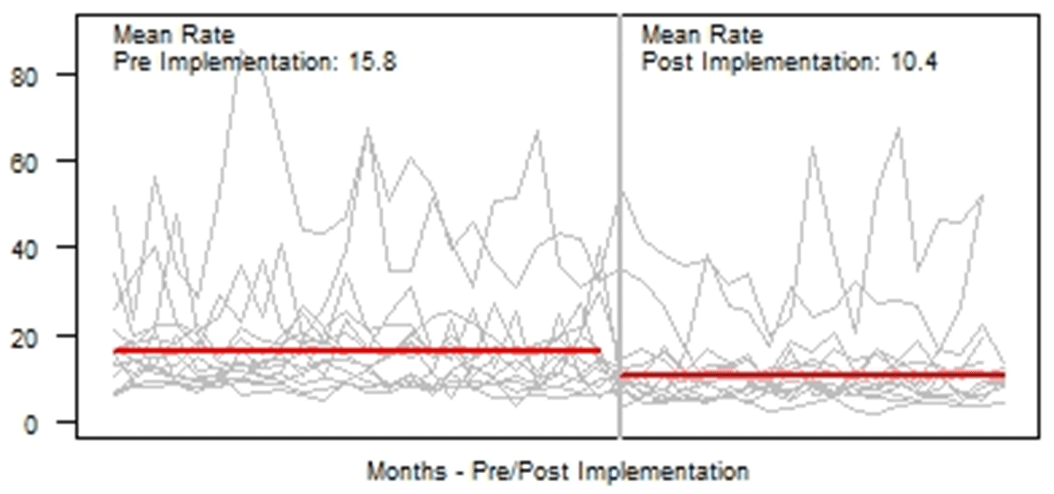
Association of Antibiotic Duration and Outcomes among NICU Infants with Invasive Staphylococcus aureus Infections
- Areej Bukhari, Ibukunoluwa Akinboyo, Kanecia Zimmerman, Danny Benjamin, Veeral Tolia, Rachel Greenberg
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s27-s28
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background:Staphylococcus aureus is the second-leading cause of late-onset sepsis among infants in US neonatal intensive care units (NICUs). Management of S. aureus bacteremia and meningitis in infants varies widely due to the lack of standardized guidelines. We examined the association between initial antibiotic duration and recurrent S. aureus infection or death among NICU infants with S. aureus bacteremia and/or meningitis. Methods: We conducted a retrospective cohort study of infants in Pediatrix Medical Group NICUs from 1997 to 2018 with first episode of S. aureus bacteremia and/or meningitis, identified as having at least 1 blood or cerebrospinal fluid (CSF) culture growing only S. aureus at any point during their NICU stay. Excluded infants were those not started on antistaphylococcal therapy within 2 days of positive culture, those with had endocarditis or osteomyelitis, or those who died or were discharged during or up to 1 day after antibiotic cessation. Antibiotic cessation was defined as last day of antibiotic given if followed by at least 3 days without antibiotics. Multivariable logistic regression was used to analyze the association between antibiotic duration categorized as <14 or ≥14 days) and recurrent SA infection (within 12 weeks of antibiotic cessation, prior to hospital discharge), or death (within 7 days of antibiotic cessation and at discharge). Results: Of 4,086 infants included, 3,991 (98%) had S. aureus bacteremia only and 95 (2%) had meningitis ± bacteremia. Of those with bacteremia only, 2,017 (50.5%), and 17 (18%) of those with meningitis received <14 days antibiotics (Figure 1). Longer antibiotic duration was associated with lower gestational age, methicillin-resistance, severe illness and bacteremia duration of ≥4 days (Table 1). There was a significant association between <14 days antibiotics and recurrent infection (p = 0.04) and 7-day mortality (p = 0.02) in the meningitis cohort. Infants with SA bacteremia who received ≥14 days antibiotics had reduced odds of recurrent SA infection (OR 0.24, 95% CI 0.18-0.32) and death (OR 0.33, 95% CI 0.25-0.44), adjusting for post-natal age, gestational age, sex, methicillin-resistance, severe illness and duration of bacteremia (Table 2). Conclusions: In the largest study thus far examining antibiotic duration among hospitalized infants with S. aureus bacteremia and/or meningitis, ≥14 days antibiotics was associated with decreased odds of recurrent infection or death. Further studies are needed to define the optimal treatment duration and identify clinical factors distinguishing infants able to safely receive a shorter antibiotic duration.
Funding: No
Disclosures: None
Figure 1.
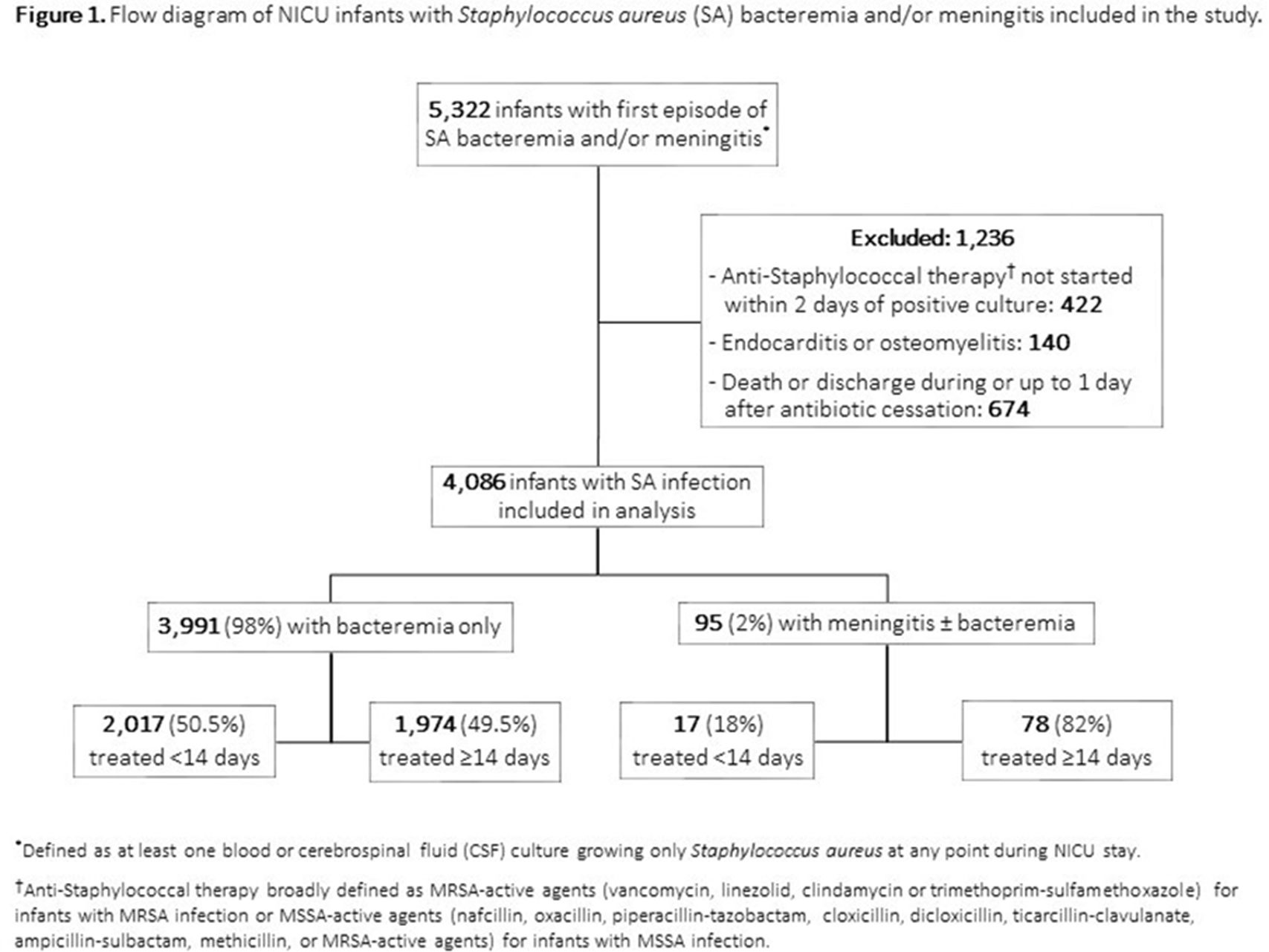
Table 1. 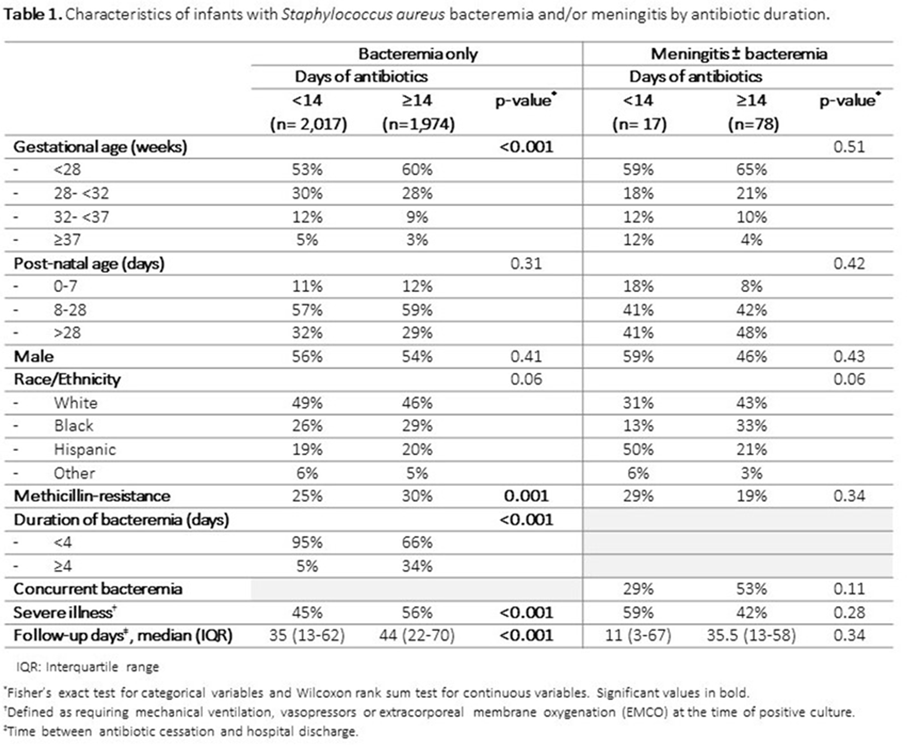
Table 2. 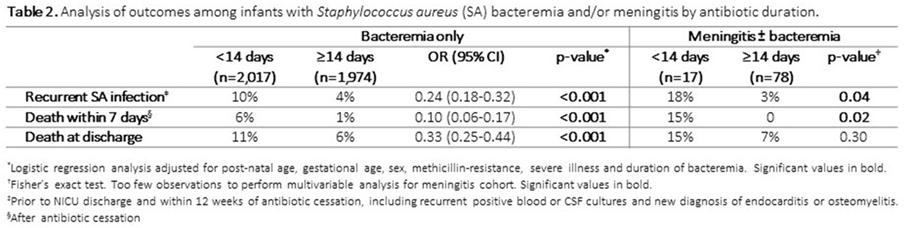
Review
Social media: A new tool for outbreak surveillance
- Averi E. Wilson, Christoph U. Lehmann, Sameh N. Saleh, John Hanna, Richard J. Medford
-
- Published online by Cambridge University Press:
- 17 November 2021, e50
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Social media platforms allow users to share news, ideas, thoughts, and opinions on a global scale. Data processing methods allow researchers to automate the collection and interpretation of social media posts for efficient and valuable disease surveillance. Data derived from social media and internet search trends have been used successfully for monitoring and forecasting disease outbreaks such as Zika, Dengue, MERS, and Ebola viruses. More recently, data derived from social media have been used to monitor and model disease incidence during the coronavirus disease 2019 (COVID-19) pandemic. We discuss the use of social media for disease surveillance.
Oral Presentation
Surveillance/Public Health
Admission and Discharge Sampling Underestimates Multidrug-Resistant Organism (MDRO) Acquisition in an Intensive Care Unit
- Sarah Sansom, Michael Lin, Christine Fukuda, Teppei Shimasaki, Thelma Dangana, Nicholas Moore, Rachel Yelin, Yoona Rhee, Lina Tabith, Jianrong Sheng, Enrique Cornejo Cisneros, John Murray, Kyle Chang, Karen Lolans, Michelle Ariston, William Rotunno, Hazel Ramos, Haiying Li, Khaled Aboushaala, Naomi Iwai, Christine Bassis, Vincent Young, Mary Hayden
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s28
-
- Article
-
- You have access Access
- Open access
- Export citation
-
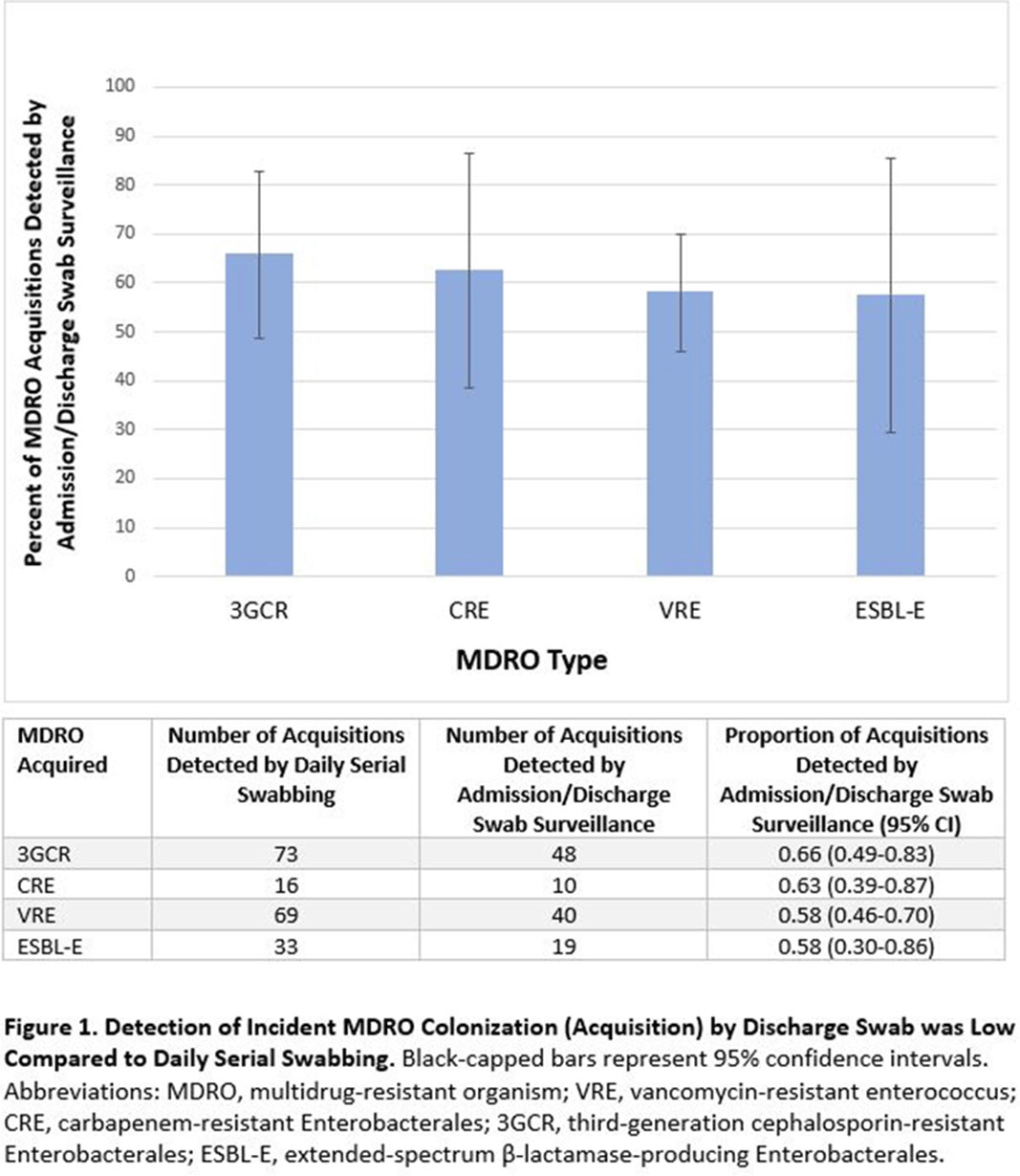
Background: Identification of hospitalized patients with enteric multidrug-resistant organism (MDRO) carriage, combined with implementation of targeted infection control interventions, may help reduce MDRO transmission. However, the optimal surveillance approach has not been defined. We sought to determine whether daily serial rectal surveillance for MDROs detects more incident cases (acquisition) of MDRO colonization in medical intensive care unit (MICU) patients than admission and discharge surveillance alone. Methods: Prospective longitudinal observational single-center study from January 11, 2017, to January 11, 2018. Inclusion criteria were ≥3 consecutive MICU days and ≥2 rectal or stool swabs per MICU admission. Daily rectal or stool swabs were collected from patients and cultured for MDROs, including vancomycin-resistant Enterococcus (VRE), carbapenem-resistant Enterobacterales (CRE), third-generation cephalosporin-resistant Enterobacterales (3GCR), and extended-spectrum β-lactamase–producing Enterobacterales (ESBL-E) (as a subset of 3GCR). MDRO detection at any time during the MICU stay was used to calculate prevalent colonization. Incident colonization (acquisition) was defined as new detection of an MDRO after at least 1 prior negative swab. We then determined the proportion of prevalent and incident cases detected by daily testing that were also detected when only first swabs (admission) and last swabs (discharge) were tested. Data were analyzed using SAS version 9.4 software. Results: In total, 939 MICU stays of 842 patients were analyzed. Patient characteristics were median age 64 years (interquartile range [IQR], 51–74), median MICU length of stay 5 days (IQR, 3–8), median number of samples per admission 3 (IQR, 2–5), and median Charlson index 4 (IQR, 2–7). Prevalent colonization with any MDRO was detected by daily swabbing in 401 stays (42.7%). Compared to daily serial swabbing, an admission- and discharge-only approach detected ≥86% of MDRO cases (ie, overall prevalent MDRO colonization). Detection of incident MDRO colonization by an admission- or discharge-only approach would have detected fewer cases than daily swabbing (Figure 1); ≥34% of total MDRO acquisitions would have been missed. Conclusions: Testing patients upon admission and discharge to an MICU may fail to detect MDRO acquisition in more than one-third of patients, thereby reducing the effectiveness of MDRO control programs that are targeted against known MDRO carriers. The poor performance of a single discharge swab may be due to intermittent or low-level MDRO shedding, inadequate sampling, or transient MDRO colonization. Additional research is needed to determine the optimal surveillance approach of enteric MDRO carriage.
Funding: No
Disclosures: None
Figure 1.
Original Article
Current practices and evaluation of barriers and facilitators to surgical site infection prevention measures in Jimma, Ethiopia
- Leigh R. Berman, Andrew Lang, Beshea Gelana, Samuel Starke, Dawd Siraj, Daniel Yilma, Daniel Shirley
-
- Published online by Cambridge University Press:
- 17 November 2021, e51
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective:
Surgical site infections (SSIs) greatly burden healthcare systems around the world, particularly in low- and middle-income countries. We sought to employ the Systems Engineering Initiative for Patient Safety (SEIPS) model to better characterize SSI prevention practices and factors affecting adherence to prevention guidelines at Jimma University Medical Center (JUMC).
Design:Our cross-sectional study consisted of semistructured interviews designed to elicit perceptions of and barriers and facilitators to SSI prevention among surgical staff and observations of current preoperative, perioperative, and postoperative SSI prevention practices in surgical cases. Interviews were recorded, manually transcribed, and thematically coded within the SEIPS framework. Trained observers recorded compliance with the World Health Organization’s SSI prevention recommendations.
Setting:A tertiary-care hospital in Jimma, Ethiopia.
Participants:Surgical nurses, surgeons, and anesthetists at JUMC.
Results:Within 16 individual and group interviews, participants cited multiple barriers to SSI prevention including shortages of water and antiseptic materials, lack of clear SSI guidelines and training, minimal Infection Prevention Control (IPC) interaction with surgical staff, and poor SSI tracking. Observations from nineteen surgical cases revealed high compliance with antibiotic prophylaxis (94.7%), hand scrubbing (100%), sterile gloves and instrument use (100%), incision site sterilization (100%), and use of surgical safety checklist (94.7%) but lower compliance with preoperative bathing (26.3%), MRSA screening (0%), and pre- and postoperative glucose (0%, 10.5%) and temperature (57.9%, 47.3%) monitoring.
Conclusions:Utilizing the SEIPS model helped identify institution-specific barriers and facilitators that can inform targeted interventions to increase compliance with currently underperformed SSI prevention practices at JUMC.




















