Contents
Physiology of pregnancy (cardiovascular, respiratory, renal, gastro-intestinal, liver and haematological systems)
Physiology of onset of parturition, myometrial contractility and cervical dilatation
Physiology of the third stage of labour
Lactation and uterine involution
The Cardiovascular System
The cardiovascular changes are illustrated in Figs. 1.1a and 1.1b and Table 1.1.

Figure 1.1b Maternal cardiovascular changes
Plasma volume ↑ from 2600 ml to 3800 ml
– Early in pregnancy (6–8 wk)
– No further ↑ after 32 wk
Red cell mass ↑ from 1400 ml to 1650–1800 ml
– Steady ↑ until term
– Haematocrit and haemoglobin concentration ↓
Cardiac output (CO) ↑ 40% from 4.5 l/min to ~6 l/min
– Early in pregnancy
– Plateau at 24–30 wk
– ↓ to pre-pregnancy level after delivery (variable time)
Supine hypotensive syndrome: If a pregnant woman in the third trimester lies supine, the gravid uterus may compress the inferior vena cava against her spine, impeding venous return which leads to a fall in cardiac output. She may experience a marked fall in blood pressure and may feel faint, dizzy and nauseous. This might also reduce the uterine blood flow, potentially leading to fetal distress in labour. This is known as supine hypotensive syndrome; it is quickly relieved if the woman moves to the lateral position. We also tend to position the mother in a left lateral position during Caesarean section until delivery for the same reason.
Oxygen consumption ↑ extra 30–50 ml/min
Alteration in regional blood flow
– Uterus
– Kidney
– Skin
– Breasts
– Skeletal muscles
During pregnancy, the increase in ventilation is greater than the increase in oxygen consumption. Therefore, the arterio-venous oxygen gradient ↓
At term, the distribution of the ↑ in CO (1.5 l/min):
– Uterus 400 ml/min
– Kidney 300 ml/min
– Skin 500 ml/min
– 300 ml/min to gastrointestinal tract (GI), breasts and others
Early in pregnancy, the extra blood supply shifts mainly to the skin and breasts
From 8 to 36 weeks
– Systolic BP ↓ 5 mmHg
– Diastolic BP ↓ 10 mmHg
Other factors which influence the blood pressure include maternal position, uterine contractions, drugs which affect the vascular tone or the cardiac function.
– HR ↑ 10–15%
– Left axis deviation 15°
– Inverted T-wave in lead III
– Q in lead III and AVF
– Non-specific ST changes
ECG changes in pregnancy are secondary to:
– Left ventricular hypertrophy and dilatation
– No change in the contractility
– Upward displacement of the diaphragm
– The apex is shifted anterior and to the left
The Respiratory System
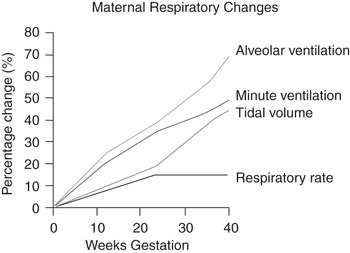
The cardiovascular changes are illustrated in Fig. 1.2.
The lung volumes in a non-pregnant normal individual are shown in Fig. 1.3. These include the tidal volume (TV), inspiratory reserve volume (IRV), expiratory reserve volume (ERV), residual volume (RV), total lung capacity (TLC), vital capacity (VC) and functional residual capacity (FRC).
Ventilation ↑ by 40% (from the first trimester)
Progesterone stimulates respiratory centre both directly (stimulates the respiratory centre) and indirectly (reduce the threshold of the respiratory centre to carbon dioxide)
Progesterone is a bronchodilator
Breathing is more diaphragmatic than thoracic
Airway resistance ↓
Tidal volume ↑, not respiratory rate
No change in the vital capacity
Residual volume ↓ 200 ml
Expiratory reserve volume ↓
Both progressively ↓ (by 20% at term)
IRV ↓ early and ↑ late in pregnancy
The total lung capacity ↓ 200 ml
No change in forced expiratory volume 1 (FEV1) or peak flow rate
Lung compliance is unaffected
Chest compliance ↓ especially in lithotomy
70% of pregnant women experience subjective dyspnoea
In view of the fact that pregnancy is a pro-coagulant state, the risk of pulmonary embolism is increased
The oxygen consumption ↑ (50 ml/min at term)
– Fetus 20 ml/min
– ↑ CO 6 ml/min
– ↑ renal work 6 ml/min
– ↑ metabolic rate 18 ml/min
The changes in the lung volumes during pregnancy in comparison to those in a non-pregnant individual are illustrated in Fig. 1.4. Table 1.2 demonstrates the difference in ventilation in pregnancy, labour and the non-pregnant state.
The normal range of the arterial blood gases (ABG) is shown in Fig. 1.5.
PCO2 ↓ to 31 mmHg
PaO2 ↑ to 14 kPa during the third trimester and then falls to <13.5 kPa at term (↑ CO unable to compensate ↑ oxygen consumption)
HCO3- ↓
Na ↓
Osmolarity ↓ 10 mmol/l

Figure 1.5 The normal range of the arterial blood gases
A suggested algorithm for the interpretation of the arterial blood gas (ABG) is shown in Fig. 1.6.
The Urinary System
The kidney size ↑ during pregnancy (1 cm length)
The ureters become dilated due to:
– Progesterone is a smooth muscle relaxant
– Pressure by the gravid uterus
These changes lead to pregnant women being prone to urinary tract infection
The renal blood flow ↑ from 1.2 l/min to 1.5 l/min (from the first trimester)
The glomerular filtration rate (GFR) ↑ to 140–170 ml/min
Both the renal blood flow and the GFR are 50–60% higher at term
The blood urea level ↓ from 4.3 to 3.1 mmol/l
The creatinine serum level ↓ from 73 to 47 µmol/l
Both the urate and HCO3- ↓
Mild glycosuria and proteinuria
The plasma osmolarity ↓due to the effect of:
– Progesterone
– Renin-angiotensin-aldosterone pathway
Table 1.3 demonstrates the renal function in pregnancy compared to the non-pregnant state.
The Gastrointestinal Tract
The following changes are seen during pregnancy:
Gastric relaxation
Delayed gastric emptying
Relaxation of the gastro-oesophageal sphincter
Reflux of gastric acid
– 80% of pregnant women experience heartburn at term
Pregnant women are prone to gastric aspiration
Slower bowel peristalsis; therefore, constipation is common in pregnancy
– Alkaline phosphatase ↑ 3 times the normal level (produced from the placenta)
– Cholecystokinin release ↓
– Gall bladder contractility ↓
– Pregnant women are prone to gallstones
The following occurs in obstetric cholestasis:
– Interaction between inherited and acquired abnormalities in bile salt transporters
– Itching in pregnancy
– ↑ liver enzymes and bile salts
– Similar reaction to the combined oral contraceptive pill
– Associated with intrauterine death and fetal distress in labour
The daily requirements of a number of vitamins are shown in Table 1.4.
The Haematological System
The following changes are seen in pregnancy:
↑ erythropoiesis from early pregnancy (due to ↑ erythropoietin and placental lactogen)
Physiological anaemia due to the fact that the increase in the plasma volume is more than the increase in the red cell volume
WBC ↑ and peaks after delivery. This rise is primarily in neutrophils.
The effect of pregnancy on the platelet count is debated, but in some women, there may be a modest decline by term, perhaps by as much as 25%. This fall is believed to be due to increased destruction of platelets not caused by immune factors (as happens in gestational thrombocytopenia).
Total requirement 700–1400 mg extra
Overall requirement 4 mg/day (from 2.8 mg/day in non-pregnant to 6.6 mg/day by the end of pregnancy)
The normal range of the ferritin level in the maternal serum is 15–300 µg/l (considered as an indicator of the iron stores)
↑ iron absorption (erythroid hyperplasia)
The amount absorbed depends on
Iron stores
Dietary content
Iron supplements
Evidence that iron absorption ↑ in the latter half of pregnancy
Still not enough for the needs in pregnancy and puerperium
Iron deficiency anaemia
Haemostasis in pregnancy
A list of the clotting factors is shown in Table 1.5.
Platelet function remains normal
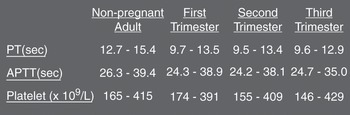
Routine coagulation screening is essentially normal (Fig. 1.7)
Remains low in labour
Returns to normal within one hour of delivery of the placenta
Evidence that the inhibition of fibrinolysis is mediated through the placenta (plasminogen activator inhibitor 2)
What stops bleeding after delivery of the placenta?
Table 1.5 A list of the clotting factors
| I Fibrinogen | VIII Anti-hemophilic factor A |
| II Prothrombin | IX Anti-hemophilic factor B or Christmas factor |
| III Tissue factor | X Stuart-Prower factor |
| IV Calcium | XI Plasma thromboplastin antecedent |
| V Proaccelerin (labile factor) | XII Hageman factor |
| VII Proconvertin (stable factor) | XIII Fibrin-stabilizing factor |
Implications of Maternal Physiological Changes on Therapeutic Drug Administration
Absorption of drugs from the gastrointestinal tract may be impaired by:
– Gastric stasis
– Poor gut motility
– Lower gastric pH (for some drugs)
The increase in plasma volume means that the volume of distribution of the drug increases, so the concentrations may be lower than expected. This is particularly important in women taking antiepileptic drugs or thyroxine. Because of the increased glomerular filtration rate, excretion of drugs mainly excreted by the kidneys will be accelerated. These changes often require doses of a drug given during pregnancy to be adjusted.
Physiology of Lactation
Fourteen days’ exposure to oestrogen followed by stimulation by prolactin is enough to establish milk production.
Prolactin is a long chain polypeptide hormone and is essential for successful lactation.
In early pregnancy, there is hyperplasia of the alveolar cells and lactiferous ducts, followed in later pregnancy by alveolar cell hypertrophy and the initiation of secretion. These changes are stimulated by the increased levels of prolactin and human placental lactogen (HPL).
During pregnancy, the high levels of oestrogen and progesterone hold this process in check, full milk production achieved only after delivery, when progesterone and oestrogen levels fall rapidly.
Milk production averages 500–1000 ml per day and is highly dependent on continued suckling (which causes the release of both prolactin and oxytocin).
In women who do not suckle, milk production gradually falls and may persist for 3–4 weeks postpartum.
In breast-feeding mothers, equilibrium is reached after around 3 weeks.
Mothers who are breast-feeding twins produce twice as much milk, i.e. ≥ 2 litres/day.
The Suckling Stimulus
The suckling stimulus sends afferent impulses to the hypothalamus, which leads to a surge of prolactin release.
This surge reaches a peak around 30 minutes after the baby is put to the breast and gradually declines to basal levels by 120 minutes.
The control of prolactin release from the anterior pituitary is primarily via prolactin inhibitory factors (PIF) from the hypothalamus which are secreted into the pituitary portal blood system.
The most important PIF is dopamine. Therefore, dopamine agonists, such as bromocriptine and cabergoline, can be used in the early puerperium to suppress milk production.
Conversely, dopamine antagonists such as metoclopramide increase prolactin levels and are sometimes used in breast-feeding women to stimulate milk production.
Thyrotropin-releasing hormone (TRH) may also play a role in stimulating prolactin production.
After the sixth postpartum week, both basal prolactin levels and the peak level following suckling gradually decline; the greater the frequency and duration of suckling, the slower the decline.
Suckling also stimulates oxytocin (octapeptide) release through afferent impulses to specialised neurons in the supraoptic and paraventricular nuclei of the hypothalamus.
The release is from the posterior pituitary.
The release of oxytocin, which typically occurs in short, one-minute bursts, may begin even before the baby is put to the breast (neuroendocrine reflex can also be initiated by the mother hearing her baby cry or even thinking about breast-feeding).
Oxytocin binds to specific receptors on the myoepithelial cells which surround the alveolar (milk-producing) cells in the breasts, and which are longitudinally arranged in the walls of the milk ducts.
Contraction of these myoepithelial cells forces the milk into the ducts; contraction of the longitudinally arranged cells in the duct walls causes them to dilate, allowing milk to flow more easily toward the nipple.
Both prolactin and oxytocin are necessary for successful breast-feeding; prolactin stimulates the production of milk while oxytocin stimulates its ejection/let down.
The Composition of Breast Milk
The composition of breast milk is listed in Table 1.6.
After delivery, the colostrum (or early milk) has a high concentration of protein relative to the concentration of lactose.
The concentration of lactose ↑ sharply and the concentration of protein ↓ over the following few days.
The main reason for this ↓ in protein concentration is dilution (in order to maintain ionic equilibrium, water is drawn into the breast, causing an ↑ in milk volume), and the total amount of protein in the milk is relatively unchanged.
The main carbohydrate in human milk is lactose. In the baby’s intestine, it is broken down by the enzyme lactase into galactose and glucose.
40% of the protein in human milk is casein, compared with 80% of cow’s milk. Other proteins include immunoglobulins and lactoferrin.
Triglycerides are the main fat found in milk and are its most variable component, which means that the estimated energy content of 75 kcal/100 mL is only an approximation.
Vitamin D deficiency can lead to rickets
Vitamin K deficiency can lead to haemorrhagic disease of the newborn.
Compared with cow’s milk, human milk has approximately one-third the concentrations of sodium and chloride. This is advantageous in babies with diarrhoea because a high solute load can exacerbate diarrhoea.
There is little iron in breast milk.
The major immunoglobulin in breast milk is IgA, with smaller amounts of IgM and IgG.
The IgA in breast milk is poorly absorbed so most stays in the baby’s intestinal tract where it protects against infection. When a mother encounters a specific pathogen in her own GI tract, plasma cells migrate from her gut to breast where they release into breast milk a specific IgA against that pathogen, thus protecting her baby.
The composition of the breast milk varies from woman to woman, over time in an individual woman and even differs between the beginning and end of the same feed.
The most important factor is the time postpartum, suggesting that the milk is adapted in a very sensitive way to the changing needs of the baby.
Any statements about the composition of human breast milk are at best averages.
Calorie Intake Required for Breast-Feeding
A breast-feeding woman requires 2950 kcal a day.
The recommended daily calorie intake is 2700 kcal (2200 kcal for the non-lactating non-pregnancy requirement plus 500 kcal toward the energy requirement of the milk).
An extra 250 kcal per day should come from the maternal fat stores.
Pregnancy during Breast-Feeding
If a woman conceives during lactation, the rapidly rising levels of oestrogen and progesterone will suppress milk production, despite the effects of the baby’s suckling.
High prolactin levels during breast-feeding tend to suppress ovulation and therefore cause amenorrhoea.
Breast-feeding is not a reliable form of contraception; at the end of a year of exclusive breast-feeding, 10% of women who do not use another form of contraception will have fallen pregnant.
Uterine Involution
Immediately after delivery of the placenta: the uterus weighs around 900 g
By seven days postpartum: the uterus weighs half that
By six weeks: almost returned to its pre-pregnancy size and weight of around 100 g
Uterine water, weight, muscle, protein and collagen all ↓ in the same proportions
Result from the rapid withdrawal of placental hormones after delivery
Three days postpartum: the superficial decidual layer becomes necrotic (shed with the lochia)
Within a week: the uterine cavity has a new endometrial layer, with the exception of the placental bed; this takes around three weeks to establish an endometrial cover
The lochia gradually ↓ over 3–6 weeks, changing in turn from red (lochia rubra) to pink (lochia serosa) to yellowish-white (lochia alba)
The Third Stage of Labour
The time from delivery of the baby until delivery of the placenta and membranes
Soon after delivery of the baby, the uterus has a strong and sustained contraction.
– ↓ the surface area of the placental bed, thus shearing off the placenta
– Helps to control bleeding from the vessels of the placental bed
It is likely that prostaglandin F2α play a major role here, as oxytocin levels do not change significantly during this time.
The Onset of Labour, Myometrial Contractility and Cervical Dilatation
The precise mechanism of the onset and maintenance of labour is still poorly understood.
During pregnancy, myometrial quiescence is maintained by pro-pregnancy factors (mainly progesterone).
Progesterone suppresses the formation of myometrial gap junctions and the effect of interleukin 8 (which causes cervical ripening).
Progesterone also decreases uterine sensitivity to oxytocin.
Antiprogesterones such as mifepristone (RU4A6) cause cervical ripening and increase myometrial contractility.
Catecholamines and relaxin also play a role in the maintenance of uterine quiescence.
During the third trimester, maternal oestrogen and corticotrophin-releasing hormone (CRH) gradually ↑. Oestradiol ↑ the concentration of oxytocin receptors in the myometrium and also ↑ oxytocin synthesis in the uterus.
CRH increases prostaglandin synthesis and may stimulate myometrial contractility.
The concentration of myometrial gap junctions increases as labour approaches.
Oestrogen promotes the formation of gap junctions.
CRH also promotes an inflammatory-type mechanism by increasing the expression of inflammatory cytokines, such as interleukin 1β and interleukin 8, and cyclo-oxygenase type II (Cox-2).
It is possible that there is a ‘functional withdrawal’ of progesterone.
– It happens only locally within the fetal membranes.
– Close to term, the dominant progesterone receptor within the uterus changes from type 1 to type 2.
Nitric oxide does not play a significant role in the onset of labour.
Neither does oxytocin; there is no significant rise in maternal oxytocin concentration immediately prior to labour (or indeed during labour).
A marked increase in oxytocin receptors in the myometrium as term approaches, so it seems certain that oxytocin plays an important role in labour, probably in combination with prostaglandins.
Nevertheless, oxytocin does not seem to be the trigger for the onset of labour.
The fetus also secretes some oxytocin (the concentration in the umbilical artery is twice that in the umbilical vein), but it is not certain if this plays a role in labour.
It is possible that the fetus triggers labour through increased cortisol release which can stimulate placental CRH synthesis.
There is a rapid rise in the activity of Cox-2 and other inflammatory cytokines at the onset of labour, leading some to compare labour to an inflammatory process.
Increased Cox-2 activity leads to an increase in prostaglandin synthesis.
The amnion and chorion secrete primarily PGE2 while the decidua favours PGF-2α.
Prostaglandin synthase inhibitors such as indomethacin may thus be used in the management of preterm labour.
Prostaglandins act on the myometrium in the uterine body to cause contractions.
Toward the end of pregnancy and in early labour, under the influence of prostaglandins and interleukin 8 (and perhaps in combination with relaxin and oestrogen), neutrophils are attracted into the cervix, where they release collagenase. This leads to gradual proteolysis of the collagen fibres in the cervix, leading to cervical ripening.
Contraction of the myometrium results from the interaction of actin and myosin. This interaction is controlled by a calcium modulated protein kinase. Communication between myometrial cells through gap junctions facilitates the coordinated contraction of the uterus.
Drugs which reduce available calcium, such as beta-agonists (e.g. ritodrine, salbutamol), thus cause uterine relaxation.
Magnesium sulphate, which inhibits calcium influx into myometrial cells, inhibits the action of myosin light chain kinase, thus causing uterine relaxation.
Calcium channel blockers also inhibit calcium influx through the cell membrane and are used for tocolysis.
Once labour has started, there are multiple feedback mechanisms which further increase prostaglandin and cytokine activity; this process is currently poorly understood.
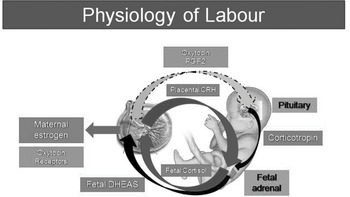
The organs and the mechanisms involved in the physiology of labour, as well as the feto-maternal interaction, are shown in Figs. 1.8 and 1.9.

Figure 1.9 The feto-maternal interaction involved in the physiology of labour