INTRODUCTION
Clostridium difficile infection (CDI) is considered primarily a nosocomial infection with the major risk factors being advanced age, prior hospitalization, and use of antibiotics [Reference Bartlett1]. However, recent studies suggest that up to 30% of CDI cases may be community associated [Reference Karlstrom2–Reference Søes4]. Wilcox et al. found that one third of patients with community-associated CDI (CA-CDI) had not been to a hospital or treated with antibiotics prior to CDI [Reference Wilcox5]. Other determinants for C. difficile acquisition have been explored, including the possible role of zoonotic transmission [Reference Gould and Limbago6]. PCR ribotype 078 is the predominant type found in pigs and calves, but other PCR ribotypes have also been reported in pigs and calves as well as in other animals. PCR ribotypes seem to vary between geographical areas and animal species [Reference Keessen, Gaastra and Lipman7, Reference Rodriguez-Palacios8]. C. difficile has also been isolated from retail meat, although the rate varies considerably [Reference Gould and Limbago6, Reference Rodriguez-Palacios9, Reference Von10]. C. difficile has been described in pets, including cats and dogs, but the significance of pets as a source of CDI remains unclear [Reference Keessen, Gaastra and Lipman7, Reference Borriello11, Reference Weese12]. Both hospital- and community-based studies have found proton pump inhibitors (PPIs) as a risk factor for CDI [Reference Yearsley13–Reference Dial15], although other studies report contradictory results [Reference Hensgens16–Reference Bavishi and DuPont18]. In one study contact with children aged <2 years was statistically significantly associated to CDI [Reference Wilcox5]. Little is known about the risk of secondary cases in household contacts of CDI cases. A recent study on this topic found the relative risk to be increased for a few months, but the absolute risk to be low [Reference Pépin, Gonzales and Valiquette19]. Most studies on hospital-associated CDI as well as CA-CDI exclude children aged <2 years, leaving the question of risk factors in this group unexplored. The aim of the present study was to identify risk factors for CDI in the community, in children aged <2 years and individuals aged ⩾2 years, attending their general practitioner (GP) because of diarrhoea or other gastrointestinal symptoms.
METHODS
Study design
We conducted a prospective matched case-control study. The study included patients living in two parts of Denmark with both rural and urban areas and a total population of about 722 500. These two parts of the country were each served by a public clinical microbiological laboratory. All patients that had a faecal sample submitted for routine microbiological testing after attending their GP because of diarrhoea or other gastrointestinal symptoms, were consecutively included in the study when diagnosed. All stool samples were analysed for C. difficile irrespective of the requested diagnostic test of the GP. Cases were defined as patients with a faecal sample positive for toxigenic C. difficile by culture, i.e. patients with non-toxigenic C. difficile were excluded from the analyses. Individually matched controls were randomly selected in the laboratory database of patients with a faecal sample negative for C. difficile by culture, and within 2 months of laboratory receipt date of the case. Three controls per case were invited. The control group was selected to represent individuals comparable to CDI patients and with gastrointestinal complaints leading to the same diagnostic request. Only patients with no history of a laboratory-confirmed C. difficile infection in the last 6 months prior to the inclusion date were included. Cases and controls were matched by age, gender, and site for laboratory analyses of samples. For adults and children aged ⩾5 years, the closest match within a time window of 2 years was used. For children aged <6 months, age matching was within a time window of 6 weeks. For children aged between 6 months and 4 years the time window was 5 months. The study period was from 24 August 2009 to 28 February 2011.
Microbiological analysis
Cycloserine cefoxitin fructose agar medium (SSI Diagnostica, Denmark) was used for isolation of C. difficile and agar plates were placed in an anaerobic atmosphere (86% N2, 7% H2, 7% CO2) at 37°C for 48 h.
Salmonella spp., Campylobacter spp., Shigella spp., Yersinia enterocolitica, Aeromonas spp., Plesiomonas spp., Vibrio spp. were isolated by standard culturing procedures. Enteropathogenic (EPEC), enterotoxigenic (ETEC), shiga toxin-producing (STEC), enteroinvasive (EIEC) and attaching and effacing (A/EEC; no EPEC serotypes) Escherichia coli were identified by standard methods [Reference Blom20–Reference Persson22].
Gastroenteritis viruses were detected by real-time reverse transcription–polymerase chain reaction (rRT–PCR) using the OneStep Qiagen rRT–PCR kit (Qiagen Nordic, Denmark) and primers and probes for norovirus genogroups I and II [Reference Kageyama23], sapovirus [Reference Johnsen, Midgley and Böttiger24], rotavirus [Reference Pang25], adenovirus [Reference Logan, O'Leary and O'Sullivan26], and astrovirus [Reference Logan, O'Leary and O'Sullivan27].
Giardia intestinalis, Cryptosporidium sp., Entamoeba histolytica, E. dispar and Dientamoeba fragilis were detected by real-time PCR [Reference Stensvold and Nielsen28].
The toxin gene profile of colonies of C. difficile was identified using a 5-plex PCR directed towards tcdA, tcdB, cdtA, cdtB and 16S rDNA [Reference Persson, Torpdahl and Olsen29]. PCR ribotyping was performed, with minor modifications to the methods described previously by O'Neill et al. and Stubbs et al. [Reference O'Neill30, Reference Stubbs31].
The bacteria (except for A/EEC), virus and parasites (except for D. fragilis and E. dispar) mentioned above were defined as generally accepted enteropathogens. D. fragilis and A/EEC were defined as candidate pathogens and E. dispar as non-pathogenic.
Case definitions
Diarrhoea was defined as >3 daily unformed stools (or for children using diapers more changes of diaper than usual) and the patient's own experience of going to the lavatory more often than usual. CDI cases were defined as patients with diarrhoea or other gastrointestinal symptoms and positive by culture for toxigenic C. difficile. Origin of CDI was classified as CA-CDI in cases either without discharge from a healthcare facility (HCF) at least 12 weeks prior to onset of symptoms, or with onset of symptoms within 48 h following admission to a HCF and still without discharge from a HCF within 12 weeks before onset of symptoms. Healthcare-associated CDI (HCA-CDI) was defined as onset of symptoms at least 48 h after admission to a HCF or onset of symptoms within 4 weeks following discharge from a HCF. CDI of unknown origin was defined as onset of symptoms 4–12 weeks after discharge from a HCF. A HCF was defined as a hospital or a nursing home with skilled nursing care [Reference Kuijper, Coignard and Tull32, Reference McDonald33].
Composition of questionnaire
Patients who fulfilled the inclusion criteria received a questionnaire by mail regarding clinical presentation of gastroenteritis and comorbidities, admission to hospital, contact with outpatient clinic and childbirth within the last 6 months prior to onset of symptoms, use of non-prescription medicine, food consumption and travelling abroad within 3 months prior to onset, contact with animals in the 2 weeks before infection, occupational exposures including working in hospitals, nursing homes, schools and nurseries, contact with children aged <2 years and contact with hospitalized individuals or nursing home residents.
The questions concerning underlying illness covered the following conditions: inflammatory bowel disease, diabetes mellitus, pre-existing renal failure and/or regular dialysis, cardiovascular disease, chronic obstructive pulmonary disease, metabolic disorders, rheumatoid arthritis, haematological malignancies, malignant solid tumours, and abdominal surgery within the 2 two months before infection.
Questions regarding food consumption included consumption of beef, pork, lamb, poultry, fish, eggs, cabbage, and other vegetables, fruit, soft drinks, sweets and pastry, milk products (both pasteurized and unpasteurized), and fermented milk products.
Contact with animals included contact with dogs, cats, guinea pigs, rabbits, horses, cattle, pigs, ‘other domesticated animals’, and ‘other pets’.
Data on prescription medication for systemic use were obtained for each individual patient from the Danish Medicines Agency which maintains a national registry of all prescriptions. According to Dial et al. and Kelly the risk of CDI related to antibiotic exposure declines significantly after 6–8 weeks [Reference Dial34, Reference Kelly35]. On this basis antibiotics prescribed within 56 days prior to onset of symptoms were included in the analysis. For all other drugs the prescription date was within 120 days prior to symptom onset.
Statistical analysis
The patients were divided into two age groups (<2 years and ⩾2 years). All analyses were performed in these two age groups separately.
For matched univariable and multivariable analyses conditional logistic regression was used. Main analyses were performed with exposure variables obtained from the questionnaires and supplemented with an extended analysis of the additional data of prescription medication. Covariates statistically significant at the 90% level in the univariable analyses were included in the multivariable analysis. The outcome variables in the multivariable analysis of the medication data were adjusted for prior hospitalization and beef consumption. Test for interactions and multiple imputations of missing values were performed in the main effects model.
Additional analyses were performed in two different subgroups. In one subgroup analysis the definition of gastroenteritis was narrowed to include only cases and controls with diarrhoea. In the other subgroup analysis cases with C. difficile as the only enteropathogen in their faecal sample were included together with controls that had no generally accepted enteropathogens in their faecal sample.
Odds ratios (OR) were expressed with 95% confidence intervals (95% CI). All analyses were performed using SAS, version 9.1 (SAS Institute Inc., USA) and Stata v. 11 (StataCorp., USA).
Ethical considerations
The study was approved by the Danish Data Protection Agency (journal number: 2009-41-3534). Exemption for review by the ethical committee system was obtained from the Committee on Biomedical Research Ethics for Capital Region (protocol number: H-C-FSP-2009-006). A signed statement of consent to review data of prescription medication was obtained from all included patients or their parents if patients were aged <18 years.
RESULTS
Demographics and origin of infection
In total, 1559 patients were invited to participate. The response rate was 61% for C. difficile-positive patients (patients with non-toxigenic C. difficile and cases) and 46% for controls, i.e. 259 cases and 455 controls completed the questionnaire and were included in the study. Forty-seven per cent (121/259) of the cases were aged <2 years (Table 1). Age ranged from 13 weeks to 94 years. Table 1 shows the classification of the origin of infection. Following exclusion of non-toxigenic strains, 110 (96%) of 115 patients aged <2 years were CA-CDI and none were HCA-CDI. In 130 patients aged ⩾2 years 90 (69%) were CA-CDI and 31 (24%) were HCA-CDI.
Table 1. Demographic characteristics, origin of infection, toxin gene profile of Clostridium difficile of patients attending general practice because of gastrointestinal symptoms, Denmark, 2009–2011
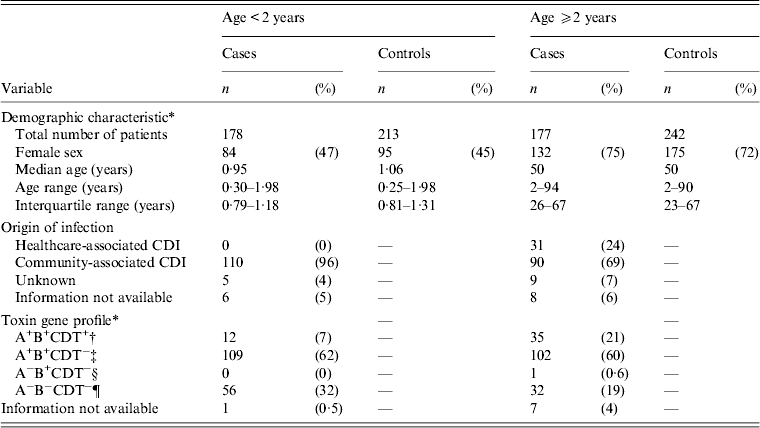
CDI, Clostridium difficile infection.
* Includes both patients with non-toxigenic and toxigenic C. difficile strains.
† A+B+CDT+, strains with toxin gene profile: tcdA+, tcdB+, cdtA+/cdtB+.
‡ A+B+CDT−, strains with toxin gene profile: tcdA+, tcdB+, cdtA−/cdtB−.
§ A−B+CDT−, strains with toxin gene profile: tcdA−, tcdB+, cdtA−/cdtB−.
¶ A−B−CDT−, non-toxigenic strains: tcdA−, tcdB−, cdtA−/cdtB−.
Co-pathogens
Faecal samples from all 810 patients were accessible for analysis of enteropathogenic bacteria, 771 (95%) for viral and 765 (94%) for parasitic analysis. Table 2 gives the numbers of co-pathogens by age group. Concomitant microorganisms, considered as generally accepted gastrointestinal pathogens, were found in 62 (53%) cases and 120 (60%) controls aged <2 years, with viruses accounting for the majority of co-pathogens, and in 30 (23%) cases and 60 (26%) controls aged ⩾2 years.
Table 2. Enteropathogens other than Clostridium difficile in the faecal sample in patients attending general practice because of gastrointestinal symptoms, Denmark, 2009–2011

EPEC, Enteropathogenic Escherichia coli; ETEC, enterotoxigenic E. coli; EIEC, enteroinvasive E. coli; A/EEC, attaching and effacing E. coli.
Risk factor analysis
Antibiotic treatment and hospitalization prior to infection were statistically significantly associated with CDI in patients aged ⩾2 years, but not in those aged <2 years (Table 3). Exclusion of children aged <2 years with a viral aetiology of diarrhoea did not result in any substantial difference in the model (data not shown).
Table 3. Univariable matched analysis of risk factors for Clostridium difficile infection on the basis of selected variables from the questionnaire from patients attending general practice because of gastrointestinal symptoms, Denmark, 2009–2011
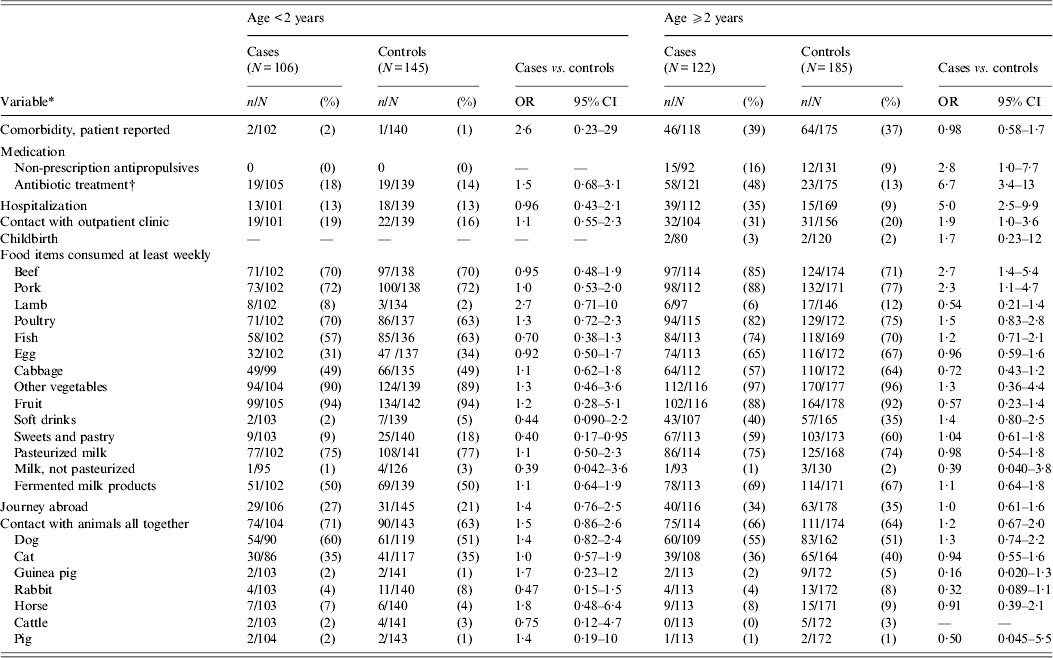
OR, Odds ratio; CI, confidence interval. All cases with toxigenic C. difficile.
* Prior to infection.
† Data from Danish Medicines Agency.
The final multivariable model for risk factors in the group of patients aged ⩾2 years contained hospitalization, use of antibiotics and consumption of beef at least weekly prior to infection (Table 4). When subdividing antibiotics, use of phenoxymethylpenicillin, dicloxacillin, and penicillins with extended spectrum remained statistically significant in the multivariable model (Table 5). No interactions between covariates were found. Multiple imputations on missing variables were performed (about 10% of patients had missing information in one or more variable) in the main multivariable analysis based on questionnaire data as well as in the supplementary analysis based on medication data. In the supplementary analysis prior hospitalization and consumption of beef were included at first, in order to adjust for these variables and consumption of beef was subsequently excluded from the model because of a P value = 0·059. Otherwise multiple imputations did not change the results in the multivariable analyses substantially and was therefore not used for the results given. Consumption of beef was still associated with CDI in patients aged ⩾2 years, when multivariable analysis was performed in the two different subgroups counting (a) only patients with diarrhoea and (b) only patients with no generally accepted enteropathogens apart from C. difficile (OR 4·5, 95% CI 1·5–14 and OR 4·6, 95% CI 1·0–20, respectively). Forty different PCR ribotypes were represented in the group of beef consumers. Of these, besides type 078, there were also types 001, 012, 014/020/077, 015, 017, 023, 027, 029, 066, 070, 106/117, and 107 in addition to a series of minor PCR ribotypes.
Table 4. Multivariable matched analysis of risk factors for Clostridium difficile infection on the basis of data from the questionnaire. Patients aged ⩾2 years attending general practice because of gastrointestinal symptoms, Denmark, 2009–2011
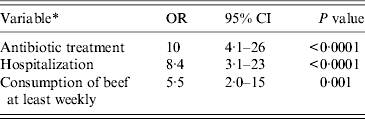
OR, Odds ratio; CI, confidence interval.
All cases with toxigenic C. difficile.
* Prior to infection.
Table 5. Multivariable matched analysis of risk factors for Clostridium difficile infection on the basis of data of prescription medications. Patients aged ⩾2 years attending general practice because of gastrointestinal symptoms, Denmark, 2009–2011
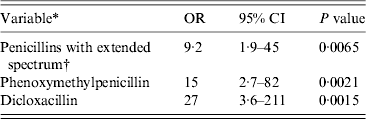
OR, Odds ratio; CI, confidence interval.
All cases with toxigenic C. difficile.
Variables adjusted for hospitalization and consumption of beef at least weekly.
* Prior to infection.
† Contains pivmecillinam, (piv)ampicillin, amoxicillin and amoxicillin/clavulanic acid.
Four per cent of cases aged ⩾2 years received a fluoroquinolone before onset of symptoms. Only one case received moxifloxacin (Table 6). No statistically significant difference was found between cases and controls regarding use of fluoroquinolones. Clindamycin use prior to onset of symptoms was found in 6% of cases in contrast to controls, where none had clindamycin prescribed. Metronidazole was positively associated with CDI in the univariable analysis, but 13/17 (76%) of these patients also received another antibiotic with the most common indication for prescription being ‘infection’. PPIs prior to infection were not found to be associated with CDI (Table 6). Non-steroid anti-inflammatory drugs (NSAIDs) other than acetylsalicylic acid (ASA) were significantly associated with CDI in patients aged ⩾2 years (Table 6). No association was found when ASA was included in NSAIDs (OR 0·7, 95% CI 0·3–1·4).
Table 6. Univariable matched analysis of prescription medications as risk factors for Clostridium difficile infection. Patients aged ⩾2 years attending general practice because of gastrointestinal symptoms, Denmark, 2009–2011
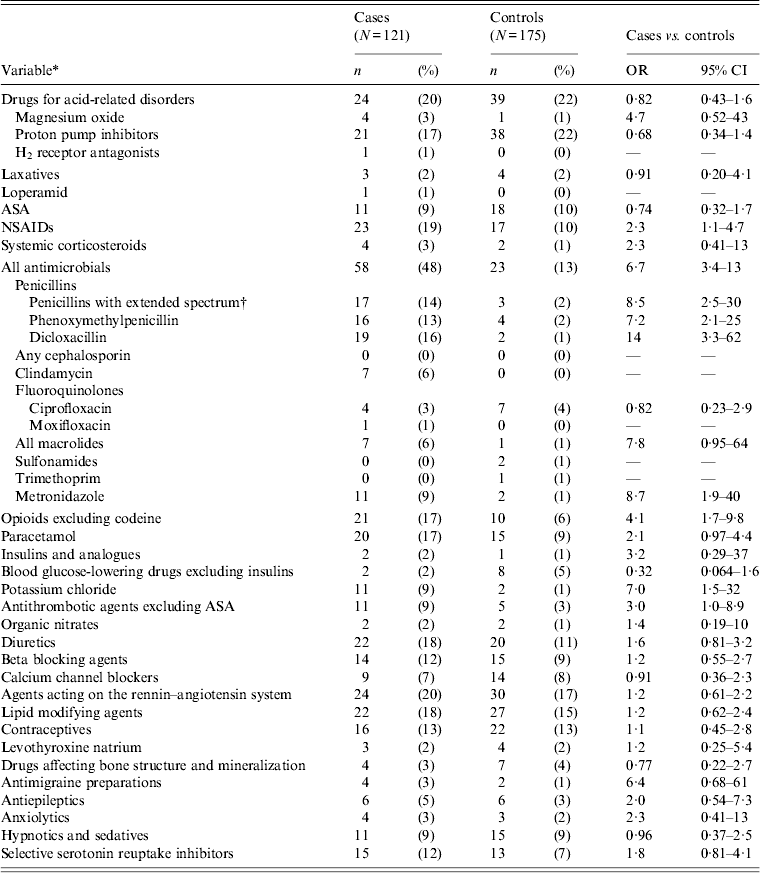
OR, Odds ratio; CI, confidence interval; ASA, acetylsalicylic acid; NSAIDs, non-steroid anti-inflammatory drugs.
All cases with toxigenic C. difficile.
* Antibiotics, except for trimethoprim, prescribed within 56 days prior to infection. Other drugs and trimethoprim prescribed within 120 days prior to infection.
† Contains pivmecillinam, (piv)ampicillin, amoxicillin and amoxicillin/clavulanic acid.
In the group of patients aged <2 years, only a few received prescription medication including antibiotics (Table 7).
Table 7. Univariable matched analysis of prescription medications as risk factors for Clostridium difficile infection. Patients <2 years of age attending general practice because of gastrointestinal symptoms, Denmark, 2009–2011
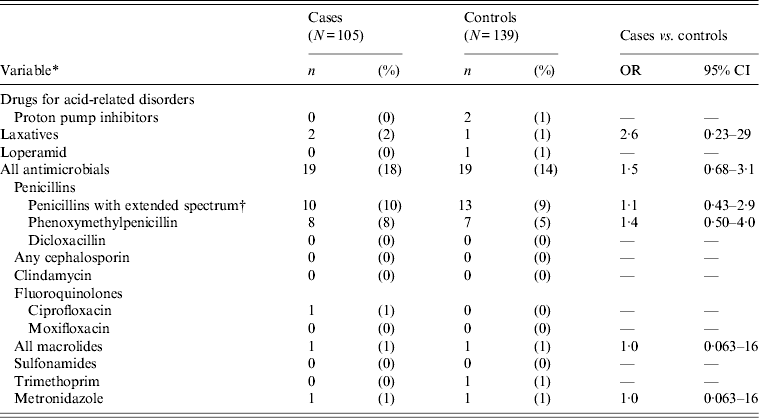
OR, Odds ratio; CI, confidence interval.
All cases with toxigenic C. difficile.
* Antibiotics, except for trimethoprim, prescribed within 56 days prior to infection. Other drugs and trimethoprim prescribed within 120 days prior to infection.
† Contains pivmecillinam, (piv)ampicillin, amoxicillin and amoxicillin/clavulanic acid.
Contact with animals was significantly associated with CDI in multivariable analysis (OR 8·1, 95% CI 1·0–64) when the group of patients aged <2 years, was set to include only cases and controls without other generally accepted enteropathogens than C. difficile.
Patients aged ⩾15 years were asked about contact with children aged <2 years, this covariate was not associated with CDI (OR 0·5, 95% CI 0·2–1·4). Neither was contact with patients admitted to hospital (OR 0·5, 95% CI 0·1–2·3) or nursing home residents (OR 1·6, 95% CI 0·7–3·6) nor occupational exposure to hospital patients (OR 0·8, 95% CI 0·2–3·3) or nursing home residents/citizens receiving skilled nursing care in their own homes (OR 2·2, 95% CI 0·6–8·1).
Patients were asked about antibiotic use in the 3 months prior to onset of symptoms. When these data were combined with the data from the Danish Medicines Agency, 24% (31/131) of cases aged ⩾2 years were neither admitted to hospital in the 6 months prior to onset of infection nor did they have any antibiotics in the 3 months prior to onset of symptoms. Notably, in the subgroup of cases aged ⩾2 years with no other enteropathogens but C. difficile in the faecal sample (97 cases), this figure fell to 16%.
Of the 259 cases with toxigenic C. difficile and the 455 controls, information of stool consistency was accessible in 255 cases and 447 controls. Of these 12 (5%) cases and 39 (9%) controls did not fulfil the diarrhoea definition, but reported other gastrointestinal symptoms such as stomach ache, nausea, weight loss or bloody faeces. The results of the univariable analyses changed only slightly when these patients were excluded (data not shown) and they were therefore maintained in the main analyses.
DISCUSSION
Narrow-spectrum antibiotics, including phenoxymethylpenicillin and dicloxacillin were identified as risk factors for CDI. This is in line with two other studies that found aminopenicillins and penicillins to be associated with CDI [Reference Wilcox5, Reference Kuntz14]. In a setting with preferential use of narrow-spectrum antibiotics, as for GPs in Denmark, it is important to be aware of the risk of CDI imposed by narrow-spectrum antibiotics. Conflicting results have been obtained in studies comparing patients with CA-CDI to individuals with or without diarrhoea and no CDI when analysing the role of PPIs as a risk factor [Reference Wilcox5, Reference Kuntz14–Reference Dial15, Reference Naggie17, Reference Kutty36]. In our study 18% and 21% of cases and controls, respectively, had been treated with PPIs. Use of PPIs in the background population was 85/1000 inhabitants in Denmark in 2010. The high frequency of individuals taking PPIs in both cases and controls reflects that use of PPIs may increase the risk of acquiring CDI as well as gastroenteritis caused by other enteropathogens and is in line with a recent study by Bavishi & DuPont [Reference Bavishi and DuPont18].
We found a marked difference in risk factor profile between cases aged <2 years and older cases. Hospitalization and use of antibiotics prior to onset of symptoms were associated with CDI in individuals aged ⩾2 years, but not in children aged <2 years. The existence of CDI in early childhood is under debate, and on this basis most studies exclude children aged <2 years. Many investigators regard toxigenic C. difficile as non-pathogenic in children aged <2 years, and high carriage rates are described in asymptomatic neonates [Reference Holst, Helin and Mardh37–Reference Viscidi, Willey and Bartlett39]. On the other hand, other studies have reported CDI in children aged <2 years [Reference Kim40–Reference Turco42] and therefore we decided to include this age group in our investigation. In contrast with older patients, we could not identify any meaningful predictors of CDI in the main analyses in this age group which indicates that the epidemiology of CDI in young children is distinct from individuals aged ⩾2 years. It might be considered whether the observed differences in risk factors for CDI between children aged <2 years and older patients aged ⩾2 years is due to a higher frequency of viral infections in the former group. However, excluding cases with concomitant virus from the analyses did not change results (data not shown), and the absence of risk factors cannot be ascribed to differences in frequencies of viral infections. Although, in a subgroup of patients aged <2 years, including only cases and controls without other generally accepted enteropathogens than C. difficile contact with animals was significantly associated with CDI in multivariable analysis. The finding could indicate contact with animals as a risk factor for CDI or for colonization with C. difficile. It can be speculated that infants' unreservedly close contact with animals is the reason for this association in just this age group and that exclusion of cases and controls with other enteropathogens than C. difficile in the faecal sample tends to strengthen the association since other enteropathogens, e.g. Campylobacter jejuni isolates from humans and pets have been demonstrated to be closely related by molecular typing [Reference Damborg43]. However, our result is hard to interpret since many different animals (both household pets and domestic animals) were included in the analysis. C. difficile has been described in household pets, including cats and dogs. Borriello et al. found that C. difficile was present in 23% of stool samples from the household pets investigated, including 49 dogs, 19 cats and one duck. Most strains were non-toxigenic [Reference Borriello11]. Weese et al. found no evidence of dogs being a significant source of household C. difficile contamination [Reference Weese12]. More studies are needed to outline this topic.
Contact with children aged <2 years has been significantly associated with CDI in one study [Reference Wilcox5]. Our study could not confirm this finding. Furthermore, we found no association between exposure to patients admitted to hospital or nursing home residents and CDI.
Consumption of beef was significantly associated with CDI in patients aged ⩾2 years. C. difficile can be detected in various farm animals including calves and pigs and it has been suggested that CDI could be a zoonosis [Reference Gould and Limbago6, Reference Rupnik44]. The bacterium has also been isolated from retail meat, although the rate varies considerably [Reference Gould and Limbago6, Reference Rodriguez-Palacios9–Reference Von10]. PCR ribotype 078 is the predominant type in pigs and calves in some studies, but PCR ribotypes seem to vary between geographical areas and animal species [Reference Keessen, Gaastra and Lipman7, Reference Rodriguez-Palacios8, Reference Rupnik44, Reference Keel45]. PCR ribotype 078 is also found in humans with CDI [Reference Goorhuis46]. We found PCR ribotype 078 in cases reporting beef consumption (four of the five type 078 cases consumed beef). However, many other PCR ribotypes of C. difficile were present in beef eaters as well, and distinct PCR ribotypes did not indicate beef per se as a source of infection. Multiple imputations on missing variables formally resulted in exclusion of beef from the model in the supplementary analysis based on medication data (P = 0·059), although it is possible that the association would have been statistically significant if more patients had been included in the study. The association of beef consumption with CDI suggests that a proportion of CDI cases are associated with beef consumption. Alternatively, theoretically the selection of controls with non-CDI diarrhoea may have introduced a bias if other food items than beef, e.g. poultry or pork caused the diarrhoea of the controls and they therefore a priori had a lower consumption of beef than the cases and the background population. However, we do not believe that this would explain the association noted here and further studies are needed to clarify whether there is a causal relationship between beef consumption and CDI.
In the group of cases aged ⩾2 years, 24% were neither admitted to hospital in the 6 months prior to onset of infection nor were they prescribed antibiotics in the 3 months prior to onset of symptoms when prescription medication data from the Danish Medicines Agency were combined with data from the questionnaire. The percentage fell to 16% when only cases with C. difficile as the only pathogen in the faecal sample were included. In a study by Wilcox et al., 35% of CDI cases neither had exposure to antibiotics nor hospitalization 4 weeks and 6 month prior to infection, respectively. Faecal samples were examined for Salmonella spp., Shigella spp., Campylobacter spp., and E. coli O157, but for no other diarrhoegenic E. coli or viruses [Reference Wilcox5]. In our study viral and parasitic analyses were performed in addition to analysis for bacterial enteropathogens. The more extended examination for enteropathogens together with the longer period of exposure to antibiotics (3 month vs. 4 weeks) prior to infection could be one of the explanations for the differences in the percentage of cases without hospitalization and use of antibiotics prior to infection in the two studies.
We found 69% of cases aged ⩾2 years to be CA-CDI. Only CDI patients who had a faecal sample submitted by their GP were included in the study. Therefore, the CDI patients in the community who were admitted directly to hospital without consulting their GP, e.g. because of severe diarrhoea, are missing from the study, leading to a possible lower proportion of community-associated cases in the study.
The study has some limitations. A matched case-control study was chosen to take possible confounding into account. The resulting loss of power may have masked other possible significant associations. The choice of gastrointestinal patients as control group was chosen to better detect C. difficile-specific predictors, but this may, on the other hand, have obscured risk factors for CDI that were in common with other infectious gastrointestinal diseases; thus it is possible that the role of PPIs may have been masked, as this will also be a risk factor for other bacterial gastrointestinal infections. The response rate was 61% and 46% for C. difficile-positive patients and controls, respectively. This should be borne in mind in terms of generalizability of the study. Still, the study population was ∼722 500 (∼13% of total population in Denmark) and included patients living in both rural and urban areas. Non-responder cases had a higher median age (59 vs. 50 years) and a higher 30-day mortality (3·6% vs. null per cent) compared to responders, indicating that non-responder cases might have had more severe CDI and/or a higher frequency of comorbidities compared to responders. For controls 30-day mortality was null per cent and 0·4% for responders and non-responders, respectively.
To conclude, our results emphasize prior hospitalization and intake of narrow-spectrum antibiotics as important predictors of CDI in the community in individuals aged ⩾2 years, but not in children aged <2 years, and suggest that a proportion of CDI cases are associated with other risk factors. The study indicates beef consumption and infants' contacts with animals as other possible risk factors. Narrow-spectrum antibiotics are the drug of choice of GPs in Denmark. This study emphasizes the importance of not neglecting the risk of CDI imposed by these antibiotics not only in the hospital setting, but also in the community.
ACKNOWLEDGEMENTS
We thank all laboratory technicians that participated in stool sample analyses at Statens Serum Institut, Copenhagen and Odense University Hospital, Odense.
DECLARATION OF INTEREST
None.








