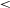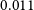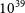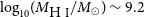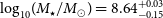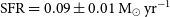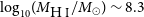2 results
The host galaxy of FRB 20171020A revisited
- Karen Lee-Waddell, Clancy W. James, Stuart D. Ryder, Elizabeth K. Mahony, Arash Bahramian, Bärbel S. Koribalski, Pravir Kumar, Lachlan Marnoch, Freya O. North-Hickey, Elaine M. Sadler, Ryan Shannon, Nicolas Tejos, Jessica E. Thorne, Jing Wang, Randall Wayth
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 40 / 2023
- Published online by Cambridge University Press:
- 11 July 2023, e029
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
The putative host galaxy of FRB 20171020A was first identified as ESO 601-G036 in 2018, but as no repeat bursts have been detected, direct confirmation of the host remains elusive. In light of recent developments in the field, we re-examine this host and determine a new association confidence level of 98%. At 37 Mpc, this makes ESO 601-G036 the third closest FRB host galaxy to be identified to date and the closest to host an apparently non-repeating FRB (with an estimated repetition rate limit of
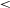 $<$
$<$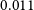 $0.011$ bursts per day above
$0.011$ bursts per day above 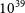 $10^{39}$ erg). Due to its close distance, we are able to perform detailed multi-wavelength analysis on the ESO 601-G036 system. Follow-up observations confirm ESO 601-G036 to be a typical star-forming galaxy with H i and stellar masses of
$10^{39}$ erg). Due to its close distance, we are able to perform detailed multi-wavelength analysis on the ESO 601-G036 system. Follow-up observations confirm ESO 601-G036 to be a typical star-forming galaxy with H i and stellar masses of 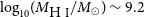 $\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 9.2$ and
$\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 9.2$ and 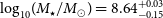 $\log_{10}\!(M_\star / M_\odot) = 8.64^{+0.03}_{-0.15}$, and a star formation rate of
$\log_{10}\!(M_\star / M_\odot) = 8.64^{+0.03}_{-0.15}$, and a star formation rate of 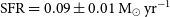 $\text{SFR} = 0.09 \pm 0.01\,{\rm M}_\odot\,\text{yr}^{-1}$. We detect, for the first time, a diffuse gaseous tail (
$\text{SFR} = 0.09 \pm 0.01\,{\rm M}_\odot\,\text{yr}^{-1}$. We detect, for the first time, a diffuse gaseous tail (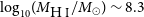 $\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 8.3$) extending to the south-west that suggests recent interactions, likely with the confirmed nearby companion ESO 601-G037. ESO 601-G037 is a stellar shred located to the south of ESO 601-G036 that has an arc-like morphology, is about an order of magnitude less massive, and has a lower gas metallicity that is indicative of a younger stellar population. The properties of the ESO 601-G036 system indicate an ongoing minor merger event, which is affecting the overall gaseous component of the system and the stars within ESO 601-G037. Such activity is consistent with current FRB progenitor models involving magnetars and the signs of recent interactions in other nearby FRB host galaxies.
$\log_{10}\!(M_{\rm{H\,{\small I}}} / M_\odot) \sim 8.3$) extending to the south-west that suggests recent interactions, likely with the confirmed nearby companion ESO 601-G037. ESO 601-G037 is a stellar shred located to the south of ESO 601-G036 that has an arc-like morphology, is about an order of magnitude less massive, and has a lower gas metallicity that is indicative of a younger stellar population. The properties of the ESO 601-G036 system indicate an ongoing minor merger event, which is affecting the overall gaseous component of the system and the stars within ESO 601-G037. Such activity is consistent with current FRB progenitor models involving magnetars and the signs of recent interactions in other nearby FRB host galaxies.
14 - 90Yttrium radioembolization for hepatocellular carcinoma
- from Section III - Organ-specific cancers – primary liver cancers
-
- By Ryan M. Hickey, Northwestern University, Riad Salem, Hadassah Hebrew University Medical Center, Robert J. Lewandowski, Northwestern University
- Edited by Jean-Francois H. Geschwind, Michael C. Soulen
-
- Book:
- Interventional Oncology
- Published online:
- 05 September 2016
- Print publication:
- 22 September 2016, pp 128-133
-
- Chapter
- Export citation
-
Summary
Radioembolization refers to the intra-arterial, transcatheter administration of micrometer-sized particles loaded with a radioisotope, most commonly yttrium-90 (Y90). Because liver tumors derive the majority of their blood supply from hepatic arteries, as opposed to the predominantly portal venous blood supply of normal liver tissue, hepatic arterial injection of Y90-labeled microspheres results in greater deposition of the spheres in tumor tissue as opposed to normal liver parenchyma. Since Y90 radioembolization provides an internal source of radiation to hepatic tumors, it is considered brachytherapy.
Preferential deposition of radioactive microspheres within tumor tissue provides for relative sparing of the uninvolved liver parenchyma from the radiation effects of Y90, thereby permitting significantly higher radiation doses than can be safely administered using external-beam radiation. The radiosensitive nature of normal liver tissue has traditionally limited the role of external-beam radiation in the treatment of primary and metastatic hepatic malignancies, as the incidence of severe radiation-induced liver disease (RILD) may exceed 50% for external-beam radiation doses greater than 35–40 Gy. However, with radioembolization, radiation doses in excess of 150 Gy can be safely administered.
Y90, a pure beta emitter with a half-life of 64.2 hours and tissue penetration of 2.5–11 mm, is incorporated into glass or resin microspheres ranging in size from 20–30 μm (glass) to 20–60 μm (resin). Glass microspheres (Therasphere, BTG International Canada, Ottawa, ON, Canada) were approved in 1999 by the US Food and Drug Administration (FDA) under a Humanitarian Device Exemption for the treatment of unresectable hepatocellular carcinoma (HCC). Resin microspheres (SIR-Spheres, Sirtex Medical, Lane Cove, Australia) were granted full premarketing approval in 2002 by the US FDA for the treatment of unresectable colorectal metastases in conjunction with intrahepatic floxuridine.
Safe and effective treatment of hepatic tumors with radioembolization requires not only the angiographic and endovascular skills critical for selective embolization procedures, but also a comprehensive understanding of radiation administration and safety, including radiation dosimetry and radiation dose modification based on tumor characteristics and a patient's clinical profile.
Patient selection
The patient selection process for Y90 radioembolization involves an assessment of the patient's burden of disease, hepatic biochemical profile, and performance status. Patients should have no extrahepatic disease and a tumor burden less than 70% of the liver volume.