Poster Presentation
Antibiotic Stewardship
Prospective Audit for Antimicrobial Use and Stewardship Practices in Intensive Care Unit at a Tertiary-Care Center in India
- Parul Singh, Purva Mathur, Kamini Walia, Anjan Trikha
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s40
-
- Article
-
- You have access Access
- Open access
- Export citation
-
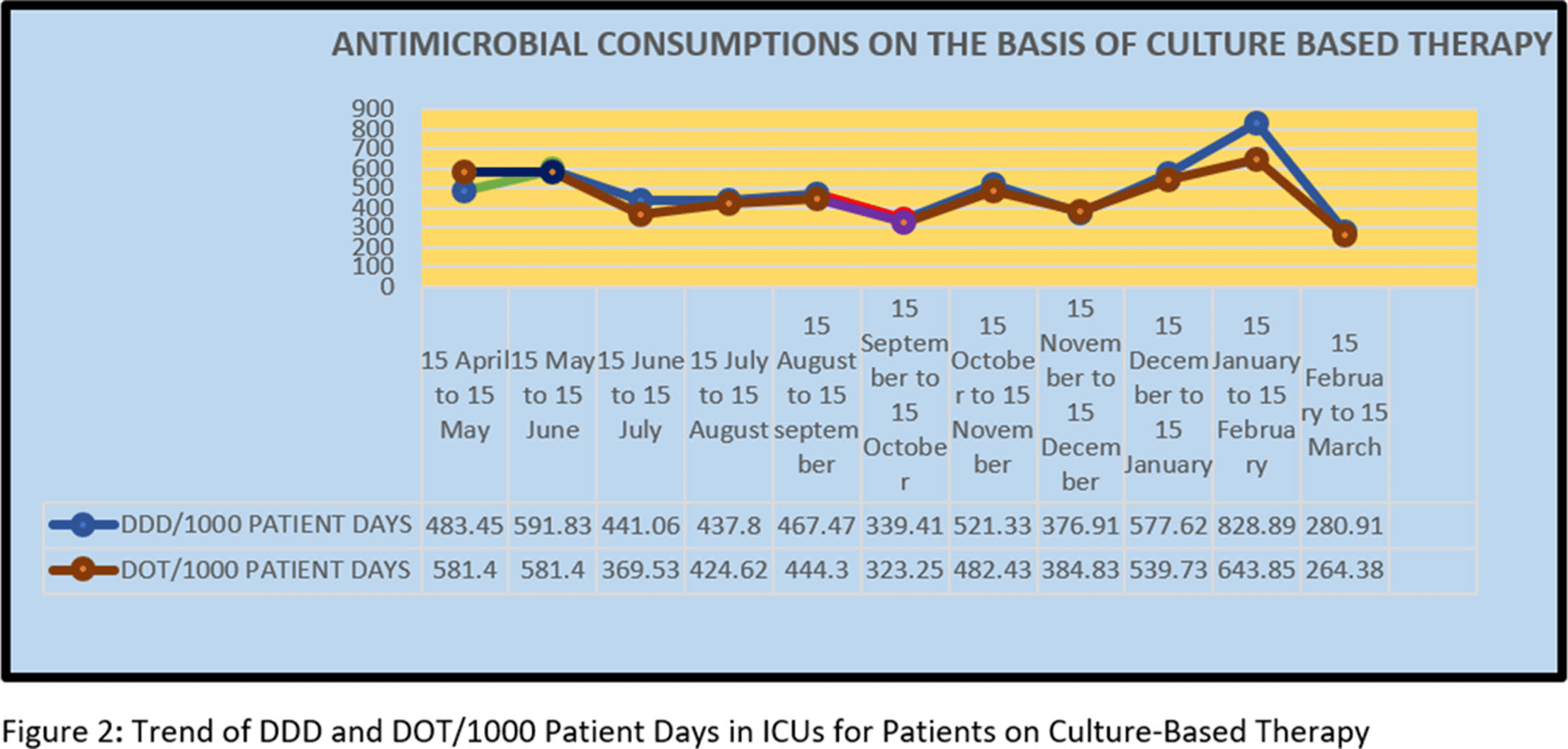
Background: Antimicrobial decision making in the ICU is challenging. Injudicious use of antimicrobials contributes to the development of resistant pathogens and drug-related adverse events. However, inadequate antimicrobial therapy is associated with mortality in critically ill patients. Antimicrobial stewardship programs are increasingly being implemented to improve prescribing. Methods: This prospective study was conducted over 11 months, during which the pharmacist used a standardized survey form to collect data on antibiotic use. Evaluation of antimicrobial use and stewardship practices in a 12-bed polytrauma ICU and a 20-bed neurosurgery ICU of the 248-bed AIIMS Trauma Center in Delhi, India. Antimicrobial consumption was measured using WHO-recommended defined daily dose (DDD) of given antimicrobials and days of therapy (DOT). Results: Antibiotics were ranked by frequency of use over the 11-month period based on empirical therapy and culture-based therapy. The 11-month DDD and DOT averages when empiric antibiotics were used were 532 of 1,000 patient days and 484 per 1,000 patient days, respectively (Figure 1). When cultures were available, DDD was 486 per 1,000 patient days and DOT was 442 per 1,000 patient days (Figure). Conclusions: The quantity and frequency of antibiotics used in the ICUs allowed the AMSP to identify areas to optimize antibiotic use such as educational initiatives, early specimen collection, and audit and feedback opportunities.
Funding: No
Disclosures: None
Figure 1.
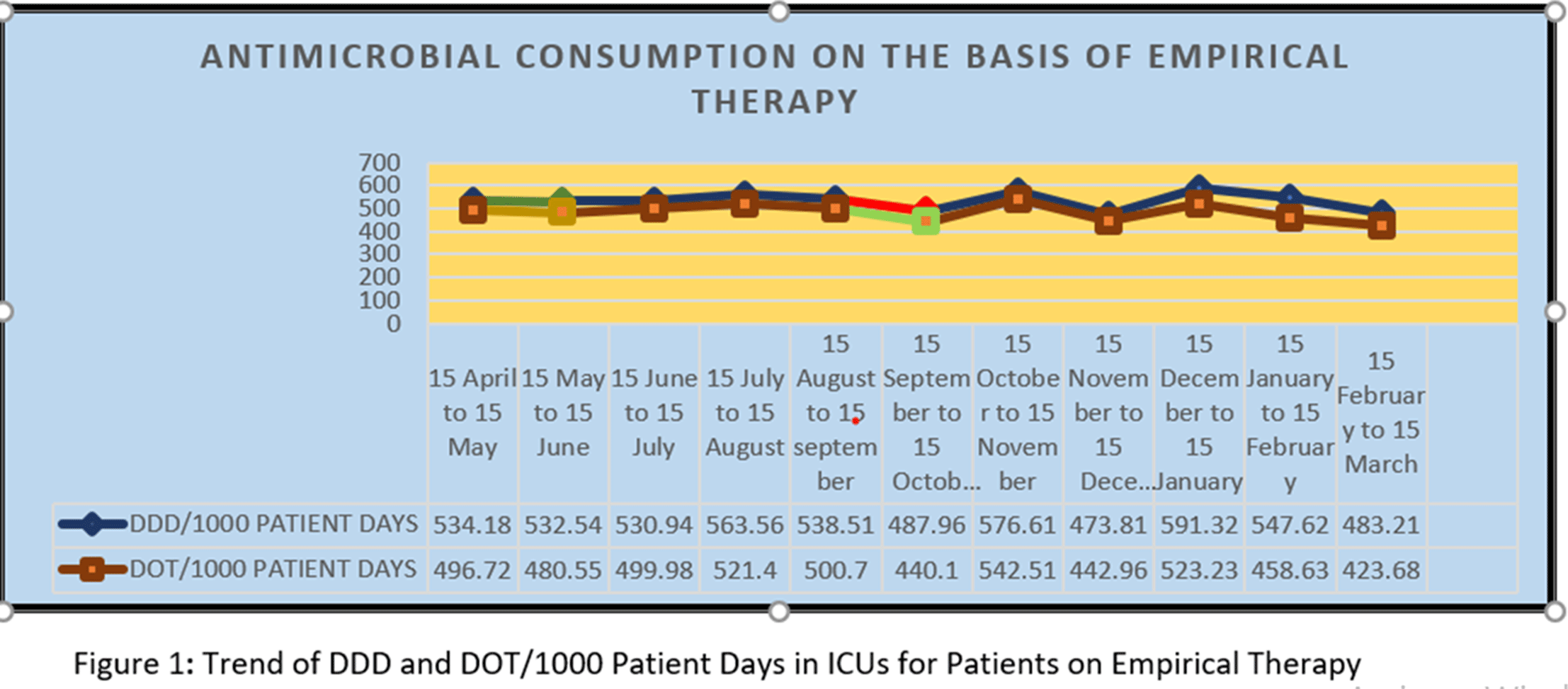
Figure 2.
Evaluating the Prevalence of Leading Practices in Antimicrobial Stewardship
- Barbara Braun, Salome Chitavi, Eddie Stenehjem, Mushira Khan, David Baker, David Hyun
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s41
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Most hospitals have a basic infrastructure in place for antimicrobial stewardship programs (ASPs). Although this is a critical first step, we need to ensure that ASPs are working to implement effective evidence-based approaches nationally. In 2018, a group of leading antibiotic stewardship organizations met and identified specific, effective, and recommended ASP activities based on current scientific evidence and their experience (Baker et al, Joint Comm J Qual Pat Saf 2019;45:517–523). To determine the extent to which hospitals are currently implementing the recommended practices, we conducted an electronic questionnaire–based assessment. Methods: A 50-item questionnaire-based assessment was sent via QualtricsTM to the hospital’s designated ASP leader. The sample comprised 992 Joint Commission accredited hospitals. The practices of interest related to (1) development of facility-specific treatment guidelines, (2) measuring appropriate use and concordance of care with these guidelines, (3) engaging clinicians while the patient is on the unit, (4) diagnostic stewardship, (5) measurement of antimicrobial utilization data, and (6) measuring hospital-acquired Clostridioides difficile infection (CDI) rates. Sampling weights were used to adjust the results for nonresponse using R software. Results: In total, 288 hospitals completed the questionnaire. Small and nonteaching hospitals were significantly less likely to respond (p < 0.005, p=0.01 respectively), however there were no differences by healthcare system membership or urban/rural location. 49% of respondents had the specialist term ASP or infectious disease (ID) in their title. Most hospitals (93.1%) had developed facility-specific treatment guidelines for specific inpatient conditions, often community-acquired pneumonia (85%), sepsis (81%), UTI (75%), and SSTI (69%). However, only 37% had formally assessed compliance with 1 or more of these guidelines. Also, 83% reported having a process for prospective audit and feedback, of which 43% do this 4–5 days per week. Similarly, 49% reported that they review all antimicrobials ordered. Recommendations are commonly given by the ASP pharmacist (69%) via some combination of telephone (78%), face-to-face (69%), text message (54%), and/or EHR alert (36%). Overall, 66% of hospitals had procedures in place to prevent inappropriate diagnostic testing for C. difficile, and 39% of hospitals had similar policies for urine specimens. Furthermore, >80% were routinely measuring days of therapy and CDI rates. Conclusions: Most hospitals have facility-specific treatment guidelines and measure CDI and days of therapy. Practices for active engagement with frontline staff in prospective audit and feedback vary widely. Greater understanding of barriers to assessing adherence to hospitals’ treatment guidelines is critical to improving this practice.
Funding: The Pew Charitable Trusts
Disclosures: None
C. difficile
Clostridioides difficile Is Not Difficult to Predict in Hospital Settings
- Kinta Alexander, Frances Petersen, Sean Brown
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s41
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background:Clostridioides difficile is a gram-positive bacteria that is the most common cause of hospital-associated infectious diarrhea among traditional and nontraditional high-risk populations. Excess healthcare costs associate with C. difficile infection (CDI) prevalence, morbidity, and mortality is shown to economically impact the US healthcare system with at least an additional $1 billion in annual cost. Exposure to antimicrobial agents resulted in increased risk for hospital-onset CDI (HO-CDI) at an inner-city hospital during 2010 and 2011. Methods: A retrospective case-control study of all persons with HO-CDI in the MICU was conducted at an inner-city hospital between January 1, 2010, and December 31, 2011. A patient was considered to have developed HO-CDI if diarrhea developed after 72 hours of admission into the MICU and a confirmed laboratory stool specimen for Clostridioides difficile infection (CDI) was obtained. A non–HO-CDI person was randomly selected using “risk set sampling.” After the application of inclusion and exclusion criteria, 88 cases were eligible for the study. Of these cases, 29 met the definition for HO-CDI, and 59 met the definition for non–HO-CDI. The relationship between antimicrobial use and the development of HO-CDI in patients in the MICU at an inner-city hospital was investigated using a logistic regression model in which the variable of total antibiotics was used as a possible predictor for predicting a positive HO-CDI. Results: Logistic regression was utilized to determine the relationships between selected study variables and presence or absence of HO-CDI. Total antibiotics was significantly related to HO-CDI. The results of this analysis showed that total antibiotics was a significant predictor for HO-CDI. The total value of the coefficient B for this predictor was 0.47, and the exponentiated value (exp[B]) of this coefficient was 1.60 (95% CI, 1.08–2.35). In this sample, patients who had 1 or more antibiotics were at a 60% greater risk of having a positive HO-CDI culture. There was a significant association between the use of metronidazole and HO-CDI (p < .001). Conclusions: Antimicrobial stewardship is an integral part of patient safety. The findings from this study were instrumental in the implementation of a fledging antimicrobial stewardship program and the use of evidence-based practices at this inner-city hospital.
Funding: No
Disclosures: None
Clinical Characteristics and Fecal Microbiome in Recurrent Versus Nonrecurrent Clostridioides difficile Infection
- Swapnil Lanjewar, Ashley Kates, Lauren Watson, Nasia Safdar
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s41-s42
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Up to 30% of patients with Clostridioides difficile infection (CDI) develop recurrent infection, which is associated with a 33% increased risk of mortality at 180 days. The gut microbiome plays a key role in initial and recurrent episodes of CDI. We examined the clinical characteristics and gut microbial diversity in patients with recurrent (rCDI) versus nonrecurrent CDI at a tertiary-care academic medical center. Methods: Stool samples were collected from 113 patients diagnosed with CDI between 2018 and 2019. Clinical and demographic data were extracted from the electronic medical record (Table 1), and 16S rRNA sequencing of the v4 region was carried out on the Illumina MiSeq using 2×250 paired-end reads. Sequences were binned into operational taxonomic units (OTUs) using mothur and were classified to the genus level whenever possible using the ribosomal database project data set version 16. Alpha diversity was calculated using the Shannon diversity index. Β diversity was calculated using the Bray-Curtis dissimilarity matrix. Differential abundance testing was done using DESeq to assess taxonomic differences between groups. A P value of .05 was used to assess significance. Results: In total, 55 patients had rCDI (prior positive C. difficile polymerase chain reaction in last 7–365 days) and 58 had nonrecurrent CDI (Table 1). Patients with rCDI had a higher frequency of organ transplant and comorbidity. No differences in α not β diversity were observed between groups. Also, 4 OTUs were more abundant in those with rCDI: Ruminococcus (n = 2), Odoribacter, and Lactobacillus. Patients with rCDI had microbiomes with greater proportions of Bacteroidetes (27% of OTUs) compared to the nonrecurrent group (18%) as well as fewer OTUs belonging to the Firmicutes phyla compared to the nonrecurrent patients (56% vs 59%). Among the rCDI patients, those experiencing 2 or more recurrences had greater abundances of Bacteroides and Ruminococcus, while those experiencing only 1 recurrence had significantly greater abundances of Akkermensia, Ruminococcus, Streptococcus, Roseburia, Clostridium IV, and Collinsella compared to those with only 1 recurrence (Table 2). Conclusions: Patients with rCDI had a more impaired microbiome than those with initial CDI. Ruminococcus OTUs have been previously indicated as a risk factor for recurrence and treatment failure, and they were significantly more abundant in those with rCDI and among those with multiple recurrences. The greatest differences in the microbiome were observed between those with 1 recurrence compared to those with multiple recurrences. Interventions for gut microbiome restoration should focus particularly on those with recurrent CDI.
Funding: No
Disclosures: None
Table 1. 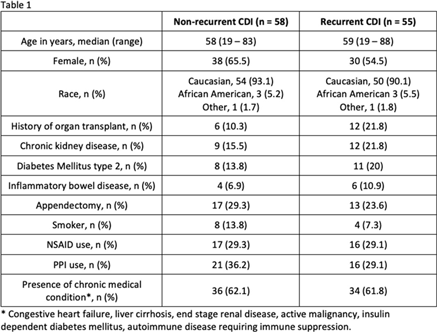
Table 2. 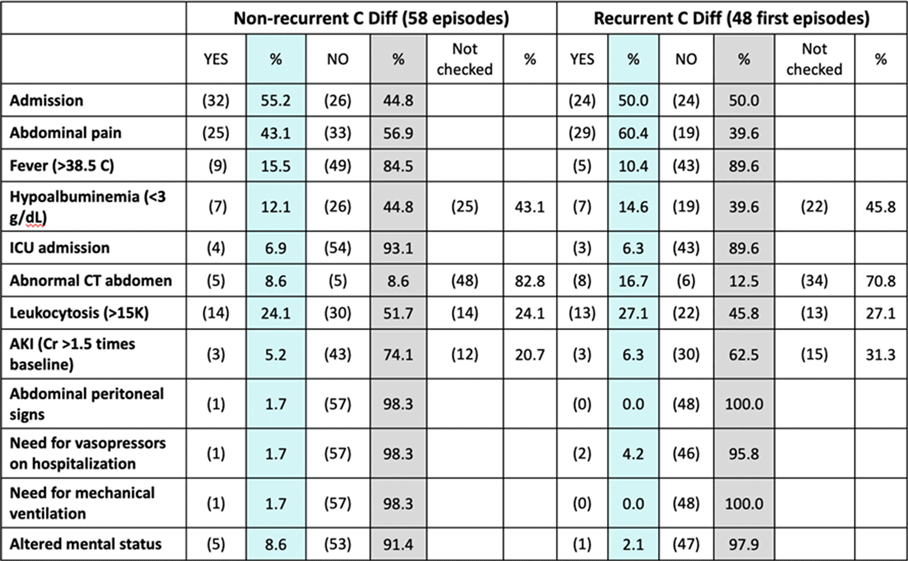
Impact of Two-Step Testing Algorithm on Reducing Hospital-Onset Clostridioides difficile Infections
- Bhagyashri Navalkele, Wendy Winn, Sheila Fletcher, Regina Galloway, Jason Parham, William Daley, Patrick Kyle, Vonda Clack, Kathy Sheilds
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s42-s43
-
- Article
-
- You have access Access
- Open access
- Export citation
-
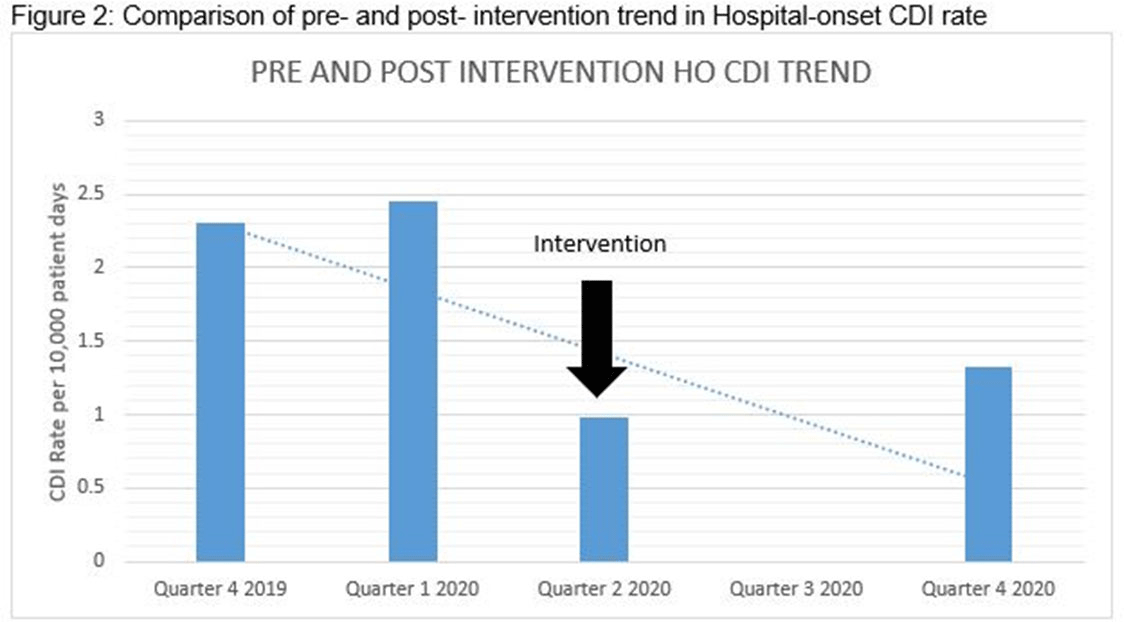
Clostridioides difficile infection (CDI) is one of the leading causes of hospital–onset infections. Clinically distinguishing true CDI versus colonization with C. difficile is challenging and often requires reliable and rapid molecular testing methods. At our academic center, we implemented a 2-step testing algorithm to help identify true CDI cases. The University of Mississippi Medical Center is a 700+ bed academic facility located in Jackson, Mississippi. Hospital-onset (HO) CDI was defined based on NHSN Laboratory Identified (LabID) event as the last positive C. difficile test result performed on a specimen using a multistep testing algorithm collected >3 calendar days after admission to the facility. HO-CDI data were collected from all inpatient units except the NICU and newborn nursery. HO-CDI outcomes were assessed based on standardized infection ratio (SIR) data. In May 2020, we implemented a 2-step testing algorithm (Figure 1). All patients with diarrhea underwent C. difficile PCR testing. Those with positive C. difficile PCR test were reflexed to undergo enzyme immunoassay (EIA) glutamate dehydrogenase antigen (Ag) testing and toxin A and B testing. The final results were reported as colonization (C. difficile PCR+/EIA Ag+/Toxin A/B−) or true CDI case (C. difficile PCR+/EIA +/Toxin A/B +) or negative (C. difficile PCR−). All patients with colonization or true infection were placed under contact isolation precautions until diarrhea resolution for 48 hours. During the preintervention period (October 2019–April 2020), 25 HO-CDI cases were reported compared to 8 cases in the postintervention period (June 2020–December 2020). A reduction in CDI SIR occurred in the postintervention period (Q3 2020–Q4 2020, SIR 0.265) compared to preintervention period (Q4 2019–Q1 2020, SIR 0.338) (Figure 2). We successfully reduced our NHSN HO-CDI SIR below the national average after implementing a 2-step testing algorithm for CDI. The 2-step testing algorithm was useful for antimicrobial stewardship to guide appropriate CDI treatment for true cases and for infection prevention to continue isolation of infected and colonized cases to reduce the spread of C. difficile spores.
Funding: No
Disclosures: None
Figure 1.
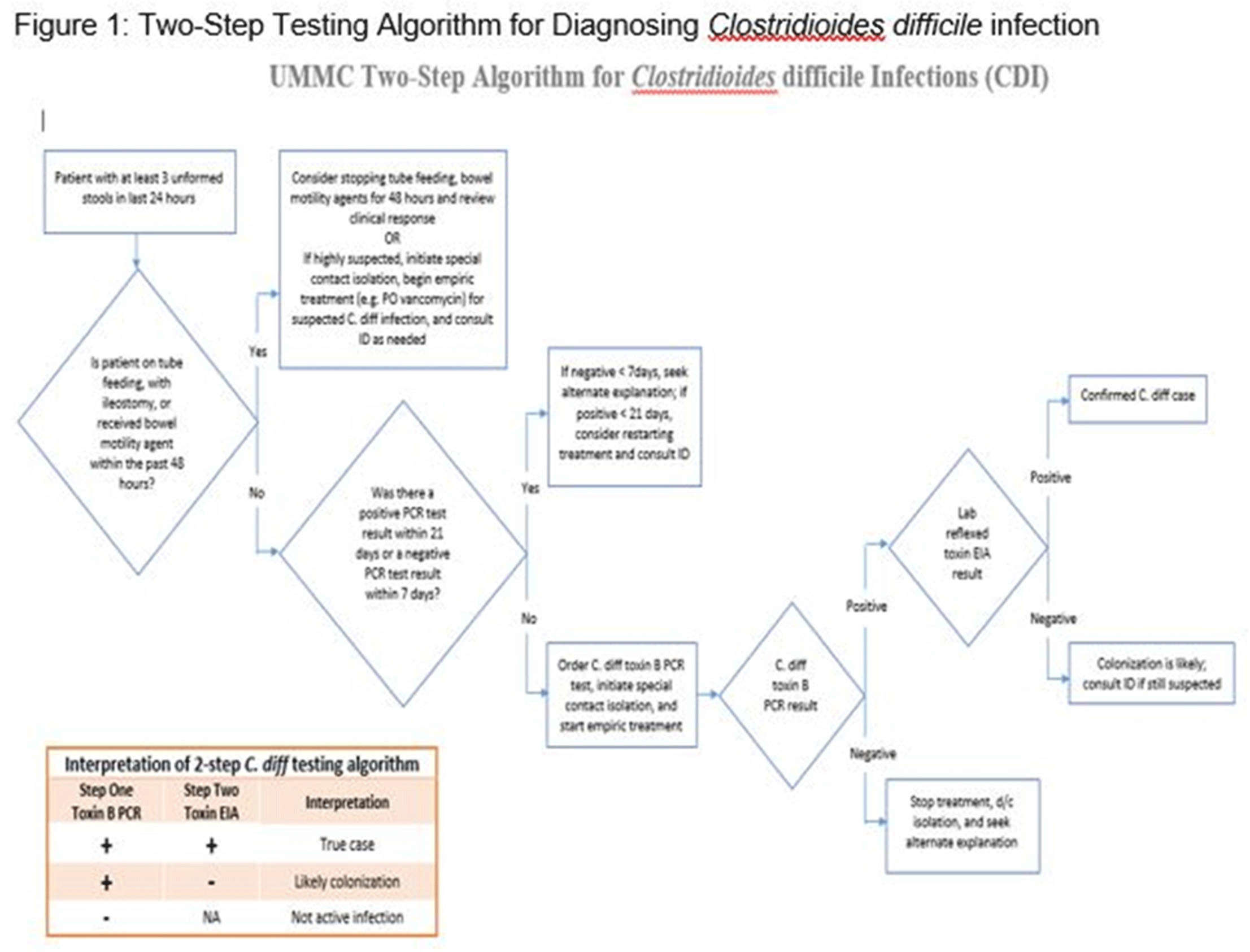
Figure 2.
Did Clostridioides difficile Testing and Infection Rates Change During the COVID-19 Pandemic?
- Armani Hawes, Payal Patel, Angel Desai
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s43
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The COVID-19 pandemic has underscored the importance of ongoing infection prevention efforts. Increased adherence to infection prevention recommendations, increased antibiotic use, improved hand hygiene, and correct donning and doffing of personal protective equipment may have influenced healthcare-associated infections (HAIs) in the United States during the pandemic. In this study, we investigated testing for Clostridioides difficile infection (CDI) and incidence during the initial surge of the pandemic. We hypothesized that strict adherence to contact precautions may have resulted in a decreased incidence of CDI in hospitalized patients during the first peak of the COVID-19 pandemic and that CDI testing may have increased even in the absence of directed diagnostic stewardship efforts. Methods: We conducted a single-center, retrospective, observational study at the Veterans’ Affairs (VA) Hospital in Ann Arbor, Michigan, between January 2019 and June 2020. We compared data on CDI tests from January 2019 through February 2020 to data from March 2020 (the admission of the first patient with COVID-19 at our institution) through June 2020. Pre-peak and peak periods were defined by confirmed cases in Washtenaw County. No novel diagnostic or CDI-focused stewardship interventions were introduced by the antimicrobial stewardship program during the study period. An interrupted time series analysis was performed using STATA version 16.1 software (StataCorp LLC, College Station, TX). Results: There were 6,525 admissions and 34,533 bed days between January 1, 2019, and June 30, 2020. Also, 900 enzyme immunoassay (EIA) tests were obtained and 104 positive cases of CDI were detected between January 2019 and June 2020. A statistically significant decrease in EIA tests occurred after March 1, 2020 (the COVID-19 peak in our region) compared to January 1, 2019–March 1, 2020 (Figure 1). After March 1, 2020, the number of EIA tests obtained decreased by 10.2 each month (95% CI, −18.7 to −1.7; P = .02). No statistically significant change in the incidence of CDI occurred. The use of antibiotics that were defined as high risk for CDI increased in the months of April–June 2020 (Figure 2). Conclusions: In this single-center study, we observed a stable incidence of CDI but decreased testing during the first peak of the COVID-19 pandemic. Understanding local HAI reporting is critical because changes in HAI reporting structures and exemptions during this period may have affected national reporting. Further research should be undertaken to investigate the effect of COVID-19 on other HAI reporting within the US healthcare system.
Funding: No
Disclosures: None
Figure 1.
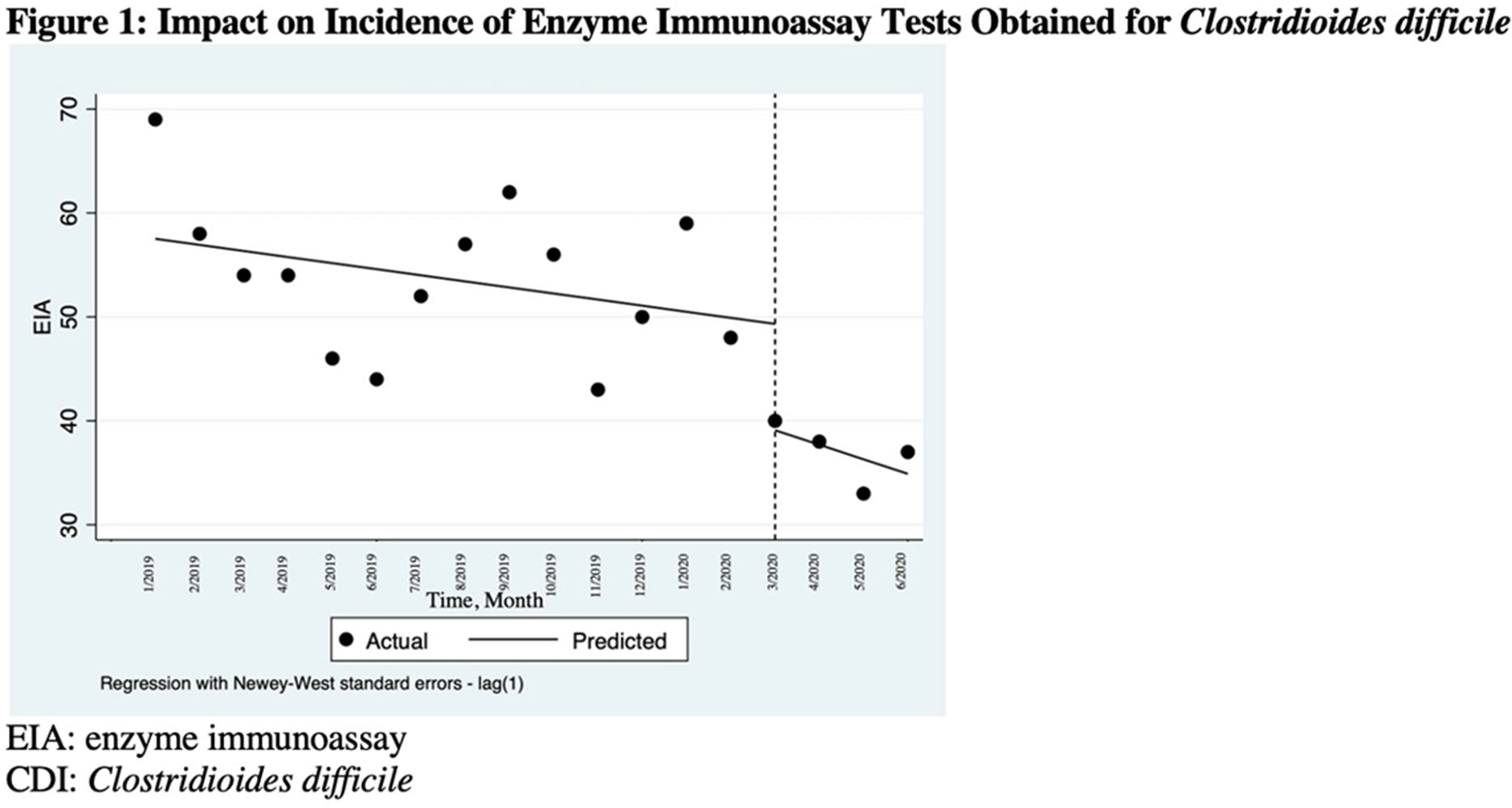
Figure 2.
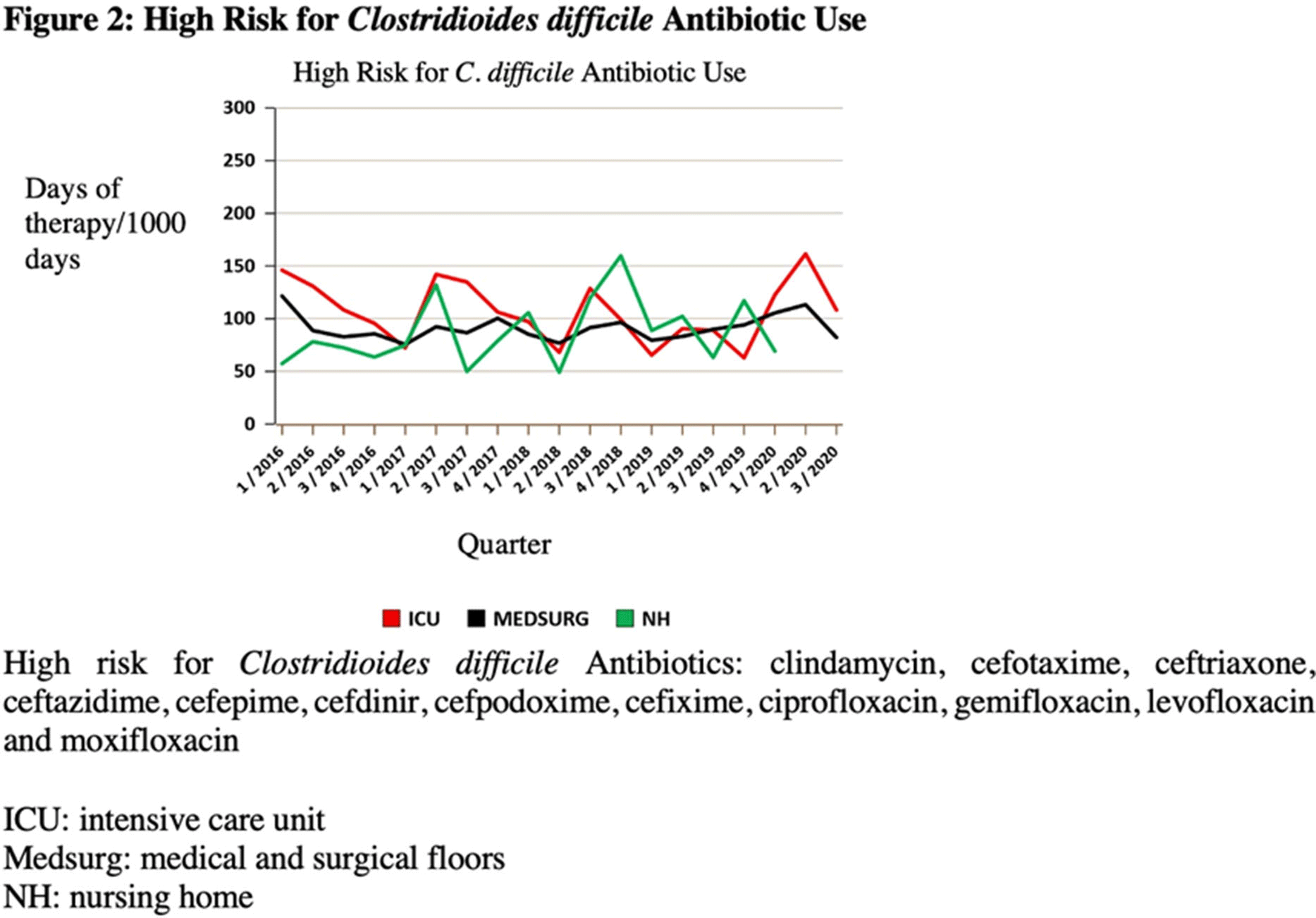
CLABSI
Inequities in CLABSI Rates in a Children’s Hospital by Race, Ethnicity, and Language Preference
- Caitlin McGrath, Matthew Kronman, Danielle Zerr, Brendan Bettinger, Tumaini Coker, Shaquita Bell
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s43-s44
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Systemic racism results in health inequities based on patient race, ethnicity, and language preference. Whether these inequities exist in pediatric central-line–associated bloodstream infections (CLABSIs) is unknown. Methods: This retrospective cohort study included patients with central lines hospitalized from October 2012 to June 2019 at our tertiary-care children’s hospital. Self-reported race, ethnicity, language preference, demographic, and clinical factors were extracted from the electronic health record. The primary outcome was non–mucosal barrier injury (non-MBI) CLABSI episodes as defined by the NHSN. CLABSI rates between groups were compared using χ2 tests and Cox proportional hazard regression. We adjusted for care unit, age, immunosuppressed status, diapered status, central-line type, line insertion within 7 days, daily CLABSI maintenance bundle compliance, number of blood draws and IV medication doses, and need for total parental nutrition, extracorporeal membrane oxygenation, and renal replacement therapy. In mid-2019, we engaged stakeholders in each care unit to describe preliminary findings and to identify and address potential drivers of observed inequities. Results: We included 337 non-MBI CLABSI events over 230,699 central-line days (CLDs). The overall non-MBI CLABSI rate during the study period was 1.46 per 1,000 CLDs. Unadjusted CLABSI rates for black or African American (henceforth, “black”), Hispanic, non-Hispanic white, and Asian (the 4 largest race or ethnicity groups by CLDs) patients were 2.74, 1.53, 1.42, 1.24 per 1,000 CLDs, respectively (P < .001) (Table 1). Unadjusted CLABSI rates for patients with limited-English proficiency (LEP) and English-language preference were 1.98 and 1.38 per 1,000 CLDs, respectively (P = .014). After adjusting for covariates, the hazard ratio (HR) point estimate for CLABSI rate remained higher for black patients (HR, 1.50; 95% CI, 0.99–2.28) and patients with LEP (HR, 1.33; 95% CI, 0.87–2.05), compared to the reference group based on largest CLD. The differences in CLABSI rate by race or ethnicity and language were more pronounced in 2 of our 6 care units. Stakeholder engagement and analysis of hospital data revealed opportunities on those units for improved (1) interpreter utilization and (2) line maintenance observation practices by race/ethnicity and language preference (data not shown). These findings and CLABSI rates over time by race/ethnicity and language preference (Figures 1 and 2) were shared with frontline staff. Conclusions: In our children’s hospital, CLABSI rates differed based on patients’ self-reported race, ethnicity, and language preference, despite controlling for factors commonly associated with CLABSI. Identifying inequities in CLABSI rates and mitigating their determinants are both essential to the goal of achieving equitable care.
Funding: No
Disclosures: None
Table 1. 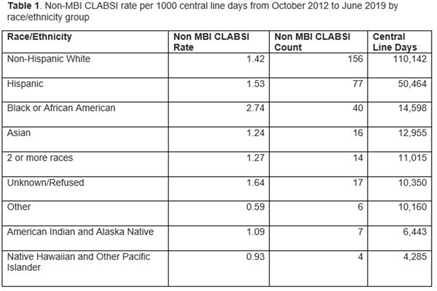
Figure 1.

Figure 2.

COVID-19
Use of COVID-19 Serologic Testing in Healthcare Workers with Acute Respiratory Tract Infection
- Amy Ray
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s44
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Diagnostic tests for COVID-19 are in high demand. Serologic assays are of interest as diagnostic adjuncts to SARS-CoV-2 quantitative polymerase chain reaction (PCR); however, many of the commercially available assays have limited validation data and clinical utility is unknown. We describe the utilization of SARS-CoV-2 IgG enzyme-linked immunosorbent assay (ELISA) for healthcare workers with acute respiratory tract infection (ARTI) who underwent SARS-CoV-2 PCR testing. Methods: The MetroHealth System is the largest public hospital system in Ohio, employing ~8,000 staff. COVID-19 detection began in early March 2020. EDI novel coronavirus COVID-19 IgG ELISA (KT-1032) targeting antibody response to viral nucleocapsid was obtained for diagnostic and seroprevalence analyses. Manufacturer reports of sensitivity and specificity of the assay are 100% and 99%, respectively. A 2-part test strategy for employees with symptoms of ARTI was implemented. Qualifying symptoms for SARS-CoV-2 PCR testing included fever and either cough or shortness of breath. Additional symptoms were included to reflect expanding knowledge of COVID-19. Employees who underwent SARS-CoV-2 PCR testing (Luminex ARIES) were offered serologic testing on day 14 following PCR result. Education accompanied the offer for serologic testing as well as the receipt of test result to aide interpretation. Results: From April 16, 2020, through July 6, 2020, 588 employees underwent PCR testing. Overall, 70 cases of COVID-19 were detected. Of the 197 employees who opted for serologic testing, IgG positivity was 12.6%. The mean time to IgG collection following PCR result was 30 days (range, 10–79). Using PCR results obtained in the clinical setting of ARTI as the diagnostic gold standard, IgG was 84.6% sensitive and 98.2% specific (Figure 1). Conclusions: In a population of symptomatic healthcare workers, SARS-CoV2 IgG testing was specific for COVID-19 diagnosis. Sensitivity was inadequate compared to the positive predictive agreement of 90% or greater required for US Food and Drug Administration emergency use authorization. In a low-prevalence environment for COVID-19 (<5%), a positive SARS-CoV-2 IgG has a low positive predictive value, which may falsely imply immunity and may negatively affect infection prevention practices.
Funding: No
Disclosures: None
Figure 1.

COVID-19 and Ventilator-Associated Event Discordance
- Kelly Cawcutt, Mark Rupp, Lauren Musil
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s45
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The COVID-19 pandemic has challenged healthcare facilities since its discovery in late 2019. Notably, the subsequent COVID-19 pandemic has led to an increase in healthcare-acquired infections such as ventilator associated events (VAEs). Many hospitals in the United States perform surveillance for the NHSN for VAEs by monitoring mechanically ventilated patients for metrics that are generally considered to be objective and preventable and that lead to poor patient outcomes. The VAE definition is met in a stepwise manner. Initially, a ventilator-associated condition (VAC) is met when there an increase in ventilator requirements after a period of stability or improvement. An IVAC is then met when there is evidence of an infectious process such as leukocytosis or fever and a new antimicrobial agent is started. Finally, possible ventilator-associated pneumonia (PVAP) is met when there is evidence of microbial growth or viral detection. Since the beginning of the COVID-19 pandemic, our hospital has seen an increase in VAEs, which is, perhaps, not unexpected during a respiratory illness pandemic. However, the NSHN definitions of VAE, and PVAP in particular, do not account for the novelty and nuances of COVID-19. Methods: We performed a chart review of 144 patients who had a VAE reported to the NHSN between March 1 and December 31, 2020. Results: Of the 144 patients with a VAE reported to NHSN, 39 were SARS-CoV-2 positive. Of the 39 patients, 4 patients (10.25%) met the NHSN PVAP definition due to a positive SARS-CoV-2 PCR that was collected in the prolonged viral shedding period of their illness (< 90 days). One of the four patients also had a bacterial infection in addition to their subsequent positive COVID-19 result. All these patients were admitted to the hospital with a COVID-19 diagnosis and their initial PCR swab was performed upon admission. Conclusions: We believe that the PVAP definition was inappropriately triggered by patients who were decompensating on the ventilator due to a novel respiratory virus that was present on admission. Early in the pandemic, frequent swabbing of these patients was performed to try and understand the duration of viral shedding and to determine when it would be safe to transfer patients from isolation after prolonged hospitalization. The NSHN definition should take into consideration the prolonged viral shedding period of COVID-19 and natural history of the illness, and subsequent COVID-19 testing within 90 days of an initial positive should not require classification as a hospital-acquired PVAP.
Funding: No
Disclosures: None
Stewardship of Remdesivir Use in a Rural Community Hospital During the COVID-19 Pandemic
- Raghavendra Tirupathi, Melissa Gross
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s45
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Remdesivir was granted EUA followed by full FDA approval for treatment of hospitalized COVID-19 patients on October 22, 2020, based on the results from the ACTT1 trial. Remdesivir use was initially restricted to infectious disease (ID) physicians in our hospital with prescription needing formal ID consultation until complete approval. Due to increasing case counts in our hospital, a decision was made to allow intensivists and hospitalists the authorization to prescribe remdesivir in a phased manner. In this retrospective study, we assessed the impact of phased-in prescribing on remdesivir utilization and days of therapy of antimicrobials. Methods: Remdesivir prescribing was streamlined by real-time institutional guidelines developed by a COVID-19 treatment committee constituting ID and other clinicians. Eligibility for remdesivir included positive SARS-CoV-2 PCR test, severe disease defined as persistent hypoxia (<94% oxygen saturation on room air), requiring supplemental oxygen and/or on mechanical ventilation (MV) for <72 hours, and symptom onset of <10 days. We retrospectively reviewed cohorts of 3 periods during which remdesivir was prescribed. In the first cohort A, between October 23, 2020, and November 12, 2020, remdesivir was restricted to ID physicians with formal ID consultation. Cohort B comprised inpatients between November 13, 2020, and December 6, 2020, when hospitalists and intensivists were allowed to prescribe remdesivir through an EMR order set after prior authorization by an ID physician via curbside or telephonic consultation. Cohort C, from December 7, 2020, to December 26, 2020, comprised inpatients with unrestricted prescribing of remdesivir by hospitalists and intensivists. We also evaluated antibiotic use. Results: In cohort A, SARS CoV-2 positivity was 20.3%; 64 inpatients tested positive and 35 patients (54.7%) who met the criteria were prescribed remdesivir after a formal consultation with an ID physician. In cohort B, requiring prior authorization by an ID physician, SARS-CoV-2 positivity rapidly increased to 34%; 193 patients tested positive and 97 patients (50.3%) received remdesivir. In cohort C, during unrestricted access, positivity further increased to 38%; 235 inpatients tested positive and 123 (52.5%) received remdesivir. Remdesivir use remained steady during the 3 phases of gradual de-escalation of restricted prescribing and safe handoff in the context of clear guidelines, as well as ongoing curbside education provided by ID physicians during the second phase. Cohort B demonstrated the best prescribing rates. Antimicrobial prescribing data were also collected during the 3 cohort phases (Figures 1–3). Conclusions: Remdesivir is an expensive antiviral with limited utility and maximum benefit in COVID-19 inpatients who are hypoxic but do not require mechanical ventilation. Stewardship of remdesivir with safe, gradual handoff to inpatient can be achieved without overuse.
Funding: No
Disclosures: None
Figure 1.
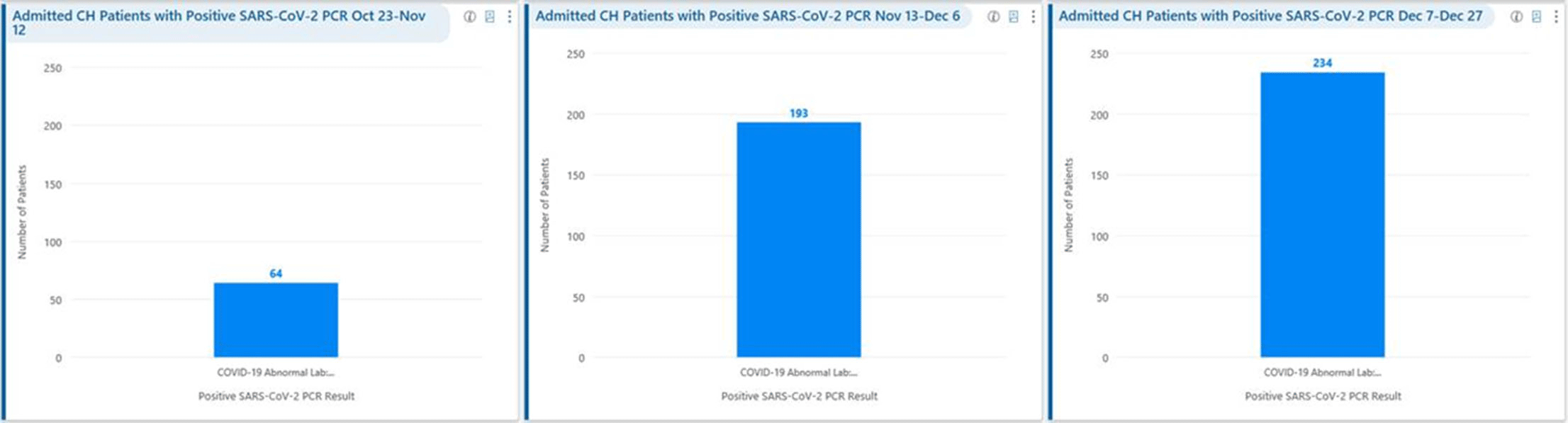
Figure 2.
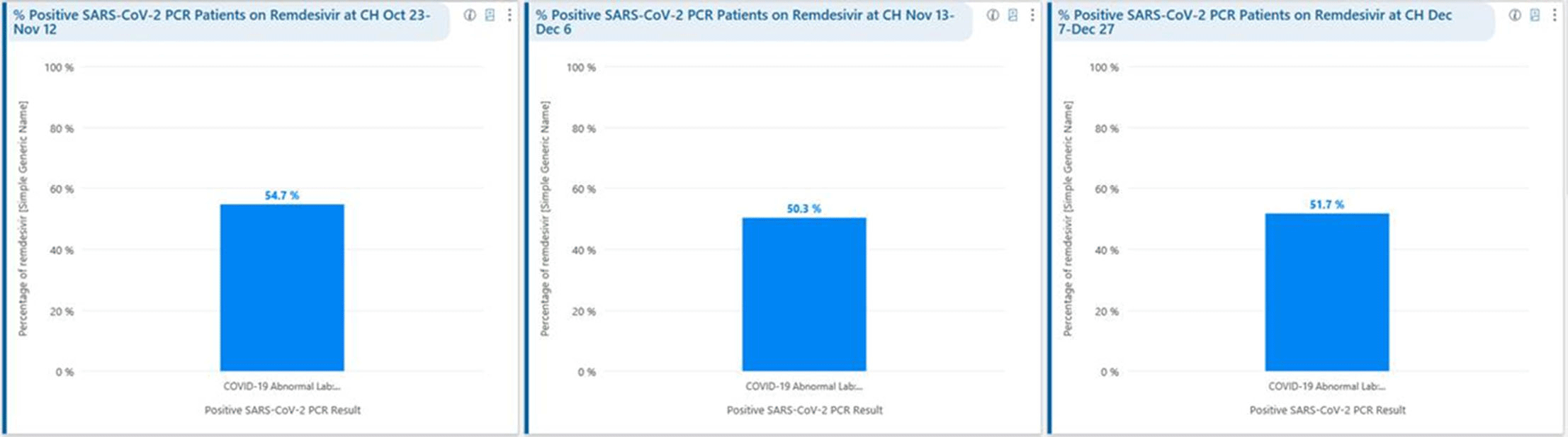
Figure 3.
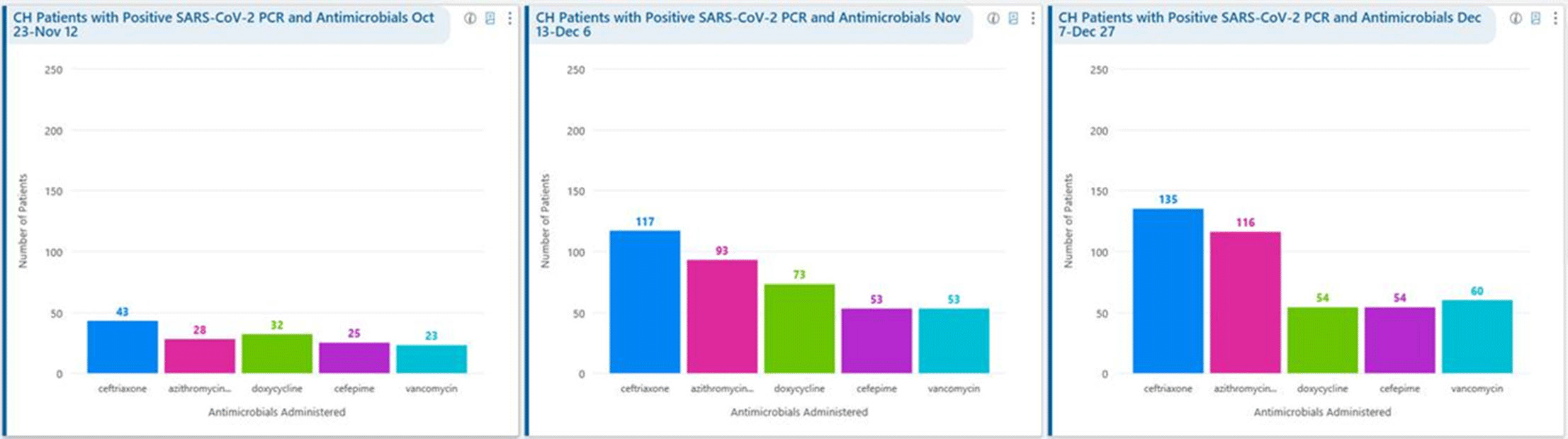
Comparing Hospital Healthcare-Associated Infection Incidence During Pre-COVID-19 Pandemic and Pandemic Eras
- Andrea Parriott, N. Neely Kazerouni, Erin Epson
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s45-s46
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Diversion of resources from infection prevention activities, personal protective equipment supply shortages, conservation (extended use and reuse) or overuse with multiple gown and glove layers, and antimicrobial prescribing changes during the COVID-19 pandemic might increase healthcare-associated infection (HAI) incidence and antimicrobial resistance. We compared the incidences of Clostridioides difficile infection (CDI), methicillin-resistant Staphyloccocus aureus bloodstream infection (MRSA BSI), and vancomycin-resistant enterococci bloodstream infection (VRE BSI) reported by California hospitals during the COVID-19 pandemic with incidence data collected prior to the pandemic. Methods: Using data reported by hospitals to the California Department of Health via the NHSN, we compared incidences in the second and third quarters of 2020 (pandemic) to the second and third quarters of 2019 (before the pandemic). For CDI and MRSA BSI, we compared the standardized infection ratios (SIRs, based on the 2015 national baseline), and we calculated the P values. No adjustment model is available for VRE BSI; thus, we measured incidence via crude incidence rates (infections per 100,000 patient days). We calculated incidence rate ratio (IRR) with 95% CI for VRE BSI. To examine the possible effect of missing data during the pandemic, we performed a sensitivity analysis by excluding all facilities that had incomplete data reporting at any time during either analysis period. Results: Incidence measures and numbers of facilities contributing data in prepandemic and pandemic periods are shown in Table 1. There were no statistically significant changes in SIRs at P = .05 for either MRSA BSI or CDI between the prepandemic and pandemic periods (MRSA BSI P = .17; CDI P = .08). Crude VRE BSI incidence increased during the pandemic compared to the prepandemic period (IRR, 1.40; 95% CI, 1.16–1.70). Excluding facilities with incomplete data had minimal effect. Conclusions: We found insufficient evidence that MRSA BSI or CDI incidence changed in California hospitals during the pandemic relative to the prepandemic period; however, there was a significant increase in the crude incidence of VRE BSI. Next, we will include interrupted time series analyses to assess departure from long-term trends, including a risk-adjusted model for VRE BSI. Additionally, we will evaluate for changes in central-line–associated bloodstream infection incidence and antimicrobial resistance among HAI pathogens.
Funding: No
Disclosures: None
Table 1. 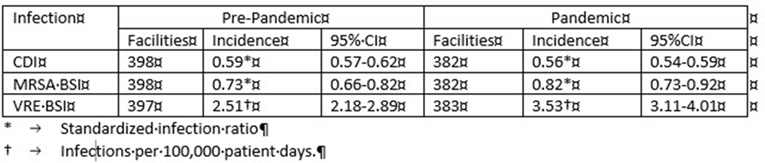
Secondary Bacterial Pneumonias and Bloodstream Infections in Patients Hospitalized with COVID-19
- Max Adelman, Divya Bhamidipati, Alfonso Hernandez, Ahmed Babiker, Michael Woodworth, Chad Robichaux, David Murphy, Sara Auld, Colleen Kraft, Jesse Jacob
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s46
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Group Name: The Emory COVID-19 Quality and Clinical Research Collaborative
Background: Patients hospitalized with COVID-19 are at risk of secondary infections—10%–33% develop bacterial pneumonia and 2%–6% develop bloodstream infection (BSI). We conducted a retrospective cohort study to identify the prevalence, microbiology, and outcomes of secondary pneumonias and BSIs in patients hospitalized with COVID-19. Methods: Patients aged ≥18 years with a positive SARS-CoV-2 real-time polymerase chain reaction assay admitted to 4 academic hospitals in Atlanta, Georgia, between February 15 and May 16, 2020, were included. We extracted electronic medical record data through June 16, 2020. Microbiology tests were performed according to standard protocols. Possible ventilator-associated pneumonia (PVAP) was defined according to Centers for Disease Control and Prevention (CDC) criteria. We assessed in-hospital mortality, comparing patients with and without infections using the χ2 test. SAS University Edition software was used for data analyses. Results: In total, 774 patients were included (median age, 62 years; 49.7% female; 66.6% black). In total, 335 patients (43.3%) required intensive care unit (ICU) admission, 238 (30.7%) required mechanical ventilation, and 120 (15.5%) died. Among 238 intubated patients, 65 (27.3%) had a positive respiratory culture, including 15 with multiple potential pathogens, for a total of 84 potential pathogens. The most common organisms were Staphylococcus aureus (29 of 84; 34.5%), Pseudomonas aeruginosa (16 of 84; 19.0%), and Klebsiella spp (14 of 84; 16.7%). Mortality did not differ between intubated patients with and without a positive respiratory culture (41.5% vs 35.3%; P = .37). Also, 5 patients (2.1%) had a CDC-defined PVAP (1.7 PVAPs per 1,000 ventilator days); none of them died. Among 536 (69.3%) nonintubated patients, 2 (0.4%) had a positive Legionella urine antigen and 1 had a positive respiratory culture (for S. aureus). Of 774 patients, 36 (4.7%) had BSI, including 5 with polymicrobial BSI (42 isolates total). Most BSIs (24 of 36; 66.7%) had ICU onset. The most common organisms were S. aureus (7 of 42; 16.7%), Candida spp (7 of 42; 16.7%), and coagulase-negative staphylococci (5 of 42; 11.9%); 12 (28.6%) were gram-negative. The most common source was central-line–associated BSI (17 of 36; 47.2%), followed by skin (6 of 36; 16.7%), lungs (5 of 36; 13.9%), and urine (4 of 36; 11.1%). Mortality was 50% in patients with BSI versus 13.8% without (p < 0.0001). Conclusions: In a large cohort of patients hospitalized with COVID-19, secondary infections were rare: 2% bacterial pneumonia and 5% BSI. The risk factors for these infections (intubation and central lines, respectively) and causative pathogens reflect healthcare delivery and not a COVID-19–specific effect. Clinicians should adhere to standard best practices for preventing and empirically treating secondary infections in patients hospitalized with COVID-19.
Funding: No
Disclosures: None
COVID-19 Contact Tracing in a Pediatric Hospital: Maximizing Effectiveness Through Specialized Team and Automated Tools
- Lindsay Weir, Jennifer Ormsby, Carin Bennett-Rizzo, Jonathan Bickel, Colleen Dansereau, Matthew Horman
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s46-s47
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: In their interim infection prevention and control recommendations for the coronavirus disease 2019 (COVID-19) pandemic, the Centers for Disease Control and Prevention (CDC) recommend that healthcare facilities have a plan to identify, investigate, and trace potential COVID-19 exposures. In an academic hospital, the scale of such tracing is substantial, given that medically complex patients can have dozens of staff contacts across multiple locations during an encounter. Furthermore, the family-centered care model employed by pediatric institutions precludes visitor exclusion, further complicating tracing efforts. Despite this complexity, tracing accuracy and timeliness is of paramount importance for exposure management. To address these challenges, our institution developed a contact-tracing system that balanced expert participation with automated tracing tools. Methods: Our institution’s contact-tracing initiative includes positive patients, parents and/or visitors, and staff for the enterprise’s inpatient, procedural, and ambulatory locations at the main campus and 4 satellites. The team consists of 11 staff and is overseen by an infection preventionist. For positive patients and parents and/or visitors, potentially exposed staff are automatically identified via a report that extracts staff details for all encounters occurring during the patient’s infectious period. For positive staff, trained contact tracers call the staff member to determine whether mask and distancing practices could result in others meeting CDC exposure criteria. Any potentially exposed healthcare workers (HCWs) receive an e-mail that details exposure criteria and provides follow-up instructions. These HCWs are also entered into a secure, centralized tracking database that (1) allows infection prevention and occupational health staff to query and identify all epidemiologic links between traced patients, parents and/or visitors, and staff, and (2) initiates staff enrollment in a twice-daily symptom tracking system administered via REDCap. Potentially exposed patients and parents and/or visitors are contacted directly by a hospital representative. The contact tracing team, infection prevention staff, and occupational health staff meet daily to review positive staff cases in the last 24 hours. Results: To date, the team has traced ~1,300 patients, 15 parents and/or visitors, and 700 staff. Since the start of the pandemic, tracing and contact notification for all positive cases has been conducted within 24 hours. Through these proactive tracing efforts and other institutional infection prevention initiatives, the institution only experienced 1 staff cluster (N < 15) and <5 hospital-onset patient cases. Conclusions: Equipping a trained group of contact tracers with automated tracking tools can afford infection prevention and occupational health departments the ability to achieve and sustain timely and accurate contact tracing initiatives throughout a large-scale pandemic response.
Funding: No
Disclosures: None
Transmission of COVID-19 on an Inpatient Hospital Prison Unit
- Kelsey Witherspoon, Manisha Shah, Justin Smyer, Nora Colburn, Christina Liscynesky, Courtney Hebert, Shandra Day
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s47
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Prison populations have been disproportionally affected by COVID-19, partly due to challenges related to social distancing. Data on viral transmission dynamics in inpatient prison units remain limited. The Ohio State University Wexner Medical Center (OSUWMC) has a 24-bed inpatient prison unit in collaboration with the Ohio Department of Rehabilitations and Corrections (ODRC). The unit has 5 shared rooms holding 4 patients each and 4 single-patient rooms. Several cases of inpatient transmission of COVID-19 were identified on the inpatient prison unit. Methods: An IRB-approved retrospective chart review was conducted to evaluate inpatient transmission dynamics of hospital-acquired (HCA) COVID-19. All ODRC patients admitted from March 1 to April 24, 2020, were included. Patients assigned to the prison unit during their hospital stay were evaluated for potential HCA COVID-19, defined as a positive SARS-CoV-2 test ≥4 days after admission. Patient characteristics, testing data, symptoms, aerosol-generating procedures (AGPs), and room assignments were reviewed. Healthcare workers (HCWs) and correction officers (COs) working on the unit who tested positive during this period were identified. Results: In total, 142 ODRC patients were admitted during the study period and 89% had a positive SARS-CoV-2 testing prior to or during admission. Also, 61 patients (43%) were assigned to the prison unit. Moreover, 8 patients on the unit met potential HCA COVID-19 definition with 7 linked to 3 distinct clusters. Also, 7 COs had COVID-19 (outside hospital exposure) and 5 HCWs acquired COVID-19 from patient exposure on the unit. In cluster 1, 4 patients admitted to the same room developed HCA COVID-19. A symptomatic index patient not tested on admission given an atypical presentation required CPAP and frequent nebulizer treatments. In cluster 2, 1 patient from cluster 1 was transferred to another room. The new roommate subsequently developed HCA COVID-19. In cluster 3, a symptomatic correctional officer was assigned to 2 patients in a shared room; the patients later developed HCA COVID-19. Conclusions: Three patient clusters of HCA COVID-19 on a prison unit were identified. Aerosol transmission potentially played a role in cluster 1. Inpatient transmission within the unit prompted updated guidance for ODRC admissions, including universal SARS-CoV-2 admission testing, excluding patients requiring AGPs from shared rooms, and preemptive isolation for patients from an ODRC facility experiencing a COVID-19 surge. Universal testing was quickly expanded to all inpatient admissions. HCWs and COs were also linked to inpatient transmission, highlighting the importance of strict infection control practices for patient populations who cannot socially distance.
Funding: No
Disclosures: None
Building a PPE Monitor Team as Part of a Comprehensive COVID-19 Prevention Strategy
- Shelley Summerlin-Long, Brooke Brewer, Amy Selimos, Mark Buchanan, Christa Clark, Karen Croyle, Cynthia Culbreth, Pam Del Monte, Lauren DiBiase, Lori Hendrickson, Pam Miller, Natalie Schnell, Katherine Schultz, Lisa Stancill, Lisa Teal, David Weber, Emily Sickbert-Bennett
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s47
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The use of personal protective equipment (PPE) is a critical intervention in preventing the spread of transmission-based infections in healthcare settings. However, contamination of the skin and clothing of healthcare personnel (HCP) frequently occurs during the doffing of PPE. In fact, nearly 40% of HCP make errors while doffing their PPE, causing them to contaminate themselves. PPE monitors are staff that help to promote their colleagues’ safety by guiding them through the PPE donning and doffing processes. With the advent of the COVID-19 pandemic in early 2020, the UNC Medical Center chose to incorporate PPE monitors as part of its comprehensive COVID-19 prevention strategy, using them in inpatient areas (including COVID-19 containment units and all other units with known or suspected SARS-CoV-2–positive patients), procedural areas, and outpatient clinics. Methods: Infection prevention and nursing developed a PPE monitoring team using redeployed staff from outpatient clinics and inpatient areas temporarily closed because of the pandemic. Employee training took place online and included fundamentals of disease transmission, hand hygiene basics, COVID-19 policies and signage, and videos on proper donning and doffing, including coaching tips. The monitors’ first shifts were supervised by experienced monitors to continue in-place training. Employees had competency sheets signed off by a supervisor. Results: The Medical Center’s nursing house supervisors took over management and deployment of the PPE monitoring team, and infection prevention staff continued to train new members. Eventually, as closed clinics and areas reopened and these PPE monitors returned to their regular positions, areas used their own staff to perform the role of PPE monitor. In the fall of 2020, a facility-wide survey was sent to all inpatient staff to assess their perceptions of the Medical Center’s efforts to protect them from acquiring COVID-19. It included a question asking how much staff agreed or disagreed that PPE monitors “play an important role in keeping our staff who care for COVID-19 patients safe.” Of the 626 staff who answered this question, 67.6% agreed or strongly agreed that PPE monitors played an important role in keeping staff safe. Thus far, there has been no direct transmission or clusters of COVID-19 involving HCP in COVID-19 containment units with PPE monitors. Conclusions: PPE monitors are an important part of a comprehensive COVID-19 prevention strategy. In early 2021, the UNC Medical Center posted and hired paid PPE monitor positions to continue this critical work in a sustainable way.
Funding: No
Disclosures: None
The COVID-19 Pandemic and Antibiotic Use on the United States–Mexico Border
- Sana Khan, Katherine Ellingson, Gemma Parra, Juan Villanueva, Carlos Garrido
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s47-s48
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The US–Mexico border represents a unique region of the country where antibiotics are more accessible and nonprescription treatment with antibiotics is deeply enculturated. Currently, both the United States and Mexico are experiencing widespread community transmission of SARS-CoV-2, which may have implications for antibiotic seeking and use. The objective of this study was to examine antibiotic seeking behavior as it relates to COVID-19 in the border region relative to the greater US and Mexico populations. Methods: An interdisciplinary team at The University of Arizona developed a survey to assess knowledge, attitudes, and beliefs about antibiotics along the US–Mexico border region (defined as 100 km from the border) and to compare findings from the border region to the broader US and Mexico populations. The team recruited survey participants through Amazon’s MTurk survey platform and through the distribution of recruitment flyers to community partners in Arizona and Mexico border regions from October 2020 to January 2021. Targeted recruitment was 750 through March 2021. We report here on findings from the first round of recruitment (n = 116). These participants were asked whether they had sought out antibiotics specifically as a treatment for COVID-19, as well as their general beliefs and behaviors on self-seeking antibiotics for illness. Results: As of January 24, 2021, we surveyed 116 participants: 82 (70.7%) from the United States and 34 (29.3%) from Mexico. Most participants (71.2%) were aged 25–44 years; 56.9% were male; and 50% reported Hispanic ethnicity. Of these, 13.8% lived within 100 km of the US–Mexico border. Overall, 21.6% of participants reported taking antibiotics to fight COVID-19–like illness. Of these participants, 28% obtained the antibiotics directly from a pharmacy, without a physician prescription, and 16% obtained them from an online vendor. Additionally, 33% of US respondents reported that they would be willing to travel to Mexico to obtain antibiotics if they were too difficult to obtain in the United States. Of these respondents, 55% said they would be willing to travel for >1 hour to obtain antibiotics. Conclusions: Preliminary data suggest that the COVID-19 pandemic will have widespread ramifications on antibiotic seeking behavior and could propagate antibiotic resistance. Targeted intervention strategies in the border region are necessary to mitigate the unique factors that contribute to antibiotic use in this area.
Funding: No
Disclosures: None
Risk Factors for In-Hospital Mortality from COVID-19 Among Nursing Home Patients—An Urban Center Experience
- Amit Vahia, Mamta Sharma, Leonard Johnson, Ashish Bhargava, Louis Saravolatz, Susan Szpunar
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s48
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: As the COVID-19 pandemic continues, special attention is focused on high-risk patients. In this study, we assessed the risk factors for COVID-19 mortality in nursing home patients. Methods: In this retrospective cohort study, we reviewed the electronic medical records of SARS-COV-2 PCR–positive nursing-home patients between March 8 and June 14, 2020. The primary outcome was in-hospital mortality. Risk factors were compared between those who were discharged or died using the Student t test, the Mann-Whitney U test, χ2 analysis, and logistic regression. Results: Among 169 hospitalized nursing-home patients, the case fatality rate was 43.2%. The mean age was 72.3 ± 13.8 years; 92 patients (54.4%) were male; and 112 patients (66.3%) were black. Within the first day of hospitalization, 83 (49%) patients developed fever. On admission, 24 (14.2%) patients were hypotensive. Leukopenia, lymphopenia, and thrombocytopenia were present in 20 (12%), 91 (53%), and 40 (23.7%) patients, respectively. Among the inflammatory markers, elevations in CRP and ferritin levels occurred in 79% and 24%, respectively. Intensive care admission was needed for 40 patients (23.7%). Septic shock occurred in 25 patients (14.8%). Patients over the age of 70 were more likely to die than younger patients (OR, 2.2; 95% CI, 1.2– 4.1; P = .20). Patients with a fever on admission were more likely to die than those who were afebrile (OR, 2.03; 95% CI, 1.08–3.8; P = .03). Also, 66.7% hypotensive patients died compared to 39.3% normotensive patients (OR, 3.1; 95% CI, 1.2–7.7 P = .01). Intubated patients died more often than those not intubated, 78.4% versus 33.3%, respectively (OR=7.3, p < 0.001, CI 3.1, 17.2) Factors significantly associated with death included higher mean qSofa (p < 0.001), higher median Charlson scores (0.02), thrombocytopenia (p = 0.04) and lymphocytopenia (0.04). From multivariable logistic regression, independent factors associated with death were Charlson score (OR=1.2, p=0.05), qSofa (OR=2.0, p=0.004), thrombocytopenia (OR = 3.0, p = 0.01) and BMI less than 25 (OR = 3.5, p=0.002). Conclusions: Our multivariable analysis revealed that patients with a greater burden of comorbidities, lower BMI, higher qSOFA sepsis score, and thrombocytopenia had a higher risk of death, perhaps because of severe infection despite a robust immune response.
Funding: No
Disclosures: None
Linking Staff Cases in a Hospital COVID-19 Outbreak Using Electronic Tracking Data
- Pragya Dhaubhadel, Margie Pace, Trina Augustine, Seth Hostetler, Mark Shelly
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s48
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Significant outbreaks of SARS-CoV-2 infections have occurred in healthcare personnel (HCP). We used an electronic tracking system (ETS) as a tool to link staff cases of COVID-19 in place and time during a COVID-19 outbreak in a community hospital. Methods: We identified SARS-CoV-2 infection cases through surveillance, case investigation and contact tracing, and voluntary testing. For those wearing ETS badges (Centrak), data were reviewed for places occupied by the personnel during their incubation and infectious windows. Contacts beyond 15 minutes in the same location were considered close contacts. Results: Over 6 weeks (August 10–September 14, 2020), 35 HCPs tested positive for SARS-CoV-2 by NAAT testing. In total, 18 nurses and aides were clustered on 1 hospital unit, 7 cases occurred among respiratory therapists that visited that unit, and 10 occurred in other departments. Overall, 17 individuals wore ETS badges as part of hand hygiene monitoring. ETS data established potential transmission opportunities in 17 instances, all but 2 before symptom onset or positive test result. Contacts were most often (10 of 17) in common work areas (nursing stations), with a median time of 45 minutes (IQR, 21–137). Contacts occurred within and between departments. A few COVID-19 patients were cared for in this location at the time of the outbreak. However, we did not detect HCP-to-patient nor patient-to-HCP transmission. Conclusions: Significant HCP-to-HCP transmission occurred during this outbreak based on ETS location. These events often occurred in shared work areas such as the nursing station in addition to break areas noted in other reports. ETS systems, installed for other purposes, can serve to reinforce standard epidemiology.
Funding: No
Disclosures: None
Using a Quality-Driven Approach to Maintain an N-95 Respirator Supply During a Pandemic-Driven Global Shortage
- Amy Selimos, Mark Buchanan, Lauren DiBiase, Stephen Dean, Pat Boone, Nicholas Shaheen, Emily Sickbert-Bennett Vavalle, Beth Willis
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s48-s49
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Reports of hospitals overwhelmed by COVID-19 patients created severe shortages of personal protective equipment (PPE). In this large academic medical system, we used a systematic team approach to proactively maintain an adequate PPE supply. The team consisted of staff from multiple departments including infection prevention, environmental health and safety, operational efficiency, and supply chain. The healthcare system solicited donations of PPE, and our team was tasked with developing a sustainable method to provide healthcare workers with safe and effective N-95 respirators. Respirators are normally fitted to our 6,000+ healthcare workers through a fit-testing process using 4 models of N-95s. We received >60 models, many in small quantities, posing a new level of complexity that prevented use of our typical fit-testing method. Methods: Donated respirators were manually verified on the CDC/NIOSH website to validate approval or approved alternative. A categorization system was developed, and respirators were sorted based on quality, style, and condition. User seal checks replaced qualitative fit testing due to the uncertain and quickly changing respirator supply. Staff were educated about the importance of performing a seal check to evaluate respirator fit and were provided instructions for what to do if they failed a seal check. We performed limited quantitative fit testing on a small group previously fit tested to 1 of the 4 models of N-95s normally stocked to identify the most effective alternative respirators to serve as substitute N-95s. Results: We were able to provide staff with new N-95s and delay the release of reprocessed N-95s. Overall, 18 models of respirators were tested on staff for filtration effectiveness and fit. We deemed 61% masks to be of last resort, and these were not released. We determined that 39% were acceptable as an alternative for at least 1 of our usual respirator models. However, only 3 models (17%) available in small quantities fit wearers whose size was in shortest supply. This scarcity led to the evaluation and purchase of a new respirator prototype for small N-95 wearers, which was an important success of our team’s work and for staff safety. Conclusions: Collaboration between teams from a variety of backgrounds, using both qualitative and quantitative data, resulted in a sustainable method for receiving, sorting, and evaluating donated N-95 respirators, ensuring the delivery of a steady supply of effective N-95 respirators to our staff. This quality-driven approach was an efficient and effective strategy to maintain our N-95 respirator supply during a pandemic driven global shortage.
Funding: No
Disclosures: None
Prevalence of SARS-CoV-2 Antibody in Healthcare Workers in Central Pennsylvania
- Taesung Kwon, Stacy Kenyon, Kimberly Kilheeney, Stanley Martin, Mark Shelly
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s49
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Determining the incidence of SARS-CoV-2 in healthcare workers (HCW) is important in assessing the safety of the work environment. Though of limited use in acute illness, serologic testing can detect some infections that occur undetected. We compared the prevalence of antibodies to SARS-CoV-2 to a place of work, exposure by role and department, and use of various prevention methods. Methods: Healthcare workers (HCWs) working in Geisinger Health System were offered voluntary serology through Employee Health. Before they had blood taken, they completed a brief questionnaire. Testing was conducted from June 15 to September 4, 2020. Blood was analyzed for SARS-CoV-2 immunoglobulin G (IgG) (Roche and Diasorin platforms). Results: In total, 2,295 employees and contract workers providing care at Geisinger facilities were tested. Most of this group, 2,037 (88.8%), were involved in direct patient care. In total, 101 tests returned positive, a rate of 4.4% (95% CI, 3.6%–5.3%). Of 54 HCWs with a positive NAAT for SARS-CoV-2, positive serology results were found in 48, a sensitivity of 89% (95% CI, 78%–95%). Those involved in patient care were slightly more likely to become infected, 91 of 2,037 (4.6%) compared to 10 of 258 who were not involved in patient care (3.9%; P = .68). Those with unprotected exposure to a known case of COVID-19 were more likely than those not exposed to be positive for SARS-CoV-2, 51 of 792 (6.4% vs 3.3%; P = .0008). This risk was highest for those exposed outside work (7 of 33; 21%; P = .003). HCWs working in COVID-19 units were positive at a rate of 4.0% (95% CI, 3.8%–5.4%), no more than other inpatient areas, which were 5.0% positive (95% CI, 3.8%–6.4%). HCWs working with outpatients were at slightly lower risk, 2.8% positivity (95% CI, 1.9%–4.1%). The rates of infection ranged between 3.3% and 5.0% by job category. Employees were asked about symptoms experienced since March 2020. Positive serology occurred in 39 (2.8%) of 1,414 employees who did not recall any symptoms. Symptoms related to COVID-19, except sore throat, were strongly correlated with positive serology. Conclusions: When provided a safe work environment, the risk of COVID-19 in employees is comparable to that in the surrounding communities. Persons with patient care responsibilities have an absolute risk that is marginally higher.
Funding: No
Disclosures: None


















