8 results
Factors associated with SARS-CoV-2 and community-onset invasive Staphylococcus aureus coinfection, 2020
- Kelly Jackson, Sydney Resler, Joelle Nadle, Susan Petit, Susan Ray, Lee Harrison, Ruth Lynfield, Kathryn Como-Sabetti, Carmen Bernu, Ghinwa Dumyati, Marissa Tracy, William Schaffner, Holly Biggs, Isaac See
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s84-s85
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Previous analyses describing the relationship between SARS-CoV-2 infection and Staphylococcus aureus have focused on hospital-onset S. aureus infections occurring during COVID-19 hospitalizations. Because most invasive S. aureus (iSA) infections are community-onset (CO), we characterized CO iSA cases with a recent positive SARS-CoV-2 test (coinfection). Methods: We analyzed CDC Emerging Infections Program active, population- and laboratory-based iSA surveillance data among adults during March 1–December 31, 2020, from 11 counties in 7 states. The iSA cases (S. aureus isolation from a normally sterile site in a surveillance area resident) were considered CO if culture was obtained <3 days after hospital admission. Coinfection was defined as first positive SARS-CoV-2 test ≤14 days before the initial iSA culture. We explored factors independently associated with SARS-CoV-2 coinfection versus no prior positive SARS-CoV-2 test among CO iSA cases through a multivariable logistic regression model (using demographic, healthcare exposure, and underlying condition variables with P<0.25 in univariate analysis) and examined differences in outcomes through descriptive analysis. Results: Overall, 3,908 CO iSA cases were reported, including 138 SARS-CoV-2 coinfections (3.5%); 58.0% of coinfections had iSA culture and the first positive SARS-CoV-2 test on the same day (Fig. 1). In univariate analysis, neither methicillin resistance (44.2% with coinfection vs 36.5% without; P = .06) nor race and ethnicity differed significantly between iSA cases with and without SARS-CoV-2 coinfection (P = .93 for any association between race and ethnicity and coinfection), although iSA cases with coinfection were older (median age, 72 vs 60 years , P<0.01) and more often female (46.7% vs 36.3%, P=0.01). In multivariable analysis, significant associations with SARS-CoV-2 coinfection included older age, female sex, previous location in a long-term care facility (LTCF) or hospital, presence of a central venous catheter (CVC), and diabetes (Figure 2). Two-thirds of co-infection cases had ≥1 of the following characteristics: age > 73 years, LTCF residence 3 days before iSA culture, and/or CVC present any time during the 2 days before iSA culture. More often, iSA cases with SARS-CoV-2 coinfection were admitted to the intensive care unit ≤2 days after iSA culture (37.7% vs 23.3%, P<0.01) and died (33.3% vs 11.3%, P<0.01). Conclusions: CO iSA patients with SARS-CoV-2 coinfection represent a small proportion of CO iSA cases and mostly involve a limited number of factors related to likelihood of acquiring SARS-CoV-2 and iSA. Although CO iSA patients with SARS-CoV-2 coinfection had more severe outcomes, additional research is needed to understand how much of this difference is related to differences in patient characteristics.
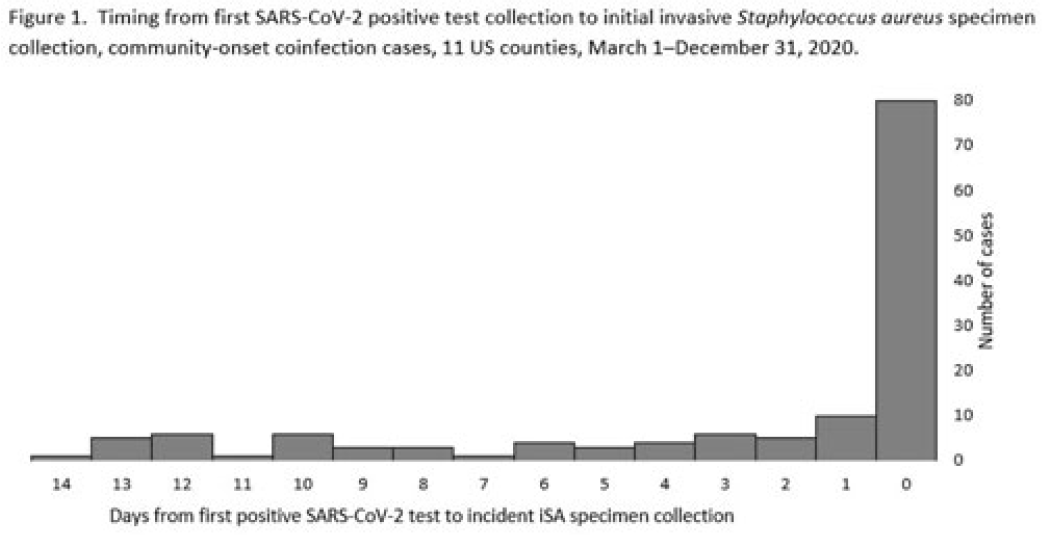
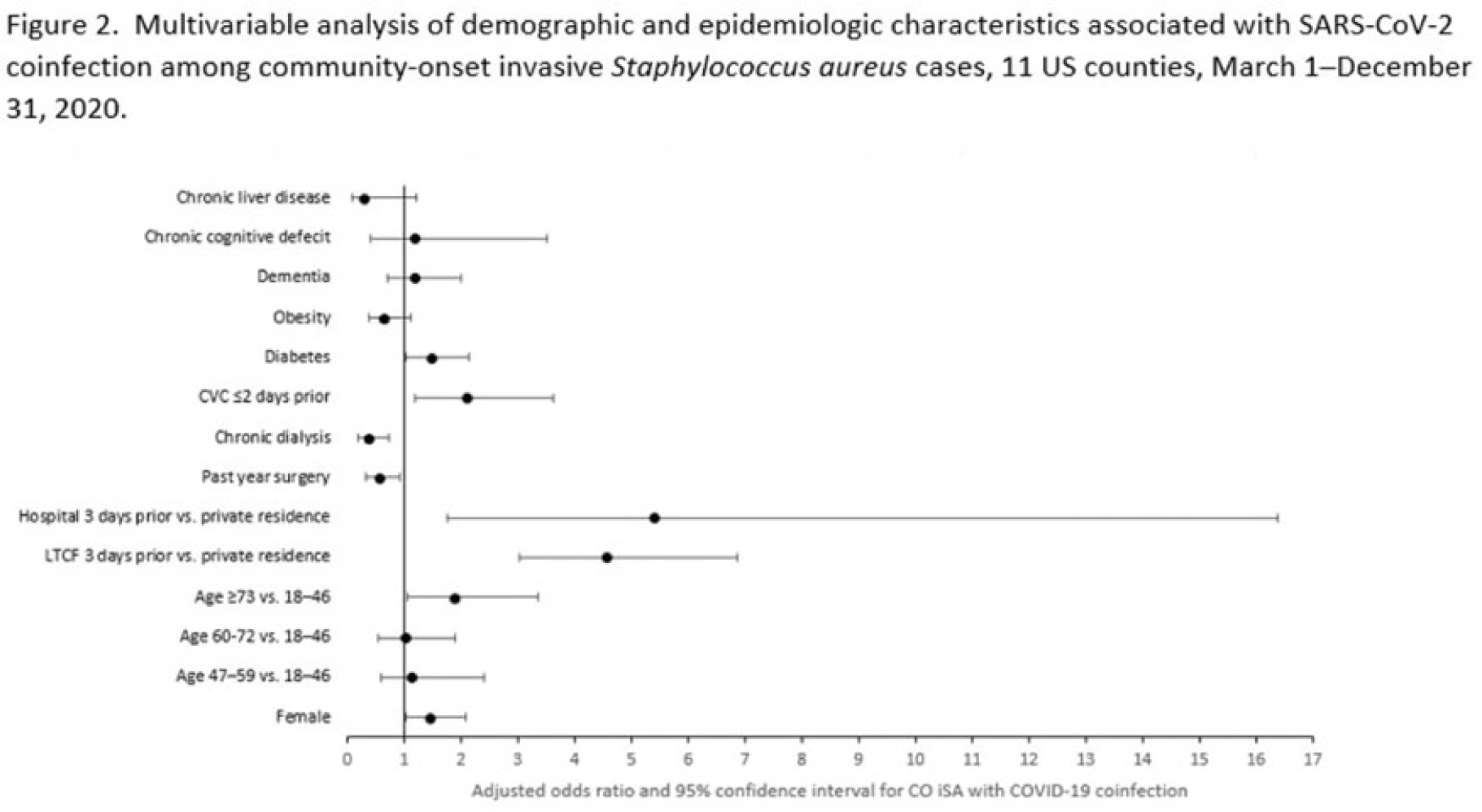
Disclosures: None
Increases in methicillin-sensitive Staphylococcus aureus bloodstream infection incidence, 2016–2019
- Kelly Jackson, Joelle Nadle, Susan Ray, Ruth Lynfield, Ghinwa Dumyati, Marissa Tracy, William Schaffner, David Ham, Isaac See
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s63-s64
-
- Article
-
- You have access Access
- Open access
- Export citation
-
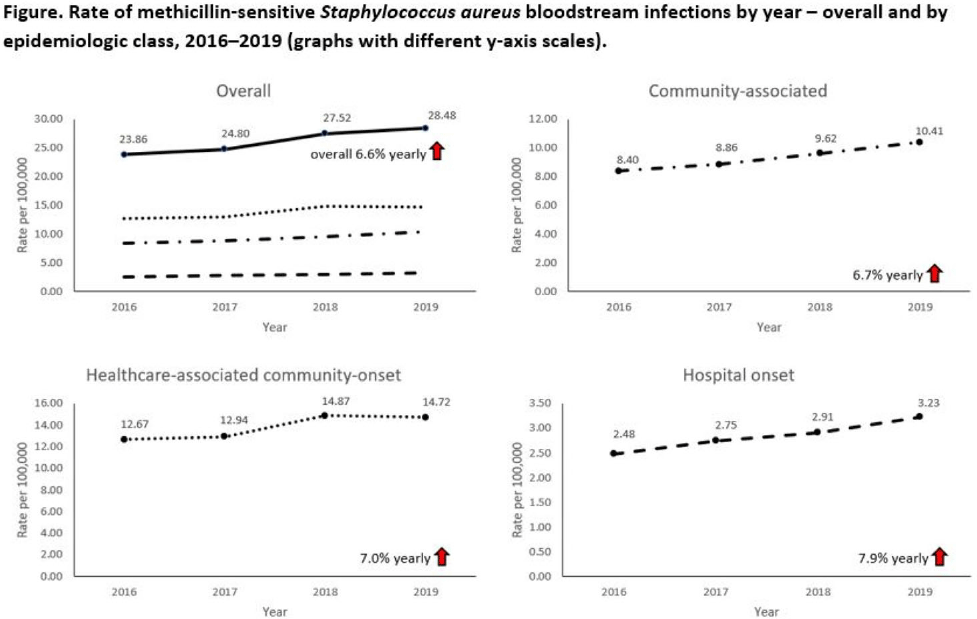
Background: Incidence of methicillin-sensitive Staphylococcus aureus (MSSA) bloodstream infections (BSIs) in the United States during 2012–2017 has been reported to have been stable for hospital-onset BSIs and to have increased 3.9% per year for community-onset BSIs. We sought to determine whether these trends continued in more recent years and whether there were further differences within subgroups of community-onset BSIs. Methods: We analyzed CDC Emerging Infections Program active, population- and laboratory-based surveillance data during 2016–2019 for MSSA BSIs from 8 counties in 5 states. BSI cases were defined as isolation of MSSA from blood in a surveillance area resident. Cases were considered hospital onset (HO) if culture was obtained >3 days after hospital admission and healthcare-associated community-onset (HACO) if culture was obtained on or after day 3 of hospitalization and was associated with dialysis, hospitalization, surgery, or long-term care facility residence within 1 year prior or if a central venous catheter was present ≤2 days prior. Cases were otherwise considered community-associated (CA). Annual rates per 100,000 census population were calculated for each epidemiologic classification; rates of HACO cases among chronic dialysis patients per 100,000 dialysis patients were calculated using US Renal Data System data. Annual increases were modeled using negative binomial or Poisson regression and accounting for changes in the overall population age group, and sex. Descriptive analyses were performed. Results: Overall, 8,344 MSSA BSI cases were reported. From 2016–2019 total MSSA BSI rates increased from 23.9 per 100,000 to 28.5 per 100,000 (6.6% per year; P < .01). MSSA BSI rates also increased significantly among all epidemiologic classes. HO cases increased from 2.5 per 100,000 to 3.2 per 100,000 (7.9% per year; P = .01). HACO cases increased from 12.7 per 100,000 to 14.7 per 100,000 (7.0% per year; P = .01). CA cases increased from 8.4 per 100,000 to 10.4 per 100,000 (6.7% per year; P < .01) (Fig. 1). Significant increases in MSSA BSI rates were also observed for nondialysis HACO cases (9.3 per 100,000 to 11.1 per 100,000; 7.8% per year; P < .01) but not dialysis HACO cases (1,823.2 per 100,000 to 1,857.4 per 100,000; 1.4% per year; P = .59). Healthcare risk factors for HACO cases were hospitalization in the previous year (82%), surgery (31%), dialysis (27%), and long-term care facility residence (19%). Conclusions: MSSA BSI rates increased from 2016–2019 overall, among all epidemiologic classes, and among nondialysis HACO cases. Efforts to prevent MSSA BSIs among individuals with healthcare risk factors, particularly those related to hospitalization, might have an impact on MSSA BSI rates.
Funding: None
Disclosures: None
Molecular Typing of Invasive Staphylococcus aureus from the Emerging Infections Program (EIP) Using Whole-Genome Sequencing
- Davina Campbell, Gillian McAllister, Kelly Jackson, Isaac See, Alison Halpin, Joseph Lutgring, Erin Epson, Susan Petit, Susan Ray, William Schaffner, Ghinwa Dumyati, Thomas Ewing, Michelle Adamczyk, Amy Gargis
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s71-s72
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: The CDC has performed surveillance for invasive Staphylococcus aureus (iSA) infections through the Emerging Infections Program (EIP) since 2004. SCCmec and spa typing for clonal complex (CC) assignment and genomic markers have been used to characterize isolates. In 2019, whole-genome sequencing (WGS) of isolates began, allowing for high-resolution assessment of genomic diversity. Here, we evaluate the reliability of SCCmec typing, spa typing, and CC assignment using WGS data compared to traditional methods to ensure that backwards compatibility is maintained. Methods:S. aureus isolates were obtained from a convenience sample of iSA cases reported through the EIP surveillance system. Overall, 78 iSA isolates with diverse spa repeat patterns, CCs, SCCmec types, and antimicrobial susceptibility profiles were sequenced (MiSeq, Illumina). Real-time PCR and Sanger sequencing were used as the SCCmec and spa typing reference methods, respectively. spa-MLST mapping (Ridom SpaServer) served as the reference method for CC assignment. WGS assembly and multilocus sequence typing (MLST) were performed using the CDC QuAISAR-H pipeline. WGS-based MLST CCs were assigned using eBURST and SCCmec types using SCCmecFinder. spa types were assigned from WGS assemblies using BioNumerics. For isolate subtyping, previously published and validated canonical single-nucleotide polymorphisms (canSNPs) as well as the presence of the Panton-Valentine leukocidin (PVL) toxin and arginine catabolic mobile element (ACME) virulence factor were assessed for all genome assemblies. Results: All isolates were assigned WGS-based spa types, which were 100% concordant (78 of 78) with Sanger-based spa typing. SCCmecFinder assigned 91% of isolates (71 of 78) SCCmec types, which were 100% concordant with reference method results. Also, 7 isolates had multiple cassettes predicted or an incomplete SCCmec region assembly. Using WGS data, 96% (75 of 78) of isolates were assigned CCs; 3 isolates had unknown sequence types that were single-locus variants of established sequence types. Overall, 70 isolates had CCs assigned by the reference method; 100% (70 of 70) concordance was observed with WGS-based CCs. Analysis of canSNPs placed 42% (33 of 78) of isolates into CC8, with 17 (52%) of these isolates classified as USA300. PVL and ACME were not accurate markers for inferring the USA300 subtype as 24% (4 of 17) of isolates did not contain these markers. Conclusions:S. aureus CCs, SCCmec, and spa types can be reliably determined using WGS. Incorporation of canSNP analysis represents a more efficient method for CC8 assignment than the use of genomic markers alone. WGS allows for the replacement of multiple typing methods for increased laboratory efficiency, while maintaining backward compatibility with historical typing nomenclature.
Funding: None
Disclosures: None
Characteristics Associated With Invasive Staphylococcus aureus Infection Rates in Nursing Homes, Emerging Infections Program
- Runa Gokhale, Kelly Jackson, Kelly Hatfield, Susan Petit, Susan Ray, Joelle Nadle, Christina B. Felsen, William Schaffner, Isaac See, Prabasaj Paul
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s60-s61
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Most invasive methicillin-resistant Staphylococcus aureus (iMRSA) infections have onset in the community but are associated with healthcare exposures. More than 25% of cases with healthcare exposure occur in nursing homes (NHs) where facility-specific iMRSA rates vary widely. We assessed associations between nursing home characteristics and iMRSA incidence rates to help target prevention efforts in NHs. Methods: We used active, laboratory- and population-based surveillance data collected through the Emerging Infections Program during 2011–2015 from 25 counties in 7 states. NH-onset cases were defined as isolation of MRSA from a normally sterile site in a surveillance area resident who was in a NH within 3 days before the index culture. We calculated MRSA incidence (cases per NH resident day) using Centers for Medicare & Medicaid Services (CMS) skilled nursing facility cost reports and described variation in iMRSA incidence by NH. We used Poisson regression with backward selection, assessing variables for collinearity, to estimate adjusted rate ratios (aRRs) for NH characteristics (obtained from the CMS minimum dataset) associated with iMRSA rates. Results: Of 590 surveillance area NHs included in analysis, 89 (15%) had no NH-onset iMRSA infections. Rates ranged from 0 to 23.4 infections per 100,000 resident days. Increased rate of NH-onset iMRSA infection occurred with increased percentage of residents in short stay ≤30 days (aRR, 1.09), exhibiting wounds or infection (surgical wound [aRR, 1.08]; vascular ulcer/foot infection [aRR, 1.09]; multidrug-resistant organism infection [aRR, 1.13]; receipt of antibiotics [aRR, 1.06]), using medical devices or invasive support (ostomy [aRR, 1.07]; dialysis [aRR, 1.07]; ventilator support [aRR, 1.17]), carrying neurologic diagnoses (cerebral palsy [aRR, 1.14]; brain injury [aRR, 1.1]), and demonstrating debility (requiring considerable assistance with bed mobility [aRR, 1.05]) (Table). iMRSA rates decreased with increased percentage of residents receiving influenza vaccination (aRR, 0.96) and with the presence of any patients in isolation for any active infection (aRR, 0.83). Conclusions: iMRSA incidence varies greatly across nursing homes, with many NH patient and facility characteristics associated with NH-onset iMRSA rate differences. Some associations (short stay, wounds and infection, medical device use and invasive support) suggest that targeted interventions utilizing known strategies to decrease transmission may help to reduce infection rates, while others (neurologic diagnoses, influenza vaccination, presence of patients in isolation) require further exploration to determine their role. These findings can help identify NHs in other areas more likely to have higher rates of NH-onset iMRSA who could benefit from interventions to reduce infection rates.
Funding: None
Disclosures: None
Antimicrobial Nonsusceptibility Among Invasive MRSA USA300 Strains by Healthcare Exposure, Three Sites, 2005–2016
- Kelly Jackson, Runa Gokhale, Davina Campbell, Amy Gargis, Susan Ray, Ruth Lynfield, William Schaffner, Joseph Lutgring, Isaac See
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s120-s121
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Incidence of community-associated (CA) and healthcare-associated, community-onset (HACO) USA300 methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections has remained unchanged in recent years. Traditionally considered a CA strain, USA300 is increasingly associated with healthcare settings. We examined whether antimicrobial nonsusceptibility among USA300 strains could distinguish epidemiologic class (community vs hospital), and whether divergences in susceptibility were occurring over time. Methods: We used data on invasive MRSA infections from active, population, and laboratory-based surveillance during 2005–2016 from 11 counties in 3 states. Invasive cases were defined as MRSA isolation from a normally sterile site in a surveillance area resident. Cases were considered hospital-onset (HO) if the culture was obtained >3 days after hospitalization and HACO if ≥1 of the following risk factors was present: hospitalization, surgery, dialysis, or residence in a long-term care facility in the past year; or central vascular catheter ≤2 days before culture. Otherwise, cases were considered CA. Sites submitted a convenience sample of clinical MRSA isolates for molecular typing and antimicrobial susceptibility testing. Molecular typing was performed by pulsed-field gel electrophoresis until 2008, when typing was inferred using a validated algorithm based on molecular characteristics. Reference broth microdilution was performed for 8 antimicrobials and interpreted based on CLSI interpretive criteria. We compared USA300 nonsusceptibility for HO and CA isolates. For antimicrobials with >5% nonsusceptibility and for which HO isolates had greater nonsusceptibility than CA isolates, we compared nonsusceptibility for HACO and CA and analyzed annual trends in nonsusceptibility within each epidemiologic class (ie, CA, HACO, and HO) using linear regression. Results: Of 17,947 MRSA cases during 2005–2016, isolates were available for 6,685 (37%), and 2,120 were USA300 (34% CA, 52% HACO, 14% HO). HO isolates had more nonsusceptibility than CA isolates to gentamicin (2.2% vs 0.6%; P = .03), levofloxacin (47.8% vs 39.7%; P = .02), rifampin (3.7 vs 1.1%; P = .01), and trimethoprim-sulfamethoxazole (3.4% vs 0.6%; P = .04). HACO isolates also had more nonsusceptibility than CA isolates to levofloxacin (50.9% vs 39.7%; P < .01). Levofloxacin nonsusceptibility increased during 2005–2016 for HACO and CA isolates (P < .01), but not among HO isolates (P = .36) (Fig. 1). Conclusions: Overall, nonsusceptibility across drugs cannot distinguish USA300 isolates causing HO versus CA disease. Although HO isolates had higher levofloxacin nonsusceptibility than CA and HACO isolates early on, USA300 MRSA HACO isolates now have levofloxacin nonsusceptibility most similar to that of HO isolates. Further study could help to explore whether increases in fluoroquinolone nonsusceptibility among CA and HACO cases may be contributing to the persistence of USA300 strains.
Disclosures: None
Funding: None
2555 Predictive cytological topography (PiCT): A radiopathomics approach to mapping prostate cancer
- Sean D. McGarry, Sarah L. Hurrell, Kenneth Ickzkowski, Anjishnu Banerjee, Kenneth Jacobsohn, William Hall, Mark Hohenwalter, Peter LaViolette, Amy Kaczmarowski, Tucker Keuter, Marja Nevalainen, William See
-
- Journal:
- Journal of Clinical and Translational Science / Volume 2 / Issue S1 / June 2018
- Published online by Cambridge University Press:
- 21 November 2018, pp. 23-24
-
- Article
-
- You have access Access
- Open access
- Export citation
-
OBJECTIVES/SPECIFIC AIMS: The objective of this study is to use machine Learning techniques to generate maps of epithelium and lumen density in MRI space. METHODS/STUDY POPULATION: Methods: We prospectively recruited 39 patients undergoing prostatectomy for this institutional review board (IRB) approved study. Patients underwent MP-MRI before prostatectomy on a 3T field strength MRI scanner (General Electric, Waukesha, WI, USA) using an endorectal coil. MP-MRI included field-of-view optimized and constrained undistorted single shot (FOCUS) diffusion weighted imaging with 10 b-values (b=0, 10, 25, 50, 80, 100, 200, 500, 1000, and 2000), dynamic contrast enhanced imaging, and T2-weighted imaging. T2 weighted images were intensity normalized and apparent diffusion coefficient maps were calculated. The dynamic contrast enhanced data was used to calculate the percent change in signal intensity before and after contrast injection. All images were aligned to the T2 weighted image. Robotic prostatectomy was performed 2 weeks after image acquisition. Prostate samples were sliced using a 3D printed slicing jig matching the slice profile of the T2 weighted image. Whole mount samples at 10 μm thickness were taken, hematoxylin and eosin stained, digitized, and annotated by a board certified pathologist. A total of 210 slides were included in this study. Lumen and epithelium were automatically segmented using a custom algorithm written in MATLAB. The algorithm was validated by comparing manual to automatic segmentation on 18 samples. Slides were aligned with the T2 weighted image using a nonlinear control point warping technique. Lumen and epithelium density and the expert annotation were subsequently transformed into MRI space. Co-registration was validated by applying a known warp to tumor masks noted by the pathologist and control point warping the whole mount slide to match the transform. Overlap was measured using a DICE coefficient. A learning curve was generated to determine the optimal number of patients to train the algorithm on. A PLS algorithm was trained on 150 random permutations of patients incrementing from 1 to 29 patients. Slides were stratified such that all slides from a single patient were in the same cohort. Three cohorts were generated, with tumor burden balanced across all cohort. A PLS algorithm was trained on 2 independent training sets (cohorts 1 and 2) and applied to cohort 3. The input vector consisted of MRI values and the target variable was lumen and epithelium density. The algorithm was trained lesion-wise. Trained PiCT models were applied to the test cohort voxel-wise to generate 2 new image contrasts. Mean lesion values were compared between high grade, low grade, and healthy tissue using an ANOVA. An ROC analysis was performed lesion-wise on the test set. RESULTS/ANTICIPATED RESULTS: Results: The segmentation accuracy validation revealed R=0.99 and R=0.72 (p<0.001) for lumen and epithelium, respectively. The co-registration accuracy revealed a 94.5% overlap. The learning curve stabilized at 10 patients with a root mean square error of 0.14, thus the size of the 2 independent training cohorts was set to 10, leaving 19 for the test cohort. DISCUSSION/SIGNIFICANCE OF IMPACT: We present a technique for combining radiology and pathology with machine learning for generating predictive cytological topography (PiCT) maps of cellularity and lumen density prostate. The voxel-wise approach to mapping cellular features generates 2 new interpretable image contrasts, which can potentially increase confidence in diagnosis or guide biopsy and radiation treatment.
Completeness of Methicillin-Resistant Staphylococcus aureus Bloodstream Infection Reporting From Outpatient Hemodialysis Facilities to the National Healthcare Safety Network, 2013
- Duc B. Nguyen, Isaac See, Nicole Gualandi, Alicia Shugart, Christi Lines, Wendy Bamberg, Ghinwa Dumyati, Lee H. Harrison, Lindsey Lesher, Joelle Nadle, Susan Petit, Susan M. Ray, William Schaffner, John Townes, Levi Njord, Dawn Sievert, Nicola D. Thompson, Priti R. Patel
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 37 / Issue 2 / February 2016
- Published online by Cambridge University Press:
- 11 November 2015, pp. 205-207
- Print publication:
- February 2016
-
- Article
- Export citation
-
Reports of bloodstream infections caused by methicillin-resistant Staphylococcus aureus among chronic hemodialysis patients to 2 Centers for Disease Control and Prevention surveillance systems (National Healthcare Safety Network Dialysis Event and Emerging Infections Program) were compared to evaluate completeness of reporting. Many methicillin-resistant S. aureus bloodstream infections identified in hospitals were not reported to National Healthcare Safety Network Dialysis Event.
Infect. Control Hosp. Epidemiol. 2016;37(2):205–207
Contributors
-
- By Yasir Abu-Omar, Matthew E. Atkins, Joseph E. Arrowsmith, Alan Ashworth, Rubia Baldassarri, Craig R. Bailey, David J. Barron, Christiana C. Burt, David Cardone, Coralie Carle, Jose Coddens, Alan M. Cohen, Simon Colah, Sarah Conolly, David J. Daly, Helen M. Daly, Stefan G. De Hert, Ravi J. De Silva, Mark Dougherty, John J. Dunning, Maros Elsik, Betsy Evans, Florian Falter, Nigel Farnum, Jens Fassl, Juliet E. Foweraker, Simon P. Fynn, Andrew I. Gardner, Margaret I. Gillham, Martin J. Goddard, Maximilien J. Gourdin, Jon Graham, Stephen J. Gray, Cameron Graydon, Fabio Guarracino, Roger M. O. Hall, Michael Haney, Charles W. Hogue, Ben W. Howes, Bevan Hughes, Siân I. Jaggar, David P. Jenkins, Jörn Karhausen, Todd Kiefer, Khalid Khan, Andrew A. Klein, John D. Kneeshaw, Andrew C. Knowles, Catherine V. Koffel, R. Clive Landis, Trevor W. R. Lee, Clive J. Lewis, Jonathan H. Mackay, Amod Manocha, Jonathan B. Mark, Sarah Marstin, William T. McBride, Kenneth H. McKinlay, Alan F. Merry, Berend Mets, Britta Millhoff, Kevin P. Morris, Samer A. M. Nashef, Andrew Neitzel, Stephane Noble, Rabi Panigrahi, Barbora Parizkova, J. M. Tom Pierce, Mihai V. Podgoreanu, Hans-Joachim Priebe, Paul Quinton, C. Ramaswamy Rajamohan, Doris M. Rassl, Tom Rawlings, Fiona E. Reynolds, Andrew J. Richardson, David Riddington, Andrew Roscoe, Paul H. M. Sadleir, Ving Yuen See Tho, Herve Schlotterbeck, Maura Screaton, Shitalkumar Shah, Harjot Singh, Jon H. Smith, M. L. Srikanth, Yeewei W. Teo, Kamen P. Valchanov, Jean-Pierre van Besouw, Isabeau A. Walker, Stephen T. Webb, Francis C. Wells, John Whitbread, Charles Willmott, Patrick Wouters
- Edited by Jonathan H. Mackay, Joseph E. Arrowsmith
-
- Book:
- Core Topics in Cardiac Anesthesia
- Published online:
- 05 April 2012
- Print publication:
- 15 March 2012, pp x-xiii
-
- Chapter
- Export citation






