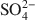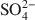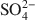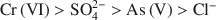2 results
Adsorption of Cr(VI) and As(V) on Chitosan-Montmorillonite: Selectivity and pH Dependence
- Jong-Hyok An, Stefan Dultz
-
- Journal:
- Clays and Clay Minerals / Volume 56 / Issue 5 / October 2008
- Published online by Cambridge University Press:
- 01 January 2024, pp. 549-557
-
- Article
- Export citation
-
Montmorillonite modified with the cationic biopolymer, chitosan, has, in weak acidic solutions, protonated amine groups which act as anion-adsorption sites. Due to the specific surroundings of the adsorption sites and diffusion paths in the interlayer of chitosan-montmorillonite, preferential adsorption of certain anions is likely. In the present study, the adsorption properties for the inorganic anions Cr(VI) and As(V) were determined, taking into account solution pH and competitive adsorption in the presence of Cl− and ${\rm{SO}}_4^{2 - }$
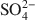 . Chitosan-montmorillonite was prepared by adding an amount of chitosan equivalent to 500% of the cation exchange capacity (CEC) at pH 5 and 75°C. The resulting anion exchange capacity was ∼0.34 molc/kg. The adsorption properties for As(V) and Cr(VI) were determined with the batch technique at pH 3 to 9. Adsorption isotherms were fitted to the Langmuir and Dubinin-Radushkevich equations and judged quantitatively by the correlation coefficient. To describe the competitive adsorption, the selectivity (S) was determined by the ratio of amounts of anions A and B adsorbed (qA/qB) in a binary system. The ionic species adsorbed, i.e. either Cr(VI) or As(V), depended on the pH, as did the degree of protonation of the amine groups, and this played a decisive role in the amount of anions adsorbed. The maximum amount of Cr(VI) adsorbed was 180 mmol/kg at pH 3.5, whereas for As(V) it was 120 mmol/kg at pH 4.0 to 5.0. The adsorption process of Cr(VI) and As(V) fit well to the Langmuir isotherm. By increasing the concentration of the competitive anion, Cl−, in solution, the amount of Cr(VI) and As(V) adsorbed remained almostconstant, whereas ${\rm{SO}}_4^{2 - }$
. Chitosan-montmorillonite was prepared by adding an amount of chitosan equivalent to 500% of the cation exchange capacity (CEC) at pH 5 and 75°C. The resulting anion exchange capacity was ∼0.34 molc/kg. The adsorption properties for As(V) and Cr(VI) were determined with the batch technique at pH 3 to 9. Adsorption isotherms were fitted to the Langmuir and Dubinin-Radushkevich equations and judged quantitatively by the correlation coefficient. To describe the competitive adsorption, the selectivity (S) was determined by the ratio of amounts of anions A and B adsorbed (qA/qB) in a binary system. The ionic species adsorbed, i.e. either Cr(VI) or As(V), depended on the pH, as did the degree of protonation of the amine groups, and this played a decisive role in the amount of anions adsorbed. The maximum amount of Cr(VI) adsorbed was 180 mmol/kg at pH 3.5, whereas for As(V) it was 120 mmol/kg at pH 4.0 to 5.0. The adsorption process of Cr(VI) and As(V) fit well to the Langmuir isotherm. By increasing the concentration of the competitive anion, Cl−, in solution, the amount of Cr(VI) and As(V) adsorbed remained almostconstant, whereas ${\rm{SO}}_4^{2 - }$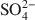 had a more pronounced competitive effect. At concentration ratios of 0.5 and 1 for ${\rm{SO}}_4^{2 - }$
had a more pronounced competitive effect. At concentration ratios of 0.5 and 1 for ${\rm{SO}}_4^{2 - }$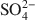 to Cr(VI) and As(V), respectively, the sorption capacity decreased by 10 and 25%, respectively. The sequence of the selectivity was: ${\rm{Cr}}\left( {{\rm{VI}}} \right) > {\rm{SO}}_4^{2 - } > {\rm{As}}\left( {\rm{V}} \right) > {\rm{C}}{{\rm{l}}^ - }$
to Cr(VI) and As(V), respectively, the sorption capacity decreased by 10 and 25%, respectively. The sequence of the selectivity was: ${\rm{Cr}}\left( {{\rm{VI}}} \right) > {\rm{SO}}_4^{2 - } > {\rm{As}}\left( {\rm{V}} \right) > {\rm{C}}{{\rm{l}}^ - }$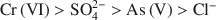 . Chitosan-montmorillonite showed a high selectivity for Cr(VI), which adsorbed chemically. Despite the lower affinity for As(V) and physical adsorption, the adsorption capacity was relatively high.
. Chitosan-montmorillonite showed a high selectivity for Cr(VI), which adsorbed chemically. Despite the lower affinity for As(V) and physical adsorption, the adsorption capacity was relatively high.
The Effect of Surface Charge and Wettability on H2O Self Diffusion in Compacted Clays
- Birgit Schampera, Stefan Dultz
-
- Journal:
- Clays and Clay Minerals / Volume 59 / Issue 1 / February 2011
- Published online by Cambridge University Press:
- 01 January 2024, pp. 42-57
-
- Article
- Export citation
-
Organo-clays are of interest in the modification of anion transport properties in engineered barriers. In the present study, surface charge and wettability were assessed for tracking changes in the effective diffusion coefficient (Deff) by the formation or suppression of bound H2O layers on the external surfaces of clays. Bentonite samples modified with three different organic cations in amounts of 0 to 400% of the cation exchange capacity were used. Diffusive transport was determined in H2O→D2O exchange experiments in a newly constructed cell adapted to the attenuated total reflectance (ATR) accessory of a Fourier-transform infrared (FTIR) spectrometer at two different dry bulk densities and various degrees of water saturation.
All organo-clay combinations showed changes in surface charge after the addition of organic cations, from a negative value of 99 mmolc/kg for the original bentonite to a maximum positive value of 230.5 mmolc/kg for hexadecylpyridinium (HDPy)-montmorillonite. The positive charge resulted from adsorption of the organic cation in excess of the CEC. Hydrophobic surface properties with contact angles >90° were obtained for HDPy-montmorillonite samples with monolayers of organic cations on the external surfaces only. Here, where hydrophobicity suppressed the formation of bound H2O layers, the largest Deff o f 2.7×10−10 m2/s was observed in the high dry bulk density range (1.0–1.5 g/cm3) under water-saturated conditions. In the low dry bulk density range (0.6–0.9 g/cm3) this effect was weakened significantly because, with increasing pore size, the effect of bound H2O layers was reduced. In the high dry bulk density range at partial water saturation (40%), diffusive transport was hindered by the small water volume. Previous work found that, in the high dry bulk density range and water-saturated state, Deff was 2.4×10−11 m2/s for the original bentonite. Deff for all hydrophilic organo-clay samples was ⩽2.1×10−11 m2/s, somewhat less than for the hydrophobic sample. In hydrophilic organo-clay samples, retardation factors that retard the value for Deff, up toa magnitude of 0.5, include an increase in dry bulk density and a decrease in water saturation. In the water-saturated state at high dry bulk densities, hydrophobic surface properties suppressing the formation of bound H2O layers can increase Deff by one order of magnitude.