3 results
Uncovering gut microbiota-mediated indirect effects of antibiotic use on Clostridioides difficile transmission
- Camden Gowler, Prabasaj Paul, Mihnea Mangalea, Daniel Nkemzi, Hannah Wolford, Sujan Reddy, Alison Halpin, Lawrence McDonald, Rachel Slayton
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s104-s105
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clostridioides difficile and multidrug-resistant organisms (MDROs) pose challenges due to treatment complexities and substantial morbidity and mortality. Susceptibility to colonization with these organisms and potential onward transmission if colonized (ie, infectivity) is influenced by the human microbiome and its dynamics. Disruptive effects of antibiotics on the microbiome imply potential indirect effects of antibiotics on C. difficile colonization. Mathematical models can help explore the relative impact of key pathways linking antibiotic use to C. difficile colonization, including the relationship between population-level antibiotic use and colonization prevalence. Methods: We built a compartmental model of long-term C. difficile colonization prevalence of nursing home residents (though malleable for any MDRO), allowing interactions between the microbiome and the colonization process. Based on proportional abundance of microbial taxa, we classified individuals into high and low α diversity groups, each further stratified into uncolonized or colonized with C. difficile. The rate of transition from the high to low microbiome diversity group was proportional to the population-level rate of antibiotic use. Transmission dynamics followed a susceptible–infectious–susceptible framework with the possibility for increased susceptibility and infectivity for the low-diversity microbiome group. First, as a comparator, we used a “null model” in which microbiome diversity did not influence host susceptibility or infectivity. Next, we sampled from realistic (literature informed) parameter ranges to analyze how the microbiome mediates the effect of antibiotics on colonization in this population. Results: Our analysis suggests that antibiotic use can catalyze colonization with C. difficile through interactions with the host microbiome, resulting in a sharp increase in colonization with a modest increase in antibiotic use (Fig 1). Increasing the population-level antibiotic use by 5% led to a median 24% increase in long-term colonization prevalence in the model (Fig 2). In contrast, increasing susceptibility or infectivity rates by 5% resulted in slightly higher increases in total colonization (27% and 29%, respectively). However, there was considerable uncertainty around these estimates, with interquartile ranges of up to 20% for some parameters (Fig 2). Conclusions: Higher population-level antibiotic use likely increases colonization by C. difficile through indirect effects of the microbiome. The increased colonization burden attributable to increasing antibiotic use may be substantial. With high uncertainty around some estimates, conducting observational studies to better understand key colonization and microbiome parameters (eg, the relative increase in susceptibility or infectivity with lower microbiome diversity) is critical for future efforts to estimate the impact of antibiotic use on colonization with C. difficile and MDROs.
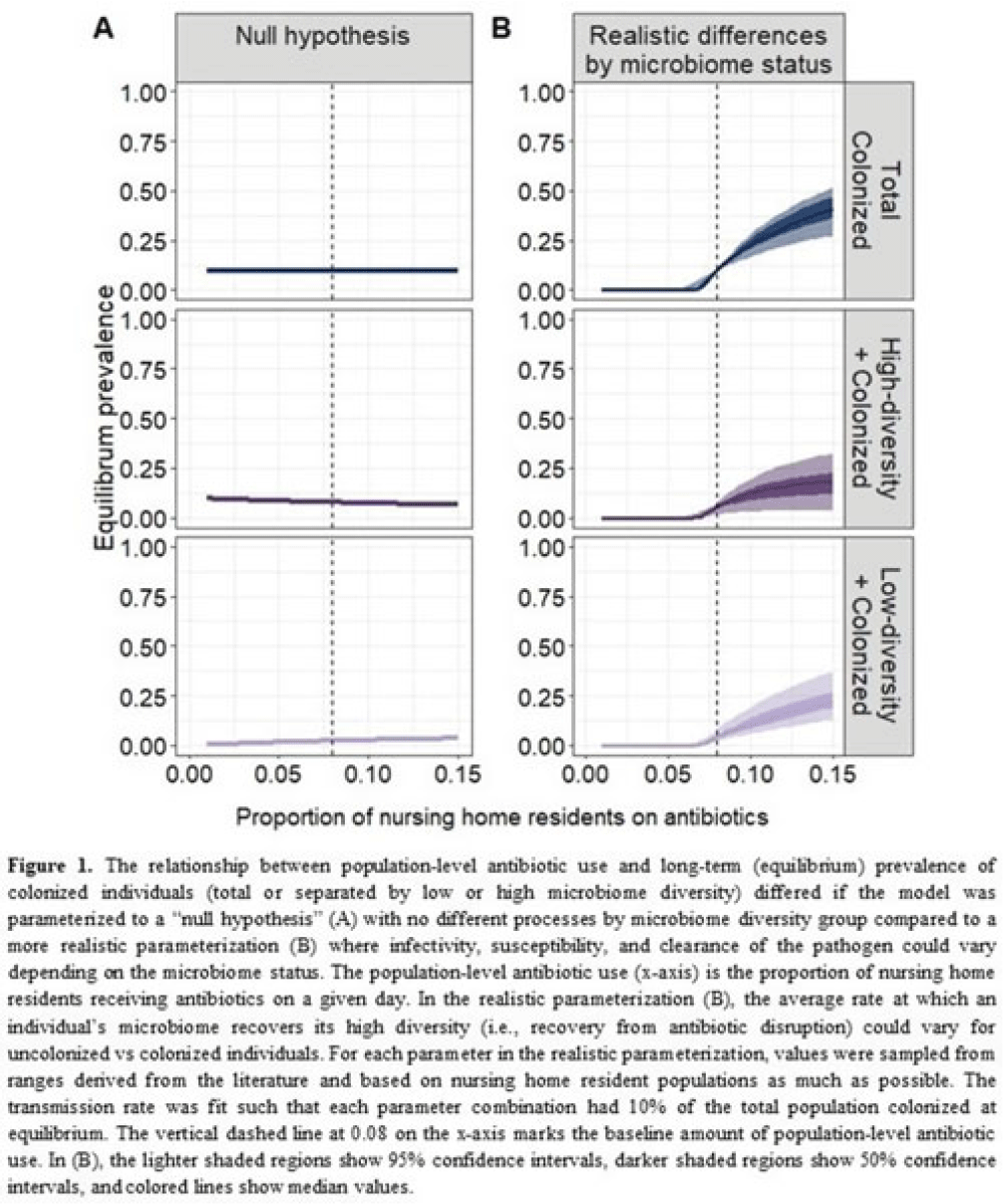
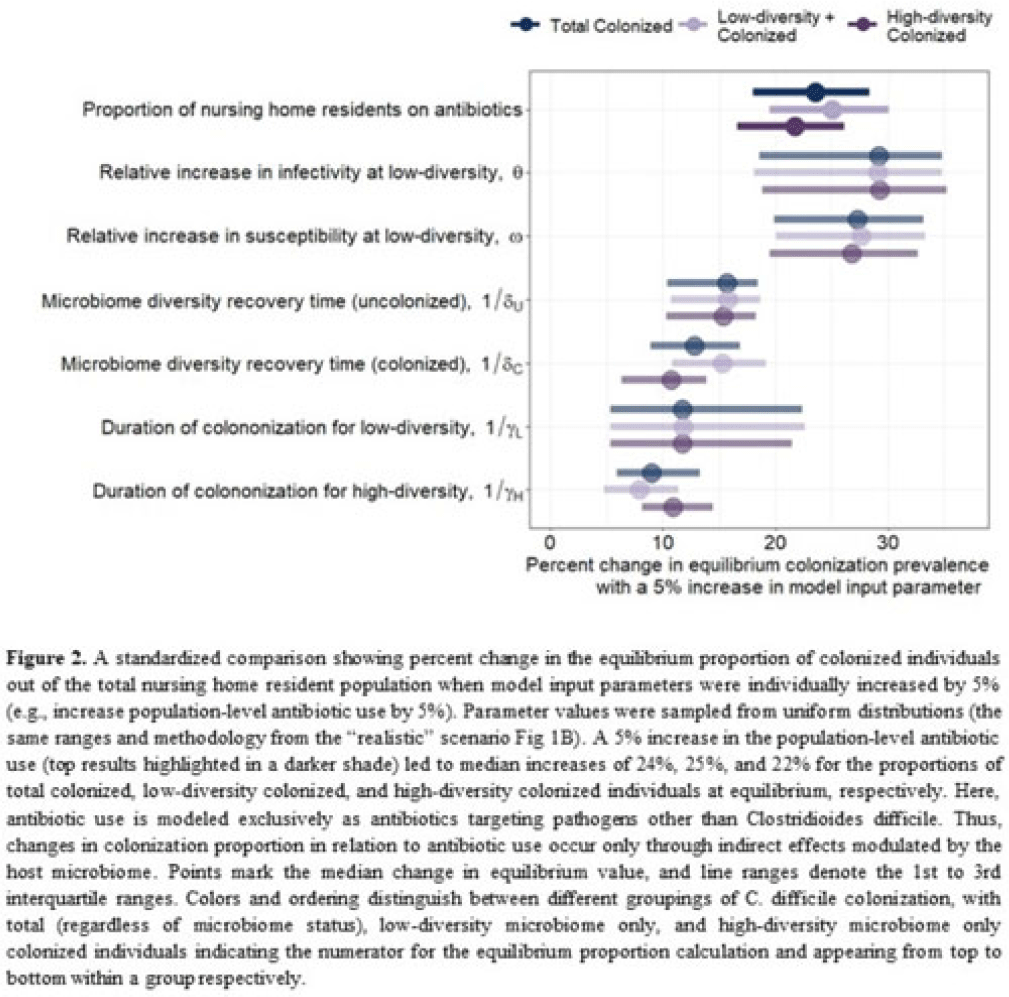
Disclosures: None
Decolonization of hospital patients may aid efforts to reduce transmission of carbapenem-resistant Enterobacterales
- Brajendra K. Singh, Prabasaj Paul, Camden D. Gowler, Sujan C. Reddy, Rachel B. Slayton
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s59-s60
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Multimodal approaches are often used to prevent transmission of antimicrobial-resistant pathogens among patients in healthcare settings; understanding the effect of individual interventions is challenging. We designed a model to compare the effectiveness of hand hygiene (HH) with or without decolonization in reducing patient colonization with carbapenem-resistant Enterobacterales (CRE). Methods: We developed an agent-based model to represent transmission of CRE in an acute-care hospital comprising 3 general wards and 2 ICUs, each with 20 single-occupancy rooms, located in a community of 85,000 people. The model accounted for the movement of healthcare personnel (HCP), including their visits to patients. CRE dynamics were modeled using a susceptible–infectious–susceptible framework with transmission occurring via HCP–patient contacts. The mean time to clearance of CRE colonization without intervention was 387 days (Zimmerman et al, 2013). Our baseline included a facility-level HH compliance of 30%, with an assumed efficacy of 50%. Contact precautions were employed for patients with CRE-positive cultures with assumed adherence and efficacy of 80% and 50%, respectively. Intervention scenarios included decolonization of culture-positive CRE patients, with a mean time to decolonization of 3 days. We considered 2 hypothetical intervention scenarios: (A) decolonization of patients with the baseline HH compliance and (B) decolonization with a slightly improved HH compliance of 35%. The hospital-level CRE incidence rate was used to compare the results from these intervention scenarios. Results: CRE incidence rates were lower in intervention scenarios than the baseline scenario (Fig. 1). The baseline mean incidence rate was 29.1 per 10,000 patient days. For decolonization with the baseline HH, the mean incidence rate decreased to 14.5 per 10,000 patient days, which is a 50.2% decrease relative to the baseline incidence (Table 1). The decolonization scenario with a slightly improved HH compliance of 35% produced a relative reduction of 71.9% relative to the baseline incidence. Conclusions: Our analysis shows that decolonization, combined with modest improvement in HH compliance, could lead to large decreases in pathogen transmission. In turn, this model implies that efforts to identify and improve decolonization strategies for better patient safety in health care may be needed and are worth exploring.
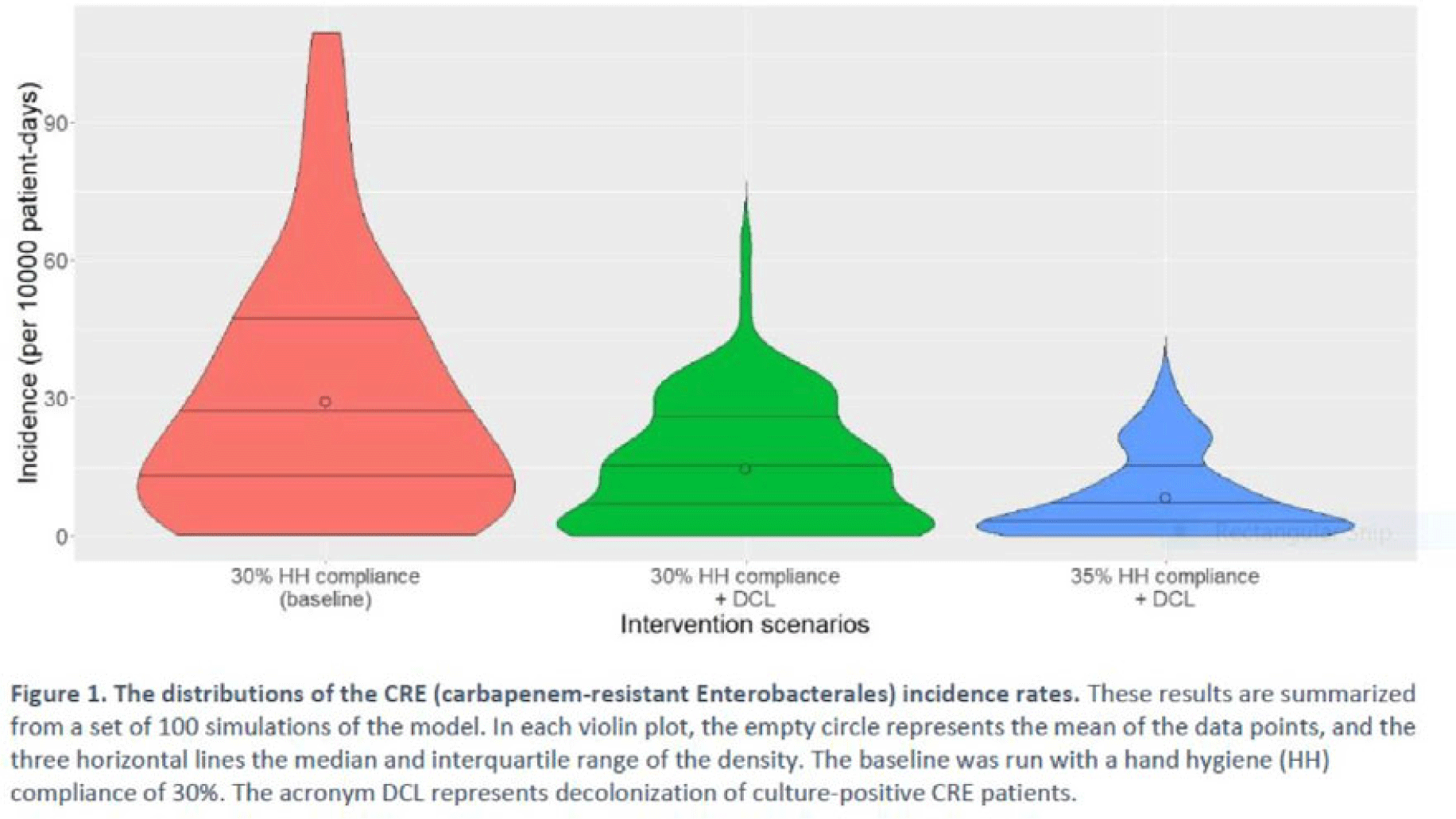
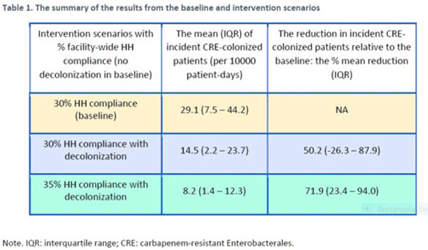
Disclosures: None
An interactive patient transfer network and model visualization tool for multidrug-resistant organism prevention strategies
- Rany Octaria, Samuel Cincotta, Jessica Healy, Camden Gowler, Prabasaj Paul, Maroya Walters, Rachel Slayton
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s120-s122
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The CDC’s new Public Health Strategies to Prevent the Spread of Novel and Targeted Multidrug-Resistant Organisms (MDROs) were informed by mathematical models that assessed the impact of implementing preventive strategies directed at a subset of healthcare facilities characterized as influential or highly connected based on their predicted role in the regional spread of MDROs. We developed an interactive tool to communicate mathematical modeling results and visualize the regional patient transfer network for public health departments and healthcare facilities to assist in planning and implementing prevention strategies. Methods: An interactive RShiny application is currently hosted in the CDC network and is accessible to external partners through the Secure Access Management Services (SAMS). Patient transfer volumes (direct and indirect, that is, with up to 30 days in the community between admissions) were estimated from the CMS fee-for-service claims data from 2019. The spread of a carbapenem-resistant Enterobacterales (CRE)–like MDROs within a US state was simulated using a deterministic model with susceptible and infectious compartments in the community and healthcare facilities interconnected through patient transfers. Individuals determined to be infectious through admission screening, point-prevalence surveys (PPSs), or notified from interfacility communication were assigned lower transmissibility if enhanced infection prevention and control practices were in place at a facility. Results: The application consists of 4 interactive tabs. Users can visualize the statewide patient-sharing network for any US state and select territories in the first tab (Fig. 1). A feature allows users to highlight a facility of interest and display downstream or upstream facilities that received or sent transfers from the facility of interest, respectively. A second tab lists influential facilities to aid in prioritizing screening and prevention activities. A third tab lists all facilities in the state in descending order of their dispersal rate (ie, the rate at which patients are shared downstream to other facilities), which can help identify highly connected facilities. In the fourth tab, an interactive graph displays the predicted reduction of MDRO prevalence given a range of intervention scenarios (Fig. 2). Conclusions: Our RShiny application, which can be accessed by public health partners, can assist healthcare facilities and public health departments in planning and tailoring MDRO prevention activity bundles.
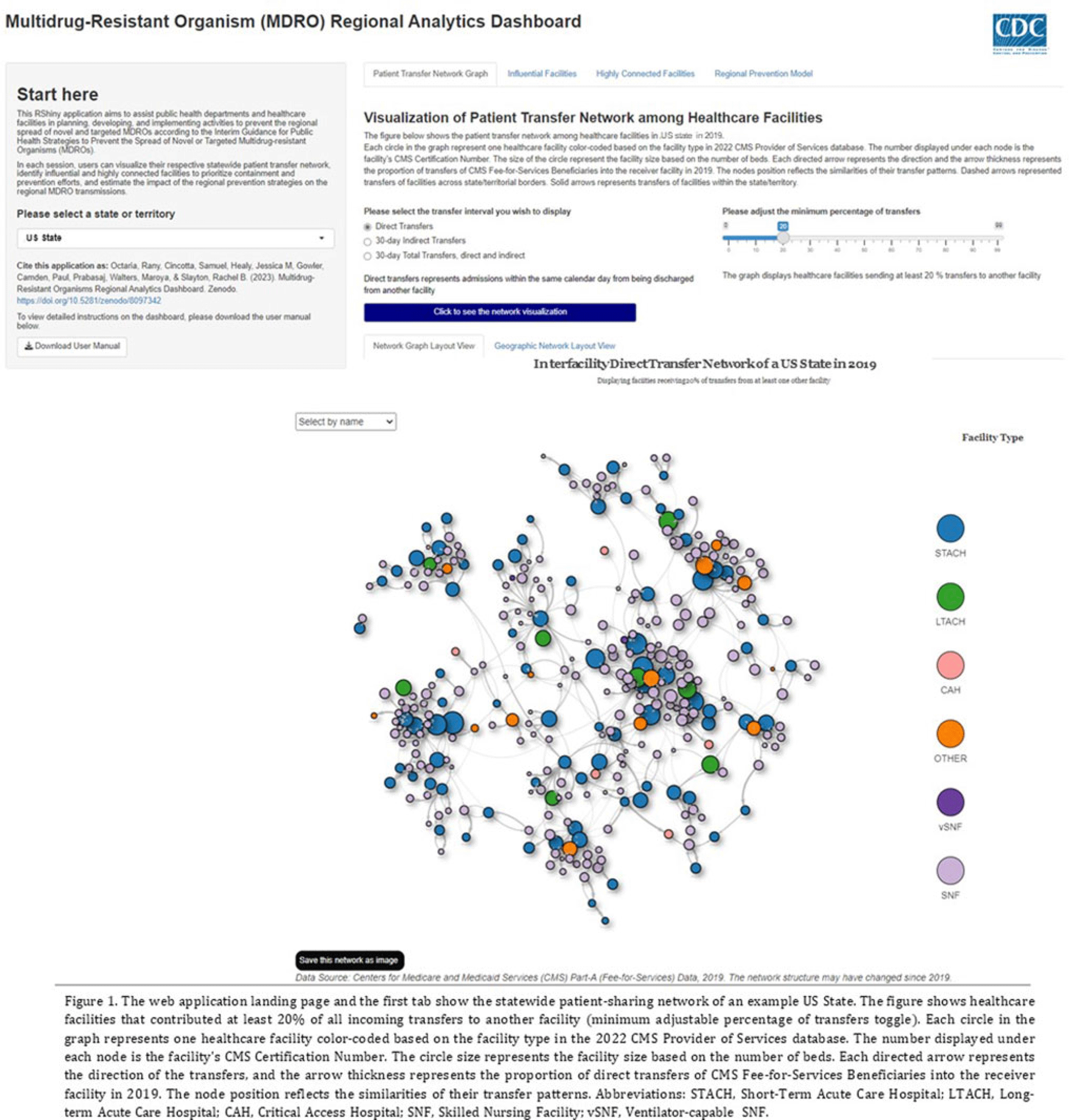
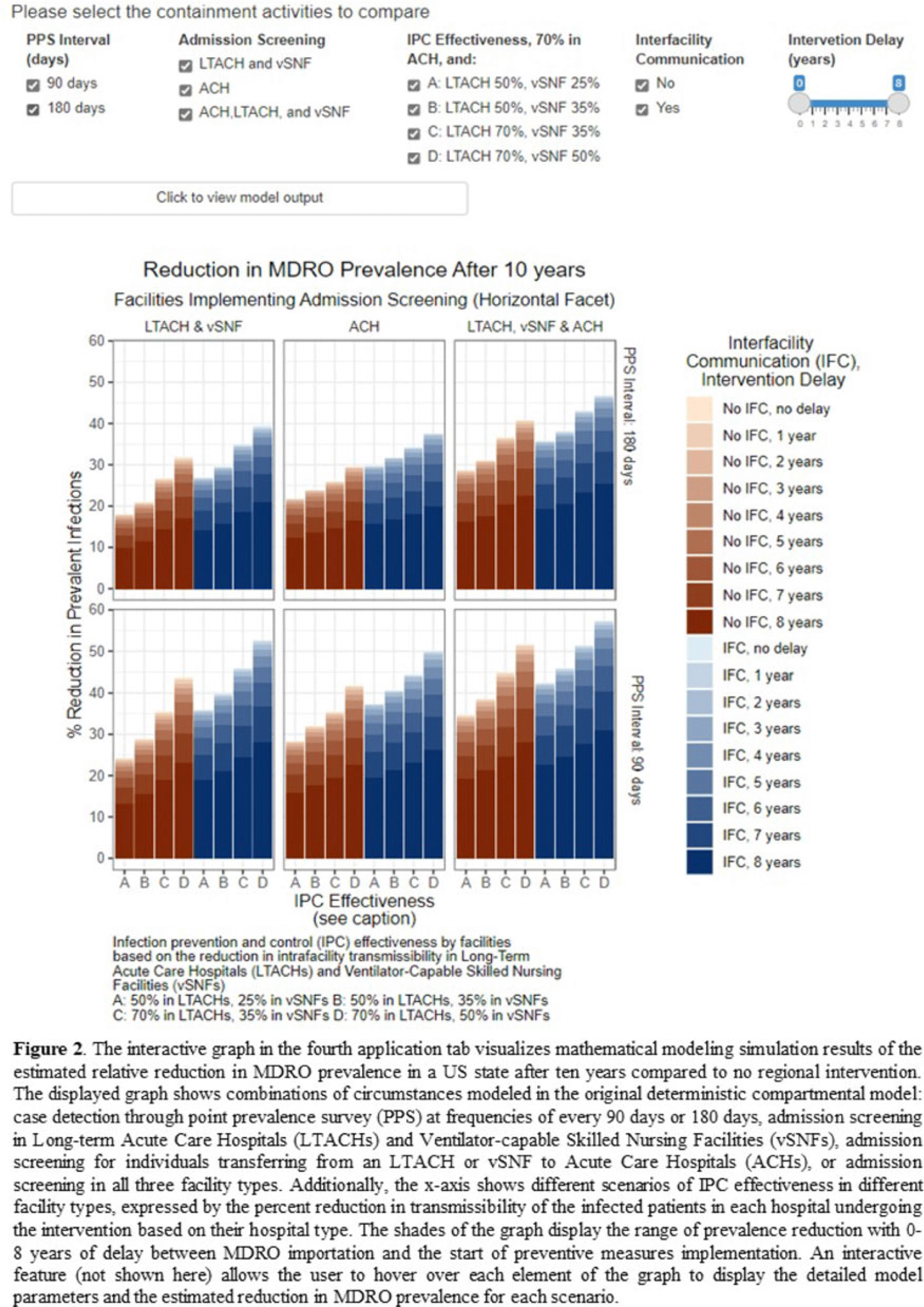
Disclosures: None









