Estimates suggest that those with serious mental illness are now dying approximately 25 years earlier than the general population and that up to 60% of premature deaths in those with serious mental illness are as a result of general medical conditions. Reference Parks, Svendsen, Singer, Foti and Mauer1 Studies have shown that not only do patients with mood disorders have more comorbid medical illnesses than mentally healthy individuals, Reference Kilbourne, Cornelius, Han, Pincus, Shad and Salloum2-Reference Ramasubbu, Beaulieu, Taylor, Schaffer and McIntyre4 but also a greater medical illness burden (a greater number of medical illnesses) seems to be associated with a more severe clinical presentation of the mood illness. Reference Beyer, Kuchibhatla, Gersing and Krishnan5 Traditionally, the high prevalence of medical illness in those with mental health problems has been viewed as a consequence of psychotropic medications and an unhealthy lifestyle. Reference Kupfer6 However, recent research has suggested that exposure to psychotropic medication does not worsen mortality risk in patients with psychiatric illness Reference Khan, Faucett, Morrison and Brown7 and that there may be underlying biological mechanisms linking mood disorder and many medical illnesses. Reference Rzhetsky, Wajngurt, Park and Zheng8,Reference Evans, Charney, Lewis, Golden, Gorman and Krishnan9
We have previously described the rates of physical disorders in large samples of patients with recurrent depression (n = 1547) and psychiatrically healthy controls (n = 884). Reference Farmer, Korszun, Owen, Craddock, Jones and Jones10 The current study will examine the rates of physical disorders in a large, well-defined, sample of patients with bipolar affective disorder. These patients were recruited and assessed using the same standardised procedures as our recurrent depression and control participants mentioned above. This will allow us to directly compare the rates of each physical illness in our newly recruited bipolar sample with our previously described recurrent depression and control samples. In addition, the current study will investigate whether the presence of medical illness in individuals with bipolar disorder is associated with a more severe bipolar illness course. This is the first study in a UK clinical population to assess rates of physical illnesses in patients with a diagnosis of bipolar disorder and make direct comparisons with unipolar and control samples.
Method
Participants
Participants were recruited at three UK sites (Birmingham, Cardiff and London) as part of ongoing molecular genetic and clinical studies of affective disorders. Reference Jones, Scott, Haque, Gordon-Smith, Heron and Caesar11,Reference Cohen-Woods, Craig, Gaysina, Gray, Gunasinghe and Craddock12 Individuals meeting DSM-IV 13 and ICD-10 14 criteria for bipolar disorder (n = 1720) were included in the study. All participants were aged 18 years or older and, because they were recruited for molecular genetic studies, they were required to be of White European ethnicity. Individuals were excluded if they: (a) had a lifetime diagnosis of intravenous drug dependency; (b) had only experienced affective illness as a result of alcohol or substance dependence; (c) had only experienced affective illness secondary to medical illness or medication; or (d) were biologically related to another study participant. After complete description of the study to participants, written informed consent was obtained.
Study assessment
Participants were interviewed using the Schedules for Clinical Assessment in Neuropsychiatry (SCAN). Reference Wing, Babor, Brugha, Burke, Cooper and Giel15 Psychiatric and family practice case-notes were reviewed for the majority of participants. Based on the SCAN interview information, and where available the case-note information, lifetime diagnoses were made according to DSM-IV and ICD-10 and ratings were made for key clinical variables (for example, age at onset, presence of psychosis). Where there was doubt regarding the diagnostic and clinical ratings the case was rated independently by two research psychologists/psychiatrists and consensus was reached. Regular interrater reliability meetings were held within and across the three sites to ensure consistency in diagnostic and ratings procedures.
Interrater reliability was formally assessed using joint ratings for a subset of 20 participants with a range of mood disorder diagnoses. Mean overall kappa statistics of 0.85 and 0.83 were obtained for DSM-IV and ICD-0 diagnoses, respectively. Mean kappa statistics and intraclass correlation coefficients (ICCs) for other key clinical variables ranged from 0.81 to 0.99 and 0.85 to 0.97, respectively.
In order to establish the lifetime presence of physical health disorders, a short self-report questionnaire was completed by participants asking whether they had ever been told by a health professional that they have any of the following 20 health problems: asthma, cancer, diabetes type 1, diabetes type 2, elevated lipids/high cholesterol, epilepsy, gastric ulcers, heart disease, hypertension, kidney disease, liver disease, memory loss/dementia, migraine headaches, multiple sclerosis, osteoarthritis, osteoporosis, Parkinson’s disease, rheumatoid arthritis, stroke, thyroid disease. Responses were scored as ‘yes’, ‘no’ or ‘uncertain’. Where an individual was coded ‘uncertain’ for a particular illness, they were excluded from analyses for that medical illness. The study received all necessary Multi-Region and Local Research Ethics Committee (MREC and LREC) approval.
The unipolar and control samples used as comparison groups have previously been described in Farmer et al. Reference Farmer, Korszun, Owen, Craddock, Jones and Jones10 In brief, participants meeting DSM-IV and ICD-10 criteria for recurrent major depression (unipolar depression: n = 1737) were recruited and assessed (as part of the Depression Case Control DeCC study) using the procedures and assessments described above for the bipolar sample. In addition, individuals in the unipolar group were excluded if they (a) had a first- or second-degree relative with a diagnosis of bipolar affective disorder, schizophrenia, schizotypal disorder, delusional disorder, acute and transient psychotic disorders or schizoaffective disorder, or (b) had ever experienced mood incongruent psychosis or psychosis outside of mood episodes.
The control group (n = 1340) were recruited to the London site via the Medical Research Council (MRC) general practice research framework (n = 846) or were staff or student volunteers of King’s College London (n = 494). Individuals were included if they were 18 years or older and were of White European origin. Control participants recruited via the MRC general practice research framework were screened using the Sham et al Reference Sham, Sterne, Purcell, Cherny, Webster and Rijsdijk16 composite index (G) of depressive and anxiety symptoms and then telephone interviewed using the Past History Schedule (PHS) Reference McGuffin, Katz and Aldrich17 to screen for lifetime absence of psychiatric disorder. The volunteers from King’s College London were interviewed in person and screened using the PHS (n = 494).
The control group also completed the short self-report medical illness questionnaire described above.
Data analysis
Rates of medical illnesses across the bipolar, unipolar and control groups.
Sociodemographic characteristics were compared between the bipolar, unipolar and control groups using chi-squared tests for categorical variables and Kruskal-Wallis tests for non-parametric continuous variables. Group comparisons of the lifetime prevalence of each of the different medical conditions were performed using chi-squared tests and pair-wise binary logistic regression analyses for each medical condition, with diagnostic group as the outcome variable. As there were significant gender differences between the three groups, gender was also entered into the logistic regression models as a cofactor. Since medical disorders occur more frequently with increasing age and there were significant differences in age at interview between the groups, age at interview was also included as a cofactor.
We used the Bonferroni method to conservatively correct for multiple testing. As there were three groups with 20 medical illnesses (3×20 = 60), P-values from the logistic regression analyses were multiplied by 60.
Rates of medical illness according to bipolar subtype
We then compared the lifetime prevalence of each of the different medical conditions in the bipolar I disorder (bipolar I) group and the bipolar II disorder (bipolar II) group using chi-squared tests and logistic regression. Again, gender and age were included as cofactors in the logistic regression models and the Bonferroni method was used to correct for multiple testing. As we were comparing 20 medical illnesses, P-values from the logistic regression analyses were multiplied by 20.
Clinical characteristics of bipolar disorder according to medical illness burden
For a subsample of the bipolar group (n = 1216), detailed clinical information regarding the course and clinical features of the bipolar illness was available. As previous studies have shown that a higher burden of medical illness is associated with a more severe illness course in bipolar disorder, we compared sociodemographic and clinical features in those bipolar participants with a high medical illness burden (history of three or more medical illnesses (bipolar+3M)) to those with no history of medical illness (bipolar+0M) using chi-squared tests for categorical variables and Mann-Whitney U-tests for non-parametric continuous variables. A cut-off of three or more medical illnesses was selected as previous studies have shown that patients with multiple conditions (defined as three or more medical conditions) are significantly more likely to have a mental illness Reference Dworkin, Von Korff and LeResche18 and that, when focusing on the number of either medical or psychiatric illnesses, the lifetime presence of three or more conditions is associated with increased disability, lower life expectancy and increased health spending. Reference Joyce, Keeler, Shang and Goldman19
A binary logistic regression analysis was carried out with group status (bipolar+3M/bipolar+0M) as the outcome variable. Gender, age at interview and the clinical features that were significantly different between the two groups (P<0.05) in the univariate analyses were entered as cofactors to establish which clinical features best predicted medical illness burden group status. All analyses were undertaken using the Statistical Procedures for the Social Sciences (SPSS) version 20 for Windows.
Results
The control group (n = 1340) was significantly younger at interview compared with the bipolar (n = 1720) and unipolar (n = 1737) groups (bipolar, unipolar>control: P<0.0001). The median age at interview (and range) in years for the bipolar, unipolar, and control groups was 47 (18-84), 47 (18-85) and 44 (18-91), respectively. The proportion of women was higher in the bipolar and unipolar groups compared with the control group (bipolar, unipolar>control: P<0.0001), with women comprising 70%, 71% and 57% of the bipolar, unipolar and control groups respectively.
Rates of medical illnesses across the bipolar, unipolar and control groups
The lifetime rates of self-reported medical illnesses in the bipolar, unipolar and control groups are shown in Table 1. Significant differences between groups were found in the lifetime prevalence of all medical illnesses with the exception of type 1 diabetes.
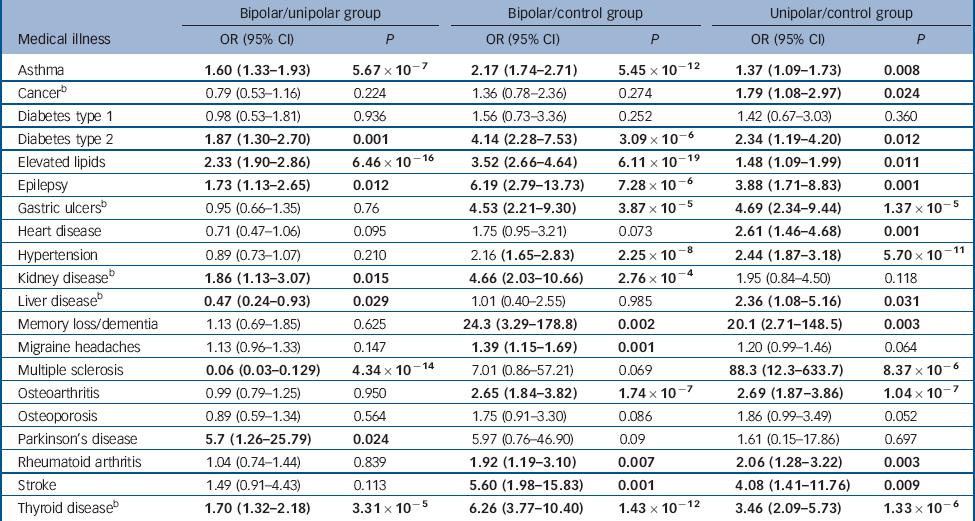
To examine differences in the rate of each medical illness between the three groups, accounting for gender and age at interview, binary logistic regression was carried out for all medical illnesses showing a significant (P<0.05) between-group difference in the univariate analyses. Diagnostic group was entered as the outcome variable and presence/absence of the medical disorder, gender and age at interview were entered as cofactors. Odds ratios, 95% confidence intervals and P-values for each logistic regression analysis are shown in Table 2. Following correction for multiple testing, using the Bonferroni method, statistically significant differences in the rates of ten medical illnesses remained between the three groups (Fig. 1).
Table 1 Lifetime rates (%) of self-reported medical illnesses in bipolar, unipolar and control groups
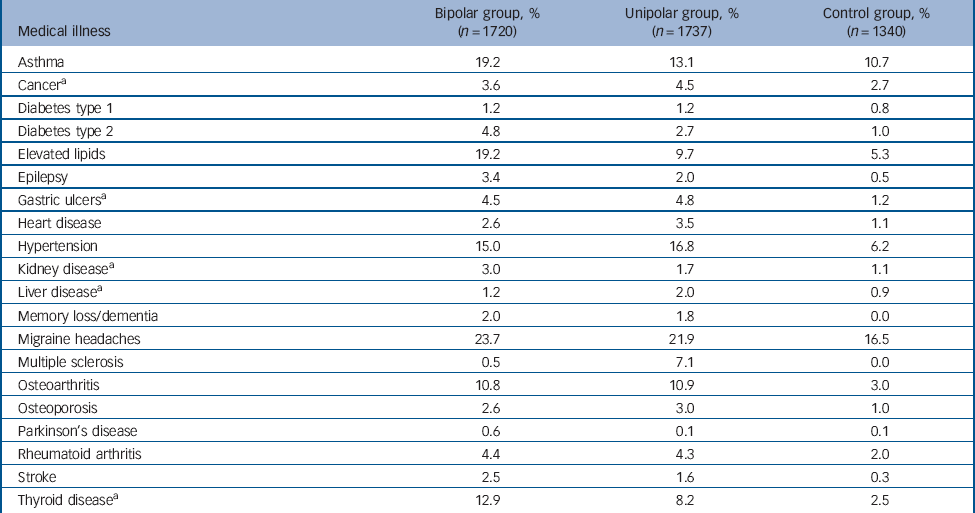
| Medical illness | Bipolar group, % (n = 1720) |
Unipolar group, % (n = 1737) |
Control group, % (n = 1340) |
|---|---|---|---|
| Asthma | 19.2 | 13.1 | 10.7 |
| CancerFootnote a | 3.6 | 4.5 | 2.7 |
| Diabetes type 1 | 1.2 | 1.2 | 0.8 |
| Diabetes type 2 | 4.8 | 2.7 | 1.0 |
| Elevated lipids | 19.2 | 9.7 | 5.3 |
| Epilepsy | 3.4 | 2.0 | 0.5 |
| Gastric ulcersFootnote a | 4.5 | 4.8 | 1.2 |
| Heart disease | 2.6 | 3.5 | 1.1 |
| Hypertension | 15.0 | 16.8 | 6.2 |
| Kidney diseaseFootnote a | 3.0 | 1.7 | 1.1 |
| Liver diseaseFootnote a | 1.2 | 2.0 | 0.9 |
| Memory loss/dementia | 2.0 | 1.8 | 0.0 |
| Migraine headaches | 23.7 | 21.9 | 16.5 |
| Multiple sclerosis | 0.5 | 7.1 | 0.0 |
| Osteoarthritis | 10.8 | 10.9 | 3.0 |
| Osteoporosis | 2.6 | 3.0 | 1.0 |
| Parkinson’s disease | 0.6 | 0.1 | 0.1 |
| Rheumatoid arthritis | 4.4 | 4.3 | 2.0 |
| Stroke | 2.5 | 1.6 | 0.3 |
| Thyroid diseaseFootnote a | 12.9 | 8.2 | 2.5 |
a. Data only available for a subset of the bipolar disorder group (n = 1216).
Rates of medical illness according to bipolar subtype
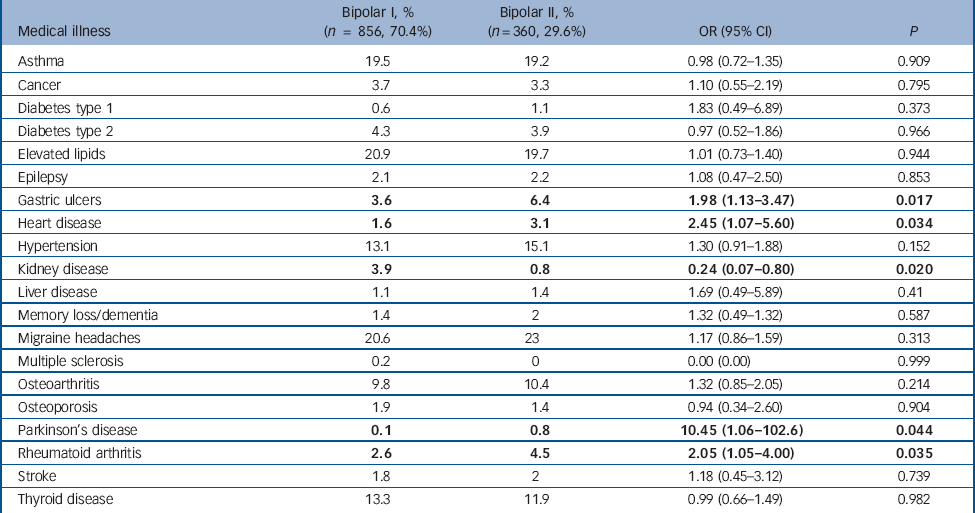
Table 3 shows the lifetime rates of self-reported medical illness according to bipolar subtype and the results of the binary logistic regression analyses with diagnostic group (bipolar I v. bipolar II) as the outcome variable and presence/absence of each medical disorder, gender and age at interview as cofactors. The rates of gastric ulcers, heart disease, Parkinson’s disease and rheumatoid arthritis were significantly higher in the bipolar II group. There was a significantly higher rate of kidney disease in the bipolar I group. These differences were not statistically significant following correction for multiple testing using the Bonferroni method.
Table 2 Binary logistic regression with diagnostic group (bipolar/unipolar, bipolar/control, unipolar/control) as the outcome variable and presence/absence of the medical disorder, gender and age at interview as cofactors (prior to Bonferroni correction)Footnote a
| Bipolar/unipolar group | Bipolar/control group | Unipolar/control group | ||||
|---|---|---|---|---|---|---|
| Medical illness | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P |
| Asthma | 1.60 (1.33-1.93) | 5.67×10–7 | 2.17 (1.74-2.71) | 5.45×10–12 | 1.37 (1.09-1.73) | 0.008 |
| CancerFootnote b | 0.79 (0.53-1.16) | 0.224 | 1.36 (0.78-2.36) | 0.274 | 1.79 (1.08-2.97) | 0.024 |
| Diabetes type 1 | 0.98 (0.53-1.81) | 0.936 | 1.56 (0.73-3.36) | 0.252 | 1.42 (0.67-3.03) | 0.360 |
| Diabetes type 2 | 1.87 (1.30-2.70) | 0.001 | 4.14 (2.28-7.53) | 3.09×10–6 | 2.34 (1.19-4.20) | 0.012 |
| Elevated lipids | 2.33 (1.90-2.86) | 6.46×10–16 | 3.52 (2.66-4.64) | 6.11×10–19 | 1.48 (1.09-1.99) | 0.011 |
| Epilepsy | 1.73 (1.13-2.65) | 0.012 | 6.19 (2.79-13.73) | 7.28×10–6 | 3.88 (1.71-8.83) | 0.001 |
| Gastric ulcersFootnote b | 0.95 (0.66-1.35) | 0.76 | 4.53 (2.21-9.30) | 3.87×10–5 | 4.69 (2.34-9.44) | 1.37×10–5 |
| Heart disease | 0.71 (0.47-1.06) | 0.095 | 1.75 (0.95-3.21) | 0.073 | 2.61 (1.46-4.68) | 0.001 |
| Hypertension | 0.89 (0.73-1.07) | 0.210 | 2.16 (1.65-2.83) | 2.25×10–8 | 2.44 (1.87-3.18) | 5.70×10–11 |
| Kidney diseaseFootnote b | 1.86 (1.13-3.07) | 0.015 | 4.66 (2.03-10.66) | 2.76×10–4 | 1.95 (0.84-4.50) | 0.118 |
| Liver diseaseFootnote b | 0.47 (0.24-0.93) | 0.029 | 1.01 (0.40-2.55) | 0.985 | 2.36 (1.08-5.16) | 0.031 |
| Memory loss/dementia | 1.13 (0.69-1.85) | 0.625 | 24.3 (3.29-178.8) | 0.002 | 20.1 (2.71-148.5) | 0.003 |
| Migraine headaches | 1.13 (0.96-1.33) | 0.147 | 1.39 (1.15-1.69) | 0.001 | 1.20 (0.99-1.46) | 0.064 |
| Multiple sclerosis | 0.06 (0.03-0.129) | 4.34×10–14 | 7.01 (0.86-57.21) | 0.069 | 88.3 (12.3-633.7) | 8.37×10–6 |
| Osteoarthritis | 0.99 (0.79-1.25) | 0.950 | 2.65 (1.84-3.82) | 1.74×10–7 | 2.69 (1.87-3.86) | 1.04×10–7 |
| Osteoporosis | 0.89 (0.59-1.34) | 0.564 | 1.75 (0.91-3.30) | 0.086 | 1.86 (0.99-3.49) | 0.052 |
| Parkinson’s disease | 5.7 (1.26-25.79) | 0.024 | 5.97 (0.76-46.90) | 0.09 | 1.61 (0.15-17.86) | 0.697 |
| Rheumatoid arthritis | 1.04 (0.74-1.44) | 0.839 | 1.92 (1.19-3.10) | 0.007 | 2.06 (1.28-3.22) | 0.003 |
| Stroke | 1.49 (0.91-4.43) | 0.113 | 5.60 (1.98-15.83) | 0.001 | 4.08 (1.41-11.76) | 0.009 |
| Thyroid diseaseFootnote b | 1.70 (1.32-2.18) | 3.31×10–5 | 6.26 (3.77-10.40) | 1.43×10–12 | 3.46 (2.09-5.73) | 1.33×10–6 |
a. Results in bold are statistically significant at P<0.05.
b. Data only available for a subset of the bipolar disorder group (n = 1216).
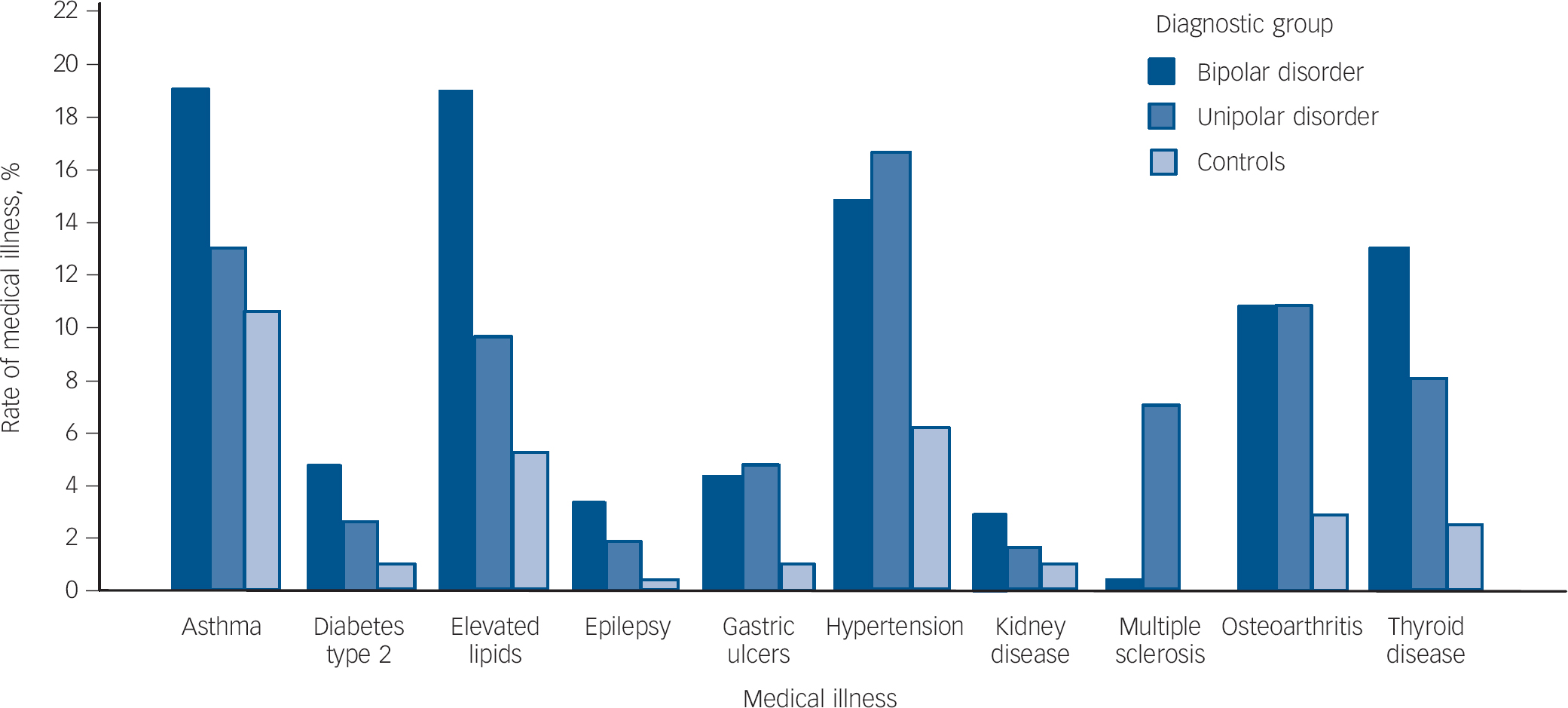
Fig. 1 Rates of medical illnesses showing statistically significant differences between groups following Bonferroni correction for multiple testing.
Statistically significant between group differences (P<0.001) following Bonferroni correction: bipolar>unipolar disorder, control group: asthma, elevated lipids; bipolar disorder >control group: diabetes type 2, epilepsy, kidney disease; unipolar, bipolar disorder>control group: gastric ulcers, hypertension, osteoarthritis; unipolar>bipolar disorder, control group: multiple sclerosis; bipolar>unipolar>control group: thyroid disease.
Clinical characteristics of bipolar disorder according to medical illness burden
In order to investigate whether a greater burden of medical illness is associated with a more severe bipolar illness course, we compared those participants who had a history of three or more medical illnesses (bipolar+3M: n = 202) with those with no medical illnesses (bipolar+0M: n = 440) (Table 4, see online Table DS1 for a more detailed version of this table). A number of variables appeared to be associated with an increase in medical illness burden. These included: a longer illness duration, a typically acute onset of mood episodes, a greater number of psychiatric in-patient admissions, deterioration in functioning, increased rates of anxiety disorder, suicide attempt, rapid cycling, and treatment with anxiolytics, mood stabilisers and electroconvulsive therapy (ECT). When controlling for age at interview, the association with each of the following three variables was no longer statistically significant: number of admissions, lifetime treatment with mood stabiliser or ECT.
Table 3 Lifetime rates (%) of self-reported medical illness according to bipolar subtype and binary logistic regression with diagnostic group (bipolar I v. bipolar II) as the outcome variable and presence/absence of the medical disorder, gender and age at interview as cofactors (prior to Bonferroni correction)
| Medical illness | Bipolar I, % (n = 856, 70.4%) |
Bipolar II, % (n = 360, 29.6%) |
OR (95% CI) | P |
|---|---|---|---|---|
| Asthma | 19.5 | 19.2 | 0.98 (0.72-1.35) | 0.909 |
| Cancer | 3.7 | 3.3 | 1.10 (0.55-2.19) | 0.795 |
| Diabetes type 1 | 0.6 | 1.1 | 1.83 (0.49-6.89) | 0.373 |
| Diabetes type 2 | 4.3 | 3.9 | 0.97 (0.52-1.86) | 0.966 |
| Elevated lipids | 20.9 | 19.7 | 1.01 (0.73-1.40) | 0.944 |
| Epilepsy | 2.1 | 2.2 | 1.08 (0.47-2.50) | 0.853 |
| Gastric ulcers | 3.6 | 6.4 | 1.98 (1.13-3.47) | 0.017 |
| Heart disease | 1.6 | 3.1 | 2.45 (1.07-5.60) | 0.034 |
| Hypertension | 13.1 | 15.1 | 1.30 (0.91-1.88) | 0.152 |
| Kidney disease | 3.9 | 0.8 | 0.24 (0.07-0.80) | 0.020 |
| Liver disease | 1.1 | 1.4 | 1.69 (0.49-5.89) | 0.41 |
| Memory loss/dementia | 1.4 | 2 | 1.32 (0.49-1.32) | 0.587 |
| Migraine headaches | 20.6 | 23 | 1.17 (0.86-1.59) | 0.313 |
| Multiple sclerosis | 0.2 | 0 | 0.00 (0.00) | 0.999 |
| Osteoarthritis | 9.8 | 10.4 | 1.32 (0.85-2.05) | 0.214 |
| Osteoporosis | 1.9 | 1.4 | 0.94 (0.34-2.60) | 0.904 |
| Parkinson’s disease | 0.1 | 0.8 | 10.45 (1.06-102.6) | 0.044 |
| Rheumatoid arthritis | 2.6 | 4.5 | 2.05 (1.05-4.00) | 0.035 |
| Stroke | 1.8 | 2 | 1.18 (0.45-3.12) | 0.739 |
| Thyroid disease | 13.3 | 11.9 | 0.99 (0.66-1.49) | 0.982 |
a. Results in bold are statistically significant at P<0.05.
Table 4 Clinical features and course of bipolar illness in participants with bipolar disorder with a high burden of comorbid medical illness (three or more medical illnesses, bipolar+3M) compared with those participants with bipolar disorder with no history of medical illness (bipolar+0M) (see online Table DS1 for a more detailed version of this table)
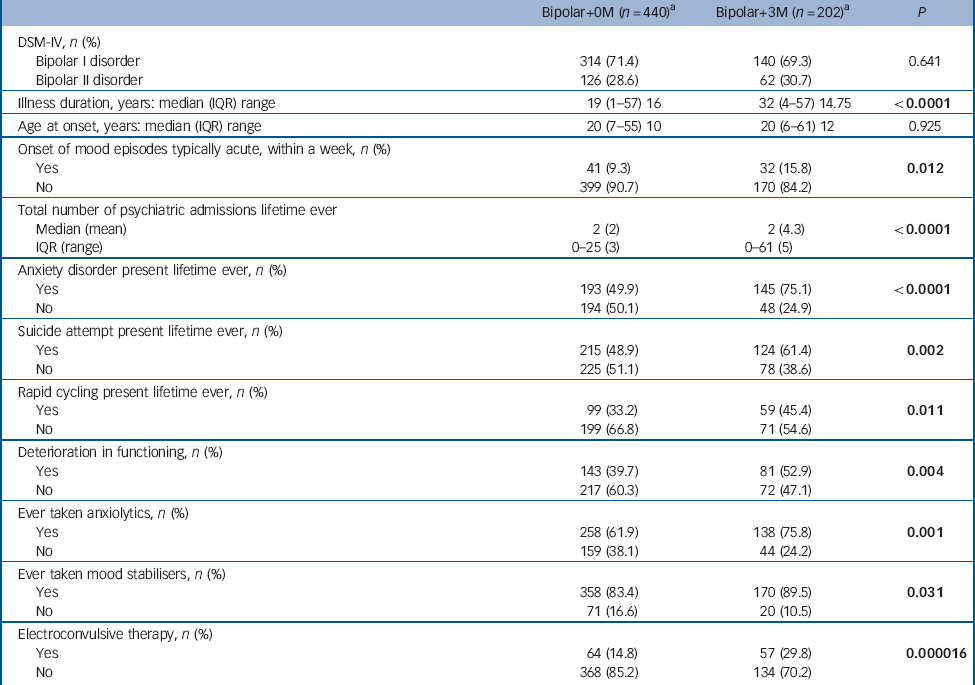
| Bipolar+0M (n = 440)Footnote a | Bipolar+3M (n = 202)Footnote a | P | |
|---|---|---|---|
| DSM-IV, n (%) | |||
| Bipolar I disorder | 314 (71.4) | 140 (69.3) | 0.641 |
| Bipolar II disorder | 126 (28.6) | 62 (30.7) | |
| Illness duration, years: median (IQR) range | 19 (1-57) 16 | 32 (4-57) 14.75 | <0.0001 |
| Age at onset, years: median (IQR) range | 20 (7-55) 10 | 20 (6-61) 12 | 0.925 |
| Onset of mood episodes typically acute, within a week, n (%) | |||
| Yes | 41 (9.3) | 32 (15.8) | 0.012 |
| No | 399 (90.7) | 170 (84.2) | |
| Total number of psychiatric admissions lifetime ever | |||
| Median (mean) | 2 (2) | 2 (4.3) | <0.0001 |
| IQR (range) | 0-25 (3) | 0-61 (5) | |
| Anxiety disorder present lifetime ever, n (%) | |||
| Yes | 193 (49.9) | 145 (75.1) | <0.0001 |
| No | 194 (50.1) | 48 (24.9) | |
| Suicide attempt present lifetime ever, n (%) | |||
| Yes | 215 (48.9) | 124 (61.4) | 0.002 |
| No | 225 (51.1) | 78 (38.6) | |
| Rapid cycling present lifetime ever, n (%) | |||
| Yes | 99 (33.2) | 59 (45.4) | 0.011 |
| No | 199 (66.8) | 71 (54.6) | |
| Deterioration in functioning, n (%) | |||
| Yes | 143 (39.7) | 81 (52.9) | 0.004 |
| No | 217 (60.3) | 72 (47.1) | |
| Ever taken anxiolytics, n (%) | |||
| Yes | 258 (61.9) | 138 (75.8) | 0.001 |
| No | 159 (38.1) | 44 (24.2) | |
| Ever taken mood stabilisers, n (%) | |||
| Yes | 358 (83.4) | 170 (89.5) | 0.031 |
| No | 71 (16.6) | 20 (10.5) | |
| Electroconvulsive therapy, n (%) | |||
| Yes | 64 (14.8) | 57 (29.8) | 0.000016 |
| No | 368 (85.2) | 134 (70.2) |
a. For some variables because of missing data n is less than n = 440 and n = 202 for BD+0M and BD+3M, respectively.
To identify the clinical characteristics of bipolar illness that best predict medical illness burden group, we entered the variables that were statistically significant at the P<0.05 level in the univariate analyses, shown in Table 4, into a logistic regression model. Age at interview and gender were also entered into the model. The clinical characteristics that best predicted high medical illness burden group were having a lifetime history of anxiety disorder (OR = 2.76, 95% CI 1.49-5.13, P = 0.001), rapid cycling mood episodes (OR = 2.25, 95% CI 1.20-4.25, P = 0.012), suicide attempt (OR = 2.39, 95% CI 1.30-4.39, P = 0.005) and mood episodes with a typically acute onset (within a week) (OR = 2.56, 95% CI 1.17-5.62, P = 0.019).
Discussion
Rates of medical illnesses across the bipolar, unipolar and control groups
The most prevalent medical conditions in the bipolar sample were migraine headache (23.7%), asthma (19.2%), elevated lipids (19.2%), hypertension (15%), thyroid disease (12.9%) and osteoarthritis (10.8%). The same six conditions were also the most prevalent conditions in the unipolar sample: migraine headache (21.9%), asthma (13.1%), elevated lipids (9.7%), hypertension (16.8%), thyroid disease (8.2%) and osteoarthritis (10.9%).
The high rates of migraine headache in these samples are consistent with previous findings. Reference Carney and Jones3,Reference McIntyre, Konarski, Soczynska, Wilkins, Panjwani and Bouffard20 The high comorbidity between affective disorder and migraine may be because of a causal relationship, with one disorder causing (or increasing risk for) the other, or may be because of shared aetiological factors. As studies show that the combination of these two disorders cause more impairment than either disorder alone Reference Lipton, Hamelsky, Kolodner, Steiner and Stewart21 it is important to make sure that the patients are screened and treated appropriately to ensure the optimal outcome for the patient.
Almost a fifth of our bipolar sample (19.2%) self-reported a lifetime history of asthma. This figure is comparable to the rate of 15.9% reported by McIntyre et al Reference McIntyre, Konarski, Soczynska, Wilkins, Panjwani and Bouffard20 in their bipolar sample. A recent study has suggested that childhood asthma increases the risk of bipolar disorder developing in adulthood. Reference Liang and Chikritzhs22 Again this leads to a number of hypotheses regarding the relationship between affective disorders and asthma in terms of aetiology and causation, with the possibility, for example, that carbon dioxide hypersensitivity and corticosteroid therapy may partly explain this association.
The increased rates of hypertension in our affective disorders samples are again consistent with previous research, although rates across studies vary according to the age ranges of participants. Reference Carney and Jones3,Reference Kilbourne, Cornelius, Han, Pincus, Shad and Salloum23,Reference Beyer, Kuchibhatla, Gersing and Krishnan24 D’Mello Reference D'Mello25 found that hypertension seemed to be related to the severity of illness in bipolar disorder with increased levels of hypertension found in those patients with higher levels of mania.
The literature concerning the relationship between mood disorders and cholesterol levels is inconsistent, with studies suggesting that lower levels are associated with current suicide risk and depressive and manic symptoms Reference Fiedorowicz, Palagummi, Behrendtsen and Coryell26 but that people with bipolar disorder are at higher risk of metabolic abnormalities including obesity and hyperlipidemia, Reference McIntyre, Danilewitz, Liauw, Kemp, Nguyen and Kahn27 consistent with the increased rates of hyperlipidaemia found in our bipolar sample. Our finding of increased rates of thyroid disorder in those with mood disorders is not surprising, given the well-established link between thyroid dysfunction and mood disorders. Reference Thomsen and Kessing28,Reference Smith, Martin, McLean, Langan, Guthrie and Mercer29 Thyroid problems are a common side-effect of the drug lithium, which is a common treatment for bipolar disorder and is also used to treat severe or treatment-resistant unipolar depression. In our bipolar sample, rates of thyroid disorder were significantly increased in those who had taken mood stabilising medication during their lifetime.
A recent study highlighted that the amount of pain people with osteoarthritis feel appears to be directly related to their mental health, Reference Wise, Niu, Zhang, Wang, Jordan and Choy30 however, our study, which found significantly higher rates of osteoarthritis in patients with mood disorders compared with controls, is the first to examine the rates of osteoarthritis in people with unipolar and bipolar disorders. Studies have shown increased rates of depression in patients with arthritis (of any type) with the temporal relationship indicating that arthritis predicts new onset of psychiatric disorder. Reference van ‘t Land, Verdurmen, Ten Have, van Dorsselaer, Beekman and de Graaf31 The results of previous studies have been conflicting in terms of investigating the relationship between osteoporosis and mood disorder. Reference Mezuk32 This is the largest study to date to examine rates of osteoporosis in bipolar disorder and we found no distinction between the bipolar, unipolar and control samples in terms of the rate of osteoporosis.
We found higher rates of kidney disease in the bipolar sample compared with controls, with rates being highest in patients with bipolar I compared with bipolar II disorder. Again, this finding is consistent with previous research Reference Smith, Martin, McLean, Langan, Guthrie and Mercer29 and is not surprising, given the adverse renal effects that have been associated with lithium therapy, Reference McKnight, Adida, Budge, Stockton, Goodwin and Geddes33 which vary from the more common reversible polyuria to irreversible kidney damage. Similarly, the increased rate of Parkinson’s disease reported in our bipolar sample compared with the unipolar sample and controls may be related to the greater use of antipsychotic medications and selective serotonin reuptake inhibitors in the bipolar sample, in that patients may be reporting symptoms occurring as a side-effect of their medication, rather than Parkinson’s disease.
Our finding of increased rates of epilepsy in the bipolar group compared with controls is consistent with previous research Reference Smith, Martin, McLean, Langan, Guthrie and Mercer29 including a study indicating that the rate of bipolar spectrum disorders in individuals with epilepsy is higher than that seen in the general population. Reference Mazza, Di Nicola, Della Marca, Janiri, Bria and Mazza34 In our study, multiple sclerosis was significantly more common in those with unipolar disorder compared with those with bipolar disorder and controls, with the rates being 7%, 0.5%, 0%, respectively; a finding not previously described. However, this finding is consistent with previous reports that have found the prevalence of major depression to be elevated in people with multiple sclerosis compared with those without multiple sclerosis and those reporting other chronic conditions. Reference Patten, Beck, Williams, Barbui and Metz35,Reference Siegert and Abernethy36 Recent studies have suggested that biological processes may account for the overlap between depression and multiple sclerosis, rather than depression simply being a psychological reaction to the burden of being ill. Reference Gold, Kern, O'Connor, Montag, Kim and Yoo37
Rates of medical illness according to bipolar subtype
To our knowledge, this is the first study to look at rates of medical comorbidities across large samples of patients with bipolar I and II disorders. In patients with bipolar disorder, the type of bipolar disorder (I or II) may be associated with an increased risk of certain medical illnesses. Participants with bipolar II disorder may be more likely to have gastric ulcers, heart disease, Parkinson’s disease and rheumatoid arthritis than patients with a diagnosis of bipolar I disorder. Participants with bipolar I disorder were significantly more likely to have had kidney disease than those with bipolar II disorder. It is important to note, however, that the statistically significant differences between the bipolar I and II groups did not remain following correction for multiple testing.
Clinical characteristics of bipolar disorder according to medical illness burden
We have also demonstrated that in those with bipolar disorder, a higher burden of medical illness is associated with certain clinical features indicative of a more severe illness course, with greater impairment in functioning. These results are in agreement with previously reported findings Reference Kemp, Gao, Chan, Ganocy, Findling and Calabrese38,Reference Soreca, Fagiolini, Frank, Houck, Thompson and Kupfer39 that increased medical burden is linked with more suicide attempts, older age and longer illness duration.
Previous reports have attributed poor physical health in people with mental ill to increased rates of smoking and alcohol misuse (and other lifestyle factors). Reference Hert, Correll, Bobes, Cetkovich-Bakmas, Cohen and Asai40,Reference Eldridge, Dawber and Gray41 In our bipolar sample, we found no statistically significant differences between the bipolar group with no history of medical illness and those with a high burden of medical illness in terms of rates of alcohol misuse or smoking. Although this finding requires further investigation it suggests that, at least in our bipolar sample, smoking and alcohol misuse may not be the most significant factors influencing susceptibility to medical illness burden. This finding is in line with the findings of Chwastiak et al, Reference Chwastiak, Rosenheck, McEvoy, Keefe, Swartz and Lieberman42 who reported no association between an increased number of medical conditions and alcohol or drug misuse in patients with schizophrenia. We found higher lifetime rates of treatment with anxiolytics, mood stabilisers and ECT in those with a higher medical illness burden, although after controlling for age at interview, this finding remained only for lifetime treatment with anxiolytics, which is consistent with the higher rates of anxiety disorders found in those with a higher medical illness burden.
The mechanisms underlying the relationship between mental and physical health disorders are complex, although evidence suggests that the causal relationships are likely to be bidirectional. Reference Ramasubbu, Beaulieu, Taylor, Schaffer and McIntyre4,Reference Evans, Charney, Lewis, Golden, Gorman and Krishnan9 The existence of a medical condition may be a stress that increases the risk of developing a mood episode/disorder. Similarly, the existence of an affective disorder may increase the risk of developing a medical condition.
Research findings to date suggest that affective disorders may share some aetiological factors in common with certain medical disorders. A pathway analysis of Wellcome Trust Case Control Consortium genome-wide association study data suggested that bipolar disorder and metabolic disorders, such as coronary artery disease and diabetes type 2, have strong genetic links and may share some common pathophysiological pathways. Reference Torkamani, Topol and Schork43 Such findings suggest that subphenotypes defined by the presence of certain comorbid medical conditions are likely to be useful in studies investigating the genetic aetiology of mood disorders. The findings of Rztetsky et al Reference Rzhetsky, Wajngurt, Park and Zheng8 suggest that many complex phenotypes (non-Mendelian disorders including both physical and psychiatric disorders) are probably rooted in genetic variation that is significantly shared by multiple disease phenotypes.
Strengths and limitations
The strengths of our study include the use of large, well-defined samples of patients, recruited from throughout the UK and assessed using standardised methodology. It is important to consider our findings in light of certain limitations. First, our rates of medical illness rely on a self-report measure. The accuracy of this self-report measure was assessed by Farmer et al, Reference Farmer, Korszun, Owen, Craddock, Jones and Jones10 where 61 patient self-reports were compared with reports from the patient’s general practitioner (GP) about the medical illnesses the patient had been treated for. Kappa statistics for self-report compared with GP report for six of the more common disorders were calculated with results as follows: asthma κ = 0.73, diabetes κ = 0.91, hypertension κ = 0.87, hypercholesterolaemia κ = 0.65, heart attack/angina κ = 0.82 and arthritis κ = 0.74. Percentage agreement between GPs and participants for the presence or absence of a medical disorder was 93%. In addition, ten of the medical illnesses included in the current study were also included in a study by Carney & Jones Reference Carney and Jones3 which examined in-patient and out-patient administrative claims in the USA. The rates of medical disorders seen in their bipolar and control samples were reassuringly similar to those seen in the current study.
A second limitation is that we do not have a consistent measure of body mass index (BMI, or of waist/hip ratio) across all of our samples. When comparing BMI between participants with unipolar depression and controls, Farmer et al Reference Farmer, Korszun, Owen, Craddock, Jones and Jones10 found that although the two groups were similarly represented in the overweight range, the unipolar depression group were significantly overrepresented in the obese range. In the Famer et al Reference Farmer, Korszun, Owen, Craddock, Jones and Jones10 study, obesity was associated with an increase in self-reported rates of hypercholesterolaemia, type 2 diabetes and myocardial infarction. These findings suggest that some of the results from the current study may be altered if we were to control for BMI. Here, BMI could be seen as a confounding factor or an intermediate phenotype with potentially shared heritable pathogenesis. Third, our sample was restricted to a single ethnic group, limiting the generalisability of our findings. Fourth, it is possible that the voluntary nature of the control sample may have resulted in an unrepresentatively healthy sample with an artificially low prevalence of medical illnesses. Finally, we did not have data on some additional lifestyle factors such as diet or activity that may be related to medical illness burden.
Implications
These findings suggest that patients with affective disorders have an increased risk of certain medical conditions. Knowledge of the most prevalent medical conditions in patients with affective disorders and developments in the prevention, detection and treatment of such illnesses in this group are essential in improving care and prognosis. It is important to raise awareness among healthcare professionals about the risks to which patients with affective disorders are exposed. Knowing which medical illnesses are likely to coexist with a mood disorder may help to improve diagnostics and management and therefore clinical and social care for patients. Early diagnostics and treatment of medical conditions are necessary to prevent mortality and unfavourable outcomes in terms of both physical and mental health.
Although the relationship between affective disorders and physical conditions is not fully understood, it is likely that some of the risk factors for comorbid psychiatric and medical illness are modifiable. Further research on this subject may help to improve quality of life, prognosis and life expectancy for those with these illnesses. Compared with individuals with no history of mental illness, individuals with bipolar disorder are at increased risk of premature death, from both natural and unnatural causes. Reference Roshanaei-Moghaddam and Katon44 A growing evidence base suggests that more integrated ways of working, with collaboration between mental health and other professionals, offers the best chance of improving outcomes for individuals with both mental health and physical conditions. Reference Yohannes, Doherty, Bundy and Yalfani45
Acknowledgements
The authors thank all of the participants who gave their time to participate in the study.










eLetters
No eLetters have been published for this article.