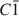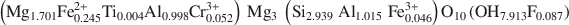4 results
Crystallographic features and cleavage nanomorphology of chlinochlore: Specific applications
- Giovanni Valdrè, Daniele Malferrari, Maria Franca Brigatti
-
- Journal:
- Clays and Clay Minerals / Volume 57 / Issue 2 / April 2009
- Published online by Cambridge University Press:
- 01 January 2024, pp. 183-193
-
- Article
- Export citation
-
Natural and synthetic micas have been used widely as substrates to study biological systems; but, as in the case of negatively charged DNA, anionic charge repulsion may render micas a less than ideal templating surface for many biological systems. The purpose of this study was to investigate the potential for the chlorite clinoclore, which contains a positively charged interlayer octahedral sheet, to serve as a substrate for DNA adsorption. The relationships between clinochlore cleavage characteristics, in terms of nano-morphology, and surface potential are investigated, as are its average crystal chemistry and topology. That the structural features of clinochlore can be used successfully to condense, order, and self assemble complex biomolecules, such as DNA is also proven.
A natural IIb-4 clinochlore [\$\end{document}
 symmetry, unit-cell parameters a = 0.53301(4); b = 0.92511(6); c = 1.4348(1) (nm); α = 90.420(3); β = 97.509(3); γ = 89.996(4) (°)] with chemical composition \$\end{document}
symmetry, unit-cell parameters a = 0.53301(4); b = 0.92511(6); c = 1.4348(1) (nm); α = 90.420(3); β = 97.509(3); γ = 89.996(4) (°)] with chemical composition \$\end{document}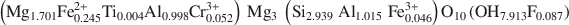 was selected. The octahedral sites of the silicate layer (<M(1)−O> = 0.2080 nm and <M(2)−O> = 0.2081 nm) are equal and occupied by Mg, whereas the octahedral sites in the interlayer M(3) and M(4) (<M(3)−O> = 0.2088 nm and <M(4) − O> = 0.1939 nm) show different sizes and are mostly completely occupied by divalent (Mg2+ and Fe2+) and trivalent (Al3+) cations, respectively.
was selected. The octahedral sites of the silicate layer (<M(1)−O> = 0.2080 nm and <M(2)−O> = 0.2081 nm) are equal and occupied by Mg, whereas the octahedral sites in the interlayer M(3) and M(4) (<M(3)−O> = 0.2088 nm and <M(4) − O> = 0.1939 nm) show different sizes and are mostly completely occupied by divalent (Mg2+ and Fe2+) and trivalent (Al3+) cations, respectively.The clinochlore cleaved surface is present in two forms: (1) the stripe type (0.40 nm in height, up to several micrometers long and ranging from some nanometers to a few microns in lateral size); and (2) the triangular type (0.40 nm in height). Both features may result either from interlayer sheets whose cleavage weak directions are related to the different M(3) and M(4) site occupancy, or from weak interlayer bonding along specific directions to the 2:1 layer underneath. The cleaved surface, particularly at the cleaved edges, presents high DNA affinity, which is directly related to an average positive surface and ledge potential.
The 2M1 dioctahedral mica polytype: A crystal chemical study
- Maria Franca Brigatti, Daniele Malferrari, Marco Poppi, Luciano Poppi
-
- Journal:
- Clays and Clay Minerals / Volume 53 / Issue 2 / April 2005
- Published online by Cambridge University Press:
- 01 January 2024, pp. 190-197
-
- Article
- Export citation
-
The structure of dioctahedral true micas such as muscovite and celadonitic muscovite (2M1 polytype, space group C2/c) is mostly affected by variations of the octahedral Al (VIAl) content. Crystals with greater Mg, Fe substitutions (i.e. celadonitic muscovite) reduce the dimensional difference between the larger trans-oriented M1 site and smaller cis-oriented M2 octahedral site. The octahedral anionic position O4 is displaced from the center of the hexagon, defined by 031 and 032 oxygen atoms (i.e. ‘octahedral hexagon’), both on and off the (001) plane. The distance between interlayer cation A and O4 is smaller in more substituted species, thus providing different orientations of the O4−H vector, as a function of VIAl. Octahedral distances (<M2−O3> and <M2−O4> are expressed as a function of cell parameters and VIA1 content, thus allowing an approximate estimate of site dimensions. These approximations are useful when a detailed structural refinement is not available. In celadonitic muscovite, the octahedral hexagon mean edge (<O31−O32>Hex) is not significantly affected by VIA1 content. The VIA1 increase produces both a decrease in cell lateral dimensions and a distorted ‘octahedral hexagon’. The decrease in a and b is consistent with a decrease of <O31−O32>Hex, whereas the distortion of the’ octahedral hexagon’ is consistent with an increase of (<031–032>Hex), because an irregular hexagon produces a longer mean edge than a regular hexagon of equal area.
The tetrahedral mean basal edge (VI<O−O>bassal) is reduced as celadonitic substitution progresses. The tetrahedral rotation angle, α was thus found to increase from celadonite to muscovite. However, in muscovite with VIAl content between 1.8 and 2.0 atoms per formula unit (a.p.f.u.), α approaches a saturation value, thus showing a proportional increase of tetrahedral and octahedral sheet lateral dimensions. Furthermore, α variation allows a coarse approximation of the threshold VIAl content, below which celadonitic substitution may not progress.
Baseline Studies of the Clay Minerals Society Source Clay Montmorillonite STx-1b
- Elena Castellini, Daniele Malferrari, Fabrizio Bernini, Maria Franca Brigatti, German Rafael Castro, Luca Medici, Adele Mucci, Marco Borsari
-
- Journal:
- Clays and Clay Minerals / Volume 65 / Issue 4 / August 2017
- Published online by Cambridge University Press:
- 01 January 2024, pp. 220-233
-
- Article
- Export citation
-
For more than forty years, The Clay Minerals Society has dispensed a set of source clays which have enabled a large number of researchers to work on similar materials. Many of these source clays remained unchanged over the years but, conversely, other clays have gone out of stock and thus were replaced. This was the fate of montmorillonite STx-1a, which was replaced by STx-1b. Although STx-1a and STx-1b share many basic chemical and mineralogical features, some minor differences exist that can affect behavior. A baseline characterization of the source clay STx-1b, which was the objective of this study, was, therefore, necessary to provide researchers a tool useful not only for new investigation but also to compare new results obtained on STx-1b with literature data on STx-1a. This characterization was gained using traditional and advanced methods that included: 1) chemical composition (major and trace elements); 2) cation exchange capacity determination; 3) thermal analyses coupled with evolved gas mass spectrometry; 4) quantitative mineralogical characterization using powder X-ray diffraction and Rietveld- RIR (Reference Intensity Ratio) refinement; 5) X-ray absorption spectroscopy at the Fe K-edge; 6) diffuse reflectance ultraviolet-visible and infrared spectroscopies; and 7) 29Si, 27Al, and 1H magic-angle spinning nuclear magnetic resonance measurements. According to this multi-analytical approach, the chemical formula for STx-1b is [4](Si7.753Al0.247) [6](Al3.281Mg0.558Fe0.136Ti0.024Mn0.002) [12](Ca0.341Na0.039 K0.061)O20(OH)4.
Characterisation of fibrous ferrierite in the rhyolitic tuffs at Lovelock, Nevada, USA
- Alessandro Zoboli, Dario Di Giuseppe, Cecilia Baraldi, Maria Cristina Gamberini, Daniele Malferrari, Giancarlo Urso, Magdalena Lassinantti Gualtieri, Mark Bailey, Alessandro F. Gualtieri
-
- Journal:
- Mineralogical Magazine / Volume 83 / Issue 4 / August 2019
- Published online by Cambridge University Press:
- 22 April 2019, pp. 577-586
-
- Article
- Export citation
-
Ferrierite is the name for a series of zeolite-group of minerals which includes three species with the same ferrierite framework (FER) crystal structure but different extra-framework cations. Recent studies have shown that ferrierite can exhibit a fibrous-asbestiform crystal habit and may possess the same properties as carcinogenic fibrous erionite. Characterisation of the ferrierite in and around a mine location will be helpful in assessing the potential for toxic outcomes of exposure in the mine and any local population.
The zeolite-rich tuff deposit of Lovelock, Nevada, USA is the largest occurrence of diagenetic ferrierite-Mg. A previous survey reported that ferrierite hosted in these rocks displays a fibrous morphology. However, these observations concerned a limited number of samples and until now there has been little evidence of widespread occurrence of fibrous ferrierite in the Lovelock deposit.
The main goal of this study was to perform a mineralogical and morphometric characterisation of the tuff deposit at Lovelock and evaluate the distribution of fibrous ferrierite in the outcrop. For this purpose, a multi-analytical approach including powder X-ray diffraction, scanning and transmission microscopies, micro-Raman spectroscopy, thermal analyses, and surface-area determination was applied.
The results prove fibrous ferrierite is widespread and intermixed with mordenite and orthoclase, although there are variations in the spatial distribution in the bedrock. The crystal habit of the ferrierite ranges from prismatic to asbestiform (elongated, thin and slightly flexible) and fibres are aggregated in bundles. According to the WHO counting criteria, most of the ferrierite fibres can be classified as breathable. While waiting for confirmatory in vitro and in vivo tests to assess the actual toxicity/pathogenicity potential of this mineral fibre, it is recommended to adopt a precautionary approach for mining operations in this area to reduce the risk of exposure.