2 results
Impact of the COVID-19 pandemic on inpatient antibiotic use in Indonesia and the Philippines
- Amara Fazal, Olivia McGovern, Garrett Mahon, Fernanda Lessa, Ma Tarcela Gler, Jemelyn Garcia, Mark Festin, Kuntaman Kuntaman, Ida Parwati, Cherry Siregar, Jay Christian, Gina de Guzman Betito, Maya Montemayor, Arleen De Leon, Emmeline Borillo, Mark Llanes, Regina Berba, Musofa Rusli, Farizal Rizky, Mariyatul Qibtiyah, Bambang Semedi, Rosantia Sarassari, Leonardus Widyatmoko, Basti Andriyoko, Adhi Sugianli, Dewi Turbawaty, Ivo Ranita, Franciscus Ginting, Made Krisna, Rahmadania Marita Joesoef, Twisha Patel
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s1-s2
-
- Article
-
- You have access Access
- Open access
- Export citation
-
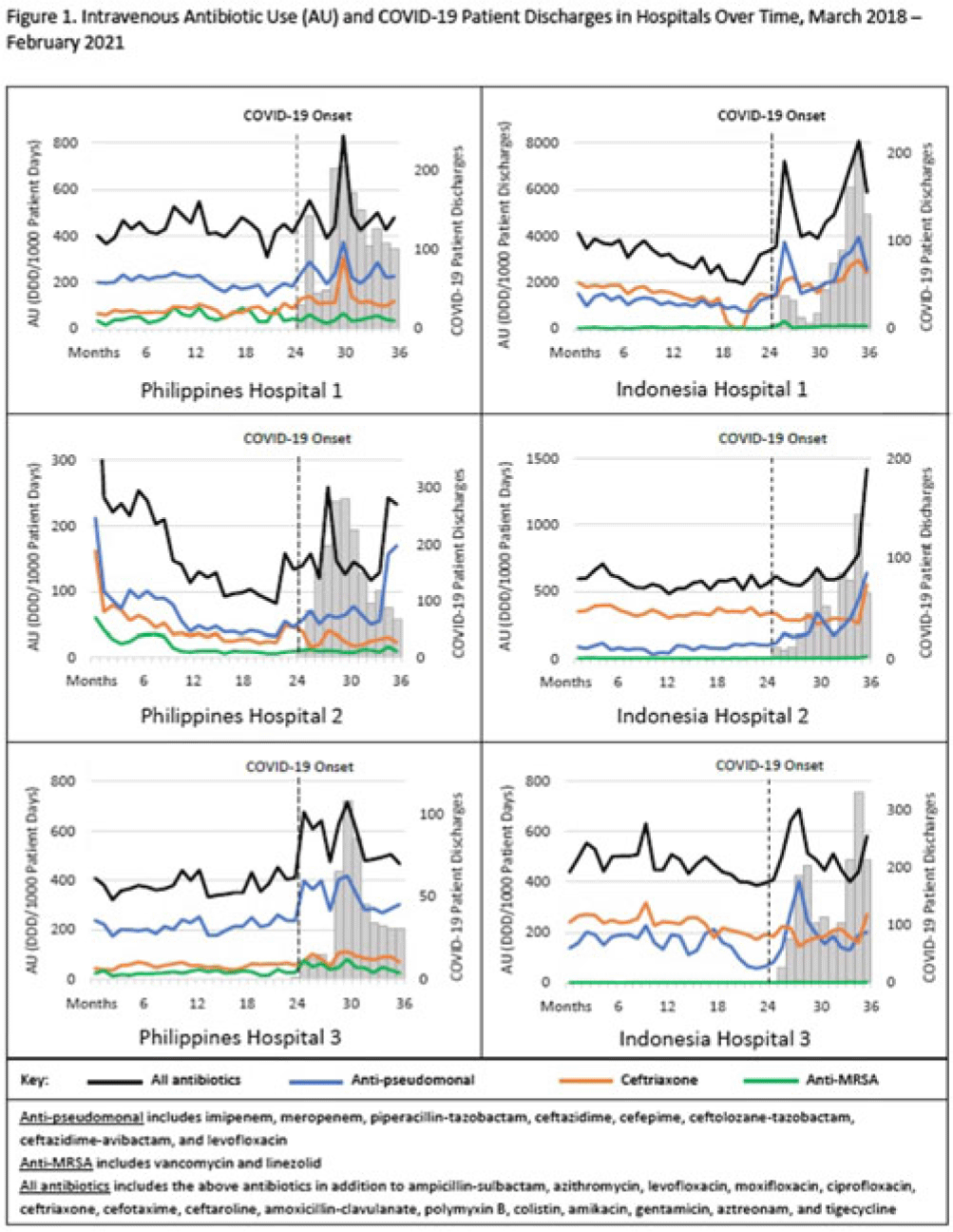
Background: The coronavirus disease 2019 (COVID-19) pandemic severely affected Southeast Asia, with >35 million cases and ~360,000 deaths. Despite relatively low rates of secondary bacterial infection among inpatients with COVID-19, several countries reported increased antibiotic use; raising concerns for worsening antimicrobial resistance. We assessed the impact of the COVID-19 pandemic on the use of antibiotics commonly used to treat respiratory infections in Southeast Asia. Methods: We evaluated intravenous antibiotic use among hospitalized adults in acute-care wards in 6 hospitals; 3 in Indonesia and 3 in the Philippines. We abstracted data on antibiotics that are commonly used to treat respiratory infections in these hospitals. We calculated antibiotic use rates for the 25 included antibiotics as monthly defined daily dose per 1,000 patient days (or patient discharges where patient days was unavailable) using data from pharmacy dispensing records and administrative records. Median antibiotic use rates for the prepandemic period (March 2018–February 2020) and the pandemic period (March 2020–February 2021) were compared, and percentage changes were calculated for (1) all 25 antibiotics combined; (2) ceftriaxone; (3) vancomycin and linezolid combined (anti-MRSA); and (4) broad-spectrum antibiotics with activity against Pseudomonas aeruginosa (anti-PSA). Monthly antibiotic use and COVID-19 patient discharges were graphed over the 36-month study period (March 2018–February 2021) to visualize trends (Fig. 1). The Wilcoxon rank-sum test was used to determine whether differences in median antibiotic use rates were statistically significant (2-tailed P < .05). Results: Overall, trends in antibiotic use were higher during months with increased COVID-19 patient discharges (Fig. 1). Use of all 25 antibiotics combined significantly increased in 4 of 6 hospitals (6.9%–63.6%) during the pandemic period compared to the prepandemic period. Ceftriaxone use significantly increased in 3 of 6 hospitals (37.1%–55.4%) and decreased in 3 of 6 hospitals (15.9%–31.9%). Anti-PSA antibiotic use significantly increased in 4 of 6 hospitals (16.1%–161.5%). Although anti-MRSA antibiotic use was low (comprising <2% of the overall included antibiotic use in Indonesia and <11% in the Philippines), use during the pandemic increased in 3 of 6 hospitals (59.8%–212.6%). Conclusions: We observed substantial increases in antibiotic use among hospitalized adults in Indonesia and the Philippines during the COVID-19 pandemic. The increased use of broad-spectrum antibiotics is concerning given the potential consequence of worsening antimicrobial resistance. Understanding how increases in antibiotic use compares to rates of bacterial infection, antimicrobial resistance, and antibiotic availability and accessibility during this time is important to contextualize results. These findings reinforce the importance of antibiotic stewardship practices to optimize antibiotic use, especially during pandemics.
Disclosure: None
PD66 Indirect Comparison Of Treatments For Metastatic Melanoma
- Alicia Aleman, Daniel Pedrosa, Magdalena Irisarri, Rafael Alonso, Ana Perez Galán, Regina Guzman
-
- Journal:
- International Journal of Technology Assessment in Health Care / Volume 34 / Issue S1 / 2018
- Published online by Cambridge University Press:
- 03 January 2019, p. 154
-
- Article
-
- You have access Access
- Export citation
-
Introduction:
Vemurafenib plus cobimetinib (VC) for the treatment of metastatic melanoma was requested to be included in the National Formulary in Uruguay. The standard of care for metastatic melanoma in Uruguay is dacarbazine. There is no published head-to-head trial assessing the effects of VC versus dacarbazine. The objective of this study was to perform an indirect comparison of the effects of dacarbazine, compared with VC, based on the results of trials that included both treatments versus the same comparator (vemurafenib alone).
Methods:We searched Pubmed and The Cochrane Library for trials comparing either VC or dacarbazine with vemurafenib. Trials were assessed in terms of risk of bias, similarity of interventions and inclusion and exclusion criteria, and comparability of characteristics of patients in the vemurafenib arm. We performed an indirect comparison using the Bucher method.
Results:From the literature search we retrieved two studies that met the inclusion criteria: a randomized clinical trial that assessed VC versus vemurafenib or placebo and another assessing dacarbazine versus vemurafenib. Both studies were similar in terms of methodological quality, inclusion and exclusion criteria, and comparability of the vemurafenib arms. However, the comparison of overall survival and progression-free survival curves for the vemurafenib arms were quite different between the two trials. At 9 months, overall survival was eighty-one percent and fifty-five percent and progression-free survival was thirty percent and fifteen percent, respectively. The indirect comparison provided the following hazard ratios: 0.24 (95% confidence interval [CI]: 0.14–0.48) for overall survival; 0.13 (95% CI: 0.09–0.19) for progression-free survival; and 0.15 (95% CI: 0.02–1.29) for grade 4 adverse events.
Conclusions:Treatment with VC increased overall survival and progression-free survival, compared with dacarbazine. Severe adverse events were less frequent with the combined therapy. However, the differences in the vemurafenib survival curves increases doubts about the accuracy of the indirect estimators of overall survival and progression-free survival.




