2 results
Impact of Two-Step Testing Algorithm on Reducing Hospital-Onset Clostridioides difficile Infections
- Bhagyashri Navalkele, Wendy Winn, Sheila Fletcher, Regina Galloway, Jason Parham, William Daley, Patrick Kyle, Vonda Clack, Kathy Sheilds
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s42-s43
-
- Article
-
- You have access Access
- Open access
- Export citation
-
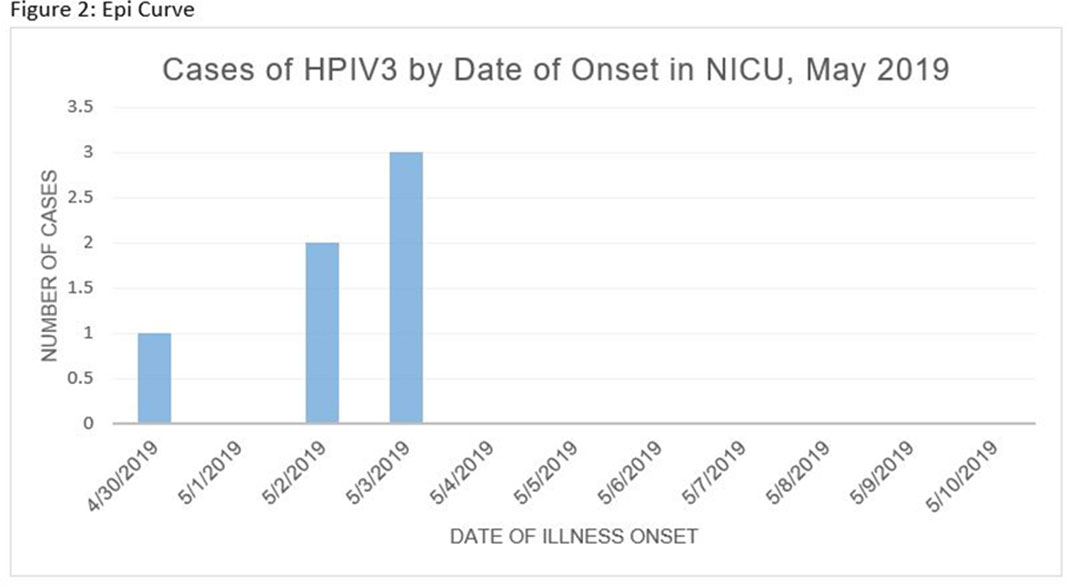
Clostridioides difficile infection (CDI) is one of the leading causes of hospital–onset infections. Clinically distinguishing true CDI versus colonization with C. difficile is challenging and often requires reliable and rapid molecular testing methods. At our academic center, we implemented a 2-step testing algorithm to help identify true CDI cases. The University of Mississippi Medical Center is a 700+ bed academic facility located in Jackson, Mississippi. Hospital-onset (HO) CDI was defined based on NHSN Laboratory Identified (LabID) event as the last positive C. difficile test result performed on a specimen using a multistep testing algorithm collected >3 calendar days after admission to the facility. HO-CDI data were collected from all inpatient units except the NICU and newborn nursery. HO-CDI outcomes were assessed based on standardized infection ratio (SIR) data. In May 2020, we implemented a 2-step testing algorithm (Figure 1). All patients with diarrhea underwent C. difficile PCR testing. Those with positive C. difficile PCR test were reflexed to undergo enzyme immunoassay (EIA) glutamate dehydrogenase antigen (Ag) testing and toxin A and B testing. The final results were reported as colonization (C. difficile PCR+/EIA Ag+/Toxin A/B−) or true CDI case (C. difficile PCR+/EIA +/Toxin A/B +) or negative (C. difficile PCR−). All patients with colonization or true infection were placed under contact isolation precautions until diarrhea resolution for 48 hours. During the preintervention period (October 2019–April 2020), 25 HO-CDI cases were reported compared to 8 cases in the postintervention period (June 2020–December 2020). A reduction in CDI SIR occurred in the postintervention period (Q3 2020–Q4 2020, SIR 0.265) compared to preintervention period (Q4 2019–Q1 2020, SIR 0.338) (Figure 2). We successfully reduced our NHSN HO-CDI SIR below the national average after implementing a 2-step testing algorithm for CDI. The 2-step testing algorithm was useful for antimicrobial stewardship to guide appropriate CDI treatment for true cases and for infection prevention to continue isolation of infected and colonized cases to reduce the spread of C. difficile spores.
Funding: No
Disclosures: None
Figure 1.
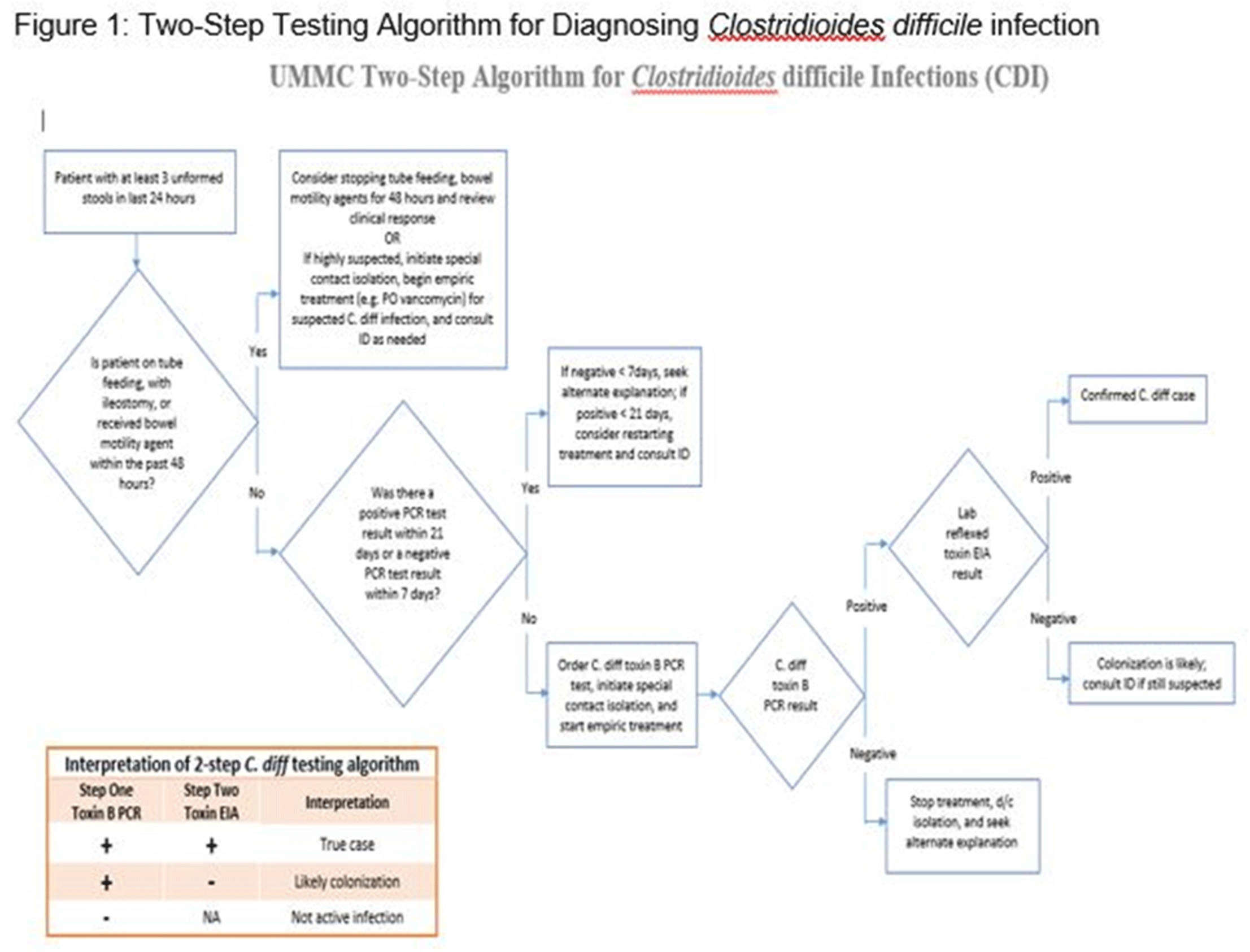
Figure 2.
Successful Control of Human Parainfluenza Type 3 Outbreak in a Level IV Neonatal Intensive Care Unit
- Bhagyashri Navalkele, Sheila Fletcher, Sanjosa Martin, Regina Galloway, April Palmer
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s26-s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Human parainfluenza (HPIV) is a common cause for upper respiratory tract illnesses (URTI) and lower respiratory tract illnesses (LRTI) in infants and young children. Here, we describe successful control of an HPIV type 3 (HPIV3) outbreak in a neonatal intensive care unit (NICU). NICU babies with new-onset clinical signs or symptoms of RTI and positive HPIV-3 nasopharyngeal specimen by respiratory pathogen panel (RPP) test on hospital day 14 or later were diagnosed with hospital-onset (HO) HPIV-3 infection. After 3 NICU babies were diagnosed with HO HPIV-3, an outbreak investigation was initiated on May 3, 2019, and continued for 2 incubation periods since the last identified case. Enhanced infection prevention measures were immediately implemented. All positive cases were placed in a cohort in a single pod of the NICU and were placed on contact precautions with droplet isolation precautions. Dedicated staffing and equipment were assigned. Environmental cleaning and disinfection with hospital-approved disinfectant wipes was performed daily. Visitors were restricted in the NICU. All employees entering the NICU underwent daily symptom screening for respiratory tract illness. All NICU babies were screened daily for respiratory tract illness with prompt isolation and RPP testing on positive screen. To determine the source of the HPIV3 outbreak, all HPIV3-positive specimens from the NICU and available temporally associated community-onset (CO) controls collected from non-NICU units were sent to the Centers for Disease Control and Prevention (CDC) for whole-genome sequencing (WGS) analysis. The first and last cases of HPIV-3 were diagnosed on May 1 and May 5, 2019, respectively. In total, 7 HO HPIV3 cases were reported: 1 in newborn nursery (NBN) and 6 in NICU. The case from the NBN was determined to be unrelated to the outbreak and the source was linked to a sick visitor. Of the 6 NICU babies, 5 had an LRTI and 1 had a URTI. Average time from admission to diagnosis was 71 days (range, 24–112). None had severe illnesses requiring intubation, and all had full recovery. No CO HPIV3 cases were reported from the NICU during the investigation. A maximum likelihood phylogenetic tree of HPIV3 WGS (Figure 1) showed that sequences from the 6 HO cases clustered together separately from the 3 CO controls, suggesting a single source of transmission, and 3 CO cases were not related to the HO cases or source of the outbreak. Early diagnosis and isolation of respiratory tract viral infections is important to prevent an outbreak. Successful control of outbreak in NICU requires prompt implementation of infection prevention measures with focus on symptom screening, cohorting, and disinfection practices.
Funding: No
Disclosures: None
Figure 1.
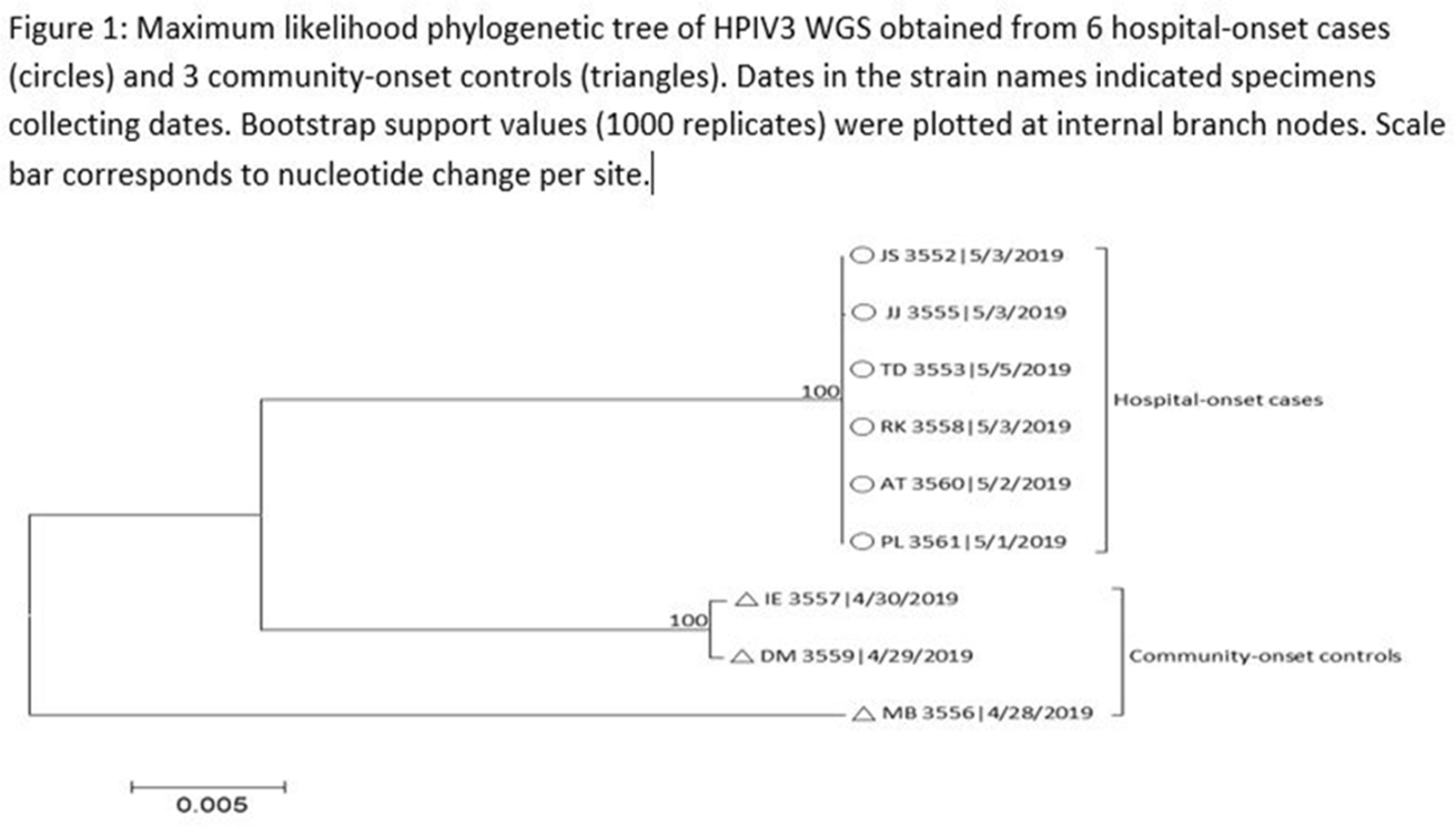
Figure 2.