3 results
COVID-19 outbreak in an acute psychiatric unit—Unique challenges and creative solutions
- Supriya Narasimhan, Sherilyn Oribello, Laura Tang, Tracey Stoll, Vidya Mony
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s94-s95
-
- Article
-
- You have access Access
- Open access
- Export citation
-
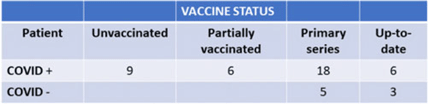
Background: We describe the management of a major COVID-19 outbreak in January 2022 during the SARS-CoV-2 omicron-variant winter surge involving the only inpatient psychiatric facility of Santa Clara County, California, which serves a population of 1.9 million. Methods: On January 14, 2022, infection prevention staff were notified of a symptomatic COVID-19 case in our locked inpatient psychiatric unit who had been admitted since October 2021. The index patient had no visitors or transfers outside the unit. The patients in this unit were noncompliant with masking and mingled with each other during meals. Initial testing identified 23 positive cases among 47 patients and 12 staff cases. Mitigating actions included closing the unit to new admissions, creating alternate care areas in the emergency psychiatric unit, and separating patients into “exposed but negative” and “infected” cohorts and housing them in “red,” and “yellow” zones, respectively. A “green” zone was created by clearance of positive cases. For the cohort exposed to COVID-19, masking was enforced by supervision, dining was scheduled in batches, and daily symptom screening and antigen testing were performed in addition to standard postexposure RT-PCR testing on day 4 and day 7. Mandatory N95 respirators and eye protection were implemented for staff on unit entry. Exposed staff followed employee health protocols for postexposure testing. Enhanced environmental control measures included terminal cleaning and UV-C disinfection of common areas and patient rooms and a thorough investigation of airflow. Discharged patients were contacted if they were residing in congregate facilities. Results: Of 47 patients, 39 (83%) tested positive for COVID-19. However, 8 patients remained negative; all 8 had received at least their primary vaccine series (Table 1). In total, 16 HCWs were SARS-CoV-2 positive in this outbreak. The outbreak officially ended 25 days after the first case. All SARS-CoV-2–positive patients had mild illness, not requiring treatment or hospitalization. We identified vaccine immune escape, staff presenteeism, patient noncompliance with masking, and comingling as major causes of transmission. We determined through contact tracing and temporality that the outbreak likely started from a positive staff member or visitor because most patients had been long-term residents. Conclusions: This outbreak was challenging due to the specialized behavioral needs of the involved patients. It was imperative to reopen this unit quickly and safely to provide psychiatric care to our county’s most vulnerable patients. Ongoing PPE education, repeated reinforcement, engagement in staff wellness to combat pandemic fatigue, and aggressive vaccination are all crucial to minimizing the impact of future outbreaks.
Disclosures: None
COVID-19 Outbreak in an Acute-Care Hospital: Lessons Learned
- Supriya Narasimhan, Vidya Mony, Tracey Stoll, Sherilyn Oribello, Karanas Yvonne, Dolly Goel
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s56-s57
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: We describe the infection prevention investigation of a cluster of 15 healthcare workers (HCWs) and 7 patients in a single non–COVID-19 unit of an acute-care hospital in September 2020. Methods: The infection prevention team was notified of 13 SARS-CoV-2–positive, symptomatic HCWs in an acute-care non–COVID-19 unit in 1 week (August 30, 2020, to September 3, 2020). In the same week, 2 patients who had been on the unit were diagnosed with nosocomial COVID-19. An epidemiologic investigation identified the exposure period to be between August 19, 2020, and September 3, 2020. The following immediate containment measures were implemented: closing the unit to new admissions, restricting float staff, moving existing patients to private rooms, mandatory masking of patients, and mandatory respirator and eye protection on unit entry for all HCWs. Exposed unit staff were tested immediately and then every 4 days until September 18, 2020. Likewise, exposed patients, including those discharged, were notified and offered testing. Hospital-wide HCW surveillance testing was conducted. Enhanced environmental control measures were conducted, including terminal cleaning and ultraviolet C (UV-C) disinfection of common areas and patient rooms and a thorough investigation of airflow. Detailed staff interviews were performed to identify causes of transmission. Multiple town hall meetings were held for staff education and updates. Results: In total, 108 total patients were deemed exposed: 33 were inpatients and 75 had been discharged. Testing identified 5 additional patient cases among 57 patients who received testing; 51 chose to self-monitor for symptoms. Staff testing identified 2 additional cases. Thus, 15 HCWs and 7 patients were linked in this cluster. The containment measures successfully ended staff transmission as of September 5, 2020. The last patient case was detected on September 10, 2020. Secondary cases were noted in 6 HCW families. We identified staff presenteeism, complacency, and socialization in break rooms and outside work as major causes of transmission. Suboptimal compliance with universal eye protection and hand hygiene (67%) were contributing factors. We determined by contact tracing and temporality that the outbreak could have stemmed from nursing home patient(s) through floating HCWs to staff on the affected unit. Directionality of transmission was from staff to patients in this cluster. Conclusions: Many facets of pandemic fatigue were apparent in this outbreak, namely, inability of HCWs to adhere to changing PPE guidance, presenteeism pressures due to workforce needs, and socialization with peers due to a false sense of security conferred by biweekly surveillance testing. Ongoing PPE education, repeated reinforcement, as well as engagement in staff wellness are crucial to combatting pandemic fatigue, conserving our workforce, and preventing future outbreaks.
Funding: No
Disclosures: None
Financial and Mortality Modeling as a Tool to Present Infection Prevention Data: What a SIR of 1.2 Means for the Hospital
- Vidya Mony, Kevin Hultquist, Supriya Narasimhan
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s64-s65
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Presenting to hospital leadership is an annual requirement of many infection prevention (IP) programs. Most presentations include current statistical data of hospital-acquired infections (HAIs) and whether the hospital has met its goals according to the National Healthcare Safety Network (NHSN) criteria. We presented HAI data in a novel way, with financial and mortality modeling, to show the impact of IP interventions to leadership not attuned to NHSN metrics. Method: We looked at 4 HAIs, their trends, and their effect on our hospital, Santa Clara Valley Medical Center (SCVMC). To estimate the impact of specific HAIs, we used 2 metrics derived from a meta-analysis by the US Department of Health and Human Services (HHS): excess mortality and excess cost. Excess mortality is defined as the difference between the underlying population mortality and the affected population mortality expressed as deaths per 1,000 population. Excess cost is defined as the additional cost introduced per patient with a specific HAI versus a similarly admitted patient without that HAI. HHS data were multiplied by the number of HAI events at SCVMC to generate estimates. Result: In our presentation, we elucidated a previously unseen cost savings and decreased mortality with 2 HAIs, central-line–associated blood stream infections (CLABSIs) and catheter associated urinary tract infections (CAUTIs), which were below NHSN targets due to IP-led interventions. We then showed 2 other HAIs, Clostridium difficile infection (CDI) and surgical site infections (SSIs), which did not meet our expected NHSN and institutional goals and were estimated to increase costs and potential mortalities in the upcoming year. We argued that proactive monies directed toward expanding our IP program and HAI mitigation efforts would cost a fraction of the impending healthcare expenditures as predicted by the model. Conclusion: By applying financial and mortality modeling, we helped our leadership perceive the concrete effect of IP-led interventions versus presenting abstract NHSN metrics. We also emphasized that without proactive leadership investment, we would continue to overspend healthcare dollars while not meeting our goals. This format of presentation gave us critical leverage to advocate for and successfully expand our IP department. Further SHEA-led cost-analysis modeling and education are needed to help IP departments promote their efforts in an effective manner.
Funding: None
Disclosures: None




