3 results
Three-day antibiotic duration in patients with pneumonia: A sixty-eight–hospital cohort
- Valerie Vaughn, Lindsay Petty, David Ratz, Elizabeth McLaughlin, Tawny Czilok, Jennifer Horowitz, Anurag Malani, Danielle Osterholzer, Scott Flanders, Tejal Gandhi
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s22
-
- Article
-
- You have access Access
- Open access
- Export citation
-
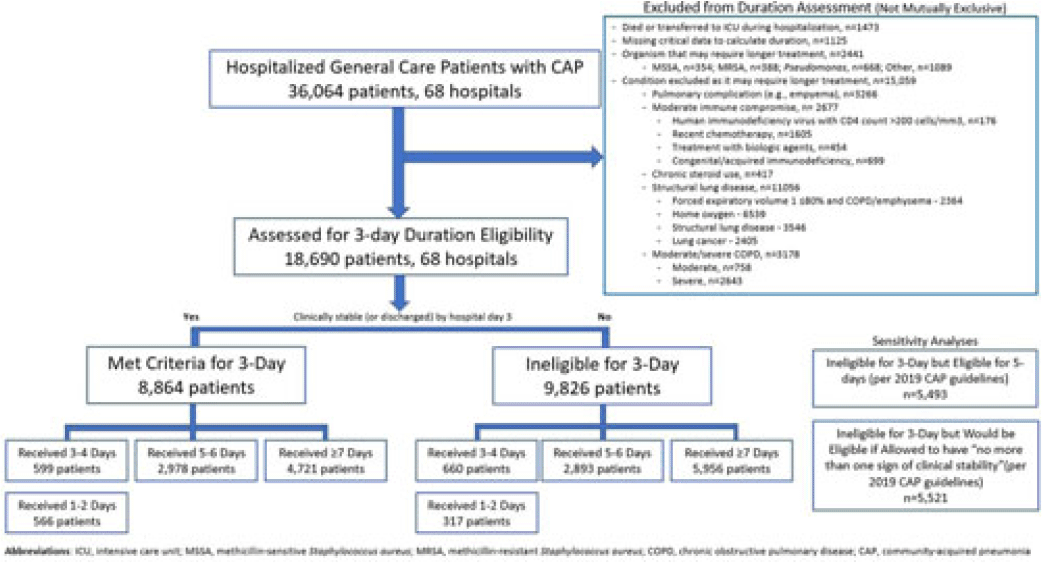
Background: Since 2019, community-acquired pneumonia (CAP) guidelines have recommended hospitalized patients be treated until clinical “stability and for no less than 5 days.” However, randomized trials have reported that, in patients who stabilize by hospital day 3, very short antibiotic durations (eg, 3 days) are noninferior to longer durations. How these trial results relate to real-world practice is unknown. Methods: Using a 68-hospital cohort study of hospitalized, general-care adults with CAP, we aimed to (1) quantify the percentage of patients who—according to trial criteria—qualify for a 3-day antibiotic duration, (2) quantify the percentage who actually received a 3-day duration, and (3) assess 30-day outcomes. Patients were considered to have CAP if they had a pneumonia discharge diagnosis and met clinical criteria for CAP. Patients with concomitant infections (including COVID-19), admission to intensive care, or severe immunocompromise were not included. Results: Between February 23, 2017, and August 3, 2022, 36,064 patients with CAP were included. Of those, 48.2% (9,826 of 36,064) were excluded due to a condition or organism ineligible for the 3-day treatment (Fig. 1). Of the 18,690 patients remaining, 52.6% (9,826) were unstable on day 3 and thus were ineligible for the 3-day treatment. Therefore, of all 36,064 patients, only 8,864 (24.6%) would be eligible under trial criteria for a 3-day treatment. Notably, 5,493 (55.9%) of 9,826 patients unstable on day 3 would be eligible for 5 days of treatment under national guidelines. In practice, use of 3–4-day treatment was rare, occurring in 599 (6.8%) of 8,864 patients eligible for a 3-day treatment versus 660 (6.7%) of 9,826 patients unstable on day 3 (P = .945). Use of 3–4-day treatment increased over time and comorbidities that could mimic CAP or a negative procalcitonin were more common in patients who received a 3–4-day treatment whereas specific symptoms of CAP were less common (Fig. 2). After adjustments, patients eligible for a 3-day duration who received a 3–4 day treatment versus a ≥5-day treatment had higher 30-day mortality (aOR, 1.87; 95% CI, 1.32–2.64) and readmission (aOR, 1.35; 95% CI, 1.17–1.56). Conclusions: Across 68 hospitals, <25% of patients hospitalized with CAP would be eligible for a 3-day antibiotic treatment. Though increasing over time, there was little use of 3–4-day treatments and, when prescribed, outcomes were worse, potentially due to CAP misdiagnosis. Given the small number of patients eligible for 3-day treatment, and the potential harm with too-short durations, it may be prudent to focus on increasing the use of 5-day treatments.
Disclosures: None
Risk Factors and outcomes associated with inappropriate empiric broad-spectrum antibiotic use in hospitalized patients with community-acquired pneumonia
- Tejal Gandhi, Lindsay Petty, Valerie Vaughn, Anurag Malani, David Ratz, Tawny Czilok, Jennifer Horowitz, Elizabeth McLaughlin, Lisa Dumkow, Stephanie Burdick, Danielle Osterholzer, Mariam Younas, Steven Bernstein, Scott Flanders
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s31-s32
-
- Article
-
- You have access Access
- Open access
- Export citation
-
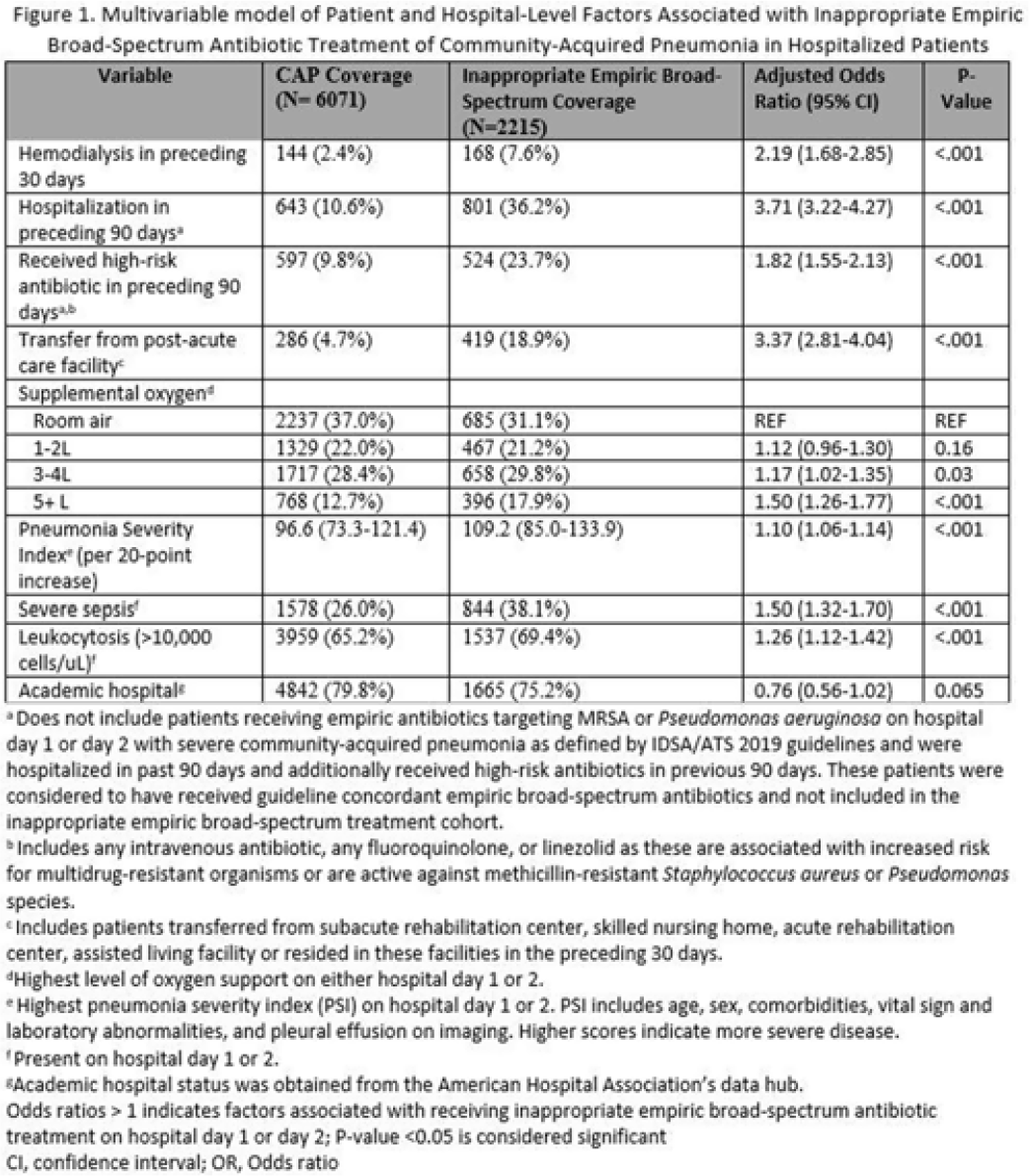
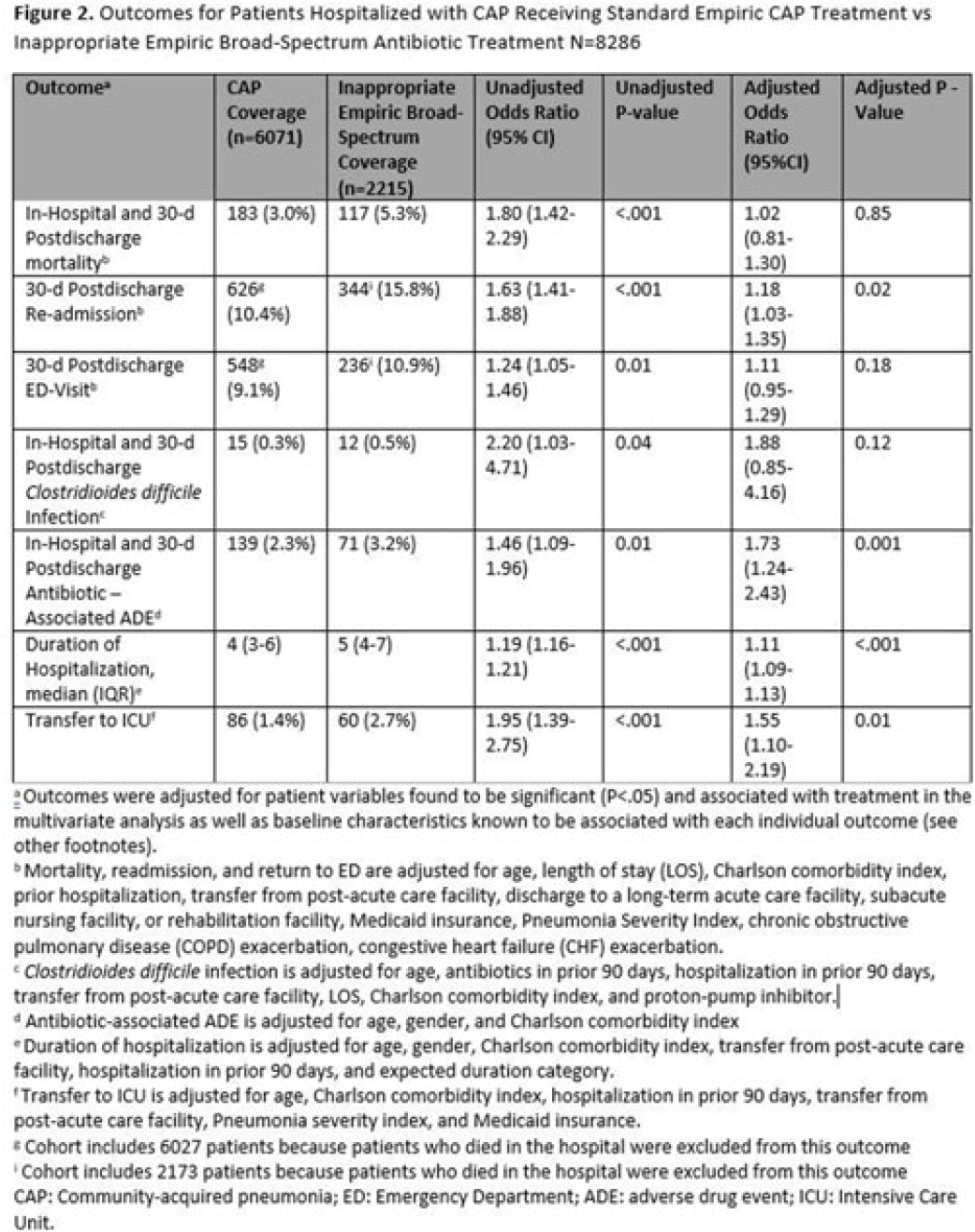
Background: Inappropriate broad-spectrum antibiotic use targeting methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa can result in increased adverse events, antibiotic resistance, and Clostridioides difficile infection. In 2019, revised ATS/IDSA community-acquired pneumonia (CAP) guidelines removed healthcare-associated pneumonia (HCAP) as a clinical entity and modified patient factors warranting empiric broad-spectrum antibiotic (BSA) use. As a result, most patients hospitalized with CAP should receive empiric antibiotics targeting standard CAP pathogens. Based on revised guidelines, we evaluated predictors and outcomes associated with inappropriate BSA use among hospitalized patients with CAP. Methods: Between November 2019 and July 2022, trained abstractors collected data on non-ICU adult medical patients admitted with CAP at 67 Michigan hospitals who received either an inappropriate empiric BSA on hospital day 1 or 2 or a standard CAP regimen. Inappropriate empiric BSA use was defined as use of an anti-MRSA or anti-pseudomonal antibiotic in a patient eligible for standard CAP coverage per IDSA guidelines. Patients with immune compromise, moderate or severe chronic obstructive pulmonary disease (COPD), pulmonary complication, or guideline-concordant treatment with BSA were excluded. Data collected included comorbidities, antibiotic use and hospitalizations in the preceding 90 days, cultures in the preceding year, signs or symptoms of pneumonia, hospital characteristics, and 30-day postdischarge patient outcomes. Data were collected through chart review and patient phone calls. Predictors of inappropriate empiric BSA were evaluated using logistic general estimating equation (GEE) models, accounting for hospital-level clustering. We assessed the effect of inappropriate empiric BSA (vs standard CAP therapy) on 30-day patient outcomes using logistic GEE models controlling for predictors associated with the outcome and probability of treatment. Results: Of 8,286 included patients with CAP, 2,215 (26.7%) were empirically treated with inappropriate BSA. The median BSA treatment was 3 days (IQR, 2.5). After adjustments, factors associated with inappropriate empiric BSA treatment included hospitalization or treatment with high-risk antibiotics in preceding 90 days, transfer from a postacute care facility, hemodialysis, support with ≥3 L supplemental oxygen, severe sepsis, leukocytosis, and higher pneumonia severity index (Fig. 1). After adjustments, patients with inappropriate empiric BSA treatment had higher readmissions 30 days after discharge, more transfers to the intensive care unit, more antibiotic-associated adverse events, and longer hospitalizations (Fig. 2). Conclusions: Patients hospitalized with CAP often received inappropriate BSA as empiric coverage, and this inappropriate antibiotic selection was associated with worse patient outcomes. To improve patient outcomes, stewardship efforts should focus on reducing inappropriate BSA use in patients hospitalized for CAP with historic HCAP risk factors or severe CAP without other guideline-directed indications for BSA.
Financial support. H.M.S. initiative is underwritten by Blue Cross and Blue Shield of Michigan.
Disclosures: None
Predictors of COVID-19 Mortality in Residents of Flint, Michigan: Effect of Age, Gender, Smoking, and Health Plan
- Mariam Younas, Danielle Osterholzer, Philip McDonald, Carlos Rios-Bedoya, Ghassan Bachuwa, Sherry Demian, Smit Deliwala, Lalida Kunaprayoon, Harini Lakshman, Thulasi Beere, Uyoyo Omaduvie
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s57-s58
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Current literature suggests that older age, hypertension, and diabetes mellitus confer a significant increased risk of mortality among patients with COVID-19. The purpose of this study is to further characterize the predictors of mortality in patients with COVID-19 in residents of Flint, Michigan, based on variables such as gender, age, smoking status, health insurance plan, and comorbidities. Methods: Hurley Medical Center, is a 443-bed public, nonprofit, teaching medical center located in Flint, Michigan. In total, 289 consecutive adult patients (aged ≥18 years) with confirmed SARS-CoV-2 infection by nasal polymerase chain reaction (PCR), admitted and discharged from our facility from March 2020 through June 2020, were retrospectively analyzed. Results: During the 4-month study period, the overall in-hospital case fatality rate (CFR) was 18% (51 of 289), with highest CFR in the age group aged 60–69 years (36%; P = .06). Nonsurvivors tended to be older with mean age of 67 years (95% CI, 61.6–71.8) versus survivors with mean age of 60 years (95% CI, 57.7–62.4). Highest mortality was seen in patients with Medicare or Medicaid as their sole health plan (39%, P = .59). Men comprised 51% (148 of 289) of the cohort with CFR of 21% versus 14% in females. Females tended to be younger with a higher body mass index (BMI) than their male counterparts (mean age of 58 years, mean BMI of 35 in women vs a mean age of 62 and BMI of 29 in men). A higher proportion of deceased were active smokers (51%; P = .02). CFR was highest in patients with hypertension (92%,; P = .15), followed by diabetes (44%; P = .85), chronic kidney disease (CKD) (31%; P = .10), obstructive sleep apnea (OSA) (28%; P = .25), asthma (22%; P = .64), and coronary artery disease (22%; P = .34). It was lowest in patients with end-stage renal disease (3%; P = .69). Conclusions: Our study suggests trends towards higher mortality with male sex, hypertension and diabetes, along with other comorbidities. Smoking seems to be a strong predictor of mortality in this cohort. Further studies are needed to ascertain the relationship between possible risk factors with COVID-19 mortality in residents of Flint, Michigan. Describing and understanding the potential risk factors is the key to improving outcomes in this population.
Funding: No
Disclosures: None






