3 results
Comparison of clinical antibiotic susceptibility testing interpretations to CLSI standard interpretations
- Erin Hitchingham, Ashley Gambrell, Raquel Villegas, Daniel Muleta
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s114
-
- Article
-
- You have access Access
- Open access
- Export citation
-
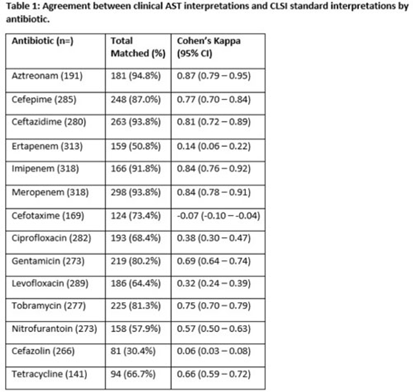
Background: Clinical antibiotic susceptibility testing (AST) interpretations based on minimum inhibitory concentrations (MIC) breakpoints are important for both clinical decision making and some reportable condition criteria. Standardization of MIC breakpoints across clinical laboratories is lacking; AST instruments are often validated for outdated Clinical and Laboratory Standards Institute (CLSI) MIC breakpoint guidelines. In this study, we analyzed the agreement between the reported clinical laboratory AST interpretations and the guideline CLSI interpretation. Methods: Clinical laboratory AST data collected from the Multisite Gram-Negative Surveillance Initiative (MuGSI) carbapenem-resistant Enterobacterales (CRE) surveillance program in Tennessee between 2019 and 2021 were utilized. MIC values from the clinical instrument were used to calculate CLSI standard interpretations following the 2019–2021 CLSI M100 guidelines. Agreement between the clinical laboratory and CLSI interpretations of the reported MIC values were measured using a weighted Cohen κ calculated in SAS version 9.4 software. Total matches were isolates with identical CLSI and clinical laboratory interpretations. Results: In total, 14 antibiotics were assessed. Of those, 9 antibiotics had at least moderate agreement (κ > 0.41) between interpretations. Agreement between the clinical laboratory and the CLSI interpretations were near perfect (κ > 0.81) for 3 antibiotics. Agreement between the clinical laboratory and the CLSI interpretations were poor for cefazolin (0.06) and ertapenem (0.14). Cefotaxime (−0.07) was the only antibiotic that suggested no agreement. Conclusions: Of the antibiotics included in the analysis, 36% had less than moderate agreement between clinical laboratory and CLSI AST interpretations. Given the increases in antimicrobial resistance globally and the emphasis placed on antibiotic stewardship, standardization across clinical AST panels should be prioritized. Inconsistencies have the potential to contribute to inappropriate antibiotic use in addition to under- or overidentification of reportable conditions, including CRE.
Disclosures: None
Identifying COVID-19 clusters in Tennessee long-term care facilities based on weekly staff vaccination rates
- Marissa Turner, Ashley Gambrell, Erin Hitchingham, Simone Godwin
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s52
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: In September 2021, the CMS mandated that long-term care facility (LTCF) healthcare workers be vaccinated for COVID-19 unless medically or religiously exempt. Vaccinating healthcare workers reduces transmission of COVID-19 among patients and workers, reducing the risk of illness among residents and patients. We examined the relationship between COVID-19 clusters and staff vaccination rates in Tennessee LTCFs. Methods: COVID-19 cluster data were collected using REDCap from January 3, 2021, to September 25, 2022, and LTCF vaccination rates were collected from the NHSN. Clusters were identified in facilities with 2 or more cases. The staff vaccination rate 2 weeks prior to the cluster was used, accounting for the lag time between vaccination dose and reaching full immunity. We selected 75% as the critical immunization threshold. The facility case rate was calculated per 100 beds. A test was performed to determine whether reaching the critical vaccination threshold was associated with cluster occurrence. The relationship between vaccination rate and case number was tested using Pearson correlation. Statistical analyses were conducted using SAS version 9.4 software. Results: The average staff vaccination rate when NHSN first required long-term care facilities to report rates rose from 47% in June 2021 to 83% in September 2022. In total, 806 clusters were identified with 20,868 combined weeks from all facilities being reported after merging facilities’ weekly vaccine percentage rates with cluster data. Most weeks from all facilities did not identify a cluster (n = 20,064, 96.15%) and did not meet the critical immunization threshold (n = 11,050, 52.95%). The association between a cluster occurring and a facility meeting the threshold was significant (χ2 = 5.41; df = 1; P 95% CI, .7327–.9740). The Pearson correlation coefficient between vaccination rate and case number was 0.05560 (P = .2894). Conclusions: There was a significant association between facilities not reaching the immunization threshold and presence of a COVID-19 cluster. The facility case rate was not correlated with staff vaccination rate; however, a limitation of this analysis was that resident vaccination was not tested. Another limitation was that medical and religious exemptions could not be differentiated. Healthcare staff should consider getting vaccinated, if able, to reduce the risk of COVID-19 and to keep staff and residents safe from COVID-19.
Disclosures: None
Characteristics of patients positive for COVID-19 and multidrug-resistant organisms in Tennessee, 2020–2021
- Carolyn Stover, Erin Hitchingham, Kristina McClanahan, Zoe Durand, Rany Octaria, Christopher Wilson, Allison Chan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s80-s81
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Multidrug-resistant organisms (MDROs) are a global threat. To track and contain the spread, the Tennessee Department of Health (TDH) performs targeted surveillance of carbapenemase-producing and pan-nonsusceptible organisms. When these MDROs are identified, TDH conducts a containment response and collects epidemiological data, which includes risk factors such as indwelling devices and previous hospitalizations. The impact of the COVID-19 pandemic on these MDROs is not well understood. Therefore, we have described the characteristics of cases positive for both COVID-19 and select MDROs. Methods: MDRO investigation data from January 1, 2020–September 30, 2021 were matched with all COVID-19 case data from the TDH statewide surveillance system, National Electronic Disease Surveillance System Base System. MDRO-positive date was defined as the specimen collection date; COVID-19 case date was first defined as the date of symptom onset and if missing, then diagnosis date, and investigation creation date, respectively. Descriptive statistics and Fisher exact tests were calculated using SAS version 9.4 software. Results: Among 336 MDRO cases, 50 had a reported SARS-CoV-2–positive result. MDRO types were Enterobacterales (CRE) (n = 31), Acinetobacter spp (CRA) (n = 18), and Pseudomonas aeruginosa (n = 1). Of these 50 cases, 20 were MDRO-positive before and 30 days after the COVID-19 case date, respectively. Of the 18 CRA cases, 16 (89%), were positive after the COVID-19 case date, compared to 13 (42%) among 31 CRE cases (P < .01). Also, 35 patients (70%) had a record of hospitalization, and 22 (63%) had their MDRO specimen collected after the COVID-19 case date (P = .37). Of these 22 patients, 4 had their MDRO specimen collected during their COVID-19 hospitalization, with an average duration from admission to MDRO collection date of 17 days (range, 4–36). Among the 50 coinfected cases, 8 died, 7 (88%) of whom were MDRO-positive after their COVID-19 case date. Data on indwelling devices at time of MDRO positivity were completed for 17 cases; 14 had an indwelling device and, among these, 13 (93%) were MDRO-positive after their COVID-19 case date. Conclusions: MDRO cases with specimen collections after COVID-19 comprised the majority of hospitalized patients, patients who died, and patients with indwelling devices compared to those with MDROs collected before their COVID-19 case date. These results show a stark difference with CRA as the most common MDRO among post–COVID-19 cases. Our data were limited by reporting gaps. We recognize that patients can remain colonized with MDROs for lengthy durations, which could have result in undetected MDRO cases prior to the COVID-19 case date. More data and analyses are needed to make targeted public health recommendations. However, these findings highlight the burden of MDROs among COVID-19 cases. including adverse health outcomes.
Funding: None
Disclosures: None




