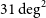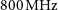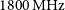104 results
Australian square kilometre array pathfinder: I. system description
- Part of
- A. W. Hotan, J. D. Bunton, A. P. Chippendale, M. Whiting, J. Tuthill, V. A. Moss, D. McConnell, S. W. Amy, M. T. Huynh, J. R. Allison, C. S. Anderson, K. W. Bannister, E. Bastholm, R. Beresford, D. C.-J. Bock, R. Bolton, J. M. Chapman, K. Chow, J. D. Collier, F. R. Cooray, T. J. Cornwell, P. J. Diamond, P. G. Edwards, I. J. Feain, T. M. O. Franzen, D. George, N. Gupta, G. A. Hampson, L. Harvey-Smith, D. B. Hayman, I. Heywood, C. Jacka, C. A. Jackson, S. Jackson, K. Jeganathan, S. Johnston, M. Kesteven, D. Kleiner, B. S. Koribalski, K. Lee-Waddell, E. Lenc, E. S. Lensson, S. Mackay, E. K. Mahony, N. M. McClure-Griffiths, R. McConigley, P. Mirtschin, A. K. Ng, R. P. Norris, S. E. Pearce, C. Phillips, M. A. Pilawa, W. Raja, J. E. Reynolds, P. Roberts, D. N. Roxby, E. M. Sadler, M. Shields, A. E. T. Schinckel, P. Serra, R. D. Shaw, T. Sweetnam, E. R. Troup, A. Tzioumis, M. A. Voronkov, T. Westmeier
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 38 / 2021
- Published online by Cambridge University Press:
- 05 March 2021, e009
-
- Article
-
- You have access Access
- HTML
- Export citation
-
In this paper, we describe the system design and capabilities of the Australian Square Kilometre Array Pathfinder (ASKAP) radio telescope at the conclusion of its construction project and commencement of science operations. ASKAP is one of the first radio telescopes to deploy phased array feed (PAF) technology on a large scale, giving it an instantaneous field of view that covers
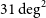 $31\,\textrm{deg}^{2}$ at
$31\,\textrm{deg}^{2}$ at 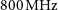 $800\,\textrm{MHz}$. As a two-dimensional array of 36
$800\,\textrm{MHz}$. As a two-dimensional array of 36 $\times$12 m antennas, with baselines ranging from 22 m to 6 km, ASKAP also has excellent snapshot imaging capability and 10 arcsec resolution. This, combined with 288 MHz of instantaneous bandwidth and a unique third axis of rotation on each antenna, gives ASKAP the capability to create high dynamic range images of large sky areas very quickly. It is an excellent telescope for surveys between 700 and
$\times$12 m antennas, with baselines ranging from 22 m to 6 km, ASKAP also has excellent snapshot imaging capability and 10 arcsec resolution. This, combined with 288 MHz of instantaneous bandwidth and a unique third axis of rotation on each antenna, gives ASKAP the capability to create high dynamic range images of large sky areas very quickly. It is an excellent telescope for surveys between 700 and 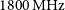 $1800\,\textrm{MHz}$ and is expected to facilitate great advances in our understanding of galaxy formation, cosmology, and radio transients while opening new parameter space for discovery of the unknown.
$1800\,\textrm{MHz}$ and is expected to facilitate great advances in our understanding of galaxy formation, cosmology, and radio transients while opening new parameter space for discovery of the unknown.
The evidence for cognitive behavioural therapy in any condition, population or context: a meta-review of systematic reviews and panoramic meta-analysis
- Beth Fordham, Thavapriya Sugavanam, Katherine Edwards, Paul Stallard, Robert Howard, Roshan das Nair, Bethan Copsey, Hopin Lee, Jeremy Howick, Karla Hemming, Sarah E. Lamb
-
- Journal:
- Psychological Medicine / Volume 51 / Issue 1 / January 2021
- Published online by Cambridge University Press:
- 18 January 2021, pp. 21-29
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
The majority of psychological treatment research is dedicated to investigating the effectiveness of cognitive behavioural therapy (CBT) across different conditions, population and contexts. We aimed to summarise the current systematic review evidence and evaluate the consistency of CBT's effect across different conditions. We included reviews of CBT randomised controlled trials in any: population, condition, format, context, with any type of comparator and published in English. We searched DARE, Cochrane, MEDLINE, EMBASE, PsycINFO, CINAHL, CDAS, and OpenGrey between 1992 and January 2019. Reviews were quality assessed, their data extracted and summarised. The effects upon health-related quality of life (HRQoL) were pooled, within-condition groups. If the across-condition heterogeneity was I2 < 75%, we pooled effects using a random-effect panoramic meta-analysis. We summarised 494 reviews (221 128 participants), representing 14/20 physical and 13/20 mental conditions (World Health Organisation's International Classification of Diseases). Most reviews were lower-quality (351/494), investigated face-to-face CBT (397/494), and in adults (378/494). Few reviews included trials conducted in Asia, South America or Africa (45/494). CBT produced a modest benefit across-conditions on HRQoL (standardised mean difference 0.23; 95% confidence intervals 0.14–0.33, I2 = 32%). The effect's associated prediction interval −0.05 to 0.50 suggested CBT will remain effective in conditions for which we do not currently have available evidence. While there remain some gaps in the completeness of the evidence base, we need to recognise the consistent evidence for the general benefit which CBT offers.
7 - Dinophyta
- from Part IV - Evolution of One Membrane of Chloroplast Endoplasmic Reticulum
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 250-297
-
- Chapter
- Export citation
-
Summary
These organisms are important members of the plankton in both fresh and marine waters, although a much greater variety of forms is found in marine members. Generally the Dinophyceae are less important in the colder polar waters than in warmer waters. The highly elaborate Dinophysales (Fig. 7.47(d), (e)) are essentially a tropical group.
A typical motile dinoflagellate (Figs. 7.1, 7.2) consists of an epicone and hypocone divided by the transverse girdle or cingulum. The epicone and hypocone are normally divided into a number of thecal plates, the exact number and arrangement of which are characteristic of the particular genus (Figs. 7.1, 7.3, 7.20(b), 7.24(b)). There is a longitudinal sulcus running perpendicular to the girdle. The longitudinal and transverse flagella emerge through the thecal plates in the area where the girdle and sulcus meet. The longitudinal flagellum projects out from the cell, whereas the transverse flagellum is wave-like and is closely appressed to the girdle. The cells can be photosynthetic or colorless and heterotrophic. Photosynthetic organisms have chloroplasts surrounded by one membrane of chloroplast E.R., which is not continuous with the outer membrane of the nuclear envelope. Chlorophylls a and c 2 are present in the chloroplasts, with peridinin and neoperidinin being the main carotenoids. About half the Dinophyceae that have been examined by electron microscopy have pyrenoids in the chloroplasts (Dodge and Crawford, 1970). The storage product is starch, similar to the starch of higher plants (Vogel and Meeuse, 1968), which is found in the cytoplasm. An eyespot may be present. The nucleus has permanently condensed chromosomes and is called a dinokaryotic or mesokaryotic nucleus.
Cell Structure
Theca
The thecal structure of motile Dinophyceae consists of an outer plasmalemma beneath which lies a single layer of flattened vesicles (Figs. 7.2, 7.3(c), 7.5) (Dodge and Crawford, 1970 ; Sekida et al., 2004). These vesicles, which normally contain cellulosic plates, give the theca its characteristic structure. The actual form and arrangement of the thecal plates varies from none in the phagotrophic Oxyrrhis marina, to very thick plates with flanges at the edges in Ceratium (Figs. 7.11, 7.48, 7.49) and Peridinium spp. (Figs. 7.2, 7.10).
Dedication
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp v-vi
-
- Chapter
- Export citation
Preface to the First Edition
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp xiii-xiv
-
- Chapter
- Export citation
-
Summary
It was that eccentric British soldier of fortune Col. Meinertzhagen, in his Birds of Arabia, who expressed the sentiment that prefaces should be kept short because few people ever read them. Accordingly, I would like to take a brief opportunity to express my gratitude to the people who offered encouragement and assistance during the preparation of this book. I would like to thank Adele Strauss Wolbarst, Robert Cnoops, Charmaine Slack, Sophia Skiordis, Caroline Mondel, Jill Keetley- Smith, Heather Edwards, Gail Arbeter, and the Lending Library at Boston Spa, England, for help while most of this manuscript was being prepared at the University of the Witwatersrand. For general encouragement while at Pahlavi (Shiraz) University and for providing assistance during the last turbulent and chaotic year of imperial rule in Iran, while the manuscript was being finished, I would like to thank Mark Gettner, Brian Coad, and Mumtaz Bokhari. When photographs or drawings have been taken directly from the original material, this is indicated by stating in the legend that it is from the original work. Most of the drawings have been redrawn to suit my tastes, and these drawings are indicated by stating that the work is after the original.
In some cases I have made drawings from photographs or have incorporated a number of drawings in one, in which case I state that the finished drawing is adapted from the original work or works. I have used the metric system in this book, and the fine-structural illustrations are expressed in micrometers (μm) and nanometers (nm).
12 - Heterokontophyta, Eustigmatophyceae
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 339-342
-
- Chapter
- Export citation
-
Summary
Eustimatophytes are yellow-green unicells that occur in freshwater, brackish water, and seawater as well as in the soil. The cells are similar to those in the Xanthophyceae, but differ in having an eyespot outside the chloroplast (Fig. 12.1) (the eyespot in the Xanthophyceae is in the chloroplast) (Hibberd and Leedale, 1970). Other characteristics of the class include a basal swelling of the tinsel flagellum adjacent to the eyespot, only chlorophyll a, chloroplasts without girdle lamellae and no peripheral ring of DNA, and chloroplast endoplasmic reticulum not connected to the nuclear envelope (Schnepf et al., 1996).
The eyespot (Figs. 12.1, 12.2) is a large orangered body at the anterior of the motile cell and is completely independent of the chloroplast. It consists of an irregular group of droplets with no membrane around the whole complex of droplets. The flagellar sheath is extended to form a T-shaped flagellar swelling at the base of the tinsel flagellum (Figs. 12.1, 12.2). This swelling is always closely appressed to the plasmalemma in the region of the eyespot. In turn, in the eyespot there is a large droplet closely applied to the plasmalemma in the area of the flagellar swelling.
The chloroplasts of the Eustigmatophyceae have chlorophyll a and β-carotene, with the two major xanthophylls being violaxanthin and vaucheriaxanthin (Whittle and Casselton, 1969 ; Antia and Cheng, 1982), the only difference in pigments compared to the Xanthophyceae being the presence of violaxanthin and the absence of antheraxanthin. Violaxanthin is the major lightharvesting pigment in the Eustigmatophyceae (Owens et al., 1987).
The Eustigmatophyceae is a monophyletic group (Andersen et al., 1998). Most of the species produce zoospores with only a single emergent flagellum (Pleurochloris magna, Fig. 12.1(d) ; Polyhedriella helvetica, Fig. 12.1(b))(Hibberd and Leedale, 1972), but there is a second basal body present, indicating that the cells had a biflagellate ancestor. The emergent flagellum is tinsel with microtubular hairs, and the flagellum is inserted subapically. Two of the algae in the class, Ellipsoidion acuminatum and Pseudocharaciopsis texensis (Fig. 12.2) (Lee and Bold, 1973), have zoospores with a long forward tinsel flagellum and a short posteriorly directed smooth flagellum.
23 - Algae and the Environment
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 492-509
-
- Chapter
- Export citation
-
Summary
It is possible to write whole books on the relationships between algae and the environment. In this chapter I have chosen a few subjects that have generated the most interest in the past couple of decades.
Toxic Algae
Algae can be harmful in two basic ways (Hallegraeff et al., 2003 ; Lassus et al., 2016).
(1) Producing large populations in the aquatic environment. Large growths of some algae (e.g., the diatom Chaetoceros (Figs. 17.36, 17.37(a)) or the prymnesiophyte Chrysochromulina (Fig. 23.1(c))) can clog the gills of fish and can be particularly a problem in aquaculture systems. Anoxic conditions, resulting in fish kills, can occur at the end of blooms of other algae (e.g., green algae) as the algae die and decompose.
(2) Production of toxins Some algae produce toxins that sicken and kill other organisms that prey on these algae. Indeed, this probably was the reason that these algae were selected for in the evolutionary process since it reduced predation by grazers (Gilbert, 1996). Filter-feeding shellfish can accumulate large quantities of these toxins as they filter the algae out of the water (Rossini and Hess, 2010). Consumption of the shellfish by man, birds, and animals results in sickness and death. The algae that produce phycotoxins are as follows.
Cyanophyceae (cyanobacteria)
• Neurotoxins anatoxin (Fig. 23.2(c)) and saxitoxin (Fig. 23.2(c)) that block the transmission of signal from neuron to neuron. These alkaloids (nitrogen-containing compounds) bind to voltage-activated Na + - channels and block influx of Na +, thereby preventing the generation of an action potential (Shimizu, 2000).
Part III - Evolution of the Chloroplast
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 77-79
-
- Chapter
- Export citation
-
Summary
The Rhodophyta (red algae) and Chlorophyta (green algae) form a natural group of algae in that they have chloroplasts surrounded by only the two membranes of the chloroplast envelope. The endosymbiotic theory of chloroplast evolution, first proposed by Mereschkowsky in 1905, is the one most widely accepted for the evolution of the chloroplast (Fig. III.1). According to this theory, a cyanobacterium was taken up by a phagocytic organism into a food vesicle. Normally the cyanobacterium would be digested by the flagellate, but by chance a mutation occurred, with the flagellate being unable to digest the cyanobacterium. This was probably a beneficial mutation because the cyanobacterium, by virtue of its lack of feedback inhibition, secreted considerable amounts of metabolites to the host flagellate. The flagellate in turn gave the cyanobacterium a protected environment, and the composite organism was probably able to live in an ecological niche where there were no photosynthetic organisms (i.e., a slightly acid body of water where free-living cyanobacteria do not grow; see Chapter 2). Pascher (1914) coined terms for this association; he called the endosymbiotic cyanobacteria cyanelles; the host, a cyanome; and the association between the two, a syncyanosis. In the original syncyanosis, the cyanelle had a wall around it. Because the wall slowed the transfer of compounds from the cyanelle to the host and vice versa, any mutation that resulted in a loss of wall would have been beneficial and selected for in evolution. As evolution progressed, these two membranes became the chloroplast envelope, the cyanome cytoplasm took over the formation of the storage product and the polyhedral bodies containing ribulose-1,5-bisphosphate carboxylase/oxygenase differentiated into the pyrenoid.
Most of the genes from the endosymbiotic cyanobacterium were transferred to the host nucleus while a small number of these genes were maintained in the resulting plastid and gave rise to the plastid genome with its associated proteinsynthesizing system. The products of many of the cyanobacterial genes transferred to the nucleus were then retargeted to the plastid to keep it functional. Approximately 3000 nuclear genes in plants encode plastid proteins, whereas the chloroplast genome contains between 100 and 120 genes. The nucleus is also capable of sensing the state of the chloroplast and to react to maintain chloroplast homeostasis.
13 - Heterokontophyta, Pinguiophyceae
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 343-344
-
- Chapter
- Export citation
Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 303-308
-
- Chapter
- Export citation
-
Summary
Algae with two membranes of chloroplast endoplasmic reticulum (chloroplast E.R.) have the inner membrane of chloroplast E.R. surrounding the chloroplast envelope. The outer membrane of chloroplast E.R. is continuous with the outer membrane of the nuclear envelope and has ribosomes on the outer surface (Fig. V.1).
The algae with two membranes of chloroplast E.R. evolved by a secondary endosymbiosis (Fig. V.1) (Lee, 1977 ; Keeling, 2013) when a phagocytic protozoan took up a eukaryotic photosynthetic alga into a food vesicle. Instead of being phagocytosed by the protozoan, the photosynthetic alga became established as an endosymbiont within the food vesicle of the protozoan. The endosymbiotic photosynthetic alga benefited from the acidic environment in the food vesicle that kept much of the inorganic carbon in the form of carbon dioxide, the form needed by ribulose bisphosphate/carboxylase for carbon fixation (see Part IV for further explanation). The host benefited by receiving some of the photosynthate from the endosymbiotic alga. The food vesicle membrane eventually fused with the endoplasmic reticulum of the host protozoan, resulting in ribosomes on the outer surface of this membrane, which became the outer membrane of the chloroplast E.R. Through evolution, ATP production and other functions of the endosymbiont's mitochondrion were taken over by the mitochondria of the protozoan host, and the mitochondria of the endosymbiont were lost. The resulting cytology is characteristic of the extant algae in the Chlorarachniophyta and Cryptophyta, which have a nucleomorph representing the degraded endosymbiotic nucleus, as well as storage product produced in what remains of the endosymbiont cytoplasm.
The type of chloroplast E.R. that exists in the Heterokontophyta and the Prymnesiophyta resulted from further reduction. The nucleomorph was completely lost and storage product formation was taken over by the host. The resulting cell had two membranes of chloroplast envelope surrounding the chloroplast. Outside of this was the inner membrane of chloroplast E.R. that was the remains of the plasma membrane of the endosymbiont. Outside of this was the outer membrane of chloroplast E.R. which was the remains of the food vesicle membrane of the host. Most of the protein synthesis of the endosymbiont was taken over by the nucleus of the host (Martin, 2010).
11 - Heterokontophyta, Synurophyceae
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 334-338
-
- Chapter
- Export citation
-
Summary
The Synurophyceae are closely related to the Chrysophyceae (Ariztia et al., 1991). The Synurophyceae differ, however, from the Chrysophyceae in the following: the Synurophyceae have chlorophylls a and c 1 (Andersen and Mulkey, 1983), the flagella are inserted into the cell approximately parallel to one another (Fig. 11.1), there is a photoreceptor near the base of each flagellum, there is no eyespot, and the contractile vacuole is in the posterior portion of the cell (Lavau et al., 1997 ; Andersen et al., 1999). Chloroplast endoplasmic reticulum is present in a few species, but absent in most. The cells usually are covered by bilaterally symmetrical scales.
In the Synurophyceae, scales composed of silica commonly occur outside the cell (Figs. 11.1, 11.2, 11.3). The scales are bilaterally symmetrical and are formed in a silica deposition vesicle. The membrane of the silica deposition vesicle (the silicalemma) controls the shape of the scale along with proteins and glycoproteins that adhere the developing scale to the silicalemma (Schultz et al., 2001). The presence of germanium in the medium results in inhibition of scale formation (Klaveness and Guillard, 1975). The scales are carried in the scale vesicle to the plasma membrane where the plasma membrane and the scale vesicle fuse, releasing the scales outside the cell (Beech et al., 1990). The scales are held next to the cell in an organic envelope (Ludwig et al., 1996), which is either hyaline or yellow-brown, the latter appearance being due to the impregnation of iron salts. The scales of the Synurophyceae are commonly composed of a number of parts, such as the dome, shield, and bristle of Mallomonas (Lavau and Wetherbee, 1994) (Figs. 11.2, 11.3(c), (d)). The scales of the Synurophyceae are overlapped precisely so that the anterior end of one scale overlaps the right margin of the scale to its left (Leadbeater, 1990). The scales are cemented together to form a scale case by the organic envelope. This precise arrangement of scales differs from the loosely arranged scales of the Chrysophyceae.
Analysis of lake sediments often reveals the presence of the silicified scales of the Synurophyceae as well as the silicified frustules of diatoms (Smol et al., 1984 ; Dixit et al., 1999).
Index
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 526-535
-
- Chapter
- Export citation

Phycology
- 5th edition
- Robert Edward Lee
-
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018
-
- Textbook
- Export citation
-
Phycology is the study of algae, the primary photosynthetic organisms in freshwater and marine food chains. Since the publication of the first edition in 1981, this textbook has established itself as a classic resource on this subject. Aimed at upper-level undergraduate and graduate students in phycology, limnology and biological oceanography, this revised edition maintains the format of previous editions, whilst incorporating the recent developments in the field such as: the potential and challenges of producing algae biofuel; the proliferation of algal toxins; and the development of new molecular tools and technologies on ancestry, phylogeny, and taxonomy of algae.
10 - Heterokontophyta, Chrysophyceae
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 318-333
-
- Chapter
- Export citation
-
Summary
The algae in the Heterokontophyta usually have cells with an anterior tinsel and posterior whiplash flagellum (Fig. 10.1). The plastids contain chlorophylls a and c along with fucoxanthin. The storage product is usually chrysolaminarin in cytoplasmic vesicles.
Based on molecular evidence (Horn et al., 2007 ; Kai et al., 2008 ; Schmidt et al., 2012 ; Yang et al., 2012), the algae in the Heterokontophyta can be divided into three clades.
Clade 1. Chrysophyceae, Synurophyceae, Eustigmatophyceae, Pinguiophyceae, Synchromophyceae.
Clade 2. Bacillariophyceae, Bolidophyceae, Dictyochophyceae, Pelagophyceae.
Clade 3. Phaeophyceae, Raphidophyceae, Xanthophyceae, Phaeothamniophyceae, Chrysomerophyceae, Schizocladiophyceae, Aurearenophyceae.
Since some of these classes have only a few representatives, and because this is a basic textbook, I have chosen not dealt with the algae in the Synchromophyceae, Chrysomerophyceae, Schizocladiophyceae, and Aurearenophyceae. The following classes are covered here (Andersen, 2004):
Chrysophyceae (golden-brown algae) (Chapter 10)
Synurophyceae (Chapter 11)
Eustigmatophyceae (Chapter 12)
Pinguiophyceae (Chapter 13)
Dictyochophyceae (silicoflagellates) (Chapter 14)
Pelagophyceae (Chapter 15)
Bolidophyceae (Chapter 16)
Bacillariophyceae (diatoms) (Chapter 17)
Raphidophyceae (chloromonads) (Chapter 18)
Xanthophyceae (yellow-green algae) (Chapter 19)
Phaeothamniophyceae (Chapter 20)
Phaeophyceae (brown algae) (Chapter 21).
CHRYSOPHYCEAE
The Chrysophyceae are distinguished chemically by having chlorophylls a, c 1, and c 2 (Andersen and Mulkey, 1983) and structurally by two flagella inserted into the cell perpendicular to each other, one photoreceptor on the short flagellum that is usually shaded by an eyespot in the anterior portion of the chloroplast, contractile vacuoles in the anterior portion of the cell, chloroplast endoplasmic reticulum, and radially or biradially symmetrical silica scales (if they are present). The storage product is chrysolaminarin. Many members of the class produce statospores enclosed in a silicified wall with a terminal pore. The Chrysophyceae/Synurophyceae probably originated about 268 Ma (million years ago) (Brown, 2010).
Most of the species in the Chrysophyceae are freshwater species, and occur in soft waters (low in calcium). Many of the freshwater species are in the plankton of lakes where they are present in abundance. The coccoid and filamentous genera are found mostly in cold springs and brooks, where they occur as gelatinous or crustous growths on stones and woodwork.
18 - Heterokontophyta, Raphidophyceae
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 397-400
-
- Chapter
- Export citation
-
Summary
The Raphidophyceae, or chloromonads, have chlorophylls a and c, and two membranes of chloroplast endoplasmic reticulum. The anterior flagellum is commonly tinsel, whereas the posterior flagellum is naked (Figs. 18.1, 18.2, 18.3). The freshwater species of the Raphidophyceae are green, whereas the marine forms are yellowish and contain the carotenoid fucoxanthin (Vesk and Moestrup, 1987). The closest relatives of the Raphidophyceae are the Eustigmatophyceae and the Chrysophyceae (Cavalier-Smith and Chao, 1996).
Marine species are euryhaline and eurythermic (tolerant of a wide salinity and temperature range) and occur in temperate and subtropical waters worldwide (Tobin et al., 2013). Marine genera are Chattonella (Fig. 18.3), Fibrocapsa (Fig. 18.1(b)), and Heterosigma (Fig. 18.1(a)). Many of the marine species produce neurotoxic compounds that are similar to brevetoxin (Fig. 18.2). Uptake of the toxin by fish results in depolarization of nerves supplying the heart. This reduces the heart rate, thereby lowering blood pressure, which in turn affects the transfer of oxygen to the gill lamellae, creating hypoxic conditions that lead to fish mortality (Tyrrell et al., 2001).
Toxic red-tide blooms of the marine Chattonella antiqua and Heterosigma carterae (Taylor, 1992) have occurred in the Seto Inland Sea in Japan (Watanabe et al., 1988). These red tides occurred in the summer when a salinity and temperature stratification occurred at a depth of 5–10 m. There was little mixing of waters above and below the stratified layer resulting in the upper layer being deficient in nutrients while the bottom layer was anaerobic. Heterosigma carterae flourishes under these conditions because it has a daily vertical migration of 10–15 m and this allows the alga to move between the stratified layers. This migration enables H. carterae to use the nutrients in the lower layers, and light and oxygen in the upper layer, resulting in the red-tide blooms of the organism. The migration is correlated with the production and degradation of cytoplasmic fat particles (Wada et al., 1987).
Heterosigma carterae has a wide salinity tolerance in culture (3–50%) and loses its motility at temperatures below 10 °C. At 5–10 °C, the alga forms non-motile masses capable of surviving in continuous darkness for up to 15 weeks (Han et al., 2002).
15 - Heterokontophyta, Pelagophyceae
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 351-353
-
- Chapter
- Export citation
-
Summary
The Pelagophyceae are a group of basically unicellular algae that are cytologically similar to the Chrysophyceae, in which they were previously classified (Andersen et al., 1993). The cells are very small, members of the ultraplankton (cells less than 2 μm) and appear as small spheres with indistinct protoplasm under the light microscope. Recent studies on the sequences of small-subunit RNA nucleotides in these algae have shown them to be closely related to each other and distinct from other members of the Heterokontophyta (Saunders et al., 1997). While these algae have been shown to be distinct from other members of the Heterokontophyta based on molecular data, they do not have cytological or morphological characters which are very different from other members of the phylum.
Pelagomonas calceolata is a very small (1.5 μm. 3 μm) ultraplanktonic marine alga with a single tinsel flagellum and basal body, and a single chloroplast and mitochondrion (Fig. 15.1(c)) (Andersen et al., 1993). Another member of the marine ultraplankton is Pelagococcus subviridis, a green-gold spherical non-motile cell (2.5–3.0 μm) with a single chloroplast, mitochondrion, and nucleus (Fig. 15.1(a)) (Vesk and Jeffery, 1987).
Members of the class are economically important because some of the algae produce “brown tides” (Ong et al., 2010). Aureoumbra lagunensis (Fig. 15.1(b)) is the causative agent of brown tides in Texas (DeYoe et al., 1997), while Aureococcus anophagefferens forms brown tides along the coasts of New Jersey, New York, and Rhode Island. The numbers of cells in brown tides can be so large that they can exclude light from the benthic eelgrass (Zostera marine), resulting in elimination of the eelgrass. The larvae of the bay scallop feed off eelgrass, and the bay scallop industry was virtually wiped out for a number of years after a brown tide in the waters off the northeast United States (Nicholls, 1995).
Aureoumbra lagunensis (Fig. 15.1(b)) is able to grow at its maximum rate at salinities as high as 70 PSU (practical salinity units ; seawater is about 35 PSU). Few algae are able to survive these hypersaline conditions. In addition, the surface of the cells are covered with a slime layer that reduces predation (Liu and Buskey, 2000).
2 - Cyanobacteria
- from Part II - The Prokaryotic Algae
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 32-76
-
- Chapter
- Export citation
-
Summary
The Cyanophyceae or blue-green algae are, today, usually referred to as the cyanobacteria (blue-green bacteria). The term cyanobacteria acknowledges that these prokaryotic algae are more closely related to the prokaryotic bacteria than to eukaryotic algae.
It has been hypothesized that the cyanobacteria evolved in freshwater at some time before 2.50 billion years ago (bya) (Blank, 2013) and that they spread into the marine environment at about the time of the Great Oxidation Event (GOE) (about 2.35 bya) where, through photosynthesis, the cyanobacteria raised oxygen levels in the atmosphere, enabling the evolution of aerobic life and dramatically changing life on the planet (Schirrmeister et al., 2014). The argument for a freshwater evolution of the cyanobacteria (Blank, 2013) involves sucrose synthesis, which originated in the cyanobacteria. Sucrose synthesis is strongly associated with a low-salinity environment.
Cyanobacteria have chlorophyll a (some also have chlorophyll b or d), phycobiliproteins, glycogen as a storage product, and cell walls containing amino sugars and amino acids. At one time, the occurrence of chlorophyll b in cyanobacteria was used as a criterion to place the organisms in a separate group, the Prochlorophyta. Modern nucleic-acid sequencing, however, has shown that chlorophyll b evolved a number of times within the cyanobacteria and the term Prochlorophyta has been discarded (Palenik and Haselkorn, 1992 ; Urback et al., 1992).
Morphology
The simplest morphology in the cyanobacteria is that of unicells, free-living (Figs. 2.14(c)) or enclosed within a mucilaginous envelope (Figs. 2.38, 2.43(a), (b)). Subsequent evolution resulted in the formation of a row of cells called a trichome (Fig. 2.12). When the trichome is surrounded by a sheath, it is called a filament (Fig. 2.12). It is possible to have more than one trichome in a filament (Figs. 2.43(e), 2.45(b)). The most complex thallus is the branched filament (Fig. 2.45(a)). Such a branched filament can be uniseriate (composed of a single row of cells) or multiseriate (composed of one or more rows of cells).
22 - Prymnesiophyta
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 470-491
-
- Chapter
- Export citation
-
Summary
The Prymnesiophyta are a group of uninucleate flagellates characterized by the presence of a haptonema between two smooth flagella. The Prymnesiophyta have two membranes of chloroplast endoplasmic reticulum, as do the Cryptophyta and the Heterokontophyta, but differ in having flagella without mastigonemes. Molecular data also show that the Prymnesiophyta are distinct from the Cryptophyta and Heterokontophyta (Bhattacharya and Ehlting, 1995 ; Medlin et al., 1994). Until 1962, the organisms were considered part of the Chrysophyceae, at which time Christensen split them off into a separate class, the Haptophyceae (named after the presence of the haptonema). The Haptophyceae was a descriptive name and not based on a genus in the class; thus the name was later changed to Prymnesiophyceae, based on the genus Prymnesium (Fig. 22.6) (Hibberd, 1976). The fossil record of the Prymnesiophyceae is known from the Carboniferous (approximately 300 000 000 years ago) (Faber and Preisig, 1994 ; Jordan and Chamberlain, 1997).
The cells are commonly covered with scales. In many cases, the scales are calcified, thereby producing coccoliths. The chloroplasts lack girdle lamellae and most contain chlorophylls a and c 1 / c 2, β-carotene, diadinoxanthin, and diatoxanthin (Zapata et al., 2004 ; Zhao et al., 2015). The storage product is primarily mannitol (Tsuji et al., 2015). The anterior end of the cell has a large Golgi apparatus and sometimes a contractile vacuole.
The Prymnesiophyceae are primarily marine organisms, although there are some freshwater representatives (Shalchian-Tabrizi et al., 2011). While diatoms dominate in nutrient-rich coastal waters, Prymnesiophyceae dominate offshore where nutrients are limiting. Prymnesiophyceae make up a major part of the marine nannoplankton and constitute about 45% of the total phytoplankton cells in the middle latitudes of the South Atlantic. They decrease in frequency toward the poles although some still occur in polar waters (Manton et al., 1977).
Cell Structure
Flagella
Most of the Prymnesiophyceae have two smooth flagella of approximately the same length (Figs. 22.1, 22.2(a)). The Pavlovales is the exception, where one flagellum is longer than the other and is usually covered by small cylindrical to club-shaped hollow scales 70 nm long and 20 nm wide (Fig. 22.2(b)) (van der Veer, 1969 ; Green and Manton, 1970).
21 - Heterokontophyta, Phaeophyceae
- from Part V - Evolution of Two Membranes of Chloroplast Endoplasmic Reticulum and the Chlorarachniophyta
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 415-469
-
- Chapter
- Export citation
-
Summary
The Phaeophyceae, or brown algae, derive their characteristic color from the large amounts of the carotenoid fucoxanthin (Fig. 21.2) in their chloroplasts as well as from any phaeophycean tannins that might be present. The chloroplasts also have chlorophylls a, c 1, and c 2. There are two membranes of chloroplast E.R., which are usually continuous with the outer membrane of the nuclear envelope. The storage product is laminarin (Fig. 21.2). There are no unicellular or colonial organisms in the order, and the algae are basically filamentous, pseudoparenchymatous, or parenchymatous. They are found almost exclusively in the marine habitat, there being only four genera containing freshwater species, that is, Heribaudiella, Pleurocladia, Bodanella, and Sphacelaria (Fig. 21.1) (Schloesser and Blum, 1980). A number of marine forms penetrate into brackish water, where they often form an important part of the salt marsh flora. These brackish water plants have almost totally lost the ability to reproduce sexually, and propagate by vegetative means only. Most of the Phaeophyceae grow in the intertidal belt and the upper littoral region. They dominate these regions in colder waters, particularly in the Northern Hemisphere, where the number of phaeophycean species is less than that of the Rhodophyceae, but the number of phaeophycean plants is much greater. In the tropics, the only place where large numbers of Phaeophyceae are found is the Sargasso Sea of the Atlantic.
The Phaeophyceae probably evolved about 260 Ma ago during the Permian Period (Brown, 2010 ; Kawai et al., 2015) from an organism in the Phaeothamniophyceae, which have motile cells similar to those in the Phaeophyceae, but lack the characteristic unilocular and plurilocular sporangia of the Phaeophyceae (Bailey et al., 1998).
Cell Structure
The cell structure (Fig. 21.3) is in many ways similar to that of the Chrysophyceae, Prymnesiophyceae, Bacillariophyceae, and Xanthophyceae, which are closely related to the Phaeophyceae. The main difference lies in the large amounts of extracellular polysaccharides surrounding the protoplast.
Cell Walls
There are no unicellular members of the Phaeophyceae, so cell walls joining cells are characteristic of the group (Yamagishi et al., 2014). Phaeophycean cell walls are generally composed of at least two layers, with cellulose making up the main structural skeleton (Kloareg and Quatrano, 1988).
Part IV - Evolution of One Membrane of Chloroplast Endoplasmic Reticulum
- Robert Edward Lee, Colorado State University
-
- Book:
- Phycology
- Published online:
- 06 August 2018
- Print publication:
- 01 March 2018, pp 231-234
-
- Chapter
- Export citation