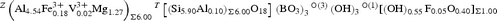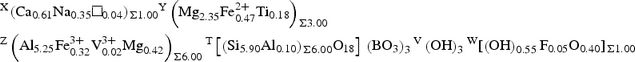2 results
Uvite, CaMg3(Al5Mg)(Si6O18)(BO3)3(OH)3(OH), a new, but long-anticipated mineral species of the tourmaline supergroup from San Piero in Campo, Elba Island, Italy
- Ferdinando Bosi, Cristian Biagioni, Federico Pezzotta, Henrik Skogby, Ulf Hålenius, Jan Cempírek, Frank C. Hawthorne, Aaron J. Lussier, Yassir A. Abdu, Maxwell C. Day, Mostafa Fayek, Christine M. Clark, Joel D. Grice, Darrell J. Henry
-
- Journal:
- Mineralogical Magazine / Volume 86 / Issue 5 / October 2022
- Published online by Cambridge University Press:
- 31 May 2022, pp. 767-776
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Uvite, CaMg3(Al5Mg)(Si6O18)(BO3)3(OH)3(OH), is a new mineral of the tourmaline supergroup. It occurs in the Facciatoia quarry, San Piero in Campo, Elba Island, Italy (42°45′04.55″N, 10°12′50.89″E) at the centre of a narrow (2–3 cm wide) vein composed of aggregates of dark brown to black tourmaline, penetrating (magnesite + dolomite)-rich hydrothermally altered metaserpentinite. Crystals are euhedral and up to 1 cm in size, brown with a vitreous lustre, conchoidal fracture and grey streak. Uvite has a Mohs hardness of ~7½, a calculated density of 3.115 g/cm3 and is uniaxial (–). Uvite has trigonal symmetry, space group R3m, a = 15.9519(10) Å, c = 7.2222(5) Å, V = 1597.3(1) Å3 and Z = 3. The crystal structure was refined to R1 = 1.77% using 1666 unique reflections collected with MoKα X-rays. Crystal-chemical analysis resulted in the empirical crystal-chemical formula $^X ({\rm Ca}_{0.61}{\rm Na}_{{0.35}} \square_{{0.04}})_{\Sigma 1.00}{}^{Y} \left( {{\rm Mg}_{1.50}{\rm Fe}^{2 + }_{0.47} {\rm Al}_{0.71}{\rm Fe}^{3 + }_{0.14} {\rm Ti}_{0.18}} \right)_{\Sigma 3.00}$
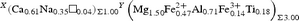 ${}^{Z} \left( {{\rm Al}_{4.54}{\rm Fe}^{3 + }_{0.18} {\rm V}^{3 + }_{0.02} {\rm Mg}_{1.27}} \right)_{\Sigma 6.00}{}^{T}\left[ {{\left( {{\rm Si}_{5.90}{\rm Al}_{0.10}} \right)}_{\Sigma 6.00}{\rm O}_{18}} \right]{\rm } \left( {\rm BO_3} \right)_3^{} {^{\rm O(3)}}\left( {\rm OH} \right)_3{}^{{\rm O}\left( 1 \right)} [\left( {\rm OH} \right)_{0.55}{\rm F}_{0.05}{\rm O}_{0.40}]_{\Sigma 1.00}$
${}^{Z} \left( {{\rm Al}_{4.54}{\rm Fe}^{3 + }_{0.18} {\rm V}^{3 + }_{0.02} {\rm Mg}_{1.27}} \right)_{\Sigma 6.00}{}^{T}\left[ {{\left( {{\rm Si}_{5.90}{\rm Al}_{0.10}} \right)}_{\Sigma 6.00}{\rm O}_{18}} \right]{\rm } \left( {\rm BO_3} \right)_3^{} {^{\rm O(3)}}\left( {\rm OH} \right)_3{}^{{\rm O}\left( 1 \right)} [\left( {\rm OH} \right)_{0.55}{\rm F}_{0.05}{\rm O}_{0.40}]_{\Sigma 1.00}$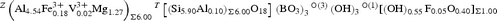
which recast in its ordered form for classification purposes is:
$$\eqalign{& ^{\rm X} ({\rm Ca}_{0.61}{\rm Na}_{0.35}\squ _{0.04})_{\Sigma 1.00}{}^{\rm Y} \left( {{\rm Mg}_{2.35}{\rm Fe}^{2 + }_{0.47} {\rm Ti}_{0.18}} \right)_{\Sigma 3.00} \cr & {}^{\rm Z} \left( {{\rm Al}_{5.25}{\rm Fe}^{3 + }_{0.32} {\rm V}^{3 + }_{0.02} {\rm Mg}_{0.42}} \right)_{\Sigma 6.00}{}^{\rm T} \left[ {{\left( {{\rm Si}_{5.90}{\rm Al}_{0.10}} \right)}_{\Sigma 6.00}{\rm O}_{18}} \right]{\rm }\left( {\rm BO_3} \right)_3{}^{\rm V} \left( {\rm OH} \right)_3{}^{\rm W} [\left( {\rm OH} \right)_{0.55}{\rm F}_{0.05}{\rm O}_{0.40}]_{\Sigma 1.00}}$$Uvite is a hydroxy-species belonging to the calcic-group of the tourmaline supergroup. The closest end-member compositions of valid tourmaline species are fluor-uvite and feruvite, to which uvite is related by the substitutions W(OH)– ↔ WF– and YMg2+ ↔ YFe2+, respectively. The occurrence of a solid-solution between uvite and magnesio-lucchesiite, according to the substitution ZMg2+ + W(OH)– ↔ ZAl3+ + WO2–, is supported by experimental data. The new mineral was approved by the IMA–CNMNC (IMA 2019-113). Uvite from Facciatoia formed by the reaction between B-rich fluids, released during the crystallisation process of LCT pegmatites, and the surrounding metaserpentinites, altered by contact metamorphism in the aureole of the Miocene Mt. Capanne monzogranitic pluton.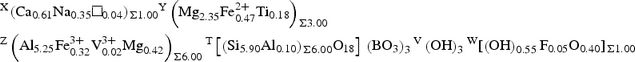
In Situ Isotopic Analysis of Uraninite Microstructures from the Oklo-Okélobondo Natural Fission Reactors, Gabon
- Mostafa Fayek, Keld A. Jensen, Rodney C. Ewing, Lee R. Riciputi
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 713 / 2002
- Published online by Cambridge University Press:
- 21 March 2011, JJ8.5
- Print publication:
- 2002
-
- Article
- Export citation
-
Uranium deposits can provide important information on the long-term performance of radioactive waste forms because uraninite (UO2+X) is similar to the UO2 in spent nuclear fuel. The Oklo-Okélobondo U-deposits, Gabon, serve as natural laboratory where the long-term (hundreds to billions of years) migration of uranium and other radionuclides can be studied over large spatial scales (nm to km). The natural fission reactors associated with the Oklo- Okélobondo U-deposits occur over a range of depths (100 to 400 m) and provide a unique opportunity to study the behavior of uraninite in near surface oxidizing environments versus more reducing conditions at depth. Previously, it has been difficult to constrain the timing of interaction between U-rich minerals and post-depositional fluids. These problems are magnified because uraninite is susceptible to alteration, it continuously self-anneals radiation damage, and because these processes are manifested at the nm to μm scale. Uranium, lead and oxygen isotopes can be used to study fluid-uraninite interaction, provided that the analyses are obtained on the micro-scale. Secondary ionization mass spectrometry (SIMS) permits in situ measurement of isotopic ratios with a spatial resolution on the scale of a few μm. Preliminary U-Pb results show that uraninite from all reactor zones are highly discordant with ages aaproaching the timing of fission chain reactions (1945±50 Ma) and resetting events at 1180±47 Ma and 898±46 Ma. Oxygen isotopic analyses show that uraninite from reactors that occur in near surface environments (δ18O= −14.4‰ to −8.5‰) have reacted more extensively with groundwater of meteoric origin relative to reactors located at greater depths (μ18O= −10.2‰ to −7.3‰). This study emphasizes the importance of using in situ high spatial resolution analysis techniques for natural analogue studies.